Abstract
Neisseria meningitidis (N. meningitidis) may cause sepsis and meningitis. N. meningitidis with a mutated lpxL1 gene has five, instead of six, acyl chains in the lipid A moiety. Compared with patients infected with the wild type (wt) meningococcus, patients infected with the lpxL1 mutant have a mild meningococcal disease with less systemic inflammation and less coagulopathy. Circulating tissue factor (TF), the main initiator of coagulation, has a central role in the development of coagulation disturbances during sepsis. To study how TF was influenced by the lpxL1 mutant, human primary monocytes and whole blood were incubated with the lpxL1 mutant or the wt meningococcus (H44/76). Monocyte and microvesicle (MV)-associated TF expression and TF-dependent thrombin generation were measured. In both purified monocytes and whole blood, our data show that the lpxL1 mutant is a weaker inducer of monocyte and MV-associated TF compared with the wt. Our data indicate that low levels of circulating TF may contribute to the reduced coagulopathy reported in patients infected with lpxL1 mutants.
Introduction
Neisseria meningitidis (N. meningitidis) is a frequent commensal of the human upper respiratory tract. 1 Meningococcal colonization can be asymptomatic, but the bacteria may also cause meningitis and sepsis, which are conditions with high morbidity and mortality. In invasive meningococcal disease, massive bacterial growth in the circulation may lead to disseminated intravascular coagulation (DIC). 1 Tissue factor (TF), the main initiator of blood coagulation, 2 is important in the pathogenesis of DIC as the TF/factor VIIa complex may initiate massive downstream coagulation activation.3,4 During DIC secondary to meningococcal sepsis, TF may be present on both blood-borne monocytes and monocyte-derived microvesicles (MVs).5–7 MVs are small membrane vesicles (<1 µm) that are shed as a response to either activation or apoptosis. 8 They may carry the negatively charged phospholipid phosphatidylserine, which together with TF makes them highly procoagulant. 4 Patients with severe meningococcal sepsis have elevated levels of circulating MVs, 7 and the procoagulant activity of these MVs has been shown to be associated with the plasma levels of bacterial LPS. 6
LPS in the Gram-negative bacterial outer membrane are able to up-regulate TF on the surface of the monocyte, 9 and the pro-inflammatory cytokine IL-6 appears to mediate the in vivo expression of TF. 10 LPS are sensed by TLR4 in combination with CD14 and MD-2, and this receptor complex initiates intracellular signaling cascades and subsequent activation of the innate immune system. 11 The lipid A part of LPS is essential for recognition by the TLR4 receptor complex, and the acylation pattern of the lipid A largely determines the biological activity of the LPS.12,13 Therefore, genetic modification of the lipid A biosynthesis pathway in N. meningitidis has been important in the search for improved vaccines with reduced toxicity and maintained adjuvant properties.12,14 A N. meningitidis mutant has been made by insertional inactivation of the lpxL1 gene, which is required for addition of a secondary acyl chain to the lipid A molecule. This lpxL1 mutant has five acyl chains in the lipid A portion (penta-acylated) instead of six acyl chains as the wild type (wt; hexa-acylated), and a decreased toxicity, despite retained adjuvant activity. 15
Recently, it has been shown that this lpxL1 mutant is also found naturally among patients with meningococcal disease.16–19 In patients with chronic meningococcemia, 47% of the bacterial isolates were found to have a mutation in the lpxL1 gene, and they had much lower in vitro IL-6-inducing capacities than the wt strains. 16 Two large studies on invasive meningococcal disease (meningitis/sepsis) in the Netherlands showed that 7–9% of the patients were infected with lpxL1 mutants.17,18 Fransen et al. showed that spontaneously occurring lpxL1 mutants induced low levels of pro-inflammatory cytokines (IL-6, TNF-α and IL-1β) in human cell lines and PBMCs. Also, patients infected with the lpxL1 mutants had a milder clinical phenotype with less systemic inflammation than the patients infected with the wt meningococci. 18 Multivariate analyses in the study by Rodenburg et al. showed that the lpxL1 mutant did not influence the amount of petechiae in these patients, 17 whereas Fransen et al. reported reduced coagulopathy, as defined by higher platelet counts on admission and lower frequency of rash, compared with patients infected with wt strains. 18
While it is well documented that the N. meningitidis lpxL1 mutant has reduced capacity to induce inflammation both in vitro and in vivo, it has not been published how the lpxL1 mutant influences the expression and activity of TF, the main initiator of coagulation. In this in vitro study, we wanted to investigate if the N. meningitidis lpxL1 mutant, with penta-acylated lipid A, induces less TF than the hexa-acylated wt meningococcus. To mimic a meningococcal infection, human primary monocytes and whole blood were incubated with titrations of penta- and hexa-acylated N. meningitidis, and monocyte- and MV-associated TF expression (mRNA and protein) and TF-dependent thrombin generation were measured.
Materials and methods
Ethics statement
The project was approved by the Regional Committee for Medical Research Ethics, South-Eastern Norway Regional Health Authority, Norway.
Materials
TBS and BSA were purchased from Sigma Aldrich (St. Louis, MO, USA). Thrombin Calibrator and FluCa-kit were from Thrombinoscope BV (Maastricht, the Netherlands). Mouse anti-human CD142-PE Abs(clone HTF-1), mouse IgG1 κ isotype control Abs(clone MOPC 21), 7-AAD, Annexin V-FITC, Annexin V Binding Buffer and BD FACS™ Lysis Solution were all from BD Biosciences (San Jose, CA, USA). Mouse anti-human CD14–APC Abs(clone RM052) was from Beckman Coulter (Brea, CA, USA). The hybridoma cells TF8-5G9 were a kind gift from Professor James H Morrissey (University of Illinois, College of Medicine, Urbana, IL, USA), and Dr Elisabeth Paus (Department of Medical Biochemistry, Oslo University Hospital, Oslo, Norway) produced the TF8-5G9 (IgG1) anti-TF Ab.
Bacterial strains
N. meningitidis strain H44/76 (B:15:P1.7,16 with immunotype L3,7,9) is an international reference strain initially isolated from a Norwegian patient with fulminant septicemia.
20
The N. meningitidis (H44/76) lpxL1 mutant was made by insertional inactivation of the lpxL1 gene, which is required for the addition of a secondary acyl chain to the lipid A molecule.
