Abstract
Neisseria meningitidis causes sepsis with coagulopathy. The present study evaluated the tissue factor (TF)-inducing capacity of bacterial LPS in different presentation forms, i.e. membrane-bound LPS versus purified LPS, and of non-LPS components of N. meningitidis. By using a wild-type N. meningitidis, a mutant N. meningitidis lacking LPS (LPS-deficient N. meningitidis), purified LPS from N. meningitidis and Escherichia coli, we measured TF-expression and TF-activity on human monocytes and microparticles (MPs). The effect of TF-modulators, such as phosphatidylserine (PS), tissue factor pathway inhibitor (TFPI) and recombinant IL-10 (rhIL-10) was investigated. In plasmas from meningococcal patients, fibrinopeptide A (FPA), LPS and IL-10 were quantified. Monocytes and MPs exposed to purified LPS or wild-type N. meningitidis had much higher TF-activity than monocytes and MPs exposed to LPS-deficient N. meningitidis (clot formation assay). Incubation with wild-type N. meningitidis, but also LPS-deficient N. meningitidis, resulted in TF-expression on monocytes (flow cytometry, qRT-PCR). Increased cellular TF-activity is associated with coincident surface-exposure of PS and the number of monocytes positive for both PS and TF was significantly higher for monocytes exposed to wild-type N. meningitidis (7.6%) compared with monocytes exposed to LPS-deficient N. meningitidis (1.8%). Treatment with rhIL-10 reduced monocyte- and MP-associated TF-activity, the number of monocytes positive for both TF and PS, and microvesiculation. Patients with meningococcal septicemia had significantly higher levels of LPS, FPA and IL-10 than patients with distinct meningitis. Our results indicate that LPS from N. meningitidis is crucial for inducing TF-activity, but not for monocyte- and MP-associated TF-expression. TF-activity seems to require coincident expression of TF and PS on monocytes, and LPS induces such double-positive monocytes.
Introduction
The levels of LPS from Neisseria meningitidis in blood from patients with meningococcal disease are closely associated with the clinical presentation and outcome, suggesting that LPS may play a prominent role in the development of the disease. 1 In a previous study, 2 we identified several thousands of particularly LPS-sensitive genes in human monocytes induced by the wild-type reference strain H44/76 N. meningitidis, but not with the LPS-deficient knock-out mutant (LPS-deficient N. meningitidis) which completely lacked LPS. 3 Wild-type N. meningitidis, i.e. the LPS-containing strain, is a potent inducer of tissue factor (TF) mRNA in human monocytes. 2 TF is the main initiator of blood coagulation 4 and is known to be essential in the development of coagulopathy in meningococcal disease. 5 The active TF/factor VIIa (FVIIa) cell surface complex initiates blood coagulation in bacteremia and fulminant sepsis by limited proteolysis and activation of factor IX (FIX) and factor X (FX), which subsequently leads to the generation of thrombin, fibrin deposition and platelet activation. 6 However, TF can be detected at the cell surface without initiating coagulation – a phenomenon denoted as encrypted TF. The molecular mechanism underlying activation of encrypted TF is still obscure, but it has been associated with the expression of phosphatidylserine (PS) at the cell surface. 7 – 9 PS is normally sequestered on the inner leaflet of the cell membrane, 10 whereas re-localization of PS to the cell surface is associated with TF decryption and activation of the coagulation system and with the release of procoagulant microparticles (MPs).7,11 MPs are small (<1 µm) membrane vesicles released from cells upon activation or apoptosis. 11 MPs may disseminate TF and procoagulant phospholipids, and can be incorporated into developing thrombi. 12 TF-positive MPs are highly procoagulant and have been linked to thrombosis in a variety of diseases, such as sepsis, cancer, acute coronary syndromes and diabetes mellitus. 13 – 16
The activity of TF can be modulated by tissue factor pathway inhibitor (TFPI), which functions either by neutralizing FXa directly or by a feedback inhibition of the FVIIa/TF complex in the presence of FXa. 17 Endothelial cells are supposed to be the major site of TFPI production with constitutive synthesis, expression on the cell surface and release of the active protein into the circulating blood, thereby regulating TF-induced coagulation. 17
IL-10, the major anti-inflammatory cytokine, is a physiologic inhibitor that may counterbalance many LPS responses.18,19 Previously, IL-10 has been shown to down-regulate TF expression and thrombin generation on monocytes in response to purified LPS from Escherichia coli. 18 Likewise, IL-10 has also been found to inhibit both apoptosis and microvesiculation of human monocytes. 20
LPS is the most potent inflammation-inducing component of Gram-negative bacteria. Membrane-bound LPS in N. meningitidis stimulates the TLR4 and its accessory protein, MD2, in human monocytes. 2 In contrast, non-LPS molecules in the bacterial membrane are known to trigger other arms of the TLR system. 21 By using the unique knock-out mutant of N. meningitidis, completely lacking LPS, it has been possible to delineate the contribution of non-LPS molecules in the inflammatory reaction in different cells including human monocytes, whole blood and large animals.2,21,22 Different presentation forms of LPS, i.e. naturally-occurring LPS integrated in the outer membrane of intact bacteria versus isolated, purified LPS, have also been shown to induce different inflammatory responses. 23 TF plays a major role in the cross-talk between inflammation and coagulation. 6 In the context of coagulation, high concentrations of purified LPS is often used to induce TF expression, 24 which may not entirely simulate the in vivo situation.
The objective of this study was to evaluate the contribution of LPS and of non-LPS components of N. meningitidis in generating TF expression and TF activity on human monocytes and their monocyte-derived MPs. TF activity on MPs isolated from whole-blood was also investigated. In addition, the effect of TF modulators, such as PS, TFPI and IL-10, was measured. The role of LPS was further demonstrated in plasma from patients with meningococcal disease where fibrinopeptide A (FPA) levels, 25 an evidence of thrombin activity, 26 were compared with IL-10 27 and LPS concentrations. 1
Materials and methods
Ethics statement
The project was approved by the Regional Committee for Medical Research Ethics, South-Eastern Norway Regional Health Authority, Norway.
Reagents
Roswell Park Memorial Institute medium (RPMI) 1640 was from Gibco (Paisley, UK). FCS, heat-treated (56°C, 30 min) before use, was from BioWhittaker Inc. (Walkersville, MD, USA). These reagents had endotoxin levels <0.25 EU/ml, as measured by a Limulus amoebocyte lysate (LAL) assay. 28 Penicillin and streptomycin (cat. no. P0906) were from Sigma (St Louis, MO, USA). Anti-TF mAbs (TF9-5B7 and TF8-5G9, IgG1) were kind gifts from Professor James Morrissey (University of Illinois, College of Medicine, Urbana, IL, USA). Anti-TFPI polyclonal Abs (Product no. 4901) was purchased from American Diagnostica Inc. (Stamford, CT, USA). Thrombin-stimulated HUVEC cells were kindly provided by Marie-Christine Mowinkel, Anders Dahm and Per Morten Sandset (Department of Haematology, Oslo University Hospital, Ullevål, Norway). Recombinant IL-10 (rhIL-10) (cat. no. 217-IL) was purchased from R&D Systems Europe Ltd (Abingdon, UK). Phycoerythrin (PE)-conjugated F(ab′)2 Fragment of Rabbit Anti-Mouse immunoglobulins was from DAKO (Glostrup, Denmark). FITC-labeled annexin V, recombinant annexin V, 7-amino-actinomycin D (7-AAD), allophycocyanin (APC)-labeled anti-CD14 (clone MØP9), APC mouse IgG2b k isotype control and annexin V binding buffer containing 10 mmol/l Hepes (pH 7.4), 140 mmol/l NaCl and 2.5 mmol/l CaCl2 were all purchased from Becton Dickinson Biosciences (Trondheim, Norway).
