Abstract
The potential effect of prenatal LPS exposure on the postnatal acute phase response (APR) to an LPS challenge in heifers was determined. Pregnant crossbred cows were separated into prenatal immune stimulation (PIS) and saline groups (Control). From these treatments, heifer calves were identified at weaning to subsequently receive an exogenous LPS challenge. Sickness behavior scores (SBS) were recorded and blood samples were collected at 30-min intervals from −2 to 8 h and again at 24 h relative to the LPS challenge. There was a treatment × time interaction for the change in vaginal temperature (VT) such that the change in VT was greater in Control than PIS from 150 to 250 min, yet it was greater in PIS than Control from 355 to 440 min and from 570 to 1145 min. There was also a treatment × time interaction for SBS such that scores were greater in Control than PIS at 0.5 h, yet were greater in PIS than Control from 2.5 to 4 h post-LPS. There was a tendency for a treatment × time interaction for serum concentrations of IL-6, which were greater in PIS than Control heifers from 5.5 to 6 h and from 7 to 8 h post-challenge. Thus, a single exposure to LPS during gestation can alter the postnatal APR to LPS in heifer calves.
Introduction
Prenatal stress has been demonstrated to influence various aspects of the postnatal stress and immune responses.1–3 The effects of prenatal stress, as reported in animal research models, are dependent upon the timing of the stress during gestation, as well as the sex of the offspring. 4 Effects on the offspring include increased hypothalamic–pituitary–adrenal axis sensitivity and behavioral changes. Prenatal stress can also alter leukocyte numbers and function, and the Ab response to subsequent vaccination.1,2 It has been demonstrated previously that repeated transportation of pregnant dams altered concentrations of glucose and NEFA in the dams, potentially due to increased epinephrine and cortisol action. 5 However, there are limited prenatal stress studies that utilize cattle as the model, none of which has directly evaluated the potential effects of prenatal immune stimulation. Lay et al. 6 demonstrated reduced cortisol clearance rates in calves from dams transported repeatedly during gestation. It is believed that the increased fetal exposure to cortisol may influence the ability of the calf to adapt to stress, and therefore affect immune responsiveness.
Our laboratory previously reported prenatal transportation can alter the postnatal acute phase response (APR) in Brahman bulls. Specifically, prenatally transported bull calves had greater rectal temperature, TNF-α and IFN-γ responses, and decreased cortisol and IL-6 responses following an LPS challenge post-weaning. 7 While typical production stressors, such as the relocation process, may alter stress and immune responsiveness in offspring, little is known about the effects of immune challenges during gestation on the offspring in cattle. Therefore, the objective of this study was to determine the potential effects of prenatal LPS exposure on the postnatal APR of heifers following an LPS challenge.
Materials and methods
Experimental design
All experimental procedures were in compliance with the Guide for the Care and Use of Agricultural Animals in Research and Teaching and approved by the Institutional Animal Care and Use Committee at the University of Florida (IACUC Study #201307950).
Pregnant crossbred cows [n = 50; 527 ± 46 kg body mass (BM)] were selected from a single herd managed similarly prior to allocation to treatments in the current study. Cows had an average parity of 5.8 ± 1.8 calvings. Cows were randomly allotted to 1 of 2 treatments: (1) prenatal immune stimulation [PIS; n = 25, administered 0.1 µg/kg BM LPS (Escherichia coli O111:B4; Sigma Aldrich, St. Louis, MO, USA) s.c. at 233 ± 19 d of gestation] and (2) saline group (Control; n = 25). Cows were administered LPS or saline on 25 September 2013. There are limited data on the effect of LPS administration on pregnant cows. Therefore, the dose of LPS administered and route of administration was selected in order to produce a physiological response in the dam, as measured by indwelling vaginal temperature devices, but not too high in order to prevent abortion of the fetus. A preliminary study was conducted on cows in the last third of gestation and confirmed this dose would not cause late-term abortions. Use of the specific LPS serotype (O111:B4 from E. coli) has been documented multiple times by the Livestock Issues Research Unit to produce a reliable and controlled response to LPS.7–9 Vaginal temperature recording devices 10 were inserted 2 d prior and removed 5 d post-LPS administration; four additional temperature-recording devices were used to record ambient temperature. Cows grazed a common bahiagrass pasture from the time of LPS administration to weaning. Calves were born October–December 2013 and birth date was recorded. At birth all calves were tagged and weighed, and male calves were surgically castrated. Day of gestation for LPS administration to dams was computed from calf birth date assuming a constant gestation length of 283 d. On 8 April 2014 calves were dewormed (fenbendazole 10% oral drench; Safe Guard, Merck Animal Health, Madison, NJ, USA), and vaccinated against Mannheimia haemolytica type A1 (One Shot; Zoetis, Florham Park, NJ, USA), infectious bovine rhinotracheitis virus, bovine viral diarrhea virus types 1 and 2, parainfluenza3 virus, bovine respiratory syncytial virus (Bovi-Shield Gold 5; Zoetis), Clostridium chauvoei, Clostridium speticum, Clostridium haemolyticum, Clostridium novyi, Clostridium sordelli and Clostridium perfringens types B, C and D (Ultrabac 8; Zoetis). On 4 June 2014 calves were administered a booster vaccination of Bovi-Shield Gold 5 and Ultrabac 8. Heifer calves were weaned and weaning mass recorded on 7 July 2014, to allow sufficient time to halter break the heifers prior to the LPS challenge on 13 August 2014. Steer calves were weaned and weaning mass recorded on 16 July 2014.
From these treatments, heifer calves (12 PIS and 11 Control) were identified at weaning (238 ± 15 d of age) to subsequently receive an LPS challenge. Heifer calves from the Control group averaged 217 ± 5 kg BM and PIS heifers averaged 211 ± 5 kg BM. On d 0, heifers were fitted with indwelling vaginal temperature-recording devices that measured vaginal temperature continuously at 5-min intervals, 10 and jugular vein catheters. Heifers were then moved into individual pens (2.5 m × 6 m) in a covered barn. Heifers were allowed ad libitum access to feed and water throughout the study. On d 1, heifers were challenged i.v. with LPS (0.5 µg/kg BM) at 0 h (1000 h). The 0.5-µg/kg BM dose of LPS was chosen for the heifers based on previous work in weaned calves demonstrating a predictable and controlled response without producing mortality.7–9 Sickness behavior scores (SBS) were recorded and whole blood samples were collected at 30-min intervals from −2 to 8 h and again at 24 h relative to the LPS challenge at 0 h.
Whole blood for serum was collected into Sarstedt tubes containing no additive (Sarstedt, Inc., Newton, NC, USA), and were allowed to clot at room temperature for 30 min prior to centrifugation at 1500 g for 20 min at 4℃. Isolated serum was stored at −80℃ until analyzed for cytokine concentrations. Whole blood for flow cytometry and neutrophil gene expression was collected into BD vacutainer tubes containing sodium heparin (Becton Dickinson, Franklin Lakes, NJ, USA) at −1, 4 and 8 h relative to the LPS challenge and processed immediately for neutrophil extraction and flow cytometry.
Sickness behavior scores
A trained observer assessed and recorded each heifer’s SBS by visual observation following the collection of each blood sample. Heifers were scored on a scale of 1 to 4 using 0.25-unit increments. Specifically, heifers scored as 1 maintained normal maintenance behavior; heifers scored as 2 were calm but with head distended and increased respiration; heifers scored as 3 displayed clinical signs of sickness, increased respiration and drool; heifers scored as 4 were observed lying on side with labored breathing and frothing at the mouth. Intervention would occur on any heifer with a SBS of 4. Heifers were scored by the same observer throughout the study who has over 23 yr animal behavior experience and 10 yr experience in observing sickness behavior in cattle.
Cytokine analysis
Serum cytokine concentrations (TNF-α, IFN-γ and IL-6) were measured in duplicate and were determined by a custom bovine 3-plex sandwich-based chemiluminescence ELISA kit (Searchlight-Aushon BioSystems, Inc., Billerica, MA, USA). The minimum detectable concentrations were 0.5, 0.1 and 3.3 pg/ml for TNF-α, IFN-γ, and IL-6, respectively. All intra-assay coefficients of variation were < 5.6% and all inter-assay coefficients of variation were < 16.0% for all assays.
