Abstract
Neurological emergencies such as stroke and traumatic brain injury are major contributors to morbidity and mortality in critically ill patients. These conditions frequently result in alterations in cerebral haemodynamics, including raised intracranial pressure, which require timely recognition and management to optimise outcomes. Neuro point-of-care ultrasound (NeuroPOCUS), incorporating transcranial Doppler (TCD), transcranial colour-coded duplex (TCCD) ultrasound, and optic nerve sheath diameter (ONSD) measurement, offers a non-invasive, bedside means of assessing cerebral physiology and is increasingly recognised as a valuable adjunct in neurocritical care. Despite the successful adoption of point-of-care ultrasound in critical care through established accreditation pathways such as FUSIC® and CACTUS®, the UK has lacked a dedicated framework for NeuroPOCUS. To address this gap, we have developed and launched a UK-specific NeuroPOCUS accreditation programme, combining structured theoretical teaching with supervised practical training. The pathway addresses the distinct needs of both paediatric and adult populations, combining theoretical learning with practical application. Core learning materials include neuroanatomy, Doppler principles, standardised insonation techniques, and interpretation of cerebral blood flow velocities and indices such as pulsatility (PI) and resistivity (RI). Supporting resources feature videos of transcranial colour-coded Duplex (TCCD) imaging in normal subjects and clinical case examples. Participants will complete a logbook of 50 supervised cases, facilitated by remote mentorship. A novel accreditation pathway provides an opportunity for further research into the use of NeuroPOCUS in neurocritical care. This article outlines the core techniques of NeuroPOCUS, the physiological insights it offers, key clinical applications, and the proposed accreditation pathway aimed at standardising practice and clinician training in the care of critically ill patients with neurological injury or dysfunction.
Introduction
Since its introduction in the early 1980s, 1 transcranial Doppler (TCD) ultrasound has become a key tool for evaluating cerebral haemodynamics, particularly in neonatal and paediatric patients. The presence of fontanelles, and the thinness of the transtemporal bone in these populations facilitate excellent visualisation of intracranial vascular structures and brain parenchyma. 2 The appeal of TCD lies in its ability to provide real-time, bedside, and repeatable assessments of blood flow velocities in major intracranial arteries, making it invaluable in neurocritical care due to its portability, repeatability, and absence of ionising radiation.
Ultrasound penetration of the adult skull poses more challenges compared to its use in children, though still can be achieved by a skilled operator. Utilising low-frequency cardiac phased array probes can reduce the attenuation of ultrasound waves caused by bone. Specific acoustic windows, areas where the skull bone is thin enough, permit ultrasound wave penetration. Despite these adaptations, ~10%–20% of the adult population may not have an adequate acoustic window, limiting the effectiveness of TCD in some cases. 3
Historically, TCD used to be a blind technique without direct vessel visualisation. Transcranial colour-coded duplex (TCCD) ultrasound combines B-mode imaging and colour Doppler flow mapping, where the intracranial vessels are directly visualised. 4 TCCD offers several advantages over traditional TCD in adults and children by enabling the direct identification of cerebral vessels and allowing more accurate insonation. It is typically performed serially, whereas TCD is more often continuous. However, in the literature, the terms TCD and TCCD are often used interchangeably. In many cases, authors refer to TCD, when operators are in fact performing TCCD. For simplicity, we will refer to the technique as ‘TCD’ throughout this article. However, the training and accreditation process, outlined later, specifically focusses on TCCD.
Another non-invasive parameter employed in neurocritical care is the measurement of optic nerve sheath diameter (ONSD). This technique serves as a surrogate for assessing intracranial pressure (ICP), especially when invasive monitoring is unavailable or contraindicated. The optic nerve sheath is contiguous with the intracranial subarachnoid space; thus, increases in intracranial pressure can lead to dilation of the sheath, detectable via ultrasonography. Studies have demonstrated that ONSD measurement is a reliable, non-invasive method for estimating elevated intracranial pressure, with high sensitivity and specificity across various patient populations. 5
In summary, TCD along with ONSD measurement, have growing evidence of benefit as non-invasive tools in neurocritical care. They can rapidly provide information on cerebral haemodynamics and intracranial pressure, aiding in the diagnosis and management of neurological conditions in both paediatric and adult patients. However, integration into routine practice is at present limited by lack of training opportunities, and further research is required in some populations, which we will explore.
In this article we will outline the core techniques and physiological insights of NeuroPOCUS including TCD, and ONSD, as well as the specific evidence base. We discuss the clinical use of these techniques in paediatric and adult critical care, and a proposed framework for accreditation based on the novel accreditation pathway from the UK.
Rationale
TCD ultrasonography offers a real-time, non-invasive view of cerebral haemodynamics,6–8 with value that extends beyond a fixed set of conditions by enabling the detection of a wide range of physiological and haemodynamic abnormalities.9,10 Its utility is particularly crucial for assessing cerebral perfusion and its relationship with intracranial pressure (ICP), where it can provide non-invasive estimation of ICP and cerebral perfusion pressure (CPP) by analysing Doppler waveforms to reflect changes in intracranial compliance.11,12 Furthermore, TCD can identify dysfunctional cerebral autoregulation by monitoring blood flow velocity responses to blood pressure changes,13–15 and detect cerebral hyperaemia as a marker of inflammation or reperfusion injury. 16
Beyond perfusion, TCD could contribute to the structural and functional assessment of cerebral vessels; it is sensitive for identifying cerebral vasospasm through focal increases in blood flow velocities.17,18 A blunted cerebrovascular response to carbon dioxide changes, quantifiable by TCD, can indicate exhausted vascular reserve and may be able to predict poor outcomes. 19 It is also effective for screening, diagnosing, and monitoring intracranial steno-occlusive disease. 20 The modality can additionally identify abnormal flow patterns and emboli from cardiac or systemic sources, 21 including the detection of right-to-left shunts like a patent foramen ovale (PFO) through agitated saline studies, 22 the identification of micro embolic signals (MES), 23 and aiding in the diagnosis of CNS vascular malformations. 24 Finally, in its ancillary and therapeutic roles, TCD may assist in confirming cerebral circulatory arrest25,26 and also contribute to therapeutic strategies to optimise cerebral haemodynamics including targeted blood pressure management.27,28
Technique
The most commonly used acoustic window when performing TCD is via the transtemporal window. The ultrasound transducer is placed in the orbitomeatal line, over the temporal area just above the zygomatic arch and in front of the tragus of the ear with the index marker anteriorly (Figure 1). Position 1 depicted in the figure should be attempted first, before moving to 2 and 3 if MCA insonation fails. In this mesencephalic plane, visualisation of the contralateral skull is found at a depth of usually 12–15 cm in the adult patient, and at variable depths in paediatric patients (8 cm in term neonates, up to adult sizes in adolescents) and appears as a curved bright line. The next landmarks to identify are a hypoechoic butterfly structure which represent the cerebral peduncles (marked P, Figure 2) surrounded by the hyperechoic basal cisterns (marked by an asterisk). Angulating the probe cephalad by 10° brings the view to the diencephalic plane where two central hyperechoic lines, corresponding to the third ventricle, are seen with the hypoechoic thalami each side (Figure 3). Further cephalad angulation of the probe to the ventricular plane allows visualisation of the frontal horns of the lateral ventricle.
