Abstract
Necrotizing enterocolitis (NEC) is a potentially fatal comorbidity of prematurity with one in five affected requiring surgical intervention. Despite its seriousness, there is lack of objective radiographic criteria on plain abdomnial radiographs (X-ray) to guide prognosis and decision making. Point of care bowel ultrasound (BUS) provides a more dynamic assessment and more information around bowel health. However, there is lack of widespread adoption of this practice by neonatologists due to training opportunities and inconsistent support from radiologists. We present a feasibility study from UK of using point of care bowel ultrasound in conjunction with X-ray to aid diagnosis of NEC. We report that that neonatologist performed BUS when used as an additional diagnostic aid has a higher positive predictive value and specificity compared to X-rays alone. Features like absent or poor peristalsis and abnormal bowel perfusion were the most consistent pathological findings in our cohort. Wider implementation of this practice is limited by training opportunities and dedicated support from radiology team
Background settings and the problem
Necrotizing enterocolitis (NEC) is a fatal inflammation of the intestine leading to invasive sepsis often requiring surgical intervention. It affects exclusively neonates and a specifically those born premature and of very low birth weight. The earliest and most used case definition of NEC was proposed by Bell et a. 1 in 1978 and further modified in 1986. 2 This classification uses a constellation of systemic and abdominal symptoms with findings on abdominal radiographs to prognosticate and guide management. With greater understanding of the epidemiological and pathological diversity of NEC as a disease process, the relevance of such standardized case definitions for clinical decision making have been questioned as they include characteristics with varying sensitivity and specificity like feed intolerance. 3 Radiological features play a significant role in diagnosis of NEC. Clinicians have historically depended on interpretation of plain abdominal radiographs (AXRs) for diagnosis. The existing gold standard for radiological confirmation of NEC is presence of intramural bowel gas or pneumatosis intestinalis (PI) on AXR. The detection of Pneumatosis intestinalis in NEC is variable and has been reported to be between 70% and 80% but can sometimes be missed as it is an early finding . 4 Furthermore, observational studies have shown that interpretation of PI is subjective and often labelled as ‘possible’ or ‘difficult to exclude’. 5 Diagnosis based on AXR can hence be challenging due to nonspecific nature of findings and lack of objective criteria for early identification of surgical NEC. 6 Given these limitations, there has been interest in exploring more dynamic methods of bowel assessment like point of care ultrasound. Recent meta-analysis has reported bowel ultrasound (BUS) to be effective in early identification of infants with NEC who might benefit from surgical intervention. 7
The Lancashire Women and Newborn Centre is a neonatal intensive care unit (NICU) based in a district general hospital in north-west neonatal network (NWODN) of UK, with a delivery rate of approximately 6000 per year of which >100 is born very preterm. The average incidence of NEC being 5%–6%. The existing pathway for surgical referral for NEC is through a multi-disciplinary conference call between referring clinician, paediatric surgical team and transport services. The decision-making process on surgical review is hence based on clinical interpretation of the referring team and a remote review of AXRs. Lack of defined criteria and objectivity often leads to conflicting opinions on appropriate place for management.
Aim
To develop a quality improvement initiative to use point of care ultrasound by neonatologist for confirmation of suspected NEC.
Methodology
A feasibility study was initiated between neonatology and radiology to analyse efficacy and safety of using bowel ultrasound (BUS) as and additional diagnostic tool along with abdominal plain radiograph (AXR) for suspected NEC. The implementation of the project required the following prerequisites:
Consultant radiologist with training or special interest in paediatric radiology to be nominated senior project lead with at least one programmed activity (PA) per week job planned for supervision of this project.
Neonatologist (s) with prior experience in use of point of care ultrasound to undertake focussed course or workshop on bowel ultrasound.
Develop a study protocol and a scanning protocol (Figure 1) with a reporting proforma.
Agreement within the wider neonatal clinical team to implement this practice.