15
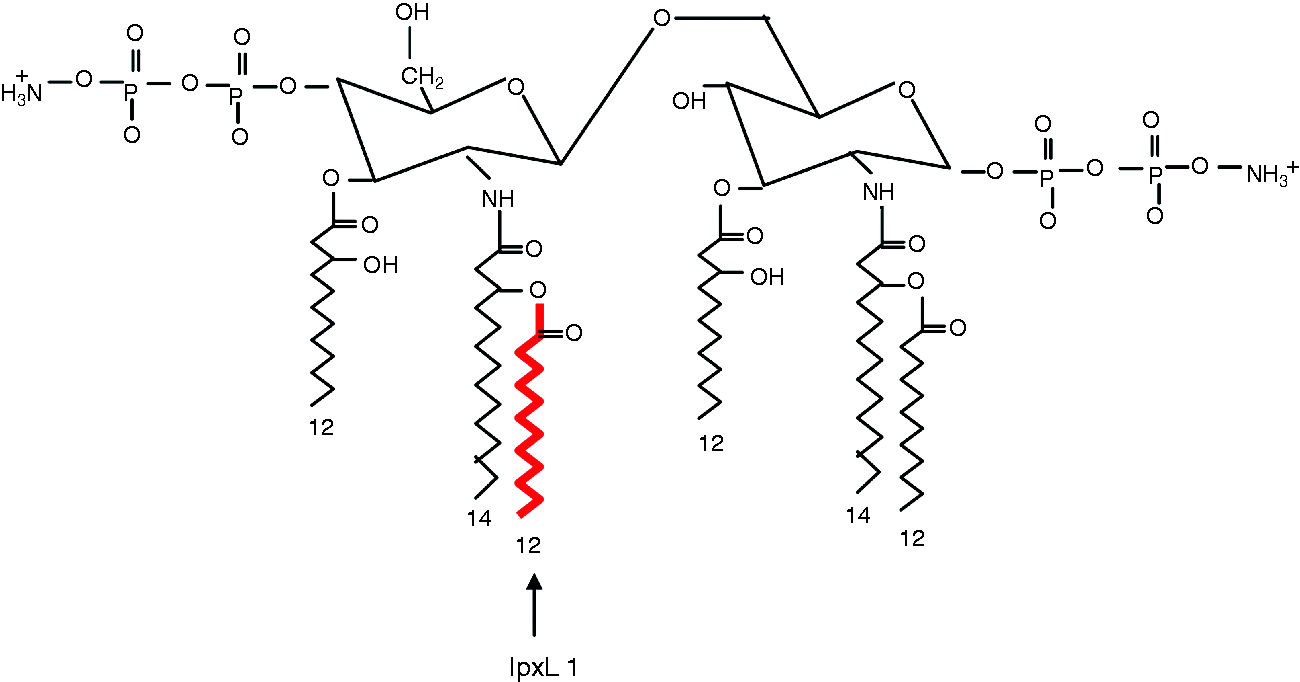
The lpxL1 mutant has five acyl chains (penta-acylated) in the lipid A portion of the LPS molecule, while the wt N. meningitidis has six acyl chains (hexa-acylated) (Figure 1). Van der Ley and van den Dobbelsteen have previously reported that the lpxL1 mutant makes normal amounts of LPS,
21
and that only the lipid A part is altered, compared with the wt.
15
Also, the laboratory-engineered lpxL1 mutant has identical cytokine-inducing activity to strains isolated from patients.
18
The LPS-deficient N. meningitidis [H44/76(pLAK33)] mutant was made by insertional inactivation of the lpxA gene, which is essential for the first step in the lipid A biosynthesis,
22
giving a viable mutant completely lacking LPS in the outer membrane. All bacterial strains used have been published previously,15,22 and were kindly provided by Arie van der Ende (Academic Medical Center, Amsterdam, the Netherlands) and Peter van der Ley (National Institute of Public Health and Environment, Bilthoven, the Netherlands), to Dominique Caugant at the National Institute of Public Health, Oslo, Norway. Here, all the bacteria were grown and heat-inactivated at 56℃ for 30 min, before being sent to our laboratory. For quantification, bacterial DNA was isolated with the MagNA Pure LC DNA isolation Kit I (Roche Diagnostics, Mannheim, Germany), and the number of bacteria in the suspension, that is, DNA copy number, was estimated by spectroscopic measurements at 260 nm (NanoDrop ND-1000; NanoDrop Technologies, Wilmington, DE, USA) combined with a genome calculator (http://cels.uri.edu/gsc/cndna.html) using a N. meningitidis genome size of 2.272.351 base pairs.
23
Illustration of N. meningitidis wt lipid A. The acyl chain missing in the lpxL1 mutant is marked in red. The illustration is modified from Fransen et al.
18
Isolation of primary human monocytes
Monocytes were isolated from 450 ml EDTA blood collected from healthy, consenting blood donors. Briefly, PBMCs were isolated using density gradient centrifugation (Lymphoprep, Axis-Shield, Oslo, Norway), followed by isolation of monocytes with a counter-current elutriation centrifuge (Avanti J26XP; Beckman Coulter). The purity of the monocytes was estimated to be > 85% by flow cytometric analysis of CD14 expression. Cryopreservation (freezing and thawing) were done as previously described. 24
Incubation and preparation of monocytes and monocyte-derived MVs
Monocytes (1 million/ml, Costar® 24-well plate with ultra-low attachment surface) were incubated (2 or 4 h, 37℃, 5% CO2) with 105/ml or 107/ml bacteria (N. meningitidis wt, N. meningitidis lpxL1 mutant or N. meningitidis LPS-deficient mutant) or vehicle (TBS). Untreated cells and cells incubated with the LPS-deficient meningococcus were included as controls. Both monocytes and bacteria were diluted in RPMI Medium 1640 (Gibco, Life Technologies, Grand Island, NY, USA) with 5% heat-inactivated FBS (BioWhittaker® FBS; Lonza, Verviers, Belgium). Cell viability was estimated in separate 96-well plates with PrestoBlue™ Cell Viability Reagent (Invitrogen, Carlsbad, CA, USA) after 4 h. After 2 h (RNA) or 4 h (flow cytometry and thrombin generation) of incubation, monocytes were pelleted at 300 g for 10 min at 4℃. For RNA analysis, the supernatant was removed and the monocytes were lysed in 350 µl Buffer RLT (RNeasy Mini Kit; Qiagen, Hilden, Germany), vortexed and cleared with Qiashredder columns (Qiagen) before storage at −80℃. For flow cytometric analysis, the supernatant was removed and the cells were re-suspended in 70 µl Annexin V Binding Buffer with 0.5% (v/v) BSA, and immediately stained. For measurement of thrombin generation, the supernatant was collected for further MV isolation, while the cell pellet was re-suspended in 1000 µl TBS with 0.5% BSA (TBSA). The cell recovery was estimated in all samples (Sysmex K-4500; Sysmex Deutschland GmbH, Norderstedt, Germany), and the cell concentration adjusted to 300,000 monocytes/ml. MVs were pelleted (17,000 g, 30 min, 20–25℃) from 800 µl of the collected supernatant. After centrifugation, the supernatant was removed and the MV pellet finally dissolved in 200 µl TBSA. We did not include an intermediate centrifugation step (2000 g) in the isolation procedure of the MVs, so the pellet probably included a mixture of both MVs and apoptotic bodies. 25 The pellets will also contain bacteria, and we have previously tested that pelleted bacteria per se are not able to initiate thrombin generation (data not shown).