Flow cytometry
The concentrations and percentages of leukocytes in whole blood collected at −1, 4 and 8 h relative to the LPS challenge were determined by flow cytometry. One ml heparinized whole blood was centrifuged at 650 g for 5 min at 4℃. RBCs were removed by mixing the cell pellet with 1 ml ice-cold hypotonic RBC lysis buffer (10.6 mM Na2HPO4, 2.7 mM NaH2PO4, pH 7.2; Fisher Scientific, Waltham, MA, USA) for 1 min on ice, followed by addition of 0.5 ml restoring buffer (10.6 mM Na2HPO4, 2.7 mM NaH2PO4, 462 mM NaCl, pH 7.2). Cells were pelleted by centrifugation at 650 g for 10 min at 4℃. The procedure was repeated twice to remove RBC. Cells were then re-suspended in 100 μl staining buffer (Dulbecco PBS, Life Technologies, Carlsbad, CA, USA), 5% heat-inactivated FBS (Hyclone, Logan, UT, USA) and 0.1% NaN3 containing 0.1 µg FITC-conjugated mouse anti-human CD14 Ab (Tük4 clone; Life Technologies). Samples were incubated in the dark on ice for 30 min, then washed with 1 ml staining buffer, followed by centrifugation at 650 g for 5 min at 4℃. Cells were then incubated with PBS containing 2% paraformaldehyde at 4℃ overnight (12–18 h), centrifuged and re-suspended in 1 ml PBS. Cells were analyzed the next day on a BD Accuri™ C6 flow cytometer (Becton Dickinson) equipped with 488 nm and 633 nm lasers. For each sample, 100 μl of sample was analyzed and cell populations were determined using FlowJo analysis software (Version 10.0.6; FlowJo, LLC, Ashland, OR, USA). Gates for CD14-positive cells were set using a non-stained control sample. The cell count and percentage of CD14-positive cells for mononuclear and granulocyte populations were determined from the event counts in the respective gates.
Neutrophil gene expression
Whole-blood samples collected into 10-ml heparin tubes were centrifuged at 1500 g for 20 min, and the plasma and buffy coats were then removed. The RBC pack was re-suspended in 10 ml cold hypotonic lysis buffer, mixed gently for 1 min, followed by addition of 5 ml restoring buffer and centrifugation at 650 g for 5 min at 4℃. The RBC lysis step was repeated twice and then remaining neutrophil pellets were placed on dry ice before storage at −80℃.
Total RNA was extracted from neutrophil samples by using Trizol Reagent (Life Technologies), and reconstituted with 50 μl RNase-free water. RNA was quantified using a BioTek Synergy HT plate reader with a Take3 Micro-Volume plate. RT-PCR was performed using the High Capacity cDNA Reverse Transcription Kit (Life Technologies) in a 20-μl reactions that contained 10 μl RNA sample, RNase inhibitor and random primers. Reactions were incubated at ambient temperature for 10 min, followed by 2 h at 37℃ and 5 min at 85℃. The cDNA samples were diluted 1:10 with DNase-free water and stored at −20℃.
Primer sequences for qPCR.
Nomenclature for the β-defensins is based on their original definitions. 37 Alternative names for BNBD genes are DEFB3, DEFB4A, DEFB7 and DEFB10.
Accesion numbers from NCBI database http://www.ncbi.nlm.nih.gov.
In vitro fibroblast challenge
Skin biopsies were obtained from the ears of heifers at 462 ± 22 d of age using an ear notcher. Prior to collection, the skin site was clipped and thoroughly cleaned with alternating application of betadine scrub and 70% alcohol. The ear-notch sample was placed into a 50-ml tube containing 30 ml transport media [PBS with antibiotic/antimycotic (100 U/ml penicillin, 100 µg/ml streptomycin and 0.25 µg/ml amphotericin (Hyclone)] and placed on ice. Samples were then shipped on ice by overnight courier to the University of Vermont. Dermal fibroblast cultures were established by collagenase digestion and cryopreserved as previously described. 11 Aliquots of cells were revived from cryopreservation, expanded and fourth passage cells were then seeded into six-well plates (2.5 × 105 cells/well) in DMEM containing 5% FBS (Hyclone), 1 × insulin-transferrin-selenium (Mediatech Inc., Herndon, VA, USA) and 1 × antibiotic cocktail. Following a 24-h incubation, media were removed and replaced with 2 ml fresh media (negative control), or media containing either 10 ng/ml recombinant bovine IL-1β (AbD Serotec, Raleigh, NC, USA) or 100 ng/ml ultra-pure LPS isolated from E. coli 0111:B4 (Sigma-Aldrich). Media was harvested 36 h later and stored at −20℃ until analysis. Some cultures did not revive from cryopreservation, leaving only eight cultures per treatment group for complete analysis. Media concentrations of IL-8 and IL-6 were determined by ELISA using commercially available kits from Mabtech and Thermo Scientific, respectively. The minimum detectable concentrations were 150 pg/ml.
Statistical analysis
Blood parameters, flow cytometry and neutrophil gene expression data were analyzed by the MIXED procedure of SAS specific for repeated measures (SAS Institute Inc., Cary, NC, USA). Treatment, time and their interaction were included as fixed effects with heifers within treatment as the subject. Due to treatment differences in basal values, vaginal temperature was further analyzed as the change in vaginal temperature relative to 0 h values. Dermal fibroblast data were analyzed using the MIXED procedure of SAS, with prenatal treatment as a fixed effect. Specific treatment comparisons were made using the PDIFF option in SAS, with P < 0.05 considered significant and 0.05 < P < 0.10 considered a tendency. All data are presented as the LSM ± SEM.