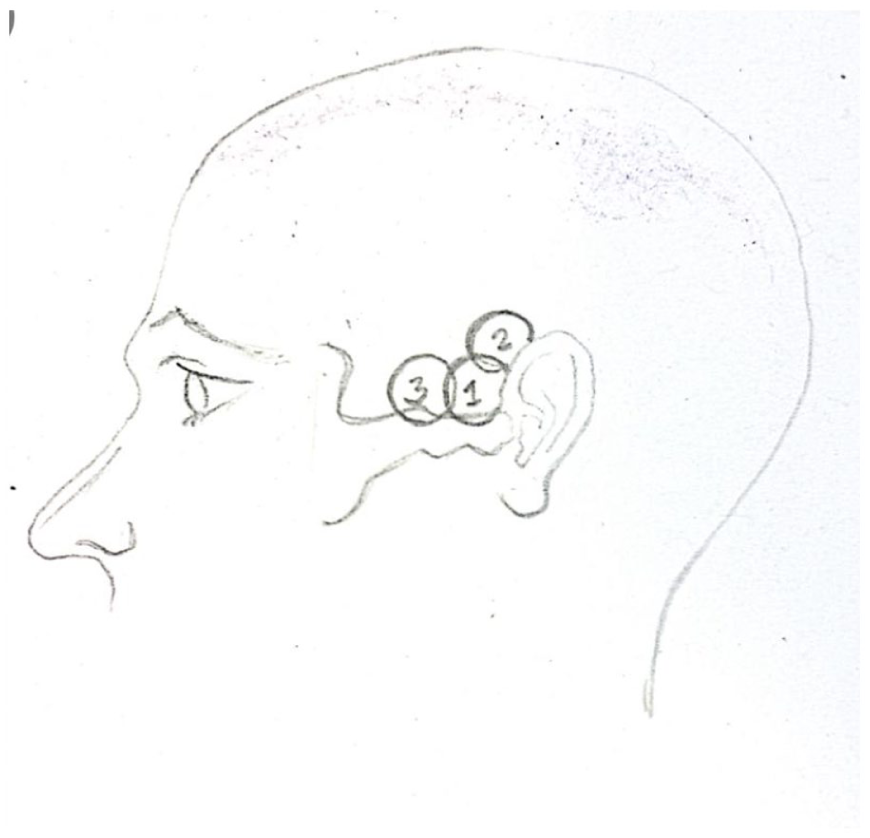
Crayon illustration by a PICU fellow. Common positioning of the transducer for the insonation of the polygon of Willis/MCA.
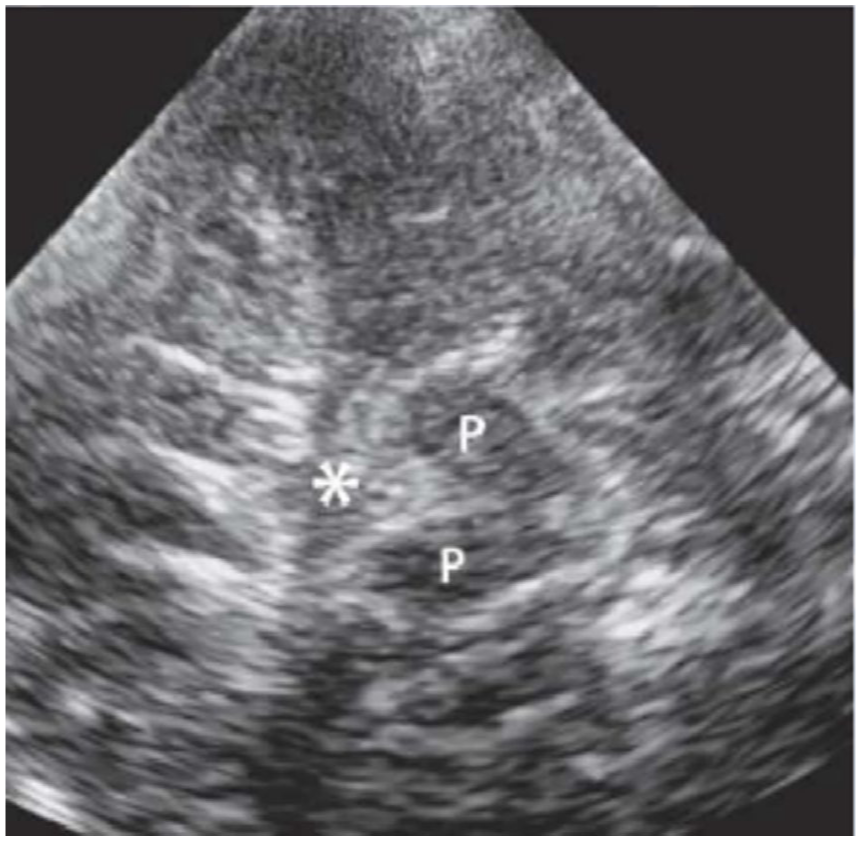
Transtemporal window mesencephalic plane.
Transtemporal window diencephalic plane.
In the mesencephalic plane, the area immediately lateral to the peduncles can then be interrogated with colour and pulsed wave Doppler to demonstrate the vessels as well as to obtain measurements (Figure 4). Cerebral haemodynamic assessment primarily relies on the mesencephalic and diencephalic planes, where the principal arteries of the circle of Willis are visualised. 29 In adults, absence of the transtemporal acoustic window is defined by failure to visualise the highly echogenic inner table of the contralateral temporal bone despite insonation depths exceeding 15 cm. 30
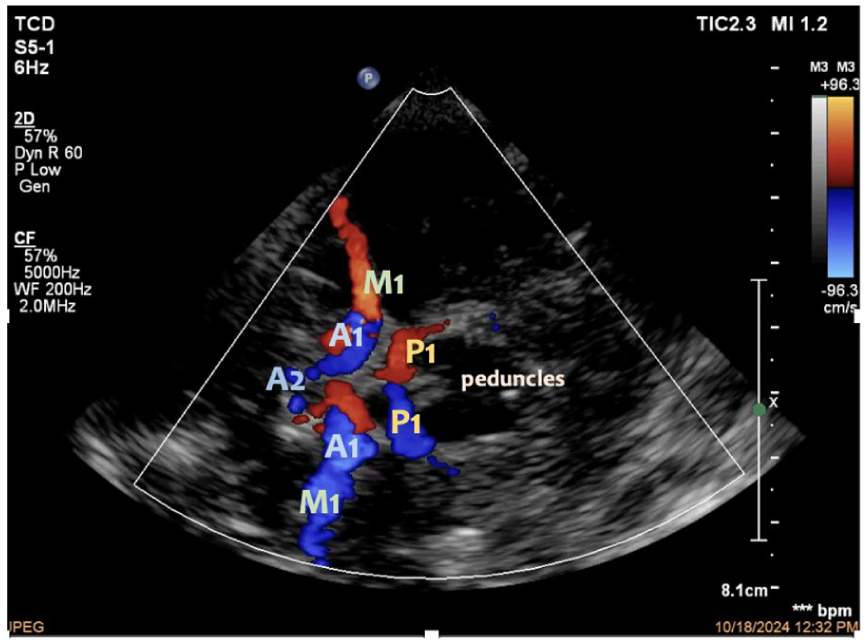
Transtemporal window demonstrating the circle of Willis.