Incubation and preparation of whole blood
Citrate-anticoagulated blood (0.109 M, Vacuette; GreinerBioOne Gmbh, Kremsmünster, Austria) was collected from healthy, consenting donors by venipuncture (21-G needle), and the first tube was discarded. Whole blood was incubated with N. meningitidis wt, N. meningitidis lpxL1 mutant, N. meningitidis LPS-deficient mutant (all at 108 bacteria/ml) or vehicle (TBS) for 4 h (37℃, 15 rpm, HulaMixer™; Invitrogen Dynal AS, Oslo, Norway). After 2 h, 500 µl whole blood was added to 1.38 ml PAXgene solution in a 3.6 ml Nunc-tube. The tubes were inverted 10 times, left in an upright position for 2 h and then frozen at −80℃ until RNA isolation. After 0 and 4 h, aliquots of whole blood were processed for flow cytometric analysis, while the rest of the blood was centrifuged twice (2500 g, 15 min, 20–25℃) to make platelet-free plasma (PFP). MVs were pelleted (17,000 g, 30 min, 20–25℃) from 500 µl PFP (prediluted with 500 µl TBS). The plasma supernatant was discarded, and the MVs were dissolved in 100 µl TBSA prior to thrombin-generation measurements.
Preparation of pooled normal plasma for measurements of thrombin generation
Blood was collected in SCAT tubes [0.109 M citrate, 50 µg/ml corn trypsin inhibitor (CTI, inhibits factor XII); Haematologic Technologies Inc., Essex Junction, VT, USA] from 10 healthy, consenting donors. We used a 21-G needle (Vacuette SAFETY Blood Collection Set + luer adapter; GreinerBioOne), and the first tube was discarded. The plasmas were prepared by centrifugation (2500 g, 15 min, 20–25℃), followed by combining of the plasmas and another centrifugation at the same conditions. The pooled normal plasma (PNP) was aliquoted and stored at −80℃ until use.
Measurement of cell- and MV-induced thrombin generation by calibrated automated thrombogram
The ability of primary monocytes or MVs derived from monocytes or whole blood to generate thrombin was measured using a modified version of the calibrated automated thrombogram (CAT), originally described by Hemker et al. 26 In brief, 20 µl monocyte or MV-suspension or thrombin calibrator were added to duplicate wells (Thermo Immulon 2HB plate; Thermo Scientific, Waltham, MA, USA) before addition of 80 µl PNP. In parallel wells, to test for TF dependency, cell and MV suspensions (20 µl) were pre-incubated with 2 μl of a mouse monoclonal anti-TF Ab TF8-5G9 (0.40 mg/ml) for 15 min (20–25℃) before addition of 80 µl PNP. We defined TF dependency as an evident reduction in the thrombin generation parameters, that is, down to the level of the untreated controls, after pre-incubation with the anti-TF Ab. The plate was incubated for 10 min at 37℃ before the reaction was initiated by automated addition of Fluobuffer containing calcium and a fluorogenic substrate. For 90 min, fluorescence was read by a Fluoroscan Ascent machine (Thermo Scientific). The thrombin-generation parameters lag time, peak, endogenous thrombin potential (ETP) and time to peak (ttPeak) were calculated by the Thrombinoscope software (Thrombinoscope BV, Maastricht, the Netherlands). Velocity index was calculated as peak (nM thrombin)/(ttpeak (min)–lag time (min)), and represents the rate of thrombin generation during the propagation phase. 27 Of note, if the thrombin-generation curve does not reach the baseline after 90 min, the ETP will not be calculated and the peak will be overestimated due to the lack of α-2-macroglobulin correction. 28
Flow cytometry analysis
Cultured monocytes were stained with 30 µl mouse anti-human CD142-PE Abs(clone HTF-1) and 5 µl Annexin V-FITC, incubated for 30 min (on ice, sheltered from light) and then washed once before 400 µl Annexin V Binding Buffer with 0.5% BSA was added. Finally, 5 µl 7-AAD was added to the cells 10 min before analysis, to allow exclusion of necrotic cells from the calculations. Monocytes were separated from lymphocytes in a forward and side scatter (SSC) dot plot after acquisition of 10,000 events. To separate TF-positive cells from TF-negative cells, a gate was set close to the main population in untreated cells, and TF (CD142) expression was reported as percent TF-positive monocytes. Pilot experiments with isotype control showed that unspecific binding was negligible (data not shown).
For whole blood, 1 µl EDTA (150 mg/ml) was added to 50 µl whole blood prior to staining, as this has been shown to reduce monocyte–monocyte and monocyte–platelet aggregation. 29 Further, the whole-blood sample was stained with 5 µl mouse anti-human CD14-APC Abs(clone RMO52) and 20 µl anti-human CD142-PE Abs(clone HTF-1). Mouse IgG1 κ PE Abs(clone MOPC 21) was used as isotype control. After 30 min at 20–25℃, the whole-blood sample was incubated with 450 µl BD FACS™ Lysis Solution for another 30 min before analysis. Two thousand monocytes were acquired in a CD14/SSC dot plot, and TF expression reported as median fluorescence intensity (MFI) after subtraction of isotype MFI. All samples were run with an BD Accuri™ C6 Flow Cytometer, and the data processed with BD Accuri™C6 software (BD Biosciences).