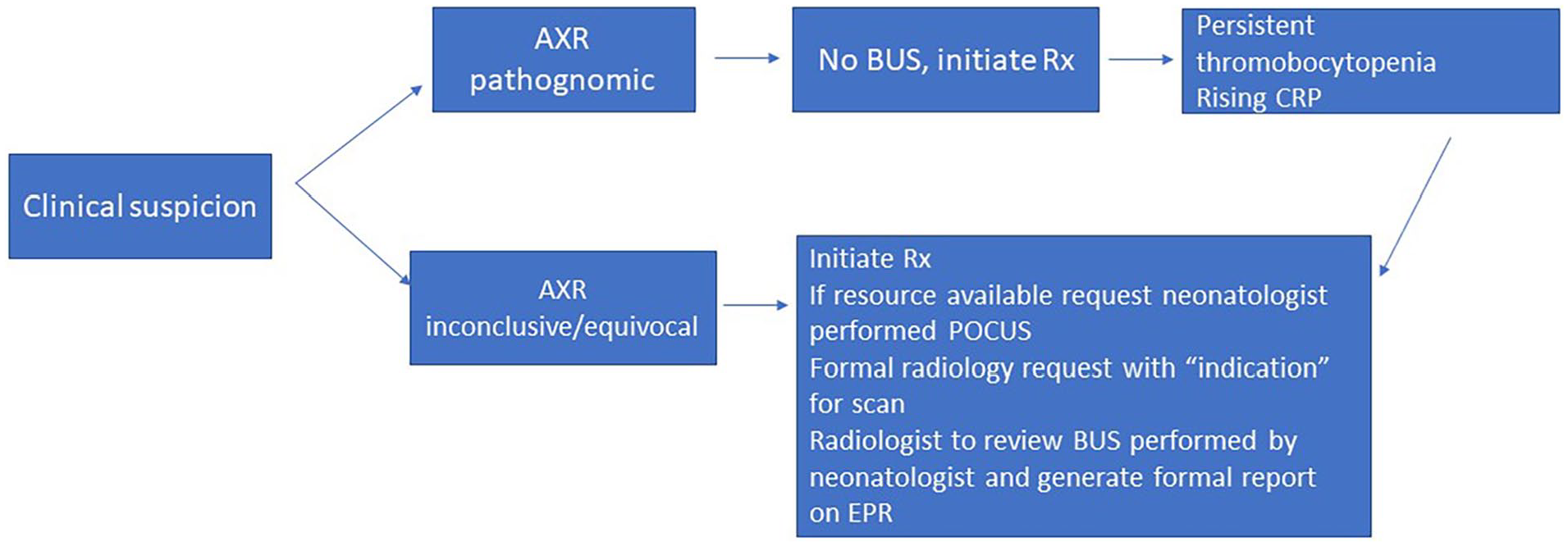
Study protocol.
The study was performed over a 24-month period; out of which in the first 12 months all patients that were initially scanned by the neonatologist were reviewed and formally reported on by consultant radiologist. In the next 12 months, all scans were performed and reported by neonatologist independently and technically difficult scans or complex scans were discussed with radiologist.
Statistical analysis
Sensitivity, specificity, positive predictive value (PPV) with confidence intervals (CI) were calculated for AXR for our sample.
No ethical approval was sought for this project as it was as agreed service improvement initiative.
Scanning protocol and reporting proforma
All patients were scanned with curvilinear 3–12 MHz and 8–18 MHz linear probe. Adequate pain control was ensured prior to the scan. Cine loop images of >1 min duration was obtained for bowel peristalsis and Doppler evaluation.