A typical TCD waveform has a systolic peak and a diastolic stepdown (Figure 5). The shape of the waveform is important to note, as changes in systolic and diastolic morphology indicate pathology. Once the waveform has been recorded calculated values for peak systolic (PSV), end diastolic (EDV), and time averaged mean flow velocities (TAM) are obtained. From these measurements, two derived measurements are calculated: the pulsatility index (PI) and the resistivity index (RI). These give an indication of the resistance to blood flow. The brain maintains a low resistance circulation to always allow preferential blood flow. Changes in the PI correspond to changes in cerebral haemodynamics. The PI value needs to be interpreted in the clinical context of the patient. The PI is typically higher in the posterior circulation (vertebral and basilar artery) as the posterior fossa is a tighter compartment. 31
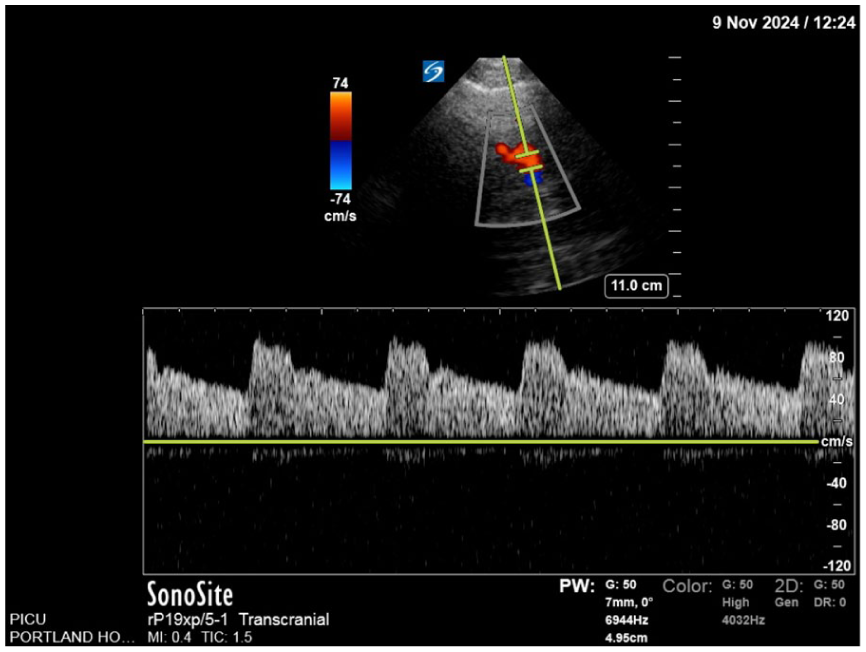
A typical TCCD waveform. PI = (PSV − EDV)/MV normal value 0.6–1.2.
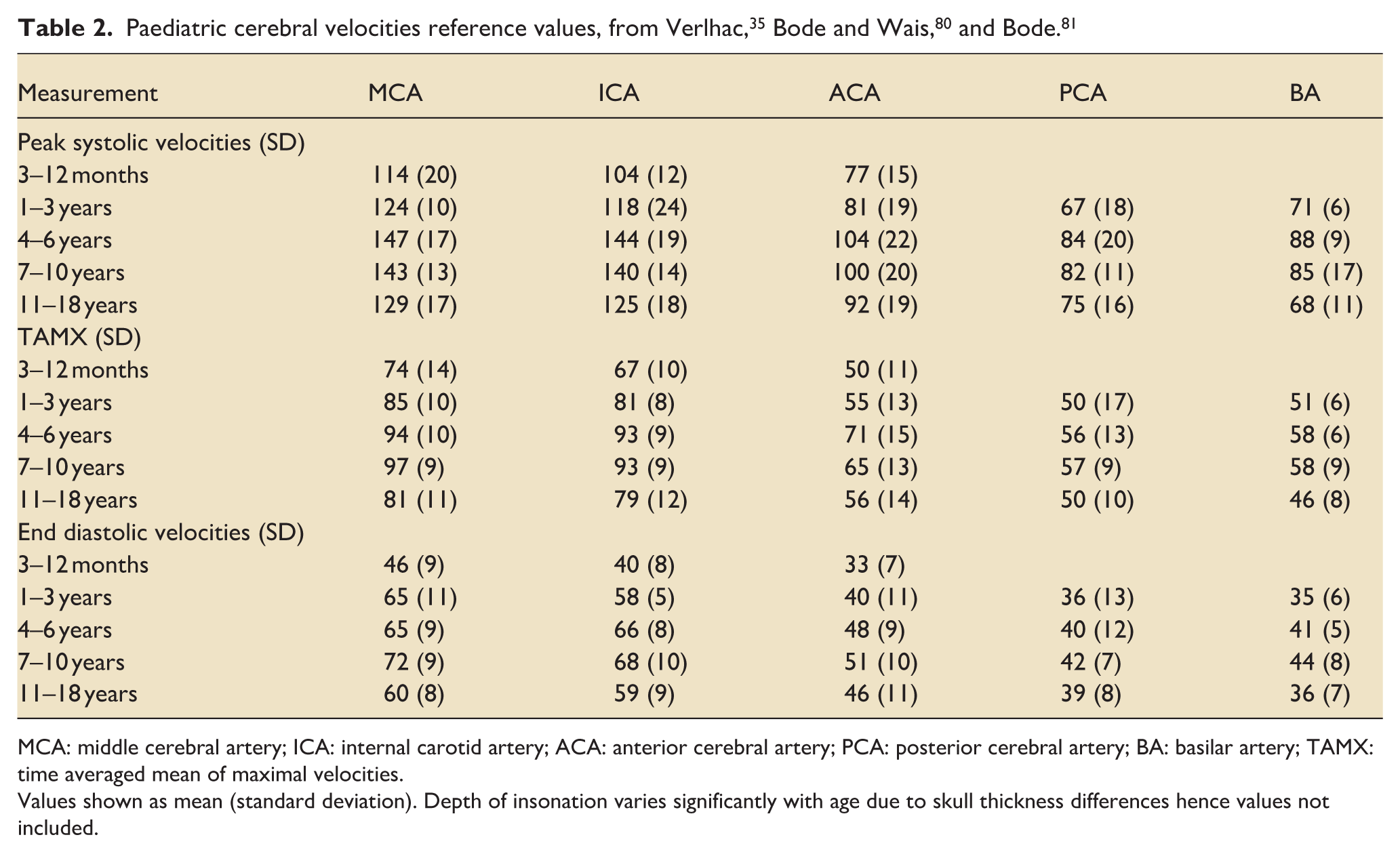
Knowledge of the depth of insonation, direction of blood flow relative to the transducer probe and the mean velocity of the flow allows correct identification of the vessel (see Table 1, including reference velocities for vessels in adults).32,33 Cerebral velocities are lower in neonatal period, increase in infancy, peak around 4–6 years old and slowly decline to adult levels thereafter (Table 2). This change parallels the higher cerebral metabolic demand in early childhood. A major implication for TCD interpretation is that absolute velocity values need to be compared to age-specific data for correct interpretation.9,34,35 As a resource for learners to facilitate clinical interpretation of paediatric TCD measurements, Plante and LaRovere have developed a standardised Z-score calculator for middle cerebral artery velocities that enables rapid comparison of patient values against multiple reference datasets. This computational tool is accessible through the OPENPediatrics educational platform. 36