RNA isolation, cDNA synthesis and real-time quantitative PCR
RNA was automatically isolated with Qiacube (Qiagen). For the cell lysates, we used the RNeasy Mini Kit with the Qiacube protocol ‘Purification of total RNA from animal tissues and cells including DNase digestion’. For the whole-blood lysates we used the PAXgene Blood RNA Kit (PreAnalytix Gmbh, Hombrechtikon, Switzerland) with the ‘PAXgene Blood RNA Part A and Part B’ Qiacube protocols. In addition, the RNeasy MinElute Cleanup Kit RNA (Qiagen) was used on RNA isolated from PAXgene tubes. RNA concentration and integrity were measured with NanoDrop ND-1000 (NanoDrop Technologies) and the Agilent 2100 Bioanalyzer system (Agilent, Santa Clara, CA, USA), respectively. For the monocyte RNA, RNA integrity numbers (RINs) were ≥ 9.0 in 44/56 samples tested, and 12 samples had RINs of 6.8–8.9. In whole-blood RNA samples, RINs were 8.3–9.5. cDNA was synthesized from 50 ng total RNA using qScript™ cDNA SuperMix (Quanta Biosciences, Beverly, MA, USA). Reverse transcription was performed with a Arktik Thermal Cycler (ThermoFisher Scientific Oy, Vantaa, Finland) for 5 min at 25℃, 30 min at 42℃ and 5 min at 85℃. Duplicate cDNA samples were combined and diluted 1:10 with RNase-free water prior to use in RT-qPCR. Gene expression of TF (Hs01076029_m1) was quantified using the ViiA™ 7 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA) and TaqMan probes. We used PPIB (Hs00168719_m1), TMBIM4 (Hs00211390_m1) and B2M (Hs99999907_m1) as reference genes. 30 The expression of TF was calculated with the ΔΔCT method, and the results expressed as fold change of TF mRNA in monocytes incubated with N. meningitidis wt or lpxL1 mutant, normalized to the reference genes and relative to unexposed monocytes. In whole blood, the crossing points for TF were > 35 (from 37.0 to undetermined) in all unstimulated samples, and the TF mRNA levels in the samples incubated with the lpxL1 mutant were therefore related to the mRNA levels in samples incubated with the wt N. meningitidis.
Statistical analysis
Data are presented as median (range) throughout the manuscript. Planned comparisons of medians were performed only between the N. meningitidis wt and the lpxL1 mutant. For this we used Wilcoxon matched-pairs signed rank test without correction for multiple comparisons (GraphPad Prism version 6.01; GraphPad Inc., La Jolla, CA, USA), and P < 0.05 was considered statistically significant.
Results
Monocyte and MV-associated thrombin generation after incubation with the N. meningitidis lpxL1 mutant or wt
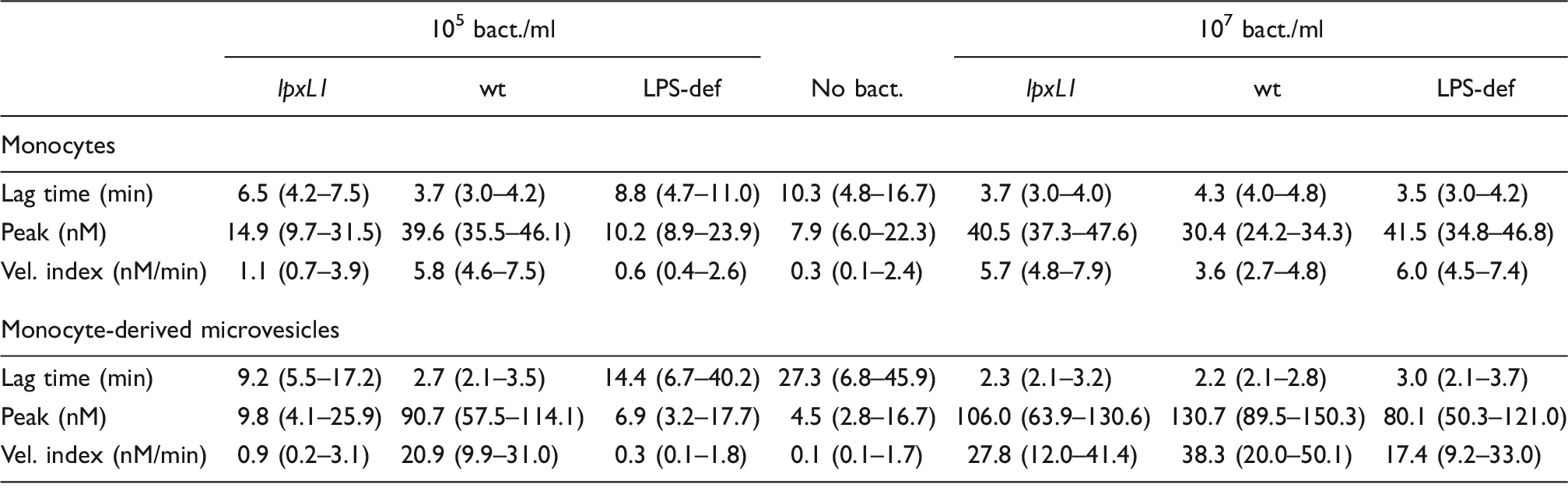
Monocytes incubated with 105 lpxL1 mutants/ml generated thrombin less efficiently than monocytes incubated with the same amount of N. meningitidis wt, as shown by the velocity indices [Figure 2, left (Mo); P < 0.05]. At 107 bacteria/ml, monocytes incubated with the lpxL1 mutant appeared to induce thrombin generation more efficiently than monocytes incubated with the wt meningococcus [Figure 2, left (Mo); P < 0.05]. Abs against TF reduced the velocity indices in all bacteria-treated monocyte samples to the levels observed for untreated cells, indicating a TF-dependent thrombin generation (data not shown). MVs derived from monocytes incubated with 105/ml of the lpxL1 mutant had significantly lower thrombin generation than MVs obtained from monocytes incubated with 105/ml of the wt meningococcus [Figure 2, right (Mo-MVs); P < 0.05]. At 107 bacteria/ml, MVs derived from monocytes incubated with the lpxL1 mutant appeared to generate thrombin almost as efficiently as MVs derived from monocytes incubated with the same amount of wt meningococci [Figure 2, right (Mo-MVs); P = 0.06]. Abs against TF reduced the velocity indices in all bacteria-treated MV samples to the levels observed for MVs released from untreated cells, indicating a TF-dependent thrombin generation (data not shown). Untreated cells and cells incubated with the LPS-deficient mutant were included as controls (Figure 2 and Table 1). The capacity of the lpxL1 mutant to induce monocyte and MV-associated TF-dependent thrombin generation was comparable to the LPS-deficient mutant. An overview of the thrombin-generation parameters velocity index, lag time and peak, for both monocytes and monocyte-derived MVs, is presented in Table 1. If thrombin generation was not detectable after 90 min, we appointed the lag time to 90 min and the peak to 0 nM thrombin.