Wild type N. meningitidis and LPS-deficient N. meningitidis
Neisseria meningitidis strain H44/76 is a serogroup B (B:15:p1.16:L,3,7,9) reference strain initially isolated from a Norwegian patient with lethal meningococcal disease. 3 The LPS-deficient N. meningitidis (LPS-deficient N. meningitidis) was obtained by insertional inactivation of the lpxA gene in the N. meningitidis strain 44/76, serogroup B and kindly provided by Liana Steegh and Peter van der Ley (NIPHE, Bilthoven, the Netherlands) to the National Institute of Public Health, Oslo, Norway, for research purposes. The meningococci were grown as previously described. 2 The bacteria suspensions were heat-inactivated at 56°C for 30 min and the number of bacteria determined by quantitative PCR. 29 The amounts of LPS in wild-type N. meningitidis and LPS-deficient N. meningitidis were quantified using the LAL assay, gas chromatography and SDS-PAGE with silver stain.23,28,30 A number of 106 wild-type N. meningitidis bacteria/ml have been shown to have activity equal to 1 ng/ml purified LPS from N. meningitidis in the LAL assay, 28 whereas 106 LPS-deficient N. meningitidis bacteria/ml have no activity at all.
Purified LPS from N. meningitidis and E. coli
Purified meningococcal LPS (N. meningitidis-LPS) was extracted and purified from N. meningitidis prototype strain H44/76 (B:15:p1.16:L,3,7,9) as previously described. 30 Escherichia coli (055:B5) (E.coli-LPS) was purchased from BioWhittaker Inc.
Isolation and incubation of monocytes and monocyte-derived MPs
PBMCs from different, consenting, healthy donors (I-VIII) (biobank material access number 908, Oslo University Hospital, Ullevål, Norway) were isolated from EDTA whole blood by density gradient centrifugation, elutriated and cryopreserved as previously described. 31 Cryopreserved human monocytes (from different donors) were thawed, re-suspended in 5% (v/v) FCS-RPMI 1640 containing 2% (v/v) penicillin/streptomycin and seeded into microtiter plates (Costar Ultralow Attachment, product code 3473). A total volume per well of 1 ml 5% FCS-RPMI containing 1 × 106 monocytes were incubated with 106 wild-type N. meningitidis/ml, 106 LPS-deficient N. meningitidis/ml, purified N. meningitidis-LPS and E. coli-LPS (final concentrations 1 ng/mL) or culture medium (5% (v/v) FCS-RPMI) for 4 h (37°C, 5% CO2) in the presence or absence of rhIL-10 (50 ng/ml). The incubation time of 4 h was determined from time course experiments of monocytes exposed to ± wild-type N. meningitidis (3, 4 and 5 h) and analysed in the clot formation assay (data not shown). The IL-10 concentration was determined from dose-response experiments of monocytes exposed to wild-type N. meningitidis and different concentrations of rhIL-10, followed by quantification of TNF-α (TNF-α EASIA, BioSource, Camarillo, CA, USA) in the supernatants. At 50 ng/ml rhIL-10, approximately 50% of the TNF-α response was inhibited (data not shown).
After 4 h of stimulation, monocytes and supernatants were separated by centrifugation (47 g, 7 min, 15°C), supernatants gently removed and monocytes either re-suspended in 1 ml 5% FCS-RPMI or lysed for RNA isolation (500 µl lysis buffer (MagNAPure, Roche), stored at –70°C). Recovered monocytes were aliquoted, counted (Advia 60 Hematology System; Bayer, Tarrytown, NY, USA), or analysed for clot forming activity (see below) and flow cytometry. For the flow cytometry analysis, monocytes were re-suspended in ice-cold, sterile, filtered (0.2 µm) Annexin V binding buffer containing 0.5% (w/v) human serum albumin (ORHA 21, Dade Behring, Liederbach, Germany). To pellet MPs, supernatants were further centrifuged (4500 g, 5 min, 20°C) to remove any contaminating cells. Subsequently, aliquots of 250 µl supernatant were centrifuged (17,000 g, for 30 min at room temperature (RT, 22–25°C)) and 225 µl of the supernatant removed. The pelleted MPs (25 µl) were further subjected to clot-forming activity or to flow cytometry analysis (see below).
Isolation of MPs from citrated whole blood
Sodium citrated (0.105 M) whole blood (40 ml) from consenting, healthy volunteers (I-IV) were exposed to wild-type N. meningitidis (106/ml), LPS-deficient N. meningitidis (106/ml), purified N. meningitidis-LPS and E. coli-LPS (final concentrations 1 ng/ml) or vehicle (LPS 0) during constant rotation (12 rpm) (MACSmix; Miltneyi Biotec GmbH, Bergish Gladbach, Germany) for 4 h at 37°C. After incubation, the blood was centrifuged at 2000 g, for 15 min at RT and plasma separated from the cell pellet and transferred to a new tube. Subsequently, aliquots of 250 µl plasma were transferred to Eppendorf tubes, centrifuged at 17,000 g for 30 min at RT, and 225 µl of the plasma removed from the remaining MP-enriched pellets. The pelleted MPs (25 µl) were further subjected to a clot formation assay.
Clot formation assay – TF activity
Twenty-five µl of monocyte suspensions or MP-enriched pellets (derived from elutriation-purified monocytes or from cells in whole blood) and 50 µl of barbital buffer (Owren) diluted in 0.9% (v/v) saline (1:10) were transferred to 96-well plates in duplicates (Cell culture cluster, Costar 3595, Corning). Pooled, platelet-free (3000 g, 30 min and 10,000 g, 10 min) citrated plasma (75 µl) was added to the wells (4 min, 37°C), and the clotting reaction initiated with CaCl2 (100 µl, 10 mmol/l, final concentration). Clot formation (time to Vmax; seconds) was monitored by kinetically measuring the optical density (340 nm) of plasma (every ninth second for 20 min) (VersaMax, Molecular Devices) and giving the clot formation time (seconds) as a measure of the TF activity. Short clot formation times indicated high TF activities, whereas long clot formation times showed low or no TF activities. Variability between experiments was monitored using longitudinal controls (aliquots of diluted human brain thromboplastin (Dade Behring)) at two different levels and resulted in a coefficient of variation (CV) of 15–20%.
To inhibit TF, a mixture (10 µl) of anti-TF mAbs (83 µg/ml TF9-5B7 and 68 µg/ml TF8-569) was added to appropriate wells 15 min before citrated plasma. TF-activities associated with monocytes (n = 3) and their corresponding MPs were also measured in platelet-poor FVII-deficient plasma (FPD7-10, Enzyme Research). To block PS, 10 µl of recombinant Annexin V (20 µg/ml) was added to appropriate wells before addition of citrated plasma. To inhibit TFPI, monocytes and monocyte-derived MPs were preincubated for 15 min at 37°C with anti-TFPI Abs (187 µg/ml) before citrated plasma was added to the wells. To estimate the contribution from the contact activation pathway, 2 μl of anti-FXII Abs (2 mg/ml, Sanquin Blood Supply) were added to appropriate wells 15 min before citrated plasma. To control that the bacteria themselves did not result in TF activity, 106 wild-type N. meningitidis or 106 LPS-deficient N. meningitidis were added to 1 ml culture medium without any monocytes and subsequently treated as samples in the clot formation assay.