Reference characteristics and velocities for vessels in adults, from Tegeler et al. 33
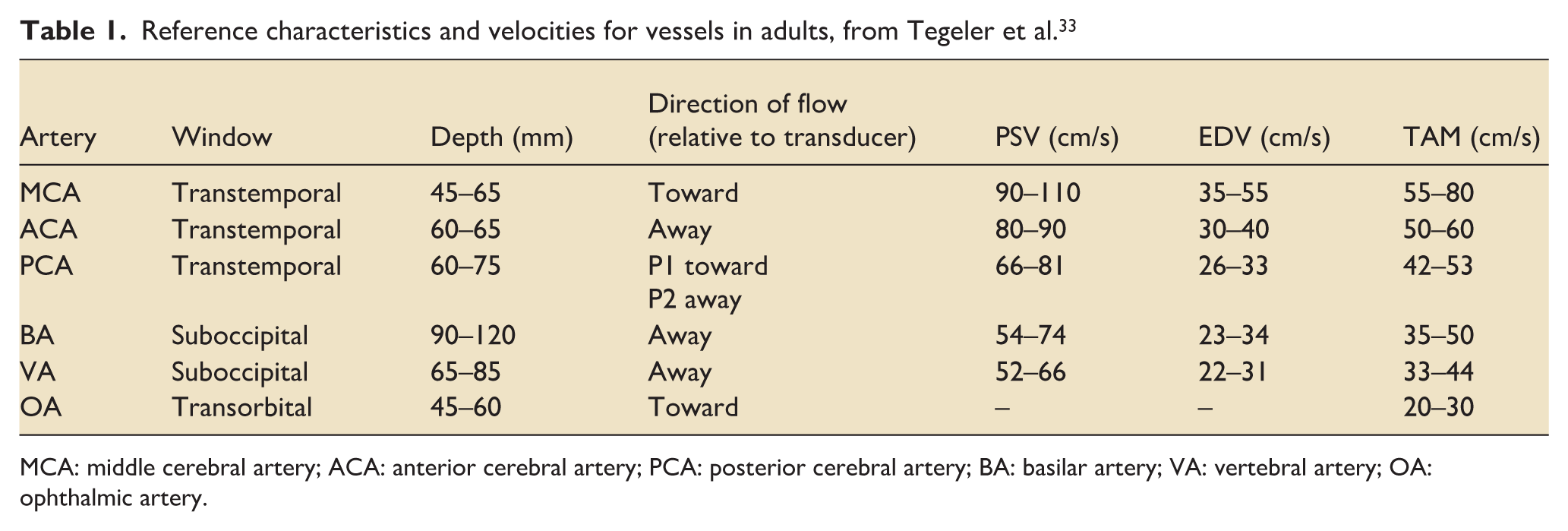
MCA: middle cerebral artery; ACA: anterior cerebral artery; PCA: posterior cerebral artery; BA: basilar artery; VA: vertebral artery; OA: ophthalmic artery.
MCA: middle cerebral artery; ICA: internal carotid artery; ACA: anterior cerebral artery; PCA: posterior cerebral artery; BA: basilar artery; TAMX: time averaged mean of maximal velocities.
Values shown as mean (standard deviation). Depth of insonation varies significantly with age due to skull thickness differences hence values not included.
In recent years, consensus guidelines have formalised the role of TCD in neurocritically unwell children and adults. Consensus standards for use of TCD in PICU were published in 2020, following a three round modified Delphi process. 34 An expert multidisciplinary panel elaborated practice recommendations for transcranial Doppler use in PICU, recommending standards for indication, technical performance and for reporting and data interpretation. In adults, the 2025 B-ICONIC (Brussels) guidelines strongly recommended the use of TCD and ONSD as part of non-invasive ICP monitoring in adults with traumatic brain injury. 37
A summary of potential adult and paediatric conditions wherein NeuroPOCUS may be used, the associated physiological processes, and relevant NeuroPOCUS parameters is illustrated in Table 3. We will expand upon these in the following subsections.
Summary of NeuroPOCUS parameters, physiological processes and relevant adult and paediatric pathologies.
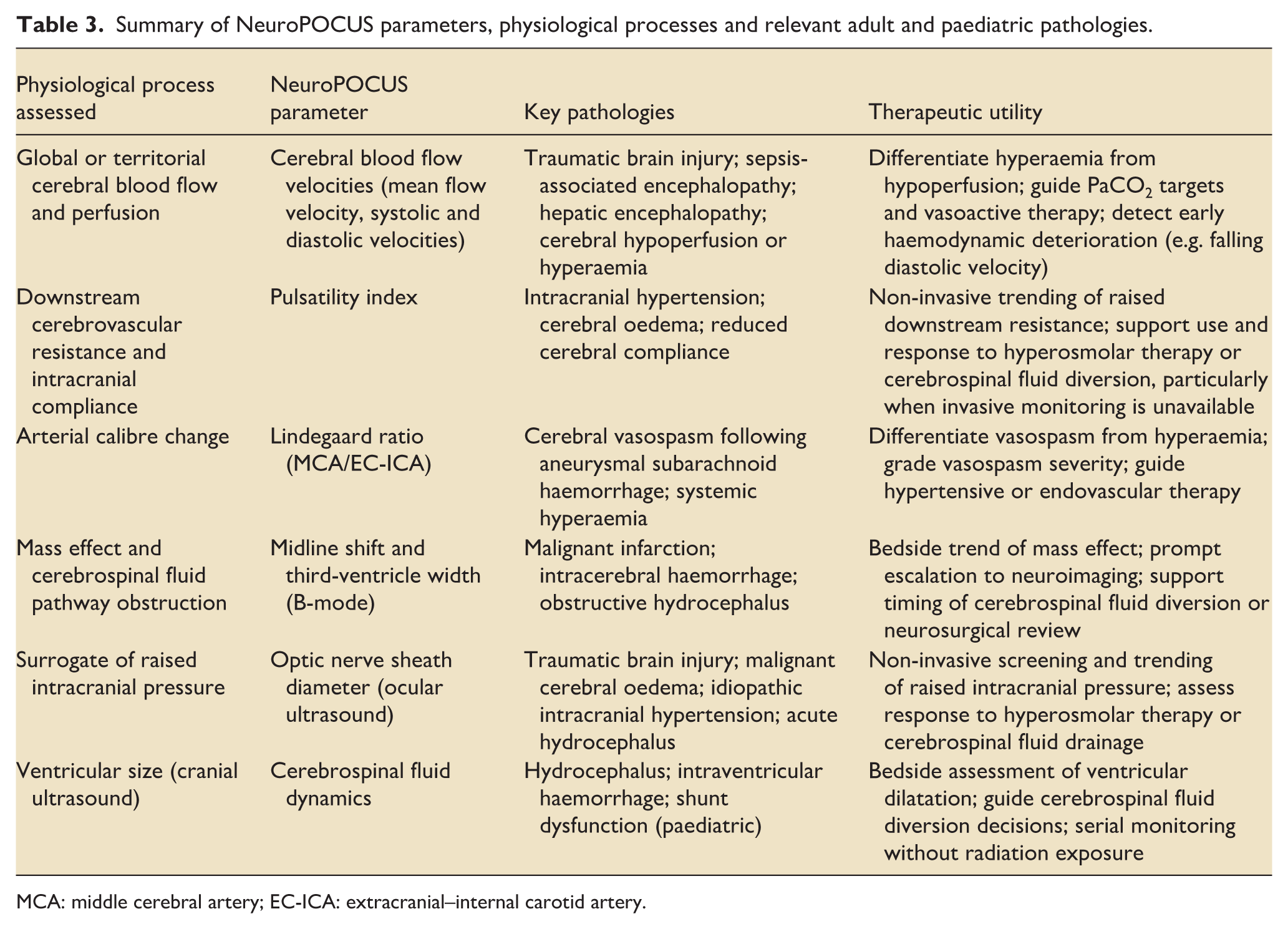
MCA: middle cerebral artery; EC-ICA: extracranial–internal carotid artery.