Thrombin generation of monocytes (Mo) and monocyte-derived microvesicles (Mo-MVs) after incubation with the N. meningitidis lpxL1 mutant or wt. Mo were exposed to N. meningitidis lpxL1, wt, LPS-deficient (LPS-def) (105 or 107/ml) or no bacteria (no bact) for 4 h. MVs were pelleted from the cell supernatant before measurements of thrombin generation. The thrombin-generation parameter velocity index of six donors is presented. Each symbol represents one donor, *P < 0.05. Thrombin-generation parameters of monocytes and monocyte-derived MVs after incubation with N. meningitidis lpxL1 mutant (lpxL1), wt, LPS-deficient mutant (LPS-def) or no bacteria (no bact). The results are presented as median (range) of six donors.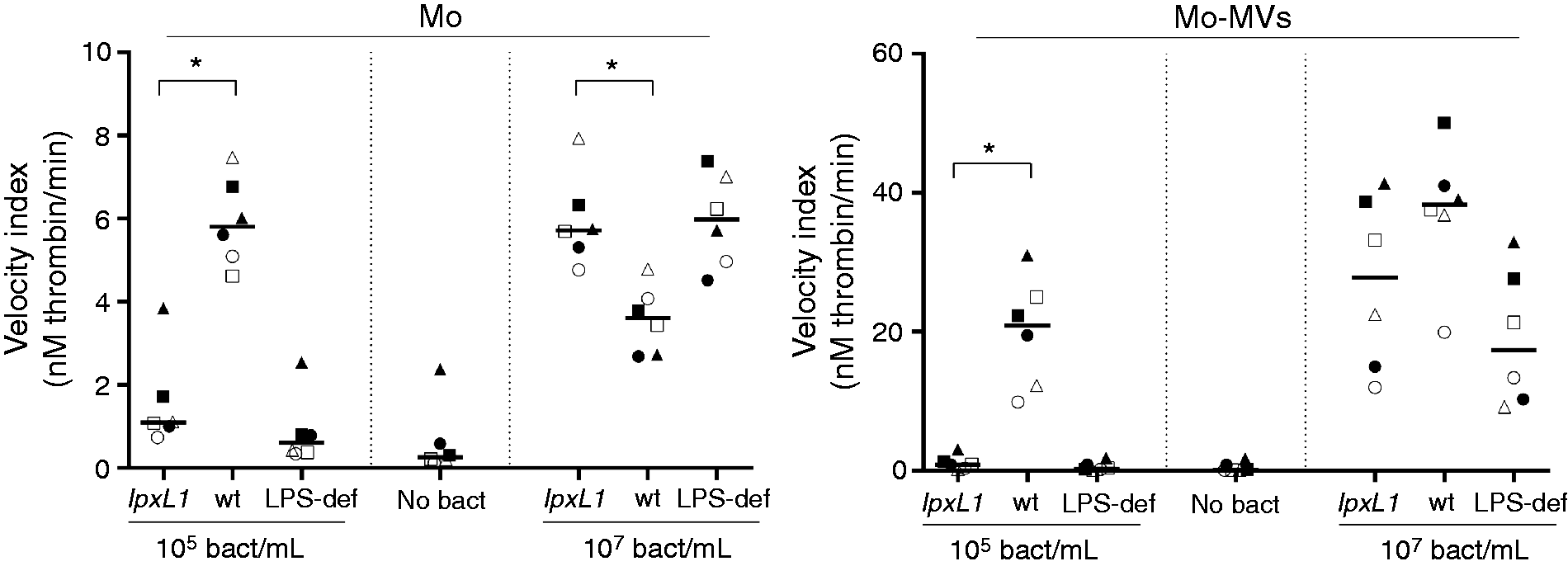
Cell viability of primary monocytes after incubation with the N. meningitidis lpxL1 mutant or wt
Monocyte viability was measured in 96-well plates with the Prestoblue cell viability assay. Monocytes incubated with 105/ml lpxL1 mutnts or wt meningococci for 4 h did not show reduced cell survival compared with untreated cells, with viabilities of 99% (94–106%) and 101% (91–109%), respectively. At 107 bacteria/ml, monocytes incubated with the lpxL1 mutant still had 96% (87–113%) viability, whereas incubation with the wt meningococcus reduced cell survival to 77% (70–87%).
Monocyte-associated TF protein expression (flow cytometry) after incubation with the N. meningitidis lpxL1 mutant or wt
After 4 h incubation, 6% (4–22%) of the untreated monocytes were TF-positive, whereas incubation with 105 lpxL1 mutants/ml resulted in 13% (6–25%) TF-positive monocytes. After incubation with the wt meningococcus (105/ml) there were 34% (27–43%) TF-positive monocytes, which is a significantly higher percentage compared with monocytes incubated with the lpxL1 mutant (Figure 3; P < 0.05). However, at high doses (107/ml), more monocytes where TF-positive when incubated with the lpxL1 mutant [34% (27–42%)] than with the wt meningococcus [21% (16–28%)] (Figure 3; P < 0.05). For the two bacterial concentrations tested, the results were comparable for the lpxL1 mutant and the LPS-deficient mutant (Figure 3). The number of apoptotic cells was estimated by Annexin V-binding. At 105 bacteria/ml, both the lpxL1 mutant and the wt N. meningitidis induced approximately 18% Annexin V-positive monocytes, which was comparable with the results for the untreated cells. At 107 bacteria/ml, the wt meningococcus induced 35% (18–52%) Annexin V-positive cells, which is significantly more than the 19% (7–33%) induced by the lpxL1 mutant (n = 6; P < 0.05). The percentage of necrotic cells (7-AAD positive) was not significantly different between monocytes incubated with 107/ml of the lpxL1 mutant [6% (3–9%)] or the wt meningococcus [9% (7–13%)].