Flow cytometry of cells
Monocytes (4 × 105) were labeled with anti-TF mAbs, FITC-labeled Annexin V (to bind PS) and 7-AAD (DNA stain, viability) and analysed as previously described. 32
Flow cytometry of MPs
Bacteria expose PS on their outer surface and have the same flow cytometric scatter properties (forward and side scatter) as MPs. Because of interference between bacteria and MPs, flow cytometry analysis of MPs was only performed on MPs released from monocytes exposed to purified N. meningitidis-LPS or to culture medium alone. MP-pellets (25 µl) were re-suspended in 225 µl ice cold Annexin V binding buffer or calcium-free PBS (control) in BD Falcon 5 ml polypropylene tubes (BD, San Jose, CA, USA). To measure CD14 on MPs, 100 µl of MP suspension was incubated 60 min on ice with Annexin V-FITC (10 µl) and anti-CD14-APC (0.5 µg/ml), or the equivalent isotype-matched control Ab (APC-conjugated Mouse IgG2b k) (0.5 µg/ml) in a final volume of 200 µl (adjusted with Annexin V binding buffer). All Ab solutions were pretreated with centrifugation at 17,000 g for 30 min at 4°C and supernatants used for staining of MPs. Samples of MPs were processed on a BD FACS Aria cell sorter and further analysed with a BD FACS DiVa Software, version 5.0.2. For quantification of MPs, 100 µl of MP suspension was diluted in Annexin V binding buffer in BD Trucount Tubes containing a known number of fluorescent beads (final volume 200 µl). The number of MPs in 1 ml was calculated according to the following formula: (MP events/beads events) × (beads per tube/sample volume).
RNA preparation
Total RNA from monocyte lysates was extracted using MagNA Pure LC RNA Isolation Kit, High Performance with the MagNA Pure LC robot according to the manufacturer’s instructions (www.roche.com), and the isolated total RNA was quantified using the Nano Drop spectrophotometer (www.swab.se).
Reverse transcription
Equal amounts of total RNA (50 ng in 13 µl H2O) were reverse transcribed in a total volume of 20 µl using oligo d(T) as primers and the reverse transcriptase Omniscript (www.qiagen.com). 33
Real-time qPCR
Gene expression of TF mRNA in monocytes was quantified using LightCycler (Roche), online detection with Fast Start Master SYBR Green and the following primers: TF Forw 5_GACCGTAGAAGATGAACGGACT-3_ and TF Rev 5_-GGAGGGAATCACTGCTTGAA-3 for TF. TFPI mRNA (Hs 00409206_m1, Applied Biosystems) was quantified using the ABI Prism 7900 HT Sequence Detection System (Applied Biosystems, Foster City, CA, USA) and TaqMan probes. The geomeans of β2microglobulin, TMPIM4 and PPIB mRNAs were used as reference genes. 34 They were quantified using LightCycler and the following primers: TMBIM4_Forw 5′ TACATGAGAGTCCTGCCTTA and TMBIM4_Rev 5'TCCAACAGCGTAAATCCAAA for TMBIM4, PPIB_Forw 5′GGTGATCTTTGGTCTCTTCGG and PPIB_Rev 5′TAGATGCTCTTTCCTCCTGTG for PPIB. The relative change of each transcript was calculated using the ΔΔ Ct method. 35 The results are expressed as relative quantities, i.e. fold change of TF and TFPI in monocytes incubated with wild-type N. meningitidis (106/ml), LPS-deficient N. meningitidis (106/ml), or purified LPS from N. meningitidis and E. coli (1 ng/ml) normalized to the reference genes and relative to monocytes incubated with only culture medium (LPS 0).
Quantifications of FPA, IL-10 and LPS in plasmas from patients with meningococcal disease
The patient samples (n = 16) were collected after informed consent was obtained from parents, relatives or patients and in accordance with guidelines approved by the Regional Medical Ethics Committee of Health Region I in Norway. Biobank material access number was 948, Oslo University Hospital, Ullevål. The blood was collected, centrifuged, plasma pipetted off and aliquoted, as described, in detail, earlier.1,28 The patients were categorized in clinical presentations as previously described. 1 Quantification of FPA was performed in plasma from heparin and Trasylol® blood as previously described 25 and the detection limit was <4 g/l. Quantification of LPS was performed in heparinized plasma25,28 with a detection limit of 0.25 endotoxin units (EU/ml). IL-10 was measured by ELISA (Biosource,) with a detection limit of 15 pg/ml. 27
Statistical analysis
The data from the flow cytometry analyses and clot formation assays are given as mean and standard deviation (SD) using SPSS PASW Statistics18 (SPSS Inc. Chicago, IL, USA). Comparison of means was performed using paired-samples t-test. Two-sided P-values<0.05 were considered statistically significant. The data-analysis on the mRNA data was performed on natural logarithm ln(x) transformed data.
Results
Recovery of cells
Monocytes exposed to wild-type N. meningitidis, LPS-deficient N. meningitidis, purified LPS from N. meningitidis and E. coli or culture medium, in the absence and presence of rhIL-10, were recovered and counted in an automated hematology instrument after 4 h. The same number of monocytes was recovered from the different inducers (data not shown).
Procoagulant activity associated with human monocytes – effects of rhIL-10
Monocytes exposed to wild-type N. meningitidis (106/ml) had a much shorter time to clot formation in the clot formation assay compared with monocytes exposed to LPS-deficient N. meningitidis (106/ml) (wild-type N. meningitidis: 66s ± 17 (mean ± SD), LPS-deficient N. meningitidis: 117s ± 30 (mean ± SD), P = 0.017, n = 5) and compared with culture medium alone (LPS 0: 152s ± 13 (mean ± SD), P = 0.001, n = 5) (Figure 1A). Incubation with purified LPS from N. meningitidis or E. coli (1 ng/ml) and wild-type N. meningitidis (106/ml) both shortened the clot formation time almost equally. Using monocytes exposed to wild-type N. meningitidis or purified LPS, treatment with rhIL-10 resulted in lengthening clot formation times (wild-type N. meningitidis P = 0.0001) to the level of unexposed monocytes. As a control, wild-type N. meningitidis and LPS-deficient N. meningitidis bacteria were added to the culture medium without any monocytes, and clot formation times were not affected (data not shown).