Clinical use in paediatric critical care patients
Traumatic brain injury
Traumatic brain injury (TBI) remains a leading cause of morbidity and mortality in paediatric populations. As illustrated by the STARSHIP study, by evaluating cerebral haemodynamics in these patients through various methods, clinicians can gain insights into the severity of the injury and the effectiveness of therapeutic intervention. 38 TCD is one potential non-invasive tool which can be used to monitor cerebral autoregulation and intracranial pressure in children with TBI.39,40 In these patients, cerebral blood flow velocities can reflect global or territorial cerebral blood flow and perfusion.
A study by Abecasis et al. demonstrated the feasibility of using a novel non-invasive method for CPP estimation (nCPP) in children with severe TBI. The study showed a good correlation between nCPP and invasive CPP, with excellent accuracy in predicting low CPP values below 70 mmHg. 39 A retrospective study involving 36 children found that TCD-derived pulsatility index (PI) and end-diastolic flow velocity (Vd) correlate with intracranial pressure, particularly in the first 24 h post-injury. 40 In a prospective study of patients with mild to moderate traumatic brain injury, Bouzat et al. found that elevated mean flow velocities in the middle cerebral artery within 48 h of admission were independently associated with worse neurological outcomes at 6 months. The findings suggest that early TCD can identify cerebrovascular dysregulation not evident on initial clinical or imaging assessment and may serve as a useful tool to stratify risk and guide management in this patient group. 41
Non-traumatic brain injury
TCD is a potentially useful bedside tool in the management of central nervous system infections and inflammatory conditions in critically ill children. In a multicentre French study, 152 non-traumatic critically ill children underwent TCD in PICU. TCD was used early (within 24 h in 74% of cases), primarily for neurological monitoring (56%), and screening for asymptomatic brain issues (24%). 42 Although intensivists found TCD clinically helpful in 86% of cases, objective concordance with normative data was low (28%), highlighting interpretative subjectivity. TCD influenced diagnosis in 72% and treatment in 36% of cases. The authors concluded that standardised protocols and outcome-focussed studies are needed to confirm its impact. 42
A retrospective study of 20 paediatric patients by Ducharme-Crevier et al., demonstrated that TCD could detect abnormal cerebral blood flow patterns, such as hyperaemia, high flow with normal PI, or low flow with elevated PI, associated with worse clinical outcomes, including increased neurologic disability and mortality. 43 TCD findings also influenced clinical decision-making in some cases, guiding further imaging, intracranial pressure monitoring, and therapeutic interventions. However, again the authors recommended their results should undergo further validation in prospective studies.
Singla et al., in their recent paper, demonstrated that in critically ill children with paediatric acute liver failure (PALF), abnormal bedside neuromonitoring findings were associated with mortality. 44 Children who died had higher MCA pulsatility index, lower MCA mean velocity, and lower reverse jugular venous oxygen saturation (SjvO2). Transcranial Doppler and SjvO2 correlate weakly with ammonia but provide real-time physiological information that ammonia alone cannot. These modalities should be considered useful adjuncts, not replacements, to clinical neurological assessment in PALF, with further research needed to determine whether acting on these measurements improves outcomes.
Hydrocephalus
In children with hydrocephalus, B mode ultrasound can support bedside assessment of shunt function and early detection of complications such as obstruction. Doppler parameters (e.g. MFV, Vd, PI) reflect changes in intracranial pressure and cerebrovascular resistance, while B-mode measurements (third-ventricle width) provide structural information and trends. In a paediatric cohort, Widehem et al. reported that third-ventricle measurement by transcranial ultrasound was feasible in 87% of cases and highly reliable versus CT (intraclass correlation coefficient 0.90–0.92). 45 In 69% of assessable scans, the technique confirmed or excluded hydrocephalus, thereby reducing the need for repeat CT. Residents attained competency after approximately five supervised examinations, underscoring transcranial ultrasound as a reliable bedside modality for monitoring shunted children and prompting timely intervention.
Clinical use in adult critical care patients
Detection of vasospasm in aneurysmal subarachnoid haemorrhage
TCD provides a bedside surrogate assessment of vasospasm in aneurysmal subarachnoid haemorrhage (aSAH) by measuring intracranial arterial flow velocities. Approximately 80% of aSAH occur within the MCA and ACA vascular territory. Mean flow velocities between 120 and 200 cm/s are indicative of vasospasm in these vessels. To distinguish elevated velocities due to vasospasm from those secondary to a hyperdynamic circulatory state (e.g. in sepsis), the Lindegaard ratio (LR) is calculated by dividing the mean flow velocity in the MCA (or ACA) by that of the ipsilateral extracranial internal carotid artery, insonated through the submandibular window. An LR > 3 suggests mild spasm, 3–6 indicates moderate spasm, and >6 denotes severe vasospasm. These velocities are influenced by physiological factors such as arterial blood pressure and arterial carbon dioxide tension; therefore, they must always be interpreted in conjunction with the clinical neurological picture.46,47
Although NICE concluded that routine TCD monitoring should not be used to guide management outside the context of clinical research, 48 recommendations in other settings have since evolved to incorporate TCD. The 2023 American Heart Association/American Stroke Association (AHA/ASA) guidelines for the management of aSAH provide a Class IIa recommendation for the use of TCD monitoring to detect cerebral vasospasm when performed by trained operators. 49 Furthermore, a recent systematic review and meta-analysis by Schenck et al. evaluating TCD-derived biomarkers for predicting delayed cerebral ischaemia (DCI) after aSAH reported that, among velocity-based indices, a mean flow velocity >120 cm/s combined with the LR yielded the highest sensitivity, specificity, and predictive values for identifying patients at risk of DCI or vasospasm. 50
Acute ischaemic stroke
Early identification of a large vessel occlusion (LVO) in acute ischaemic stroke (AIS) is critical, as it directly influences triage decisions, facilitates timely transfer to thrombectomy-capable centres, and optimises the therapeutic window for reperfusion. Bankole et al. conducted a systematic review evaluating the diagnostic accuracy of TCD for detecting AIS caused by LVO. 51 They reported that TCD derived PI demonstrated the highest diagnostic performance, with accuracy ranging from 96% to 99.2%, and concluded that TCD may support early stroke management when immediate CT angiography (CTA) is unavailable or contraindicated, such as in patients with reduced eGFR or haemodynamic instability. Similarly, Jazayeri et al., in a recent systematic review and meta-analysis, found that TCD and TCD-derived indices could be integrated into prehospital stroke pathways to enhance triage accuracy and expedite transfer of patients with suspected LVO. 52
Nedelmann et al. developed consensus recommendations on the use of TCD for assessing recanalisation following LVO. By analysing the flow dynamics of collateral circulation, they proposed the Collateral and Occlusion Grading of Intracranial Flow (COGIF) scoring system to standardise interpretation of intracranial flow obstruction. 53 They observed that a focal increase in velocity indicated intracranial stenosis, whereas elevated flow velocities along an entire arterial segment reflected hyperperfusion, typically seen immediately after recanalisation or in the presence of collateral flow across the affected territory.