Standard practise of scanning from RLQ-RUQ-LUQ-LLQ (R-Right, L-Left, U-Upper, L-Lower, Q-Quadrant) in both sagittal and transverse planes was followed. The convex probe was used to evaluate for free fluid, complex collections, and portal venous gas. The high frequency linear transducer was used to evaluate bowel characteristics: thickness, echogenicity, dilatation, peristalsis, presence of pneumatosis and perfusion. The normative values were based on data from previous cohort study 8 and published implementation framework. 9
Data collection
All scans were formally reported on electronic patient recording system. Data was collected on patient demographics, corrected gestational age and day of life when scan was performed. Formal report on any X-ray done within 12 h of the scan was reviewed and documented. Data on laboratory blood results (platelets, C-reactive protein, and lactate) done within the same 12-h window was also collected to assess correlation.
Results
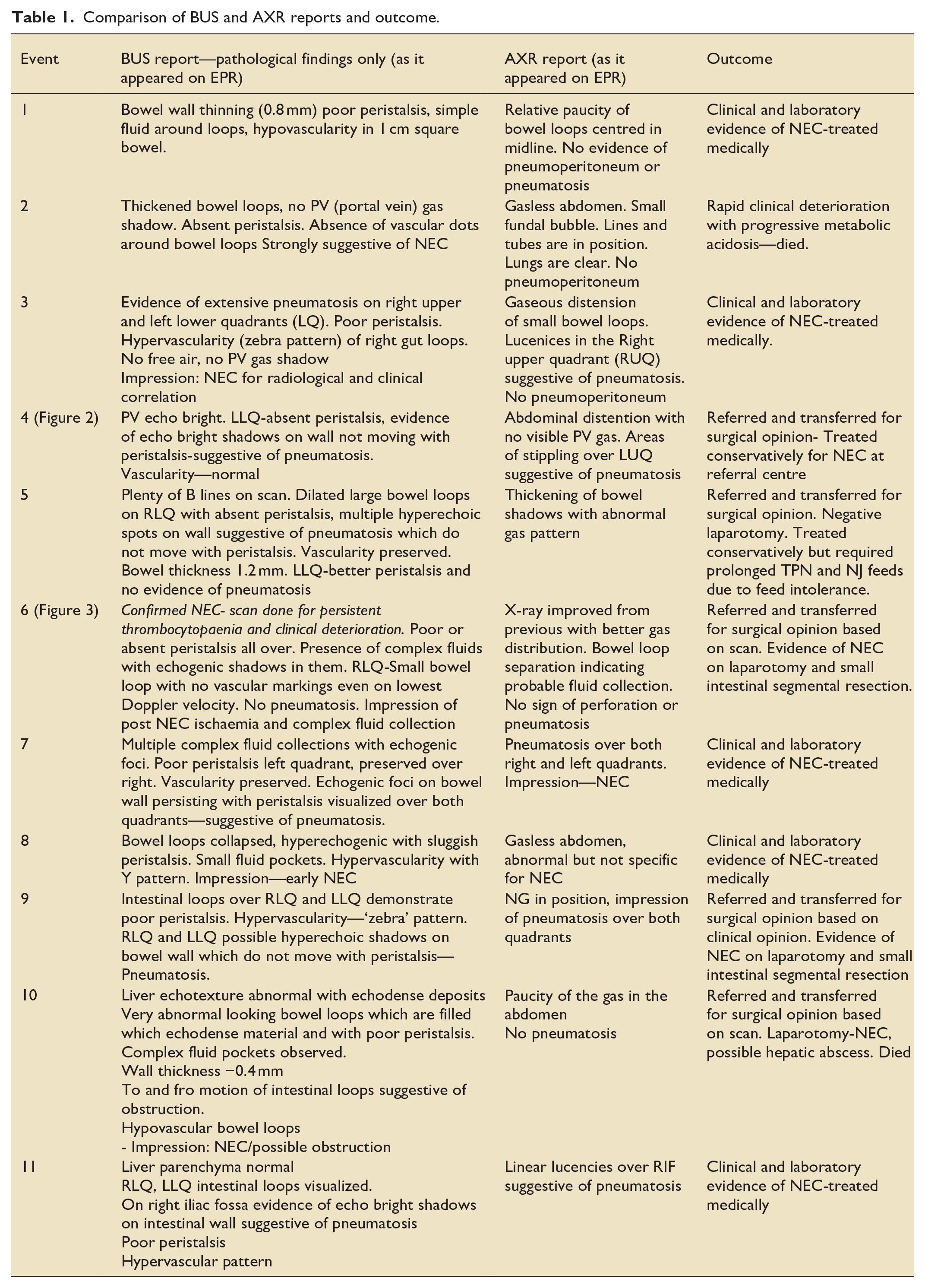
Thirty-two scans were undertaken for 28 preterm infants over a period of 24 months. Mean gestational age (GA) at birth for our cohort was 28.4 weeks. All scans had a concurrent plain AXR done within 12 h window of the scan reported by neonatologist and the radiologist. Eleven of 32 (34.3%) were pathological scans. Only 5 (45.4%) of the concurrent 11 AXRs were reported to have definite NEC with presence of pneumatosis, rest were reported to be non-specific. The positive scans and X-ray reports are described in Table 1.