Effect of rhIL-10 and TFPI on TF activity associated with Mo and MPs. Monocytes from three to five donors (Mo-donor) were exposed to wild-type N. meningitidis (wtNm), LPS-deficient N. meningitidis, purified LPS from N. meningitidis (Nm LPS) and E. coli (E. coli-LPS) or to culture medium alone (LPS 0) for 4 h in the absence and presence of rhIL-10. Monocytes and supernatants were separated by centrifugation and monocyte-associated TF activity (panel A) and MP-associated TF activity (panel B) were measured using a clot formation assay. In panel C, MP-associated TF activity of wild-type N. meningitidis-exposed monocytes were measured in the absence and presence of inhibiting Abs against TFPI. In all panels single donors are presented to display donor variation.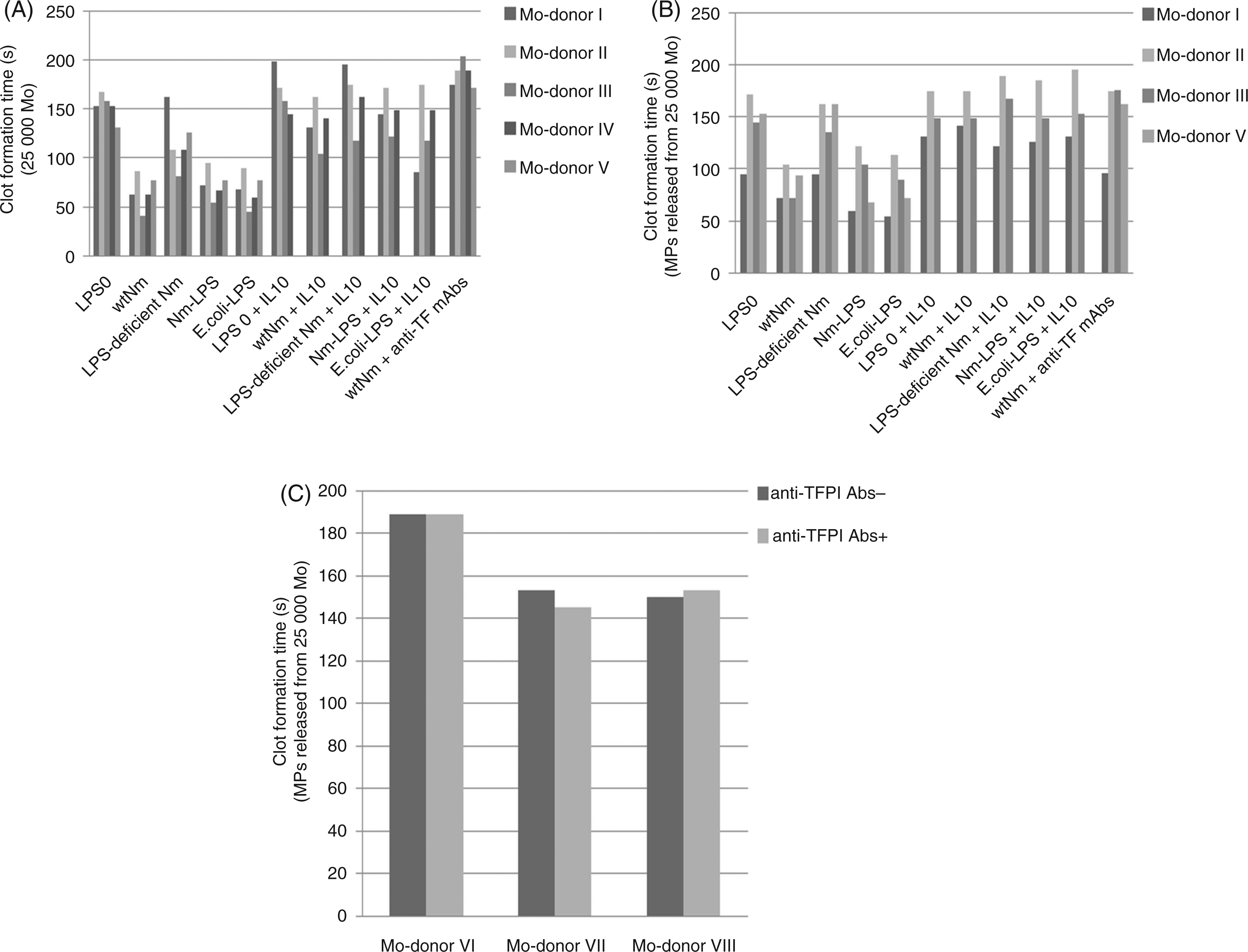
MPs released from monocytes exposed to wild-type N. meningitidis significantly shortened the clot formation times compared with MPs released from monocytes exposed to LPS-deficient N. meningitidis (wild-type N. meningitidis: 85 ± 16 s (mean ± SD), LPS-deficient N. meningitidis: 139 ± 31s (mean ± SD), P = 0.014, n = 4) and compared with culture medium alone (LPS 0 141 ± 33 s (mean ± SD; P = 0.016) (Figure 1B)). Furthermore, stimulation with membrane-bound LPS (wild-type N. meningitidis 106/ml) or purified LPS (1 ng/ml) from N. meningitidis and E. coli resulted in approximately equal MP-associated procoagulant activity. The presence of rhIL-10 during the incubation significantly lengthened the clot formation time of MPs released from monocytes exposed to wild-type N. meningitidis (P = 0.001).
Procoagulant activity associated with monocytes and monocyte-derived MPs – effects of Abs against TF, TFPI and FXII
Monocyte suspension (25 µl containing 2.5 × 104 monocytes) exposed to wild-type N. meningitidis showed a clot formation time of 66s ± 17 (mean ± SD) (n = 5) (Figure 1A). The clot formation times of centrifugation-enriched MP-pellets released to the cell culture medium from 2.5 × 105 monocytes during stimulation with wild-type N. meningitidis, were 85s ± 16 (mean ± SD; n = 4) (Figure 1B). Comparison of clot formation times associated with monocytes and MPs showed that the monocytes have much higher procoagulant activity than their corresponding MPs under our conditions. The presence of anti-TF mAbs (Figure 1A, B) or use of platelet-poor FVII-deficient plasma (data not shown) in the clot formation assay significantly lengthened the clot formation times associated with LPS-stimulated monocytes and their monocyte-derived MPs – indicating that TF was essential for clot formation. The presence of anti-TFPI Abs in the clot formation assay did not modulate TF activity exerted by human monocytes (data not shown) or MPs (Figure 1C). In control experiments, TF activity associated with thrombin-stimulated HUVEC cells were increased in the presence of anti-TFPI Abs, which indicated that the anti-TFPI Abs were inhibitory (data not shown). Using wild-type N. meningitidis-exposed monocytes, clot formation times in the presence of Annexin V (to block PS) were very long (>150 s). The presence of anti-FXII Abs had a negliable effect on the clot formation times associated with monocytes ± exposed to wild-type N. meningitidis or culture medium.
Procoagulant activity associated with MPs derived from whole blood – effects of rhIL-10
MPs derived from whole blood exposed to wild-type N. meningitidis (106/ml) had shorter clot formation times in the clot formation assay as compared with whole blood exposed to LPS-deficient N. meningitidis (106/ml) (P = 0.010), 1 ng/ml purified LPS from N. meningitidis (P = 0.036) and E. coli (P = 0.018), or to cell culture medium (P = 0.018) (Figure 2). The presence of rhIL-10 during the incubation significantly lengthened the clot formation time associated with MPs derived from wild-type N. meningitidis-exposed whole blood (P = 0.007). Using anti-TF mAbs, clot formation time associated with MPs derived from wild-type N. meningitidis-stimulated whole blood were lengthened to the level of unexposed blood, indicating that TF was essential for clot formation.