Monocyte TF protein surface expression after incubation with the N. meningitidis lpxL1 mutant or wt. Monocytes were exposed to N. meningitidis lpxL1, wt, LPS-deficient (LPS-def) (105 or 107/ml) or no bacteria (no bact) for 4 h. Monocyte-associated TF protein surface expression was analyzed using flow cytometry. Percent TF-positive cells of six donors are presented. Each shape represents one donor, *P < 0.05.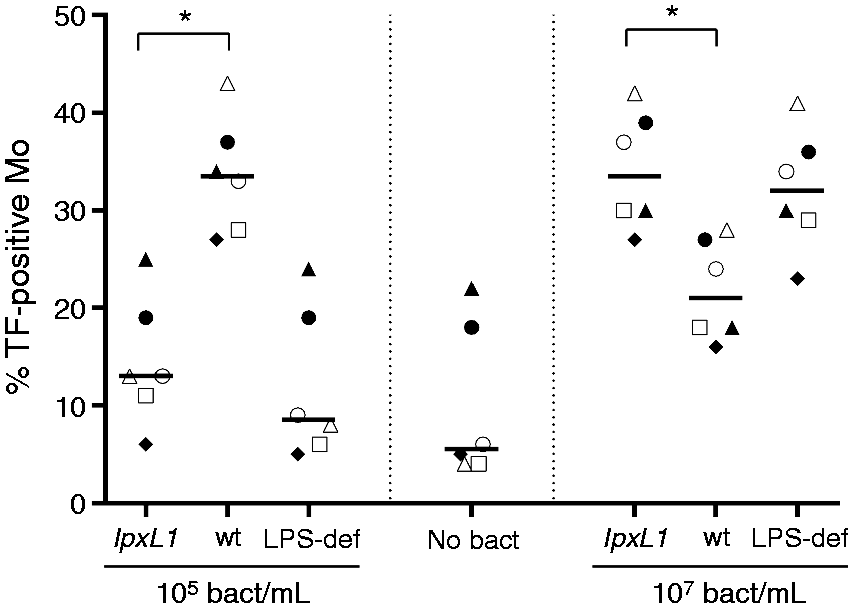
TF mRNA expression in monocytes after incubation with the N. meningitidis lpxL1 mutant or wt
TF mRNA levels were normalized to reference genes, and reported as fold change compared with untreated cells incubated for 2 h. Incubation of monocytes with 105 lpxL1 mutants/ml for 2 h increased the TF mRNA level 2-fold (1–4-fold), whereas incubation with 105/ml of the wt meningococcus increased the TF mRNA level 21-fold (4–58-fold). At 107 bacteria/ml, the TF mRNA level was increased 31-fold (5–97-fold) in the lpxL1 mutant-incubated monocytes, and 36-fold (7–171-fold) in the wt-incubated monocytes (Figure 4). For the two bacterial concentrations tested, the results were comparable for the lpxL1 mutant and the LPS-deficient mutant.
TF mRNA expression in monocytes after incubation with the N. meningitidis lpxL1 mutant or wt. Monocytes were exposed to N. meningitidis lpxL1, wt, LPS-deficient (LPS-def) (105 or 107/ml) or no bacteria (no bact) for 2 h, before RNA isolation and RT-qPCR. TF mRNA expression was calculated with the ΔΔCT method, and expressed as fold change of TF mRNA relative to unexposed monocytes. All samples were normalized to three reference genes. Each shape represents one donor, *P < 0.05.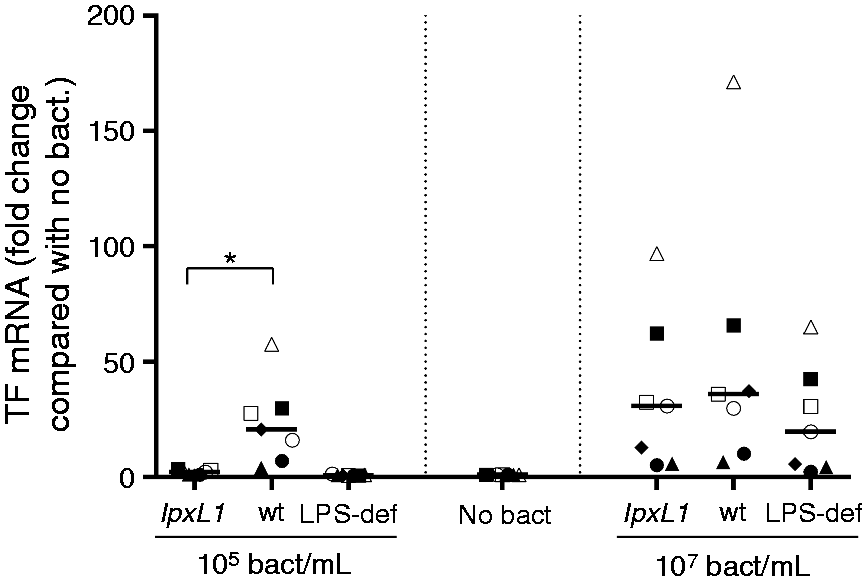
Thrombin generation of MVs derived from whole blood after stimulation with the N. meningitidis lpxL1 mutant or wt
MVs isolated from whole blood after incubation with the N. meningitidis lpxL1 mutant (108/ml) were less potent inducers of thrombin generation than MVs from whole blood stimulated with the same amount of the wt meningococcus (see thrombin generation curves from one donor in Figure 5, upper left). The median velocity index was significantly reduced [0.7 (0.4–1.3) nM thrombin/min vs. 4.4 (2.0–7.7) nM thrombin/min], the lag time longer [21.3 (9.0–31.3) min vs. 4.7 (4.0–6.4) min] and the peak reduced [14 (11–21) nM thrombin vs. 43 (23–60) nM thrombin], with P < 0.05 for all parameters. MVs obtained from whole blood incubated with vehicle had a median velocity index of 0.9 (0.3–2.0) nM thrombin/min, a median lag time of 32.7 (23.2–40.2) min and a median peak of 18 (9–26) nM thrombin. MVs obtained from whole blood incubated with the LPS-deficient mutant had a median velocity index of 0.8 (0.3–1.4) nM thrombin/min, a median lag time of 24.0 (13.8–33.0) min and a median peak of 15 (10–21) nM thrombin (Figure 5). Pre-incubation of the MVs with an anti-TF Ab prior to the initiation of thrombin generation reduced the velocity index, prolonged the lag time and decreased the peak to the level of untreated samples, indicating a TF-dependent, MV-associated thrombin generation (Figure 5). We used a bacterial concentration of 108/ml in the whole-blood experiments as MVs isolated from whole blood after incubation with lower concentrations of the wt meningococcus displayed very low thrombin generation in pilot experiments (data not shown).