Malignant MCA syndrome
Malignant MCA syndrome is a term used to describe the deterioration of GCS and neurological symptoms following an MCA infarction, which is attributed to the space-occupying vasogenic oedema caused by the infarcted area, potentially leading to midline shift (MLS), and raised intracranial pressure. In addition to standard TCD assessments of MCA flow velocity and changes in PI, transcranial ultrasound can be used to quantify MLS. 54 To calculate MLS, the transtemporal acoustic window in the diencephalic plane is used to identify the third ventricle. Bilateral measurements in millimetres are taken from the start of the ultrasound beam at the top of the screen to the centre of the third ventricle (between the hyperechoic lines). The difference between the two measurements divided by two gives the MLS, that is, (right side − left side)/2. An MLS > 2.5 mm on transcranial ultrasound is considered significant and has been shown to have good correlation with CT imaging. Transcranial ultrasound can be used to monitor trends and hence prompt escalation to neuroimaging, and support timing of interventions. Of note, in the post decompressive craniectomy patient, this method of measuring MLS is inaccurate. 55
Traumatic brain injury
The 2025 B-ICONIC guidelines made a strong recommendation for the use of TCD, in particular the PI, and ONSD as part of non-invasive ICP monitoring in adults with TBI. 37 The recommendation for use of PI was based on seven studies performed predominantly in adults with TBI, which broadly described PI as demonstrating high accuracy and correlating well with invasive measures of ICP in this population.56,57 In one retrospective study of 68 adults with TBI, PI was strongly correlated with ICP of 15–20 mmHg (r = 0.705, p < 0.001) and ICP of ⩾20 mmHg (r = 0.716, p < 0.001). 58 In a separate study of 93 adults (75 of whom had TBI), PI and RI were also significantly positively correlated with intracranial pressure (r = 0.90 and 0.89; p < 0.001). 56
Cerebral hyperperfusion syndrome post carotid endarterectomy surgery
Cerebral hyperperfusion syndrome (CHS) clinically presents as a triad of ipsilateral headache, seizure, and focal neurological symptoms occurring in the absence of cerebral ischaemia. It is accompanied by post-operative hypertension in almost all patients. If left untreated the hyperperfusion causes cerebral oedema and MLS. In CHS, hyperperfusion is defined as a >100% increase in MCA velocity. Baseline measurement of MCA velocity before induction of anaesthesia is a quick non-invasive assessment and allows continued measurement, as CHS can appear immediately post operatively or up to a month later. Given the signs of CHS are similar to stroke, the MCA velocity is a useful diagnostic differential in this clinical scenario. 59
Other indications (adult and paediatric)
CNS inflammatory and infectious conditions
TCD can be used to monitor cerebral blood flow alterations in infectious or inflammatory conditions such as meningitis, encephalitis, and encephalopathy. 44 In such cases, it serves as a bedside adjunct to infer cerebral hypoperfusion and guide therapeutic decisions. Although it cannot diagnose cerebral oedema directly, a recent observational study in adults confirmed that TCD was able to detect early haemodynamic changes suggestive of cerebral oedema in patients with high-risk or limited access to conventional imaging, including those with systemic infections and altered mental status. 60
Cardiac surgery and ECMO
Neurological complications such as stroke, delirium, cognitive dysfunction can occur during and after cardiac surgery. TCD allows evaluation of cerebral haemodynamics in real-time and show which mechanism is involved in their development, either by embolism, thrombosis, hypoperfusion, or hyperperfusion, during the perioperative and postoperative setting. 61 For ECMO patients, transfer for CT imaging is highly resource-intensive and logistically challenging. In this context, TCD may offer a useful bedside surveillance modality, particularly as ECMO patients are at increased risk of intracerebral haemorrhage (ICH), where the incidence is reported to be in the range of 1.8%–21% depending on cannulation – venoarterial or venovenous ECMO.62,63 The ability to use a non-invasive surveillance tool like TCD could enable earlier detection of ICH, as shown in one paediatric study where children with ICH had higher than normal cerebral blood flow velocities noted for days prior to clinical recognition of bleeding. 64
Post-cardiac arrest management
Avoiding significant neurological morbidity after cardiac arrest is a key goal of neurocritical care. TCD may help in monitoring cerebral haemodynamics and guiding therapeutic strategies post-resuscitation. The 2020 ILCOR guidelines emphasise the importance of maintaining adequate cerebral perfusion. 65 In a systematic review, Scholefield et al. found limited but emerging evidence suggesting that TCD measurements, particularly low mean flow velocities and elevated pulsatility index in the first 72 h after paediatric cardiac arrest, may be associated with poor neurological outcomes. 66 In a prospective study in adults, Reichenbach et al. found that TCD parameters, particularly elevated peak systolic velocities, measured on days 5–7 after cardiac arrest were significantly associated with poor neurological outcomes at 6 months. Early TCD findings (days 1 and 3) were not predictive, suggesting that delayed cerebral haemodynamic changes may carry greater prognostic value than initial assessments. 67 Overall, most available studies to date are small, observational, and heterogeneous, so more robust data are needed before TCD can be routinely recommended for prognostication.
Cerebral circulatory arrest
TCD may prove to be a valuable tool as part of the assessment of brain death in adults and children by detecting cerebral circulatory arrest. Increasing ICP causes decreasing CPP until critical closing pressure (CrCP) is reached. CrCP is the arterial pressure below which cerebral blood flow (CBF) approaches zero. The step-wise changes in CBF can be observed via transtemporal TCD of the MCA flow in an emergency setting (Figure 6). Oscillatory flow/reversed diastolic flow or systolic spikes, indicate the absence of cerebral blood flow. 40 To confirm cerebral circulatory arrest, all anterior and posterior circulation vessels bilaterally should be insonated with extracranial circulation through ICA insonation submandibularly to demonstrate physiological forward flow bilaterally. At least two studies should be done 30 min apart to confirm circulatory cerebral arrest.