TF activity in whole blood in the absence and presence of rhIL-10. Whole blood from four donors (WB-donor) were exposed to wildtype N. meningitidis (wtNm), LPS-deficient N. meningitidis, purified LPS from N. meningitidis (Nm-LPS) and E. coli (E.coli-LPS) or to culture medium (LPS 0) for 4 h in the absence and presence of rhIL-10. Cells and plasmas were separated and TF activity was measured in MP-enriched pellets from plasma using a clot formation assay. Single donor data are presented to display donor variation.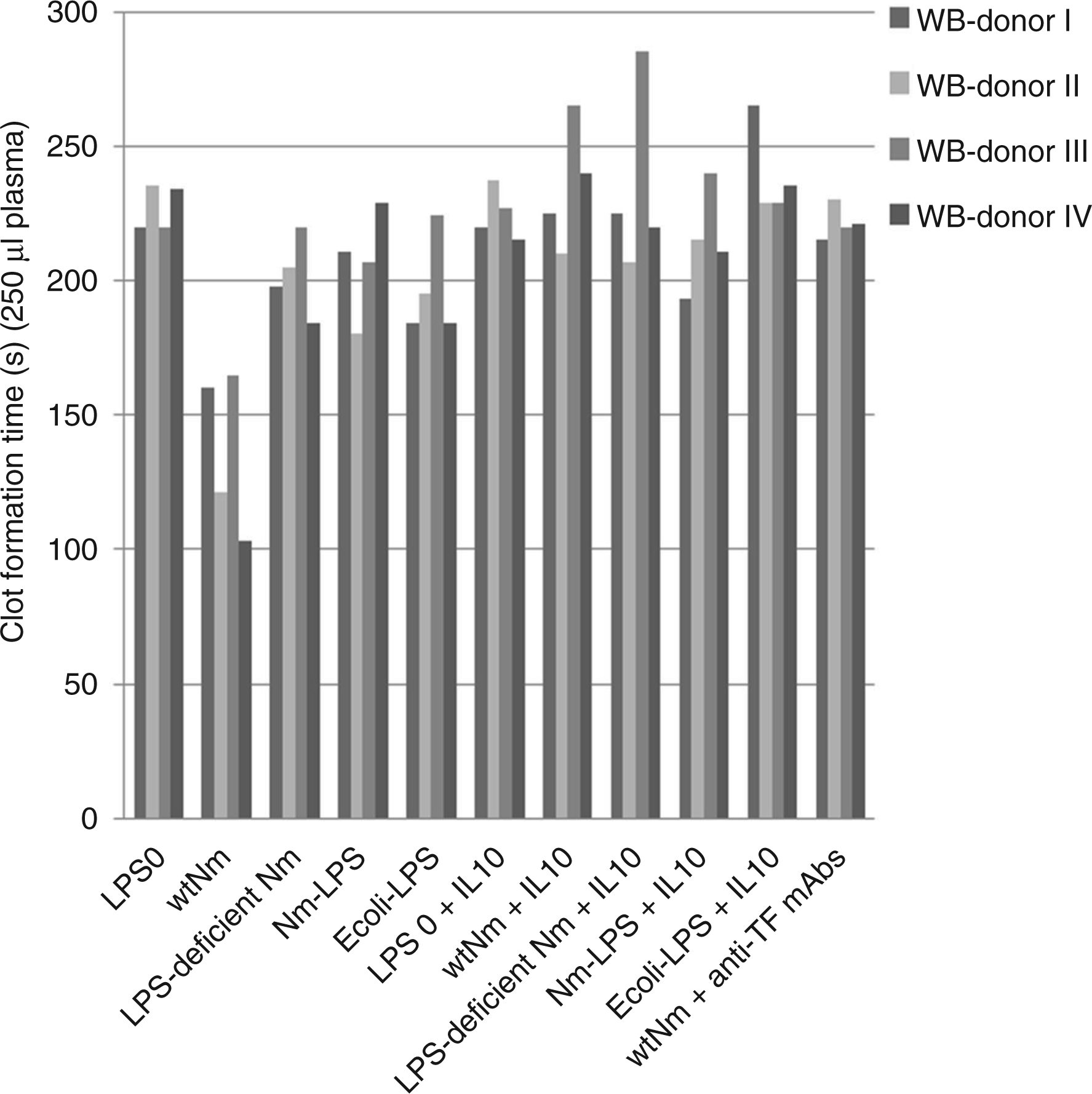
Flow cytometric analysis of TF, PS and 7-AAD expression on the surface of monocytes – effects of rhIL-10
Incubation of monocytes with wild-type N. meningitidis (106/ml) resulted in 40.4% TF-positive monocytes (Figure 3, panel C, upper quadrants); of these 7.6% were positive for both TF and PS (panel C, upper right quadrant). Monocytes exposed to LPS-deficient N. meningitidis also expressed TF (26.6%) (panel E, upper quadrants), but very few of these monocytes were positive for both TF and PS (1.8%) (panel E, upper right quadrant). Incubation with rhIL-10 decreased TF expression on monocytes exposed to wild-type N. meningitidis from 40.4% to 28.3% (compare panels C and D, upper quadrants), whereas the number of monocytes positive for both TF and PS was reduced from 7.6% to 2.9% (compare panels C and D, upper right quadrants).
Flow cytometry analysis of TF and PS expression and of late apoptosis/necrosis on monocytes in the absence and presence of rhIL-10. Monocytes from five donors were exposed to culture medium (LPS 0; panels A and B), wild-type N. meningitidis (wtNm; panels C and D), or LPS-deficient Nm (panels E and F) for 4 h in the absence and presence of rhIL-10. Subsequently, monocytes were triple-labeled with anti-TF mAbs, Annexin V (to bind PS) and 7-AAD (to stain DNA viability) and analysed by flow cytometry. Dot plot from one representative experiment is shown. TF (PE-labeled anti-TF mAbs) is plotted on the Y-axis and PS (FITC-labeled Annexin V) on the X-axis. TF-positive cells were defined as events above the ∼95 percentile of monocytes incubated in the absence of LPS (panel A, upper quadrants). Quadrant distributions with percent positive cells in each quadrant are displayed as mean ± standard deviation (SD) of five different experiments. Late apoptotic/necrotic cells (PS+, 7-AAD+) are represented by red spots.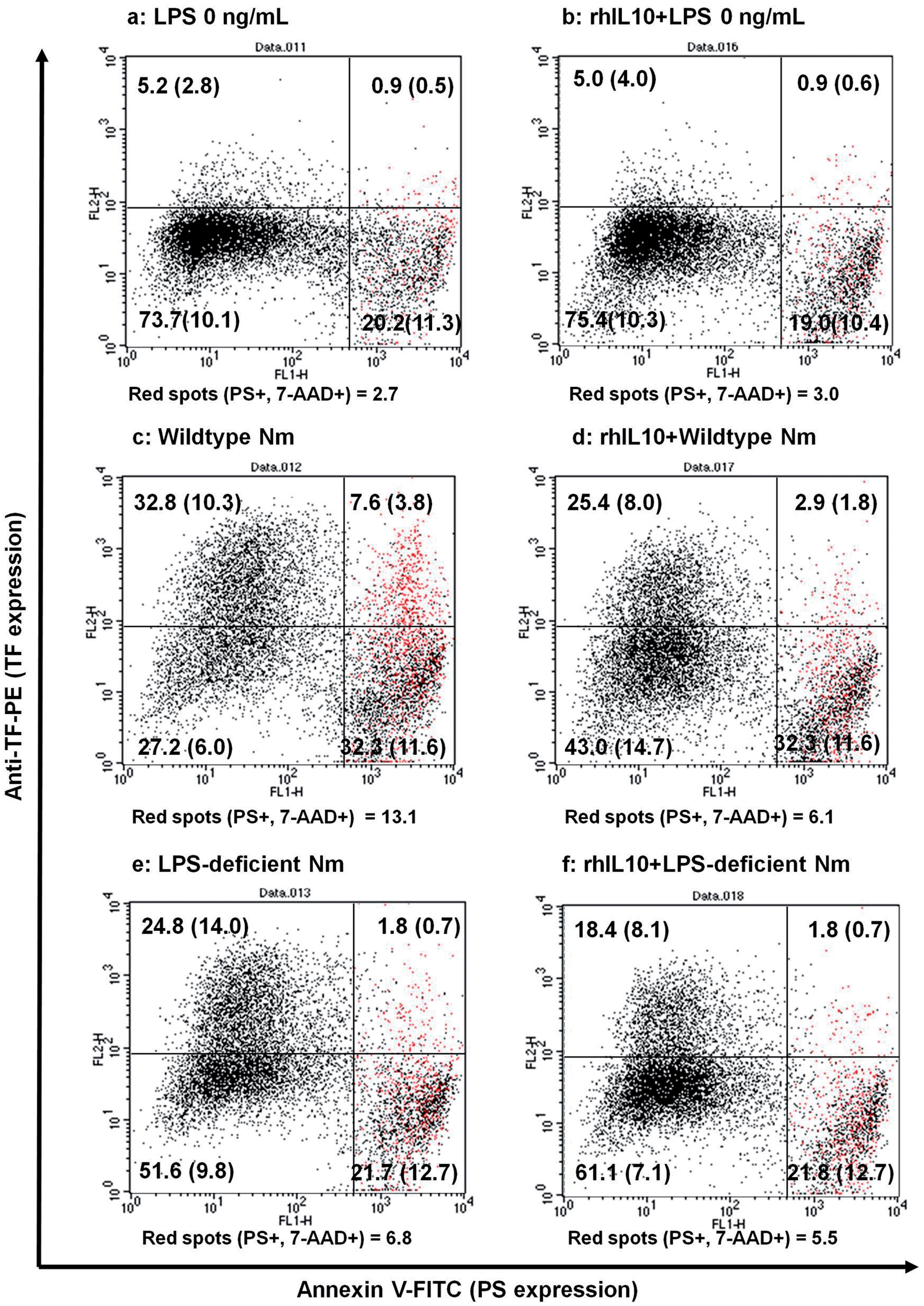
Incubation of monocytes with wild-type N. meningitidis (106/ml) increased the number of late apoptotic and necrotic (PS- and 7-AAD-positive) monocytes from 2.7% to 13.1% (compare panels A and C, red spots). In the wild-type N. meningitidis-exposed monocytes the presence of rhIL-10 decreased the number of late apoptotic/necrotic monocytes from 13.1% to 6.1% (compare panels C and D, red spots). When monocytes were exposed to purified LPS from N. meningitidis or E. coli, monocytes were distributed in the four different quadrants as when they were exposed to wild-type N. meningitidis (data not shown).