Thrombin generation of MVs derived from whole blood after incubation with the N. meningitidis lpxL1 mutant or wt. Citrate- anticoagulated blood was exposed to N. meningitidis lpxL1, wt, LPS-deficient (LPS-def) (108/ml) or no bacteria (no bact) for 4 h. MVs were pelleted from platelet-free plasma and incubated ± anti-tissue factor Abs (anti-TF abs) before measurements of thrombin generation. Thrombin-generation curves from one representative donor are shown (upper left). Three thrombin-generation parameters are presented for six different donors: velocity index (upper right), lag time (lower left) and peak (lower right). Flat curves, i.e. no visible thrombin generation, were appointed lag times of 90 min and peaks of 0 nM thrombin. Each shape represents one donor, *P < 0.05.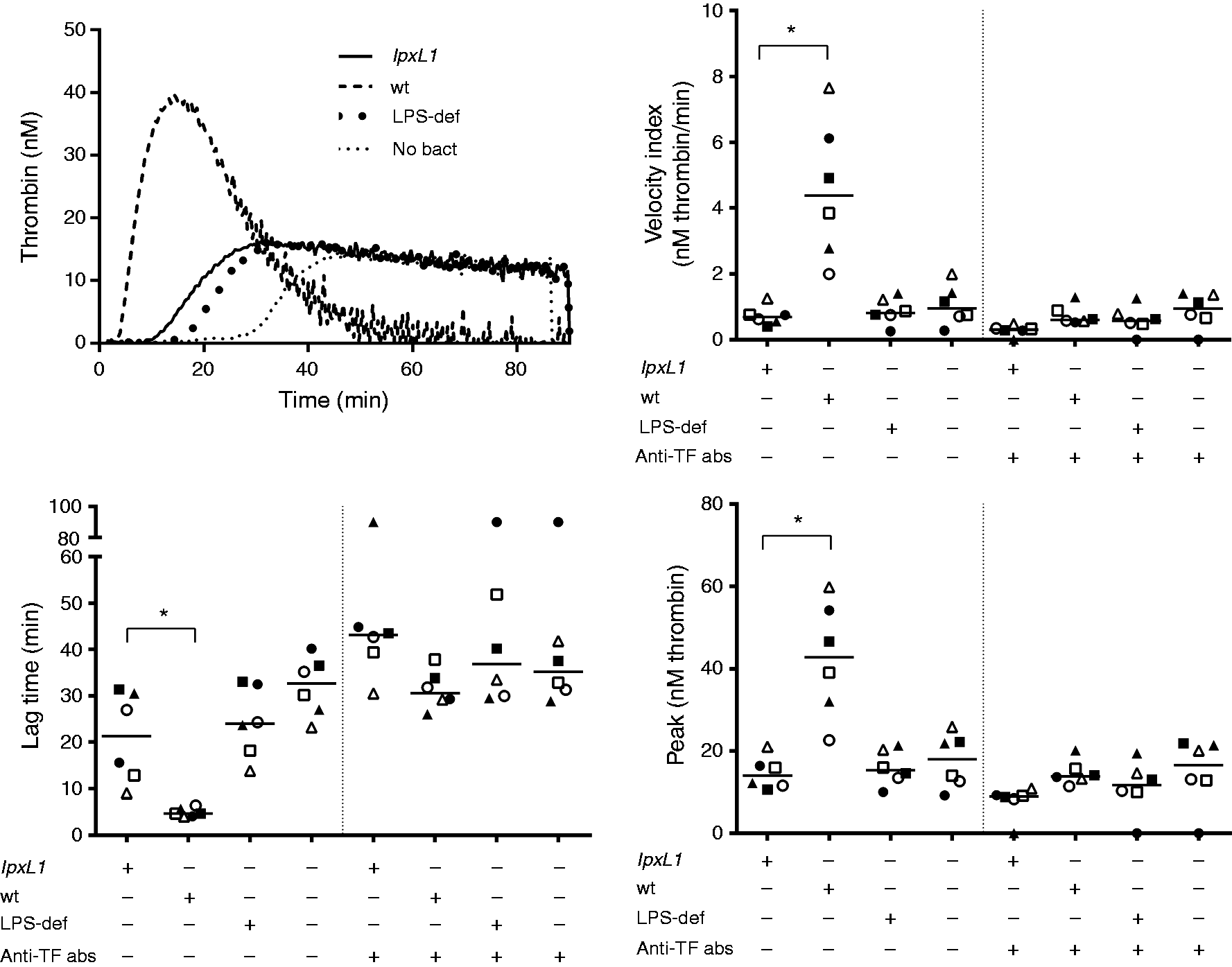
Monocyte TF protein expression in whole blood after stimulation with the N. meningitidis lpxL1 mutant or wt
Monocytes in whole blood incubated with 108/ml of the N. meningitidis lpxL1 mutant had a significantly reduced TF surface expression compared with monocytes in whole blood incubated with the wt meningococcus, with median (range) MFI of 1917 (812–3146) and 4243 (2532–5632), respectively (P < 0.05; Figure 6). Monocytes in whole blood incubated with vehicle or the LPS-deficient mutant had a median (range) MFI of 103 (0–205) and 1802 (1092–2063), respectively.