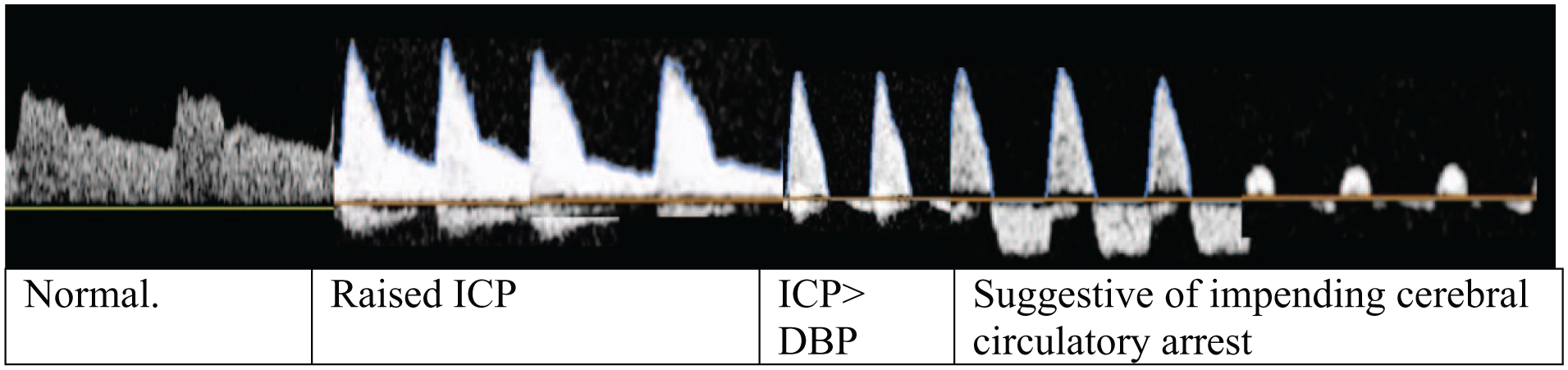
Step wise changes in MCA waveform shape as intracranial pressure increases.
Meta-analyses have described high sensitivity (90%–95%) and specificity (98%–99%) of TCD for brain death diagnosis. 68 However, TCD in this scenario is evaluating cerebral circulatory arrest rather than brainstem function, and so should be used with caution as an ancillary test for diagnosing death by neurological criteria.25,47,69 The World Brain Death Project recommends against using TCD as an ancillary test in paediatric patients due to insufficient validation data in this population. 25
ONSD and raised intracranial pressure
The sheath of the optic nerve is a continuation of the intracranial meningeal layers, enclosing a subarachnoid compartment, that is, continuous with the intracranial subarachnoid space. The anterior retrobulbar perioptic subarachnoid space is surrounded by orbital fat and soft tissues, rendering it more compliant than the intracranial segment, which is constrained by bony structures. In the absence of obstruction to CSF flow, increases in CSF pressure are transmitted along the optic nerve sheath and preferentially result in distension of this anterior, more compliant segment, providing the physiological basis for optic nerve sheath diameter (ONSD) measurement. 70
A high frequency (7.5–12 MHz) linear probe is used with the ophthalmic setting selected, according to the ALARA (as low as reasonably achievable) principle – the principle that ultrasound energy exposures should be minimised as much as possible. The anechoic globe is seen with the optic nerve sheath behind it. The ONSD is measured 3 mm from the back of the globe and across between the external borders of the hyperechoic area surrounding the optic nerve (green lines, Figure 7).70,71 The 3 mm marker from the back of the globe is where the optic nerve sheath is most distensible. Measuring the ONSD in both the transverse and the sagittal planes in both eyes and averaging the measurement allows for a more accurate assessment. Visualisation of the central retinal artery, as it runs through the centre of the optic nerve sheath, aids the identification of the optic nerve sheath from ultrasound artefact caused by the lamina cribrosa.
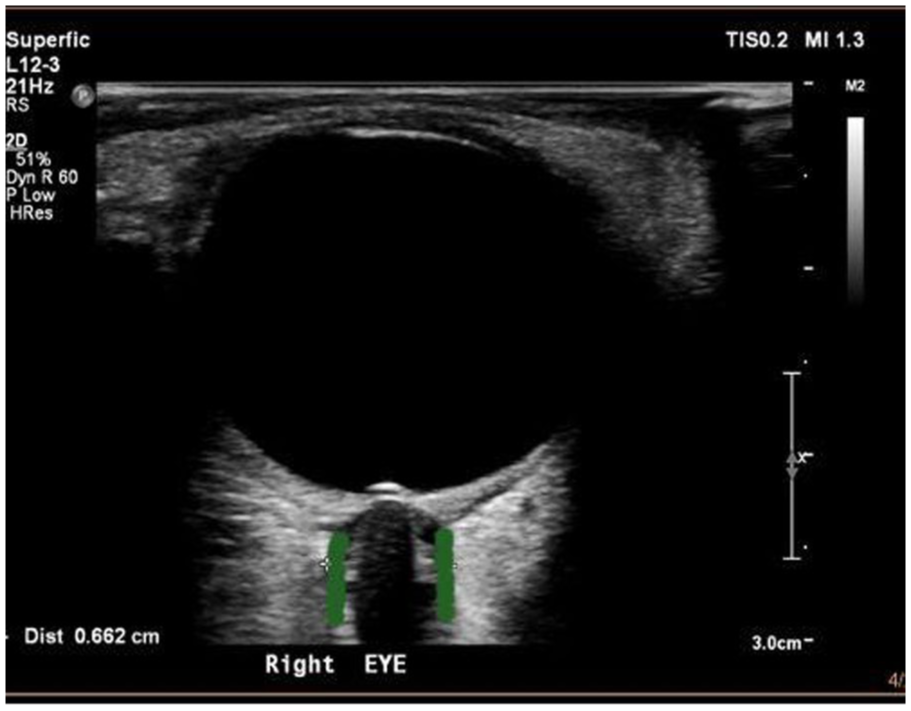
Ultrasound anatomy of ONSD. Green lines represent ONSD.
An international consensus statement has established standardised quality criteria for ONSD ultrasound to enhance its reliability in detecting elevated ICP. The panel recommends using a linear probe, acquiring measurements in the axial/coronal plane, and measuring 3 mm posterior to the globe, at least two measurements per eye should be averaged. These guidelines aim to improve accuracy, consistency, and comparability across clinical settings. 70
The normal ONSD in the adult population ranges between 5.6 and 5.9 mm depending on gender and ethnicity. An ONSD above 6 mm is suggestive of an intracranial pressure >20 mmHg. In children, ONSD increases with age, particularly during infancy, and normative values show substantial overlap across age groups. Available data derive largely from small, heterogeneous cohorts using different ultrasound techniques and reference standards, and robust age-specific normal ranges have not been firmly established. 72 Although early studies suggested upper limits of ~4.0 mm in infants and 4.5 mm in older children, more recent paediatric studies demonstrate considerable variability in ONSD values and propose different thresholds for detecting raised intracranial pressure, reflecting differences in age, pathology, methodology, and reference standards.72,73 Consequently, until large, methodologically robust paediatric datasets are available, ONSD measurements should be interpreted in the context of age, clinical assessment, serial changes, and complementary neuromonitoring rather than reliance on absolute cut-off values.