Results
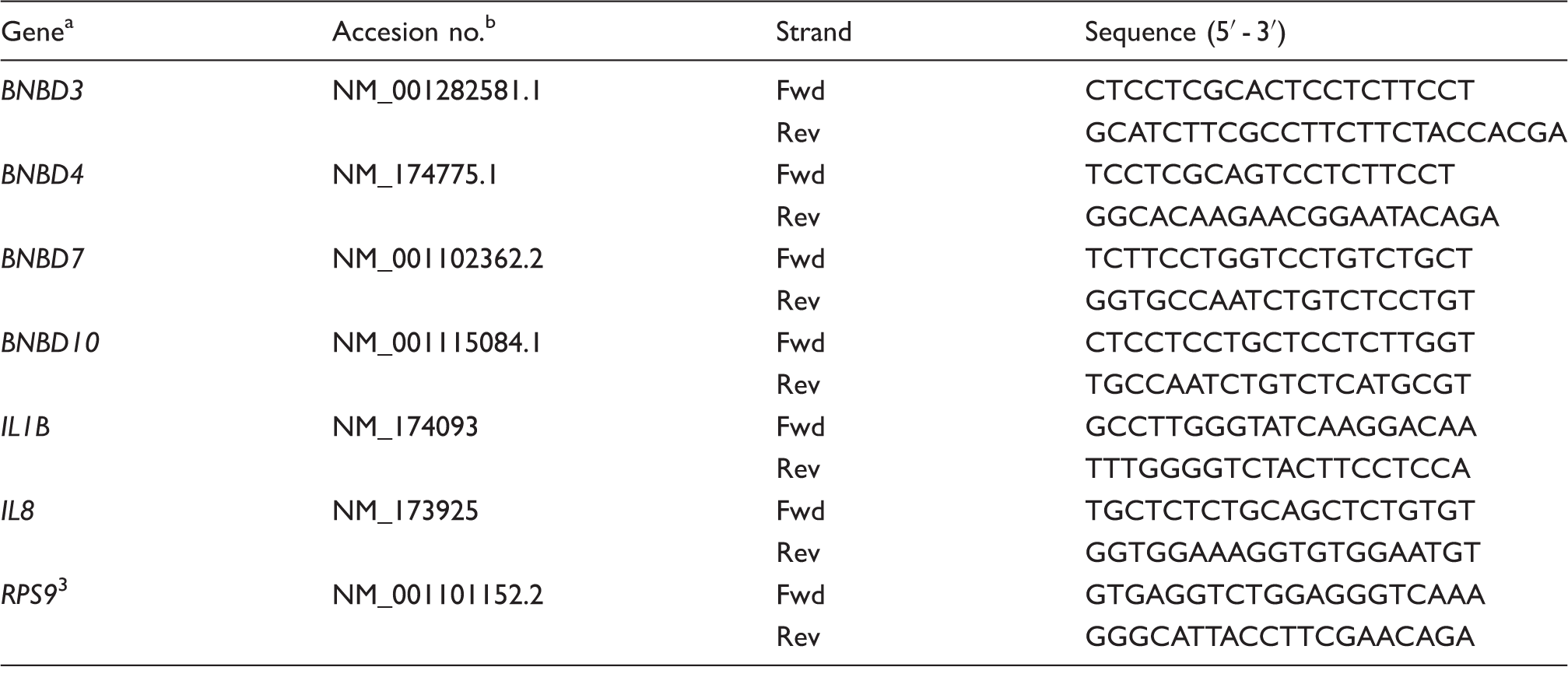
There was a treatment × time interaction (P < 0.001) for vaginal temperature. Specifically, vaginal temperature was greater in Control than PIS heifers from −115 to 285 min (P ≤ 0.04), at 660 min (P = 0.04), at 670 min (P = 0.05), from 685 to 720 min (P ≤ 0.04), from 745 to 1040 min (P ≤ 0.05) and from 1050 to 1075 min (P ≤ 0.05; Figure 1). There was also a time (P < 0.001) and treatment effect (P < 0.001) where Control heifers had greater overall vaginal temperature compared with PIS heifers (39.11 vs. 39.09 ± 0.004℃, respectively). Upon further analysis, a treatment effect was observed (P < 0.001) for vaginal temperature in the 12-h baseline period prior to the LPS challenge. Specifically, vaginal temperature was greater in Control heifers (38.92 ± 0.01℃) as compared with PIS heifers (38.85 ± 0.01℃). Due to the treatment differences prior to the challenge, vaginal temperature was analyzed as the change in vaginal temperature relative to 0 h values. There was a treatment × time interaction (P < 0.001) for the change in vaginal temperature relative to 0 h. The change in vaginal temperature was less in PIS heifers from 150 min to 250 min, yet was greater from 355 to 440 min, from 575 to 1125 min and from 1140 to 1145 min compared with Control heifers (P ≤ 0.05). There was also a time (P < 0.001) and a treatment effect (P < 0.001) such that the change in vaginal temperature was greater in PIS heifers (0.36 ± 0.01℃) than in Control heifers (0.10 ± 0.01℃).
Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on the vaginal temperature response to a postnatal LPS challenge (0.5 µg/kg BM). Control n = 11, PIS, n = 12. Data are presented as the LSM. SEM is ± 0.09 for Control and ± 0.08 for PIS.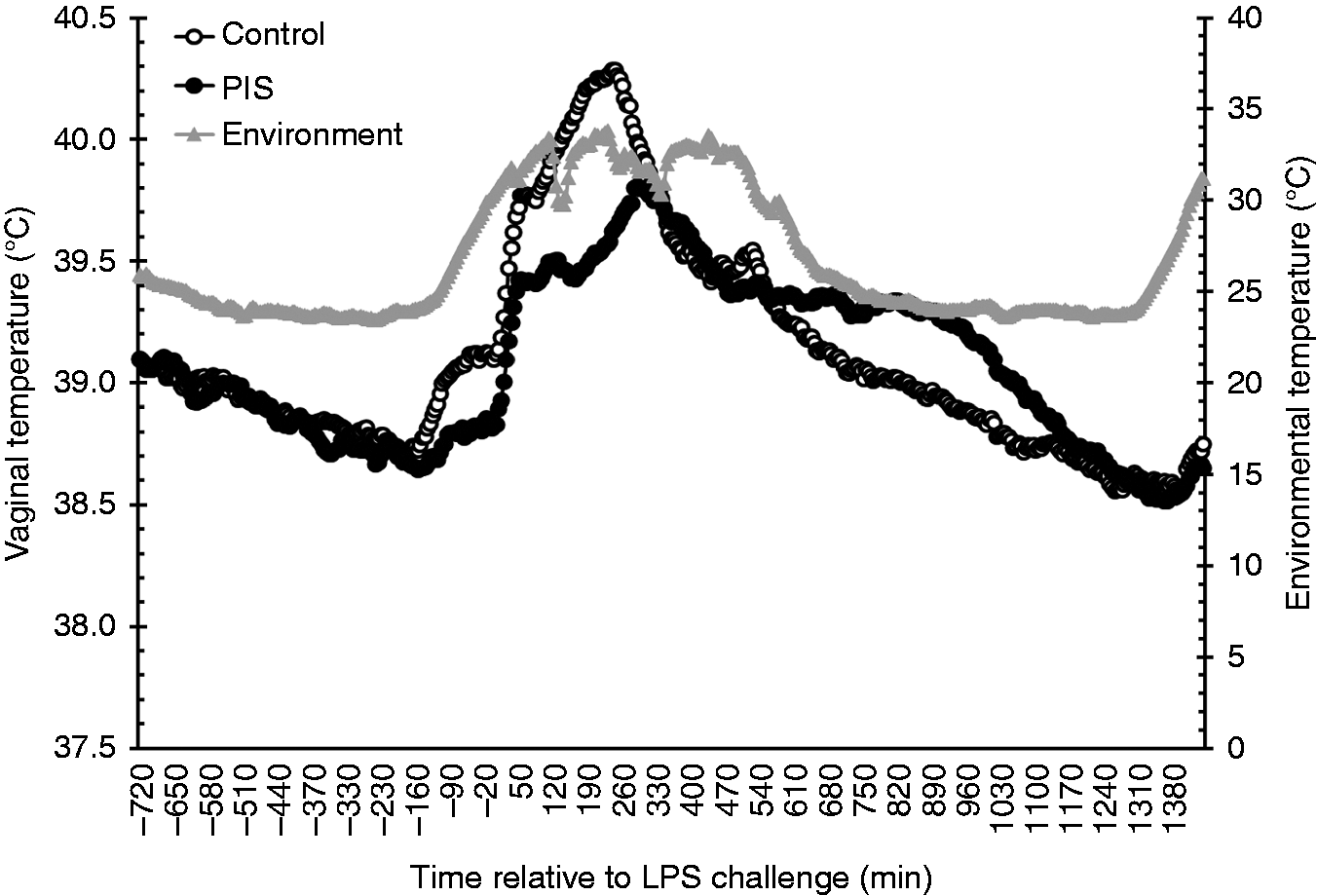
Sickness behavior scores were affected by a treatment × time interaction (P = 0.021; Figure 2). While sickness behavior was greater Control heifers at 0.5 h (P = 0.040), an extended sickness behavior was observed in PIS heifers such that SBS were elevated from 2.5 to 4 h in PIS heifers compared with Control heifers (P ≤ 0.05). Thus, there were time (P < 0.001) and treatment effects (P = 0.002), with PIS heifers producing greater SBS (1.15 ± 0.01) than Control heifers (1.10 ± 0.01).
Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on the sickness behavior response to a postnatal LPS challenge (0.5 µg/kg BM). Control n = 11, PIS, n = 12. Data are presented as the LSM ± SEM. *Treatments differ P ≤ 0.05.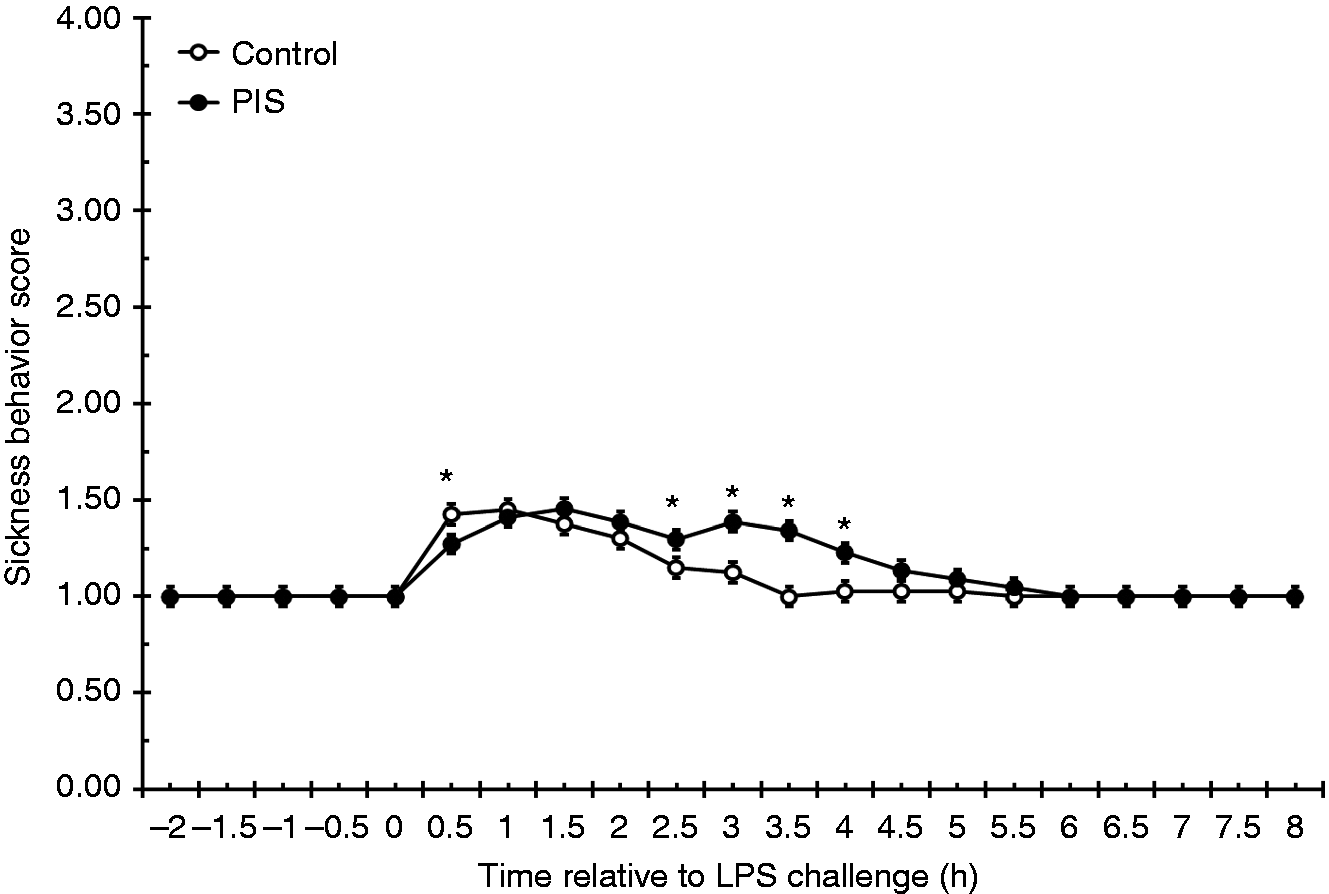
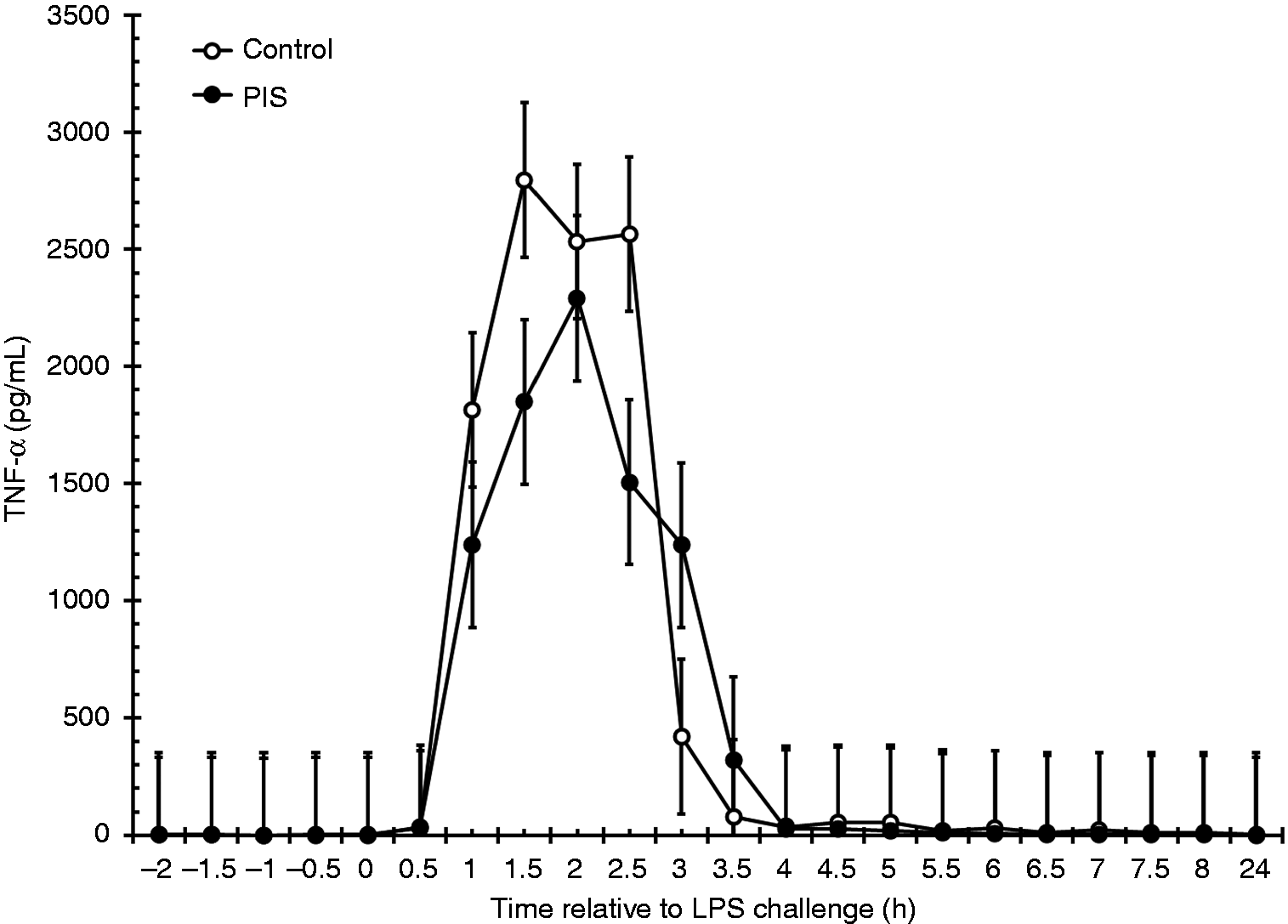
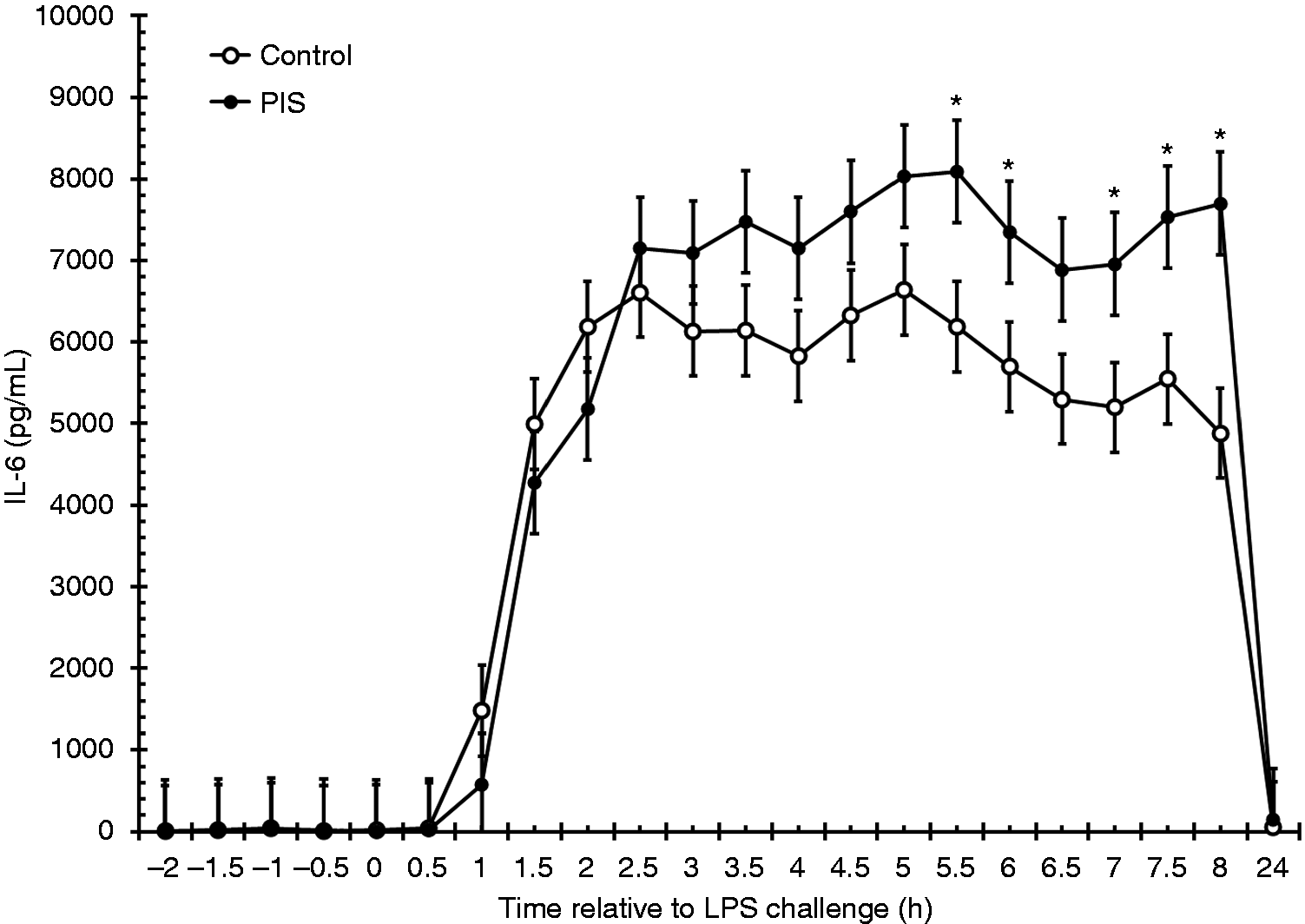
Serum concentrations of IFN-γ were not affected by treatment (P = 0.327) or by a treatment × time interaction (P = 0.598). However, there was an effect of time (P < 0.001; Figure 3), where IFN-γ increased at 3 h post-LPS (P = 0.050; 0 h vs. 3 h), and remained elevated until 4 h post-LPS (P = 0.486; 0 h vs. 4 h). Similarly, there was no effect of treatment (P = 0.396) or treatment × time (P = 0.912) for TNF-α, but there was an effect of time (P < 0.001; Figure 4). Concentrations of TNF-α increased within 1 h (P < 0.001; 0 h vs. 1 h), and remained elevated until 3.5 h post-challenge (P = 0.563; 0 h vs. 3.5 h). There was also a tendency (P = 0.056) for a treatment × time interaction for serum IL-6 concentrations, such that IL-6 was greater in PIS heifers from 5.5 to 6 h (P ≤ 0.050) and from 7 h to 8 h (P ≤ 0.037; Figure 5). There was a treatment (P < 0.001) and time (P < 0.001) effect, with PIS heifers producing greater serum IL-6 (4512 ± 134 pg/ml) than Control heifers (3788 ± 118 pg/ml).
Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on the IFN-γ response to a postnatal LPS challenge (0.5 µg/kg BM). Control n = 11, PIS, n = 12. Data are presented as the LSM ± SEM. Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on the TNF-α response to a postnatal LPS challenge (0.5 µg/kg BM). Control n = 11, PIS, n = 12. Data are presented as the LSM ± SEM. Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on the IL-6 response to a postnatal LPS challenge (0.5 µg/kg BM). Control n = 11, PIS, n = 12. Data are presented as the LSM ± SEM. *Treatments differ P ≤ 0.05.
The number of mononuclear cells and granulocytes in blood along with the percentage of each population that were positive for CD14 were not affected by treatment (P > 0.45) or treatment × time interaction (P > 0.62). There was an effect of time on the number of mononuclear cells and granulocytes in blood (P < 0.001), and the percentage of each population that were positive for CD14 (P < 0.001), where the number of mononuclear cells and granulocytes and the percentage of mononuclear cells in both treatment groups that were positive for CD14 was substantially less at 4 h and 8 h post-LPS challenge compared with −1 h pre-challenge (P < 0.001, −1 h vs. 4 h). In contrast, the percentage of granulocytes positive for CD14 in both groups increased from just over 10% at −1 h to approximately 70% at 4 h (P < 0.001, −1 h vs. 4 h; Figure 6) and 50% at 8 h relative to LPS challenge (P < 0.001, −1 h vs. 8 h; Figure 6). There was a tendency for a treatment × time interaction for the number of CD14 + monocytes in blood, where the PIS group had fewer CD14 + monocytes than the Control group at −1 h (P = 0.012) but no difference at 4 h (P = 0.993) and 8 h (P = 0.747) post-LPS challenge.
Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on the peripheral blood leukocyte profile during a postnatal LPS challenge (0.5 µg/kg BM). (A) Representative flow plots for blood samples collected at –1 h, 4 h and 8 h relative to the postnatal LPS challenge showing the gating criteria for mononuclear cells, granulocytes and cells positive for FITC-conjugated anti-CD14. (B) Cell counts/ml of blood and percentage of CD14-positive cells are presented as the LSM ± SEM. Control n = 11, PIS, n = 12. *Treatments differ P ≤ 0.05.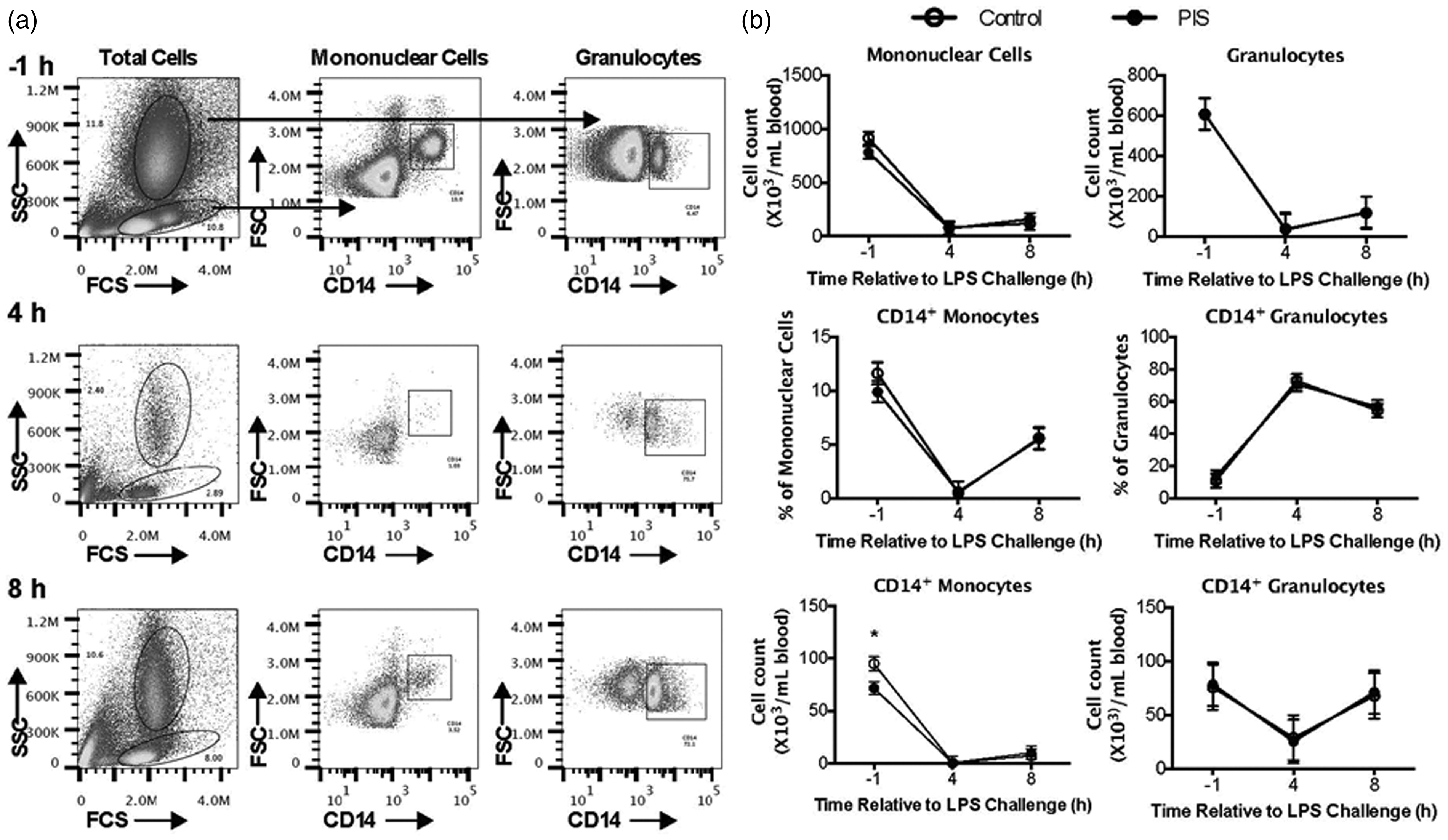
Expression of the host defense gene BNBD3 in neutrophils was not affected by time (P = 0.407) or treatment × time interaction (P = 0.112). However, at 8 h post-LPS challenge neutrophil BNBD3 expression was threefold greater in the PIS group than the Control group (P = 0.051) and there was a tendency for BNBD3 to increase with time in the PIS group (P = 0.058; −1 h vs. 8 h) but not the Control group (P = 0.356, −1 h vs. 4 h; P = 0.498, −1 h vs 8 h). BNBD4 expression in neutrophils was not affected by treatment (P = 0.426) or treatment × time interaction (P = 0.731), but increased approximately 20-fold at 4 h post-LPS challenge in both groups (P < 0.001, −1 h vs. 4 h) and remained elevated at 8 h post-LPS challenge (P < 0.001, −1 h vs. 8 h). There also was an effect of time on neutrophil BNBD7 expression (P = 0.023), and even though there was not a treatment × time interaction for BNBD7 (P = 0.235), BNBD7 only increased in neutrophils from the PIS group (P = 0.002, −1 h vs. 8 h; Figure 7), while it was not affected in the Control group (P = 0.480, −1 h vs. 4 h; P = 0.445 −1 vs. 8 h; Figure 7). In addition, at 8 h post-LPS challenge neutrophil BNBD7 expression was threefold greater in the PIS group than the Control group (P = 0.047). Expression of BNBD10 in neutrophils was affected by time (P < 0.001) and treatment × time interaction (P = 0.002). Expression of BNBD10 was less in the PIS group compared with the Control group at −1 h (P < 0.009; Figure 7), not different between groups at 4 h (P = 0.360; Figure 7) but threefold greater in the PIS group compared with the Control group at 8 h post-LPS challenge (P = 0.016; Figure 7). Neutrophil BNBD10 expression increased substantially ( > 60-fold) from −1 h to 4 h post-LPS challenge in both groups (P < 0.001, −1 h vs. 4 h; Figure 7), and while it did not change from 4 h to 8 h in the Control group (P = 0.162, 4 h vs. 8 h; Figure 7), it continued to increase from 4 h to 8 h in the PIS group (P = 0.048; 4 h vs. 8 h; Figure 7).
Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on expression of bovine neutrophil β-defensin 3 (BNBD3), BNBD4, BNBD7, BNBD10, IL1B and IL8 during a postnatal LPS challenge (0.5 µg/kg BM). Neutrophils were collected from Control (n = 11) and PIS (n = 12) heifers at –1 h, 4 h and 8 h relative to postnatal LPS challenge. Data are presented as the LSM ± SEM expression of each gene relative to ribosomal protein S9 (RPS9) gene expression. *Treatments differ P ≤ 0.05, **Treatments differ P < 0.01.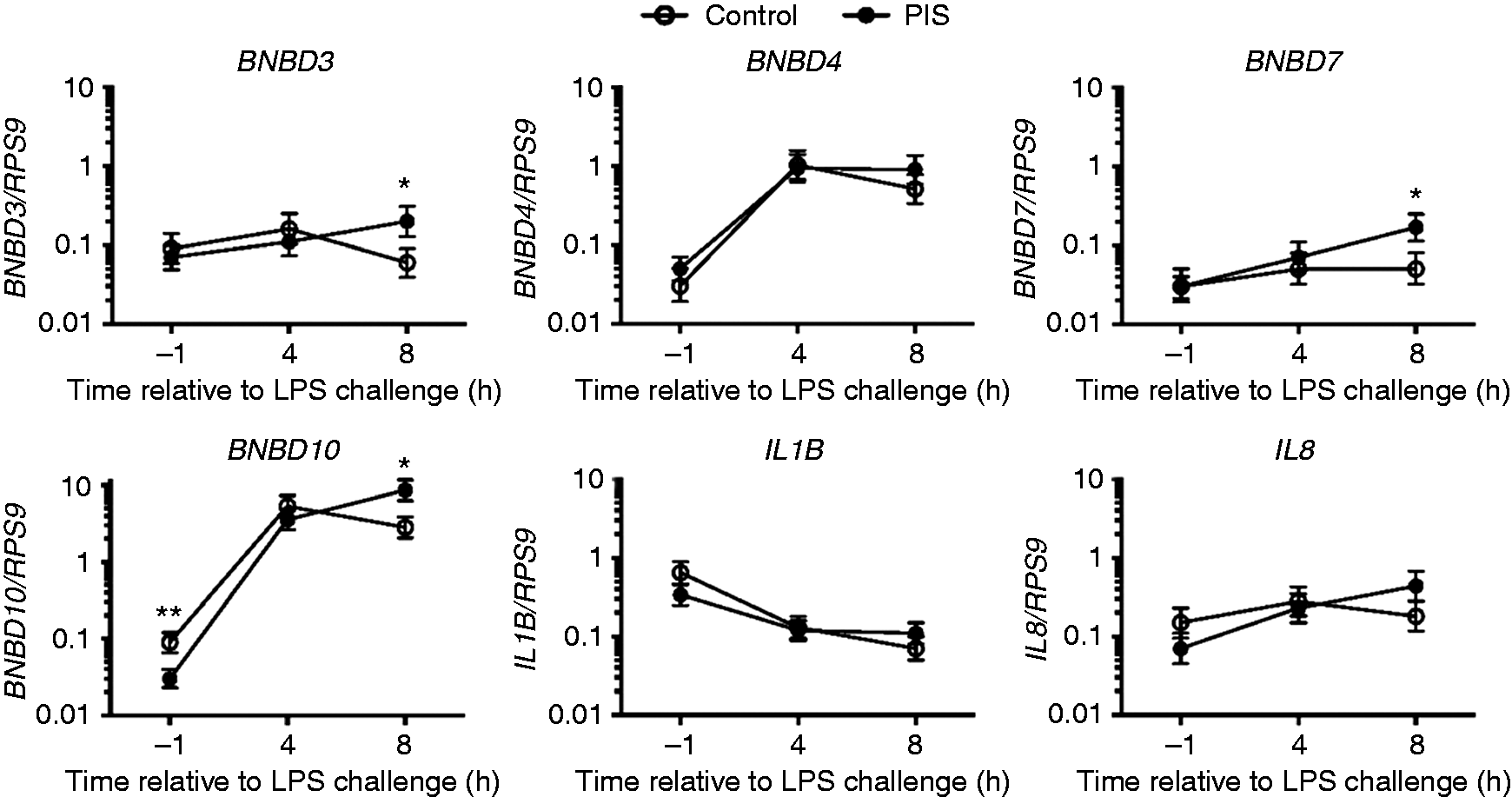
Expression of the IL1B gene in neutrophils decreased with time in both groups (P < 0.001), such that it was ninefold less at 8 h post-LPS challenge compared with –1 h in the Control group (P < 0.001; –1 h vs. 8 h; Figure 7) and threefold lower at 8 h post-LPS challenge compared with –1 h in the PIS group (P = 0.013; −1 h vs. 8 h; Figure 7). There were no differences in IL1B between groups at any time point (P > 0.15; Figure 7) and there was not a treatment × time interaction for IL1B (P = 0.273). There was a tendency for an effect of time on IL8 expression in neutrophils (P = 0.155; Figure 7) but no effect of treatment (P = 0.998) or treatment × time interaction (P = 0.155). However, while IL8 increased nearly eightfold from –1 h to 8 h post-LPS challenge in the PIS group (P = 0.004; –1 h vs. 8 h; Figure 7) it was not increased in the Control group (P = 0.356, –1 h vs. 4 h; P = 0.844, –1 h vs 8 h; Figure 7).