Monocyte TF protein surface expression in whole blood after incubation with the N. meningitidis lpxL1 mutant or wt. Citrate-anticoagulated blood was exposed to N. meningitidis lpxL1, wt, LPS-deficient (LPS-def) (108/ml) or no bacteria (no bact) for 4 h. Monocyte TF protein surface expression was analyzed by flow cytometry, and reported as MFI after subtraction of isotype MFI. Each shape represents one donor, *P < 0.05.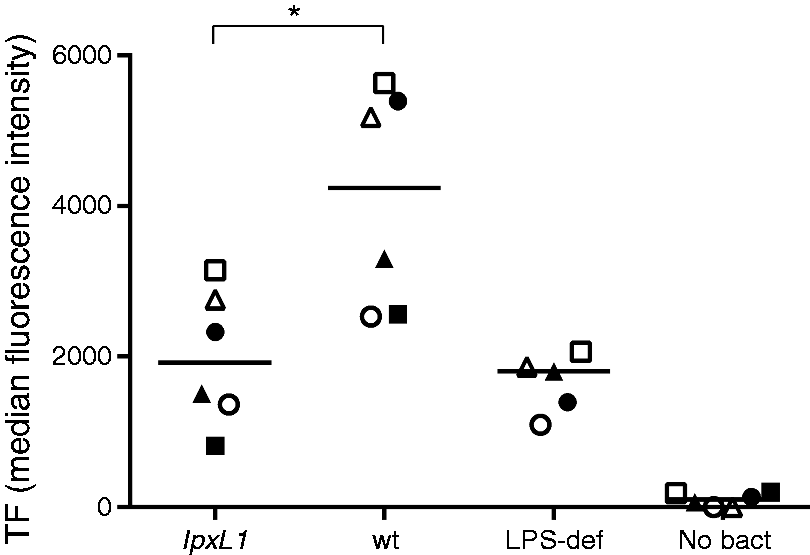
TF mRNA expression in whole blood after stimulation with the N. meningitidis lpxL1 mutant or wt
Compared with whole blood incubated with the wt meningococcus, TF mRNA levels in whole blood incubated with 108 lpxL1 mutants/ml for 2 h was 48% (10–78%) reduced (P < 0.05; n = 6). We found a similar reduction of 54% (24–66%) in whole blood incubated with 108 LPS-deficient mutants/ml (data not shown).
Discussion
The N. meningitidis lpxL1 mutant has been reported to be a weaker inducer of pro-inflammatory cytokines in vitro, and to cause a relatively mild phenotype with less systemic inflammation in patients with meningococcal disease, compared with the wt meningococcus.17,18 Inflammation and coagulation are tightly linked in sepsis, and in this in vitro study we are the first to report that the N. meningitidis lpxL1 mutant is also a weak inducer of TF, the main initiator of coagulation. To mimic meningococcal infection, human primary monocytes or whole blood were incubated with 105 to 108/ml of N. meningitidis lpxL1 mutant or wt, which are clinically relevant concentrations found in patients with meningococcal sepsis.31,32
At low bacterial concentrations, LPS are the most active components of the bacteria. 18 When purified monocytes were incubated with 105/ml of the lpxL1 mutant, monocyte-associated TF mRNA/TF protein expression and monocyte and MV-associated TF-dependent thrombin generation were lower than after incubation with equal amounts of the wt meningococcus. In line with previous publications on individual differences in the ability to respond to LPS, termed high and low responders,33,34 we observed a large inter-individual variation in monocyte TF mRNA levels, TF protein surface expression and monocyte- and MV-associated thrombin generation. At high bacterial concentrations, others have shown that non-LPS components are responsible for a substantial part of the cytokine production in monocytes and PBMCs,18,35 making the LPS structure relatively less important. In line with this, at 107/ml, we found that the lpxL1 mutant and the wt meningococcus induced the same amount of TF mRNA after an incubation time of 2 h. However, after an incubation time of 4 h, monocytes incubated with the wt meningococcus (107/ml) appeared to have less TF protein surface expression (flow cytometry) and less TF-dependent thrombin generation than monocytes incubated with the lpxL1 mutant (107/ml). In parallel, we observed a reduced viability of monocytes incubated with 107/ml of the wt meningococcus, and a concomitant increase in TF-dependent MV-associated thrombin generation. TF-positive monocytes will shed TF-bearing MVs, 36 which, by definition, are formed as a response to cell activation or apoptosis. 8 A reasonable explanation to our observations is therefore that some of the activated, TF-positive monocytes shed TF-positive MVs and die during the 4-h incubation period. When we compensate for this cell loss in our cell-based assays (flow cytometry and thrombin generation), we probably replace dead, TF-positive monocytes with monocytes from a population with fewer TF-positive cells, and thereby measure an artificially reduced monocyte-associated TF expression/activity.
Previously, Amundsen et al. have shown that a whole-blood model allows sustainable monocyte viability. 29 Using this model, the lpxL1 mutant and the LPS-deficient mutant were much weaker inducers of MV-associated TF-dependent thrombin generation and monocyte-associated TF mRNA and TF protein expression than the N. meningitidis wt, even at a high bacterial load (108/ml). In contrast to our observations, Uronen et al. reported equivalent levels of pro-inflammatory cytokines in whole blood incubated with the N. meningitidis wt and the LPS-deficient mutant at 108/ml. 37 However, this discrepancy might be related to different experimental setups, read-outs (TF vs. cytokines), and quantification (OD260nm vs. OD540nm) and inactivation (heat inactivation vs. fixation) of the meningococci.
In all our experiments, the penta-acylated lpxL1 mutant behaved more like the mutant completely deficient of LPS than like the wt meningococcus with six fatty acyl chains. Fransen et al. reported the same pattern for in vitro cytokine production (IL-6, TNF-α and IL-1β) in MM-6 cells and PBMCs. 18 They also showed that four spontaneous lpxL1 mutants behaved similarly to the constructed knockout lpxL1 mutant used in our study, and that the reduced cytokine-inducing capacity was due to impaired recognition of the penta-acylated LPS by the LPS–receptor complex (TLR4, MD-2, CD14). 18 Overall, our findings on the reduced TF activity and TF expression induced by the lpxL1 mutant are in accordance with previous reports on pro-inflammatory cytokines.18,38
In this in vitro study, we show that the N. meningitidis lpxL1 mutant, with penta-acylated lipid A, is a much weaker inducer of TF expression and TF activity in human monocytes and monocyte-derived MVs, compared with the hexa-acylated wt meningococcus. Circulating TF has a central role in the development of coagulation disturbances during sepsis, and these data support the notion that one underlying mechanism behind the reduced coagulopathy reported in patients infected with the lpxL1 mutant 18 is the reduced expression and activity of blood-borne TF, the principal initiator of the coagulation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