Systematic reviews in adults suggest that an ONSD above 6 mm has high sensitivity for raised intracranial pressure; in a systematic review and meta-analysis across 71 eligible studies, ONSD had sensitivity of 97% (95% CI, 92%–99%), and specificity of 86% (95% CI, 74%–93%) for raised ICP. 5 In a meta-analysis across 25 paediatric studies, ONSD had sensitivity and specificity of 92% (86%–96%) and 89% (77%–96%), respectively. 74
Kim et al. demonstrated ONSD is strongly dependent on eyeball transverse diameter (ETD), whereas the ONSD/ETD ratio (~0.18) was independent of sex, height, BMI, head circumference, supporting the ratio as a more stable marker than ONSD alone across individuals. 75
Hansen et al. demonstrated in cadaveric models that the maximum ONSD was 7.5–8 mm and at high pressures (45–55 mmHg), the optic nerve sheath lost its elastic recoil suggesting that under these conditions, changes in ONSD do not reliably reflect clinical response to interventions aimed at lowering intracranial pressure. 76
Irrespective of technique or value chosen, it is important to recognise that ONSD should be interpreted in the clinical context and ideally in conjunction with other non-invasive measurement of raised intracranial pressure such as TCD and automated pupillometry. 37
Accreditation
Despite the increasing uptake of POCUS in critical care and acute medical specialities and recent publications on non-invasive assessment of cerebral haemodynamics as part of a head-to-toe assessment,6,9,11,69 and the inclusion of TCD and ONSD in consensus guidelines such as B-ICONIC, 37 accreditation is not available in most countries, and to date there has been no UK accreditation training programme. European programmes overseen by ESNCH and ESICM (GenIUS) have annual hands-on workshops, as well as providing curriculum and competency guidance.77,78 Neurocritical care fellowships in America, Canada, and India incorporate TCD as part of their educational content on non-invasive neuromonitoring but some of these fellowship programmes lack sufficient ultrasound-competent faculty to successfully develop and implement an ultrasound curriculum.
To address the current gaps and advance the field, we provide an example accreditation strategy which is being implemented in the UK for paediatric and adult intensivists (see Table 4 and Figure 8). The framework targets four areas: standardisation, competency, diagnostic accuracy, and patient outcomes. The unified curriculum, defined imaging protocols, and harmonised interpretation thresholds aims to ensure that all practitioners, regardless of background or experience, develop a reliable approach to NeuroPOCUS. 79 Competency will be built and maintained through minimum supervised scan numbers, mentorship, and formal assessments. Diagnostic accuracy will be promoted via adult–paediatric case discussions and training in monthly webinars and via mentorship. Ultimately, increased number of operators will encourage research into the diagnostic accuracy of NeuroPOCUS and whether it can, for example, shorten time-to-diagnosis for vasospasm, venous congestion, and raised ICP; reduce unnecessary transfers and imaging; and support safer titration of haemodynamic and osmotherapy strategies. Implementation will include quality assurance and feedback loops to refine the curriculum and mitigate variability. Further research on the efficacy of the accreditation strategy is planned.
NeuroPOCUS – paediatric and adult accreditation – example program.

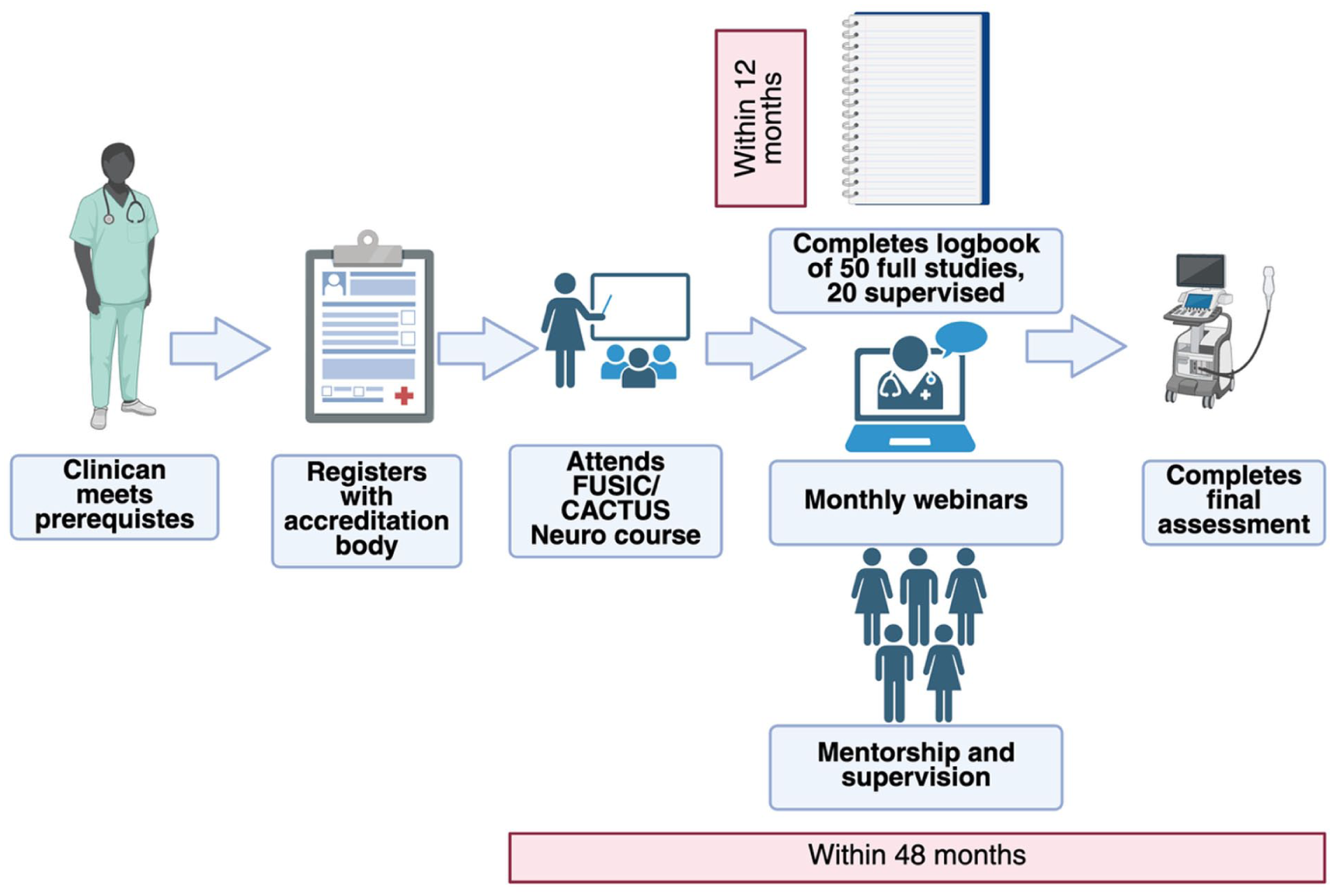
NeuroPOCUS accreditation timeline.
Conclusion
TCD and ONSD measurements have several potential benefits in neuromonitoring critically ill adults and children, by providing non-invasive bedside data that can be rapidly obtained, and readily repeated. NeuroPOCUS measurements have been correlated with clinical outcomes across a number of studies in both adults and children, and in some cases used to guide clinical management. The possibility of wider accreditation in NeuroPOCUS provides an opportunity to conduct further training and therefore, ultimately, research into the potential value of non-invasive neuromonitoring via TCD, ONSD, and other modalities. It also provides an opportunity for collaboration between experts in paediatric and adult neurocritical care practice. We recommend more research and training in NeuroPOCUS within the critical care community.
Footnotes
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.