Comparison of BUS and AXR reports and outcome.
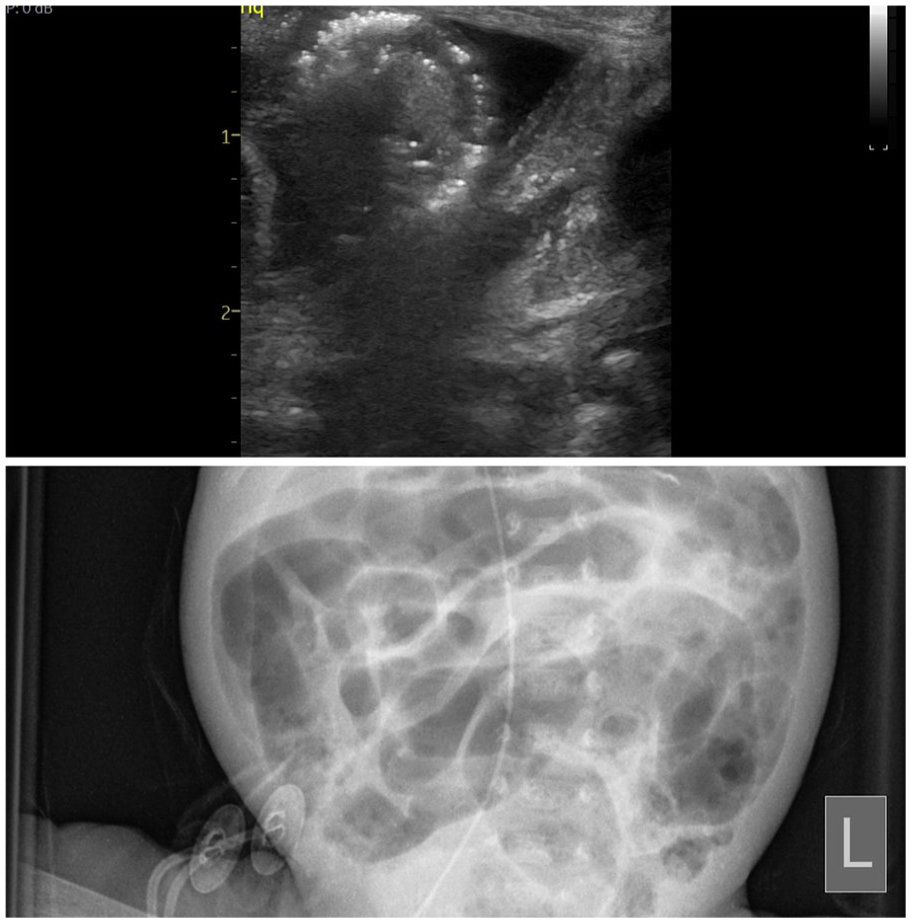
Circumferential echo-brightness on isolated gut loop demonstrating pneumatosis intestinalis. AXR was suggestive of possible stippling.