Flow cytometric analysis of CD14 and PS expression on the surface of MPs – effects of rhIL-10
Neisseria meningitidis-LPS significantly increased the number of CD14-positive and PS-positive MPs (P = 0.001) compared with incubation in culture medium alone (Figure 4). The number of CD14- and PS-positive MPs were reduced by adding rhIL-10 (P = 0.054) into the culture medium in addition to N. meningitidis-LPS.
Number of CD14- and PS-positive MPs in the absence and presence of rhIL-10. Monocytes from three donors (Mo-donor) were incubated with or without 1 ng/ml purified LPS from N. meningitidis (Nm-LPS) in the absence and presence of rhIL-10. Subsequently, MPs were pelleted from the supernatants by double centrifugation and analysed by flow cytometry. The number of CD14- and PS-positive MPs is presented on the Y-axis. Single donor data are presented to display donor variation.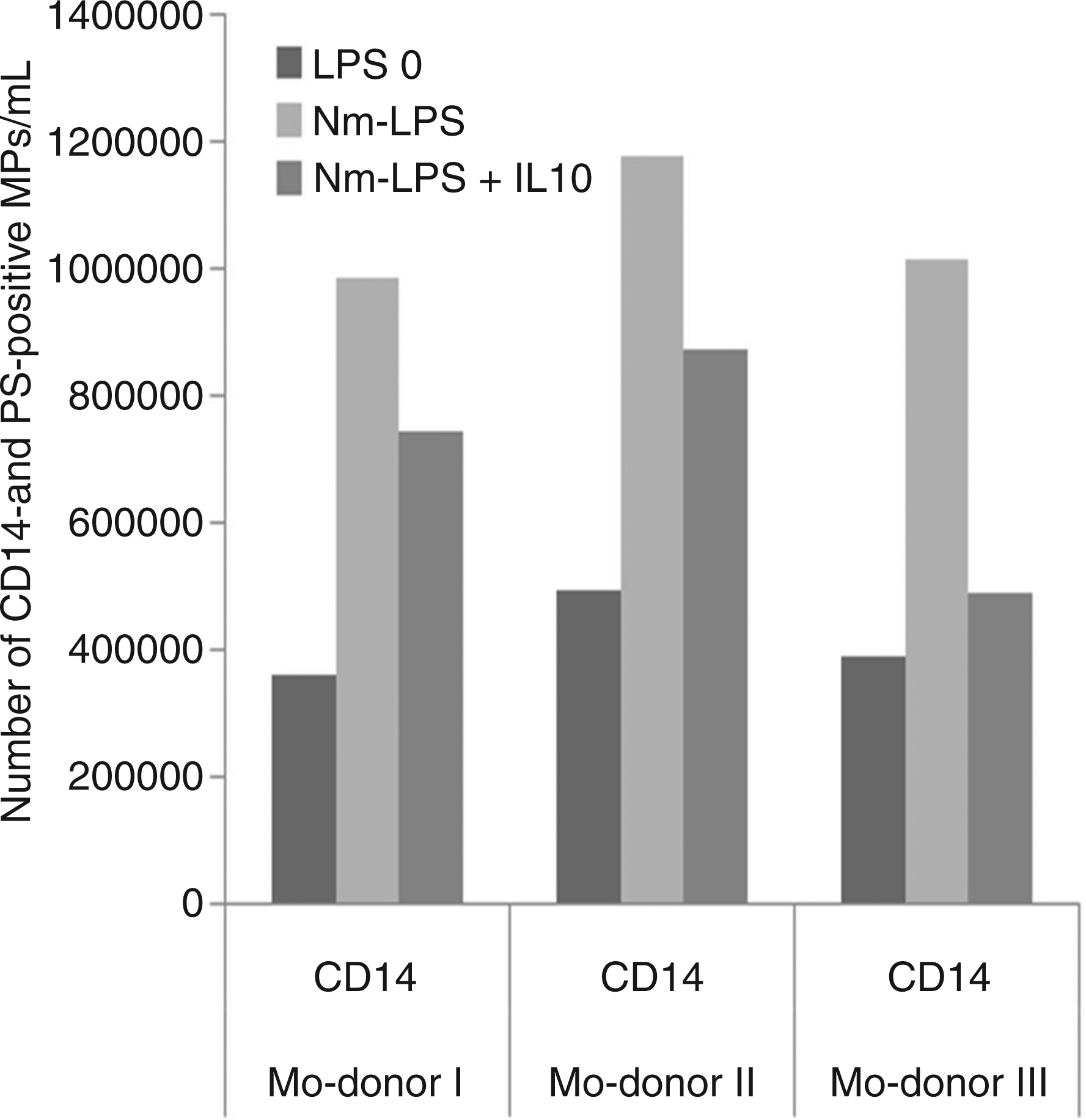
Quantification of TF- and TFPI-mRNA in monocytes by RT-qPCR – effect of rhIL-10
Compared with unexposed monocytes (LPS 0), TF-mRNA in monocytes exposed to wild-type N. meningitidis increased 19-fold (P = 0.012), LPS-deficient N. meningitidis 11-fold (P = 0.029), N. meningitidis-LPS 16-fold (P = 0.011) and E. coli-LPS 17 fold (P = 0.011) (Figure 5, panel A). When rhIL-10 was added, TF-mRNA expression declined to fivefold (wild-type N. meningitidis), fourfold (LPS-deficient N. meningitidis), eightfold (N. meningitidis-LPS) and sevenfold (E. coli-LPS) compared with unexposed monocytes respectively.