Dermal fibroblasts produced substantial quantities of IL-8 in response to LPS and even more in response to IL-1B (Figure 8), but there was no difference between treatment groups. Production of IL-6 by fibroblasts from PIS heifers was numerically less than that produced by Control heifers (Figure 8), although the difference was not significant (P = 0.15). Fibroblast response to the challenge dose of IL-1B was greater than the response to the challenge dose of LPS, but there was no effect of prenatal treatment on fibroblast response.
Effect of prenatal LPS exposure (0.1 µg/kg BM at 233 ± 19 d of gestation) on IL-8 and IL-6 concentration in culture media using a LPS or IL-1B challenge model of dermal fibroblasts. Dermal fibroblasts were collected from Control (n = 8) and PIS (n = 8) heifers at 462 ± 22 d of age. Data are presented as the LSM ± SEM.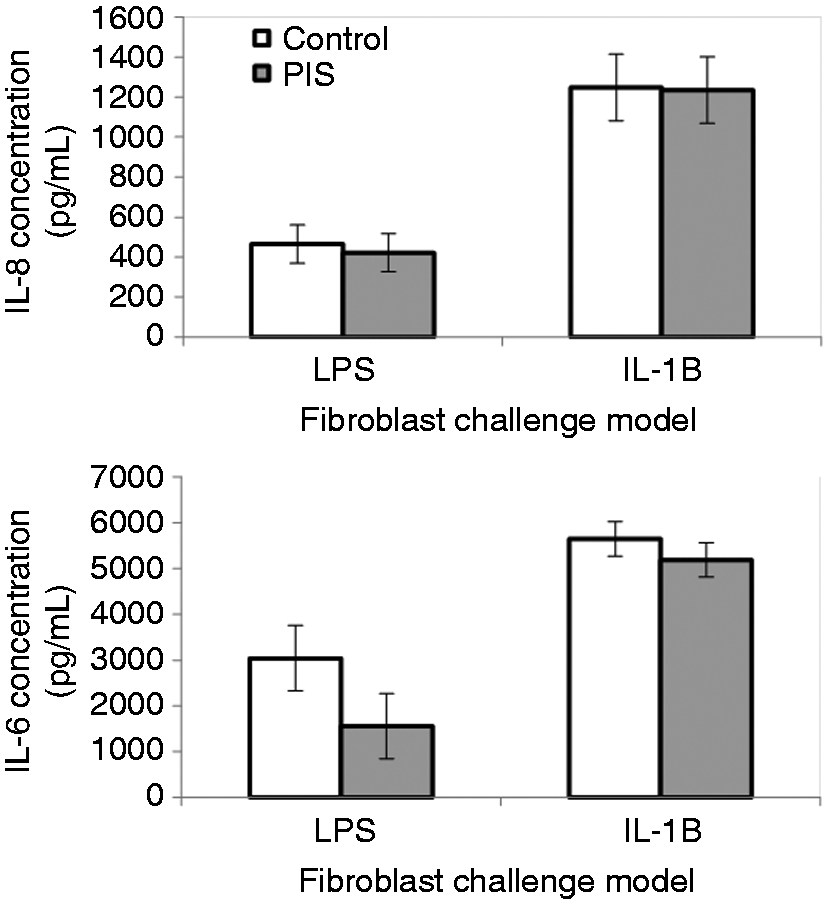
Discussion
Cattle are exposed to stressors and immunological challenges throughout the production cycle, not excluding gestation. Mature cycling cows spend most of their lives pregnant; thus, factors that can influence the cows can have implications on calves and therefore the entire herd. While multiple prenatal stress studies have been conducted utilizing rodent models, there are few studies that utilize cattle, and even fewer that have investigated the potential effects of prenatal immune stimulation. After review of the literature, this study appears to be the first to demonstrate that a single low-dose injection of LPS to cattle during late gestation can affect the postnatal APR of subsequent offspring.
Various physiological signs are monitored by producers in order to identify cattle that are ill, including body temperature and cattle behavior. Previous studies have demonstrated that administration of LPS initially results in an increase in body temperature, followed by a gradual decrease that, if followed for 12–24 h, will decrease below baseline values.8,9 These changes are similar to what was observed in the current study. Peak vaginal temperature values were greater in Control than PIS heifers, suggesting an attenuated response to LPS in the PIS heifers. Also, it is interesting to observe the extended elevation in vaginal temperature in PIS heifers in this study. A study in which sows were exposed to heat stress in utero, and subsequent piglets were exposed to heat stress postnatally, observed an increase in rectal temperature compared with control pigs. 12 Also, rats exposed to restraint stress during gestation produced a greater body temperature response to postnatal LPS exposure. 13 In contrast, LPS administration to adult rats, born from dams administered LPS in utero, had an attenuated febrile response. 14 While there are certainly species differences between rodents and cattle, in the aforementioned study the dams were given a larger does than the adult offspring, while the opposite is true for the current study. Mouihate 15 reported that there are long-lasting effects of viral and bacterial infection in utero, such as the one mimicked by administration of LPS in the current study. A study in pigs administered cortisol in utero found that a postnatal LPS challenge induced a greater ear temperature response when cortisol was administered in the first or third gestational period compared with the second gestational period or control pigs. 16 Thus, in several species, it appears that prenatal stress can influence the postnatal temperature response to an immune challenge.