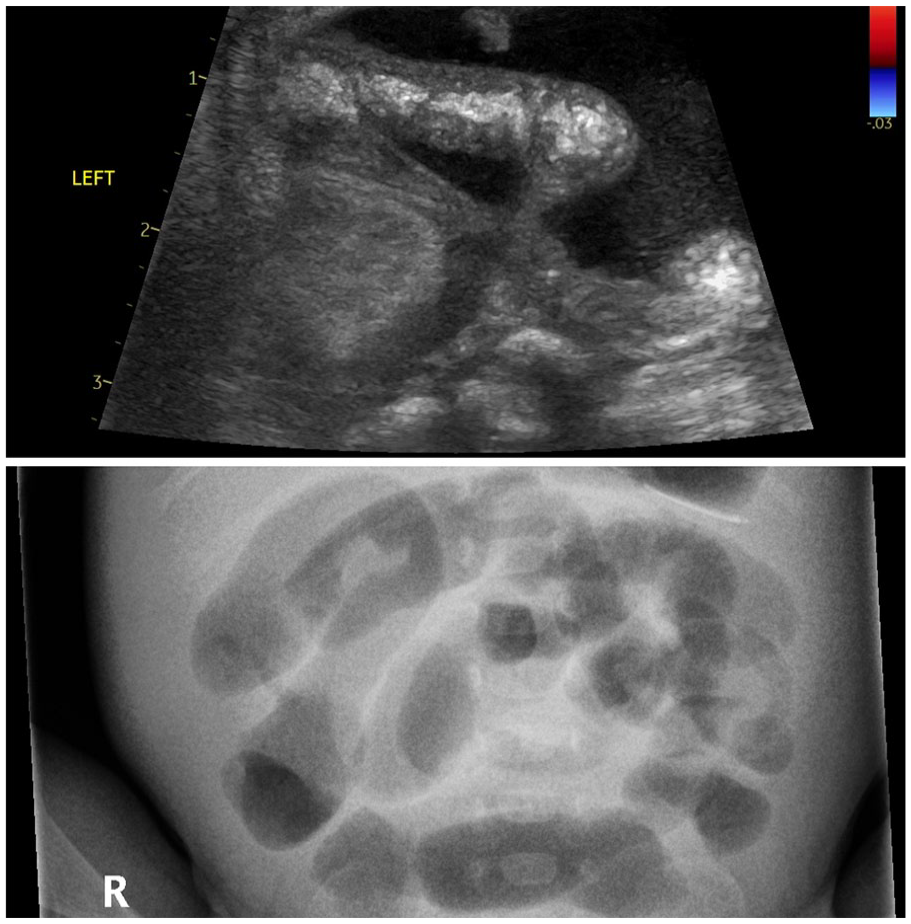
Isolated gut loop which was avascular and collection of complex fluid – complication of confirmed NEC. AXR was considered non-specific.
Six of 32 AXRs were reported to have suspected pneumatosis but all had negative BUS. All these instances had clinical improvement and did not develop NEC subsequently. In these instances, antibiotics were stopped within 72 h and feeds were safely restarted. The sensitivity for plain AXR in our cohort was 45.45% (CI 16.75%–76.62%) and PPV was 45.45% (CI 24.23%–68.47%). The specificity was 77.78% (CI 57.74%–91.38%).
Among the laboratory parameters, raised C-reactive protein showed highest correlation with a positive BUS. Ten of 11 positive scans had a clinically significant rise of CRP (>20 mg/L) providing it with a sensitivity of 90.91% (CI 58.72%–99.77%) with a positive likelihood ratio of 1.73 of having NEC when associated with a positive BUS. Approximately half of those with abnormal BUS (5/11) had thrombocytopaenia (<100 109/L) and 2/11 had a raised value of lactate (>4.0 mmol/L) within a period of 12 h before or after the scan. The combination of thrombocytopaenia and elevated CRP have a sensitivity of 62.5% (CI 24.49%–91.48%) and has a positive likelihood ratio of 1.56.