mRNA expression of TF (panel A) and TFPI (panel B) in monocytes in the absence and presence of rhIL-10. Monocytes from three donors (Mo-donor) were exposed to wild-type N. meningitidis (wtNm), LPS-deficient N. meningitidis (LPS-def Nm), purified LPS from N. meningitidis (Nm-LPS) and E. coli (E. coli-LPS) for 4 h in absence and presence of rhIL-10. Subsequently, monocytes and supernatants were separated and the monocytes harvested for RNA isolation. TF- and TFPI-mRNA were quantified by qRT-PCR. Fold changes of each transcript, presented on the Y-axis, were calculated using the ΔΔCt method and a geomean of the stably expressed reference genes b2M, TMPIM4 and PPIB mRNA.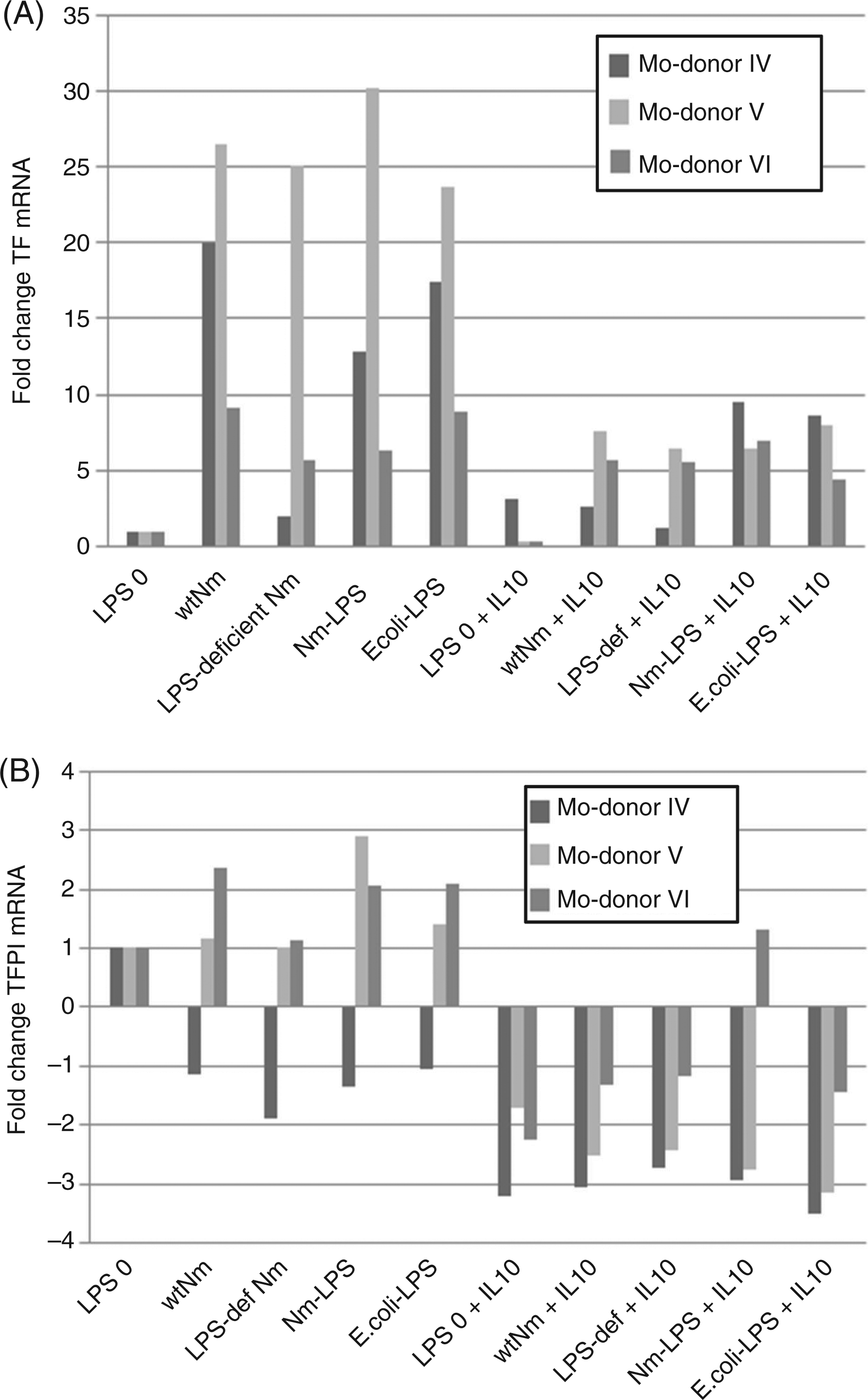
Quantifications of TFPI-mRNA in monocytes exposed to the different inducers (Figure 5, panel B) showed almost similar and low expression levels. Compared with unexposed monocytes, fold changes were 0.8 (wild-type N. meningitidis), 0.07 (LPS-deficient N. meningitidis), 1.2 (N. meningitidis-LPS) and 0.8 (E. coli-LPS). However, the presence of rhIL-10 further down-regulated these low expression levels.
Quantification of FPA, IL-10 and LPS in plasma from patients with meningococcal disease
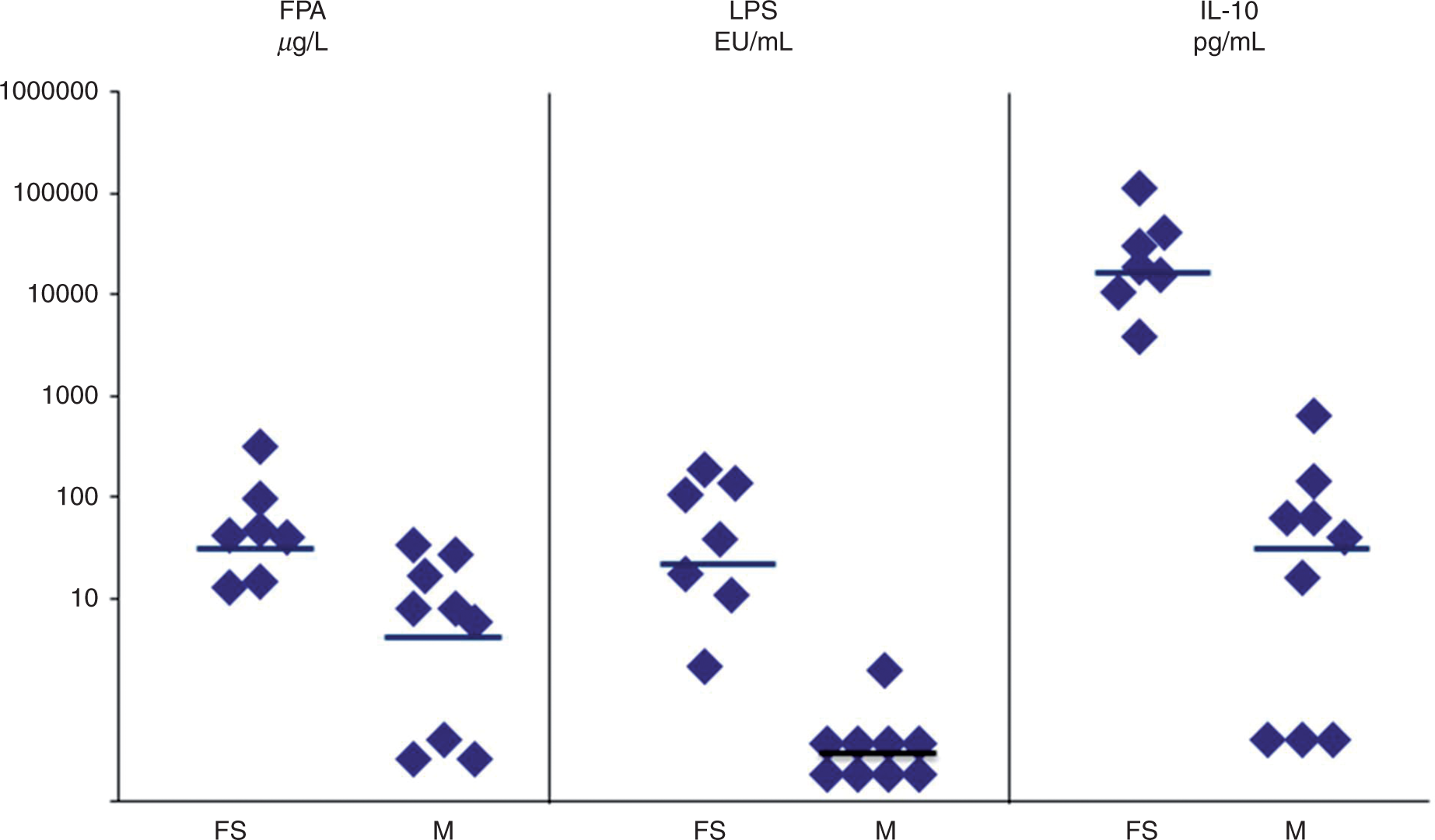
The median level of FPA in plasma from patients with fulminant septicemia was significantly higher; 41 µg/ml (range from 13–312 µg/ml; P = 0.028) than in samples from patients with distinct meningitis; median 8 µg/ml (range 1–42 µg/ml). The median level of IL-10 in patients with fulminant septicemia was significantly increased to 18,500 pg/ml (range 3900–111,000 pg/ml; P = 0.011) compared with samples from patients with distinct meningitis who had a median level of 40 pg/ml (range 10–640 pg/ml). The patients with fulminant septicemia had a median LPS level in plasma of 38 EU/ml (range 2–190 EU/ml), which was significantly higher (P = 0.0001) compared with patients with distinct meningitis; <0.25 EU/ml (range 0–2 EU/ml).