It is important to note that the changes observed in vaginal temperature were relatively small, and the question arises as to the biological significance of this change. Baseline differences in vaginal temperature between Control and PIS heifers were 0.07℃, while the overall average change in vaginal temperature response was 0.26℃ between Control and PIS heifers. However, while these differences appear minimal, it could still lead to significant consequences within the ability of cattle to recover from such a challenge. Kluger and Rothenburg 17 reported that increasing body temperature by 1℃ requires a 10–13% increase in metabolizable energy. This does not account for the energy needed for other aspects of the immune response, including activation of immune cells and production of acute-phase proteins and immunoglobulins. Thus, other more recent reports in a variety of species suggest that the amount of metabolizable energy needed for an immune response is much higher, around 30–50%,18,19 although, clearly, this is an estimate as there are no methods that can truly estimate this cost. Therefore, alterations in the febrile response, even if small, could have significant implications on the energy available for activation and maintenance of the immune response, and thus influence the recovery of an animal from such a challenge. Furthermore, a better orchestrated and efficient response to an infection, perhaps through prenatal programming, could result in reduction in use of energy resources, thus promoting a faster recovery, as suggested in companion data. 20
Coincident with changes in body temperature, behavioral indices are frequently monitored for signs of illness. Administration of LPS to cattle typically results in decreases in feed and water intake, increased time spent lying down and increased respiration rate, among other symptoms.9,21,22 Few studies have measured sickness behavior in prenatally stressed animals. Studies in pigs have found differences in behavior, including decreases in aggressive behavior in piglets born to sows exposed to social mixing during the second or third gestational periods. 23 Also, prenatally stressed Brahman bulls had increased SBS compared with control bulls. 8 In the current study, an elevated and extended sickness behavior response was observed in the PIS heifers compared with Control heifers, which is similar to the observed vaginal temperature response. Studies in prenatally stressed mice exposed to a postnatal LPS challenge observed decreases in exploratory behavior and distance traveled. 24 Additionally, Enayati et al. 25 reported a link between serum concentrations of IL-6 and cortisol concentrations in rat dams exposed to LPS during late gestation, and anxiety and depressive behaviors in male offspring, suggesting that the changes observed in dams in response to LPS during gestation altered offspring behavior. The increased sickness behaviors, as well as the elevated IL-6 and cortisol responses (reported previously 20 ) would support this link in cattle. However, the sickness behavior response observed in response to LPS in the current study was minimal, with heifers mainly displaying increases in respiration and distention of the head (i.e. scores between 1 and 2), with no severe behaviors observed. While the differences observed between Control and PIS heifers was statistically significant in the current study, whether the differences observed in the current study are of biological significance is questionable. However, combined with the other findings in this study, the differences in sickness behavior support the overall effects of PIS on the response to a postnatal challenge.
Cytokines, such as TNF-α, IL-1β and IL-6, play a major role in the initiation of fever. 15 The lack of an effect of PIS on the TNF-α response to postnatal LPS challenge is similar to what was observed in weaned pigs exposed to restraint stress in utero. 26 The increased and extended IL-6 response is very similar to the extended and increased vaginal temperature response observed in PIS heifers, which suggests that this cytokine may have a strong influence on the temperature response to LPS in the current study. Additionally, stress hormones can stimulate an increase in body temperature, which is supported by the elevated cortisol concentrations observed. 20 While elevation of body temperature is considered a necessary response to infection, as it aids in the clearance of bacterial pathogens, an extended response may be detrimental to the body such that it continues to require a large amount of energy. Kluger and Rothenburg 17 reported that a 10–13% increase in metabolizable energy is required in order to increase body temperature 1℃. Elevation of body temperature for an extended period of time will utilize a greater amount of energy, and is supported by the decreased glucose and non-esterified fatty acid and increased blood urea nitrogen observed in response to LPS in the current study, as previously reported. 20
The bovine β-defensin genes BNBD3, BNBD4, BNBD7 and BNBD10 encode for a group of host defense peptides that are bactericidal towards Gram-negative and Gram-positive bacteria. Little is known about the factors that regulate their expression in cattle. The peptides are abundant in neutrophil granules, 27 and expression of the BNBD4, BNBD7 and BNBD10 genes are upregulated in the mammary tissue during mastitis in dairy cattle. Intra-mammary LPS challenge stimulates secretion of lingual antimicrobial peptide, 28 a closely related member of the β-defensin cluster, and, in vitro, LPS also stimulates expression of two other β-defensin genes, BNBD5 and TAP, in tracheal and mammary epithelial cells.29,30 In the present study, BNBD3, BNBD4, BNBD7 and BNBD10 were increased in peripheral blood neutrophils in response to the postnatal LPS challenge. Interestingly, the prenatal LPS challenge affected expression of the BNBD3, BNBD7 and BNBD10 expression in response to the postnatal LPS challenge in a manner similar to what was observed for vaginal temperatures, sickness behavior and serum IL-6 concentrations. Whether or not increased β-defensin gene expression in peripheral blood neutrophils has any bearing on neutrophil bactericidal capabilities is far from certain, yet the data in the current study collectively seem to indicate that prenatal LPS exposure programs neutrophils to be more responsive to LPS.
Considerable animal-to-animal variation exists in the ability of their dermal fibroblasts to respond to LPS or IL-1β.11,31 Responses of this model cell type are related to in vivo intra-mammary responses to mastitis pathogens. Animals with greater responding fibroblasts develop an enhanced response to the induced infection, resulting in greater leakage of BSA into milk during the acute phase of the infection. The fibroblast responsiveness is also developmentally regulated in that fibroblasts from 16-mo-old animals are much more responsive than fibroblasts obtained at an earlier age from the same animals. 32 This age-dependent difference suggests an epigenetic component to regulation of the innate immune response that may be affected by differences in prenatal environment. In the current study, fibroblasts were collected from the prenatally challenged and Control heifers when they were approximately 15 mo of age, to determine if the prenatal challenge had generated long-term effects on the ability of this model cell type to respond to stimulation with LPS or IL-1β. Such effects could then be further investigated in additional cultures from cryopreserved cells. However, there was no significant effect of PIS on the fibroblast response to LPS due to considerable animal-to-animal variation. The lower, although not significant, in vitro fibroblast IL-6 response to LPS from the PIS heifers is in keeping with a depressing effect of prenatal LPS on the innate response to LPS that has been observed in mice. 33 In that study the mouse dams were injected with LPS on the day of zygote formation as compared with the current study’s late gestation treatment, suggesting that PIS occurring earlier in gestation may have greater effects on the innate immune system of the developing offspring.
Studies in humans have found that increased stress during gestation was associated with elevated concentrations of serum IL-6 and decreased IL-10 in women, and increased lymphocyte production of IL-6 and IL-1β. 34 This is interesting considering that the heifers exposed to PIS in utero had an increased IL-6 response to postnatal LPS challenge in the current study. Additionally, pigs exposed to restraint stress in utero and administered LPS post-weaning had greater serum IL-6 concentrations than non-stressed controls. 26 Also, a study found that pregnant ewes administered LPS at 138 d of gestation had greater IL-6 concentrations than control and non-pregnant ewes. Further, cytokines play a major role in the development of the central nervous system during gestation. 35 Thus, alterations in cytokine concentrations in the maternal environment induced by LPS administration may have profound effects on neural development during late gestation, potentially affecting neural responses associated with stress, behavior and immune function. However, further research is necessary to confirm alterations in neural responses. As discussed earlier, studies in rodents have found a link between elevated maternal IL-6 concentrations and behavior.25,36 Furthermore, after maternal immune activation in mice with Poly(I:C), Hsiao and Patterson 36 reported increased IL-6 in the placenta of pregnant mice. Therefore, prenatal exposure to LPS may have had profound effects on the immune system of calves.
Conclusion
Prenatal LPS exposure altered several aspects of the APR to postnatal LPS challenge. Specifically, PIS heifers had a prolonged increase in vaginal temperature and sickness behavior responses, had an increased and extended IL-6 response, and changes in β-defensin gene expression in isolated neutrophils. These results demonstrate that the postnatal APR can be significantly altered with a single low-dose exposure to LPS in utero. Additionally, this study warrants further investigation into the potential to program prenatally the immune system of beef cattle in a manner that would improve postnatal immune function and overall animal health.
Footnotes
Acknowledgements
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. The U.S. Department of Agriculture (USDA) prohibits discrimination in all its programs and activities on the basis of race, color, national origin, age, disability, and where applicable, sex, marital status, familial status, parental status, religion, sexual orientation, genetic information, political beliefs, reprisal, or because all or part of an individual’s income is derived from any public assistance program. (Not all prohibited bases apply to all programs.) Persons with disabilities who require alternative means for communication of program information (Braille, large print, audiotape, etc.) should contact USDA’s TARGET Center at (202) 720-2600 (voice and TDD). To file a complaint of discrimination, write to USDA, Director, Office of Civil Rights, 1400 Independence Avenue, S.W., Washington, D.C. 20250-9410, or call (800) 795-3272 (voice) or (202) 720-6382 (TDD). USDA is an equal opportunity provider and employer. The authors would like to acknowledge the technical support of JW Dailey, JR Carroll, RE Buchanan, and C Wu (USDA-ARS).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