On analysis of bowel characteristics, all pathological scans demonstrated poor or absent peristalsis. Abnormal vascularity was present in 7/11. Interestingly only 54% (6/11) of the abnormal scans demonstrated pneumatosis indicating that pneumatosis, although pathognomonic, is a transient sign of NEC. 9
Discussion
Our study highlights the importance of assessment of suspected NEC using BUS in conjunction with plain radiographs and clinical assessment. Use of ultrasound, especially when used serially provide a more dynamic assessment of the bowel status with additional anatomical information like vasculature and wall thickening. 10 Despite increasing use of ultrasound, AXR has remained the first line of investigation due to its availability, familiarity and relative ease of interpretation. Lack of expertise, experience and training supervision limits the use of BUS in the clinical scenario. In a recently published survey conducted among neonatologists and radiologists in Sweden, 92% respondents used AXR as first line investigation and majority of the neonatologists also self-reported the AXRs. 11 Only 32% units reported that ‘some’ members used BUS routinely and 14% units reported that all clinicians in their team used BUS.
Our results highlight the previously published effectiveness of BUS in clinical suspicion of NEC. In >50% of the positive cases plain AXR failed to diagnose or confirm NEC. We found abnormal peristaltic and bowel perfusion patterns to be the most common pathological findings in scans. Complex fluid collections have been reported to be sensitive for identifying surgical NEC. 12 Five infants were accepted for surgical review based on scan findings and four underwent laparotomy for NEC. Two of these had complex fluid pockets in preceding BUS and underwent resection (Figure 3).
Strengths and limitations
This is the first pilot study on use of BUS for NEC reported from the UK. The strength of this study lies in the multi-disciplinary approach where formal supervision and mentorship was undertaken for quality assurance of the scans performed by the neonatologist. Although small numbers, our study highlights the importance of dynamic assessment of bowel and identification of features other than pneumatosis intestinalis. It also indicates that with development of expertise such information can aid in decision making around surgical referrals.
The obvious limitation to this study is that it is a single centre experience with only one neonatologist and one radiologist performing these scans, so there were several missed opportunities. One of the major challenges of implementing routine point of care BUS in clinical practice lies in lack of wider engagement from both neonatal and radiology teams. This is due to relative lack of training opportunities in BUS for a neonatologist as compared to other point of care organ scans like echocardiography or cranial ultrasound. There are very few international workshops and even fewer accredited courses available to acquire and update skills for BUS. The POCUS Working Group of the European Society of Paediatric and Neonatal Intensive Care (ESPNIC) published evidence-based guidelines for use of POCUS in 2020. 13 The expert group stated that ‘POCUS is helpful to detect signs of necrotizing enterocolitis (NEC) [although for a definitive diagnosis a detailed assessment should be performed by a paediatric radiologist or a person with specific advanced ultrasound training]—agreement (quality of evidence C). Ultrasound may be a useful adjunct detecting changes consistent with NEC even when radiographs are inconclusive’.
They also went on to classify ‘level of training’ required to perform organ specific POCUS and recommended ‘medium’ level training for BUS while detailed cardiological assessment was deemed as ‘advanced’. The essential corollary to these recommendations is the support of training of neonatologist in at least basics of ultrasound and support of a paediatric radiologist with advanced knowledge of ultrasound. The medicolegal aspects of neonatologist performing BUS in UK are also unclear. Analysis and review of legal aspects of neonatologist performed echocardiography suggest that a clinician performing these scans should be able to identify normal anatomical structures and further aim to maintain competence through continuous educational process. 14 A similar approach should be adopted in training neonatologist in performing BUS.
Conclusion
Use of bowel ultrasound to confirm diagnosis of suspected NEC was safe and efficacious in our experience. It aided us in making surgical referrals, in scenarios where AXR and the clinical presentations were equivocal or non-specific. With further training of more members and provision of supervision by radiologist, a wider implementation is possible.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