Discussion
The present study evaluated if bacterial LPS in different presentation forms, i.e. membrane-bound LPS versus purified LPS, and if non-LPS components of N. meningitidis increased TF expression and TF activity on human elutriation-purified monocytes and their monocyte-derived MPs. We used concentrations of bacteria and LPS that have been in the range of what we have previously measured in meningococcal patients.1,29,36 In vitro experiments studying mechanisms of LPS often use excessively high concentrations of purified LPS from E. coli 24 which have never been documented to occur in patients.
We found that human monocytes exposed to wild-type N. meningitidis showed higher TF activity (shorter clot formation times) compared with monocytes exposed to a similar number of LPS-deficient N. meningitidis. An interesting observation is that exposure of wild-type N. meningitidis, but also LPS-deficient N. meningitidis, resulted in TF expression on monocytes, as demonstrated by flow cytometry and qRT-PCR, indicating that non-LPS molecules can induce a certain up-regulation of the TF protein. However, coincident expression of TF and PS on the monocyte surface was significantly lower in monocytes exposed to LPS-deficient N. meningitidis compared with wild-type N. meningitidis-exposed monocytes. The results support previous observations that TF may then be present on the cell surface without being fully functional, i.e. encryption of TF. 7 One proposed explanation for the restrained TF activity (decryption) is an asymmetric distribution of cell surface PS in the cell membrane.7,9 We have previously shown that, after separation of LPS-stimulated human monocytes by sorting flow cytometry, PS-positive monocytes were considerably more procoagulant than PS-negative monocytes, even though both PS-negative and PS-positive monocytes expressed cell surface TF protein. 32 The present experiments imply that monocyte-associated TF activity is dependent on the coincident expression of TF and PS on the monocyte surface. We also show that membrane-bound LPS has a dual role in augmenting cell surface TF and in re-localizing PS to the outer cell membrane.
Inflammation and coagulation are closely linked in a variety of disorders, including sepsis, and IL-10 is a major regulator of anti-inflammatory processes 19 and of thrombin generation. 18 Poietvin et al. showed that the presence of IL-10 reduced monocyte-associated TF expression and TF-bound MP release in response to purified LPS from E. coli. 18 In accordance with this study, we showed that the presence of rhIL-10 resulted in a substantial decrease in TF activity in both wild type N. meningitidis-exposed monocytes and in their MPs. In addition, we also showed that rhIL-10 reduced TF activity associated with MPs derived from citrated whole blood which had been exposed to wild type N. meningitidis. In contrast, monocyte-associated TF expression, analysed by flow cytometry and qRT-PCR, was not reduced to the same extent as TF activity by rhIL-10. By flow cytometry, we found that treatment with rhIL-10 only slightly decreased TF expression and that a substantial part of the monocytes exposed to either wild type N. meningitidis or LPS-deficient N. meningitidis, still expressed TF. On monocytes exposed to wild-type N. meningitidis, we observed that rhIL-10 only slightly decreased the overall TF expression, but evidently reduced the number of monocytes positive for both TF and PS. Thus, again, monocyte-associated TF activity seemed to be more closely related to the number of cells positive for TF and PS than to overall TF expression. 32 Both wild-type N. meningitidis-exposed monocytes and their corresponding MP-enriched pellets showed increased TF activities. Comparison of the TF activities showed that TF activity associated with monocytes was much higher than TF activity associated with their corresponding MPs. This is in line with Johnson et al. who suggested that ∼5% of the TF activity in whole blood was associated with microvesicles/microparticles. 37
In accordance with others, we showed that the fold change of monocyte-associated TFPI mRNA expression was low 17 and independent of our different inducers. It is still controversial whether TF activity on monocytes is modulated by monocyte-produced TFPI. Previous studies have shown that small amounts of surface-bound TFPI on monocytes from patients with acute myocardial infarction only partially inhibited TF activity. 38 On the other hand, using PBMC, Basavaraj et al. found that cell surface TFPI is an important regulator of procoagulant reactivity of human monocytes. 17 In a recent review using whole blood stimulated with purified E. coli LPS, Key and Mackman reported that inhibition of TFPI had no effect on TF activity. 39 In our study, neither monocyte-associated TF activity nor MP-associated TF activity were affected by the presence of inhibiting antibodies against TFPI. To our knowledge the effect of inhibition of TFPI on MP-associated TF activity has not previously been shown.
MPs derived from patients with meningococcal disease have previously been studied and documented to have procoagulant properties. 13 To mimic such an in vivo situation, we used an in vitro whole blood model to study TF activity associated with MPs derived from the different blood cells. We found that MPs derived from blood cells exposed to 106/ml wild-type N. meningitidis resulted in significantly higher TF activity than MPs derived from blood cells exposed to 106/ml LPS-deficient N. meningitidis. MPs released in whole blood after exposure to purified LPS from N. meningitidis or E. coli had significantly lower MP-associated TF activity compared with MPs released in whole blood after exposure to the whole bacteria (wild type N. meningitidis). This is in contrast to MPs released from elutriation-purified monocytes where purified LPS from N. meningitidis or E. coli induced essentially equal TF-activity. The results imply a crucial role of membrane-bound LPS in MP-associated TF-activity in whole blood. The observation is in line with Sprong et al. who found that the cytokine production was higher in whole blood exposed to wild-type N. meningitidis than in whole blood exposed to purified N. meningitidis-LPS. 40 Formation of aggregates caused by the highly hydrophobic behavior of purified N. meningitidis-LPS 30 may possibly lead to “detoxification” of purified LPS and thereby reduction of its biologic activity.
Few studies have correlated blood-borne TF in clinical samples with downstream markers of coagulation activation. To investigate whether our in vitro findings of increased TF activity in the presence of LPS reflects the in vivo situation, we compared levels of FPA, IL-10 and LPS previously quantified in plasmas from patients with meningococcal disease (fulminant septicemia with massive coagulopathy versus samples from patients with distinct meningitis and modest coagulopathy). Significantly higher levels of FPA were found in patients with high LPS levels (fulminant septicemia) than in patients with low levels of LPS (distinct meningitis), suggesting the coagulation response to be more severe in patients with high levels of LPS. In addition, we found significantly higher levels of IL-10 in patients with fulminant septicemia than in patients with meningitis. IL-10 has shown to be a major regulator of anti-inflammatory responses 19 and of monocyte-associated thrombin generation. 20 We suggest that the levels of FPA probably would have been much higher without the patientś IL-10 responses.
In summary, by using the unique bacteria model of wild-type N. meningitidis and LPS-deficient N. meningitidis, we show that membrane-bound LPS is potent but not the only bacterial component for inducing TF expression. However, LPS is crucial for eliciting TF activity observed in monocytes and MPs derived from monocytes or MPs derived from whole blood. The models show that the presence of both TF and PS on the surface of the monocyte appears to be required to induce TF activity. The results underscore the importance of measuring TF activity in addition to TF expression because the presence of the TF protein alone on the cell surface may not be sufficient to exert TF activity. Quantifications of LPS, FPA and IL-10 in plasmas from patients with meningococcal disease clearly support the observation that increased levels of LPS are associated with an activation of the coagulation system. In conclusion, our data suggest a crucial role of LPS from N. meningitidis in monocyte- and MP-associated TF activity. LPS has a dual role in inducing both TF and relocation of PS on the monocyte surface, a mechanism that seems to be important for increasing the procoagulant activity of the TF molecule.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
We thank Claus Bryn and E. Arne Høiby, National Institute of Public Health, Oslo, for providing highly purified N. meningitidis LPS and cultures of N. meningitidis H44/76 and the LPS-deficient N. meningitidis 44/76 lpxA-.
