Abstract
The use of corticosteroids in severe community-acquired pneumonia (sCAP) management is a contentious issue with current practices of United Kingdom (UK) intensivists largely unknown. To investigate this, we surveyed UK intensive care clinicians from 20 September 2024 to 19 December 2024, collecting 160 responses from 115 intensive care units (response rate 48.3%). 56.1% of responders use corticosteroids in the treatment of sCAP. There were large variabilities in practice. Hydrocortisone started within 24 h of admission 50 mg four times a day for 4–5 days was the most reported regime. The variation in practice coupled with relative equipoise requires further evaluation and guidance.
Introduction
Severe community-acquired pneumonia (sCAP) can be defined as pneumonia presenting within 48 h of hospital admission that requires intensive care unit (ICU) level care. 1 sCAP is associated with poor ICU outcomes with mortality exceeding 30%. 2 While some studies suggest potential benefit from the use of corticosteroids as an adjunct when managing patients with sCAP, there is a lack of consensus and current standards of practice are largely unknown. 3 Therefore, we designed a survey to explore practices regarding corticosteroid use for sCAP in ICUs across the UK.
Methods
The 11-question survey was developed by staff from General Intensive Care Unit (GICU), University Hospital Southampton (UHS) using a survey platform (Survey Monkey Inc., San Mateo, California). An internal pilot study was carried out prior to wider dissemination and the questions were refined based on the pilot feedback. The survey was emailed to all ICUs in the UK over a period of 3 months from 20 September 2024 to 19 December 2024.
Questions included the grade and ICU location of the respondent, whether they use steroids for sCAP treatment including: indications for use, drug choice, dose, timing of initiation, duration of therapy and method of cessation. The use of biochemical/imaging markers to guide steroid initiation and application of guidelines were also asked. Responses were taken as consent for the inclusion in data analysis and dissemination of the results. All data collection and storage were in line with UHS policy and the UK Data Protection Act (2018).
Results
The survey received 160 responses from 115 hospitals, representing 48.3% (115/238) of UK ICUs (Figure 1). 83.1% of respondents were consultants and 89.4% were based in general ICUs.
Map of unit responses across the United Kingdom. Each marker represents one intensive care unit that responded to the survey. Image created using Google Maps (2024).
56.1% of respondents use steroids in their management of sCAP. The most common indications for starting steroids were COVID-19 (82.9%), Pneumocystis jivorecii pneumonia (57.0%), bacterial aetiology (45.6%) Smaller proportions of respondents use steroids in their treatment of other viral aetiologies (26.6%), hospital-acquired (12.7%), ventilator-associated (10.4%) and aspiration-associated (7.6%) pneumonias.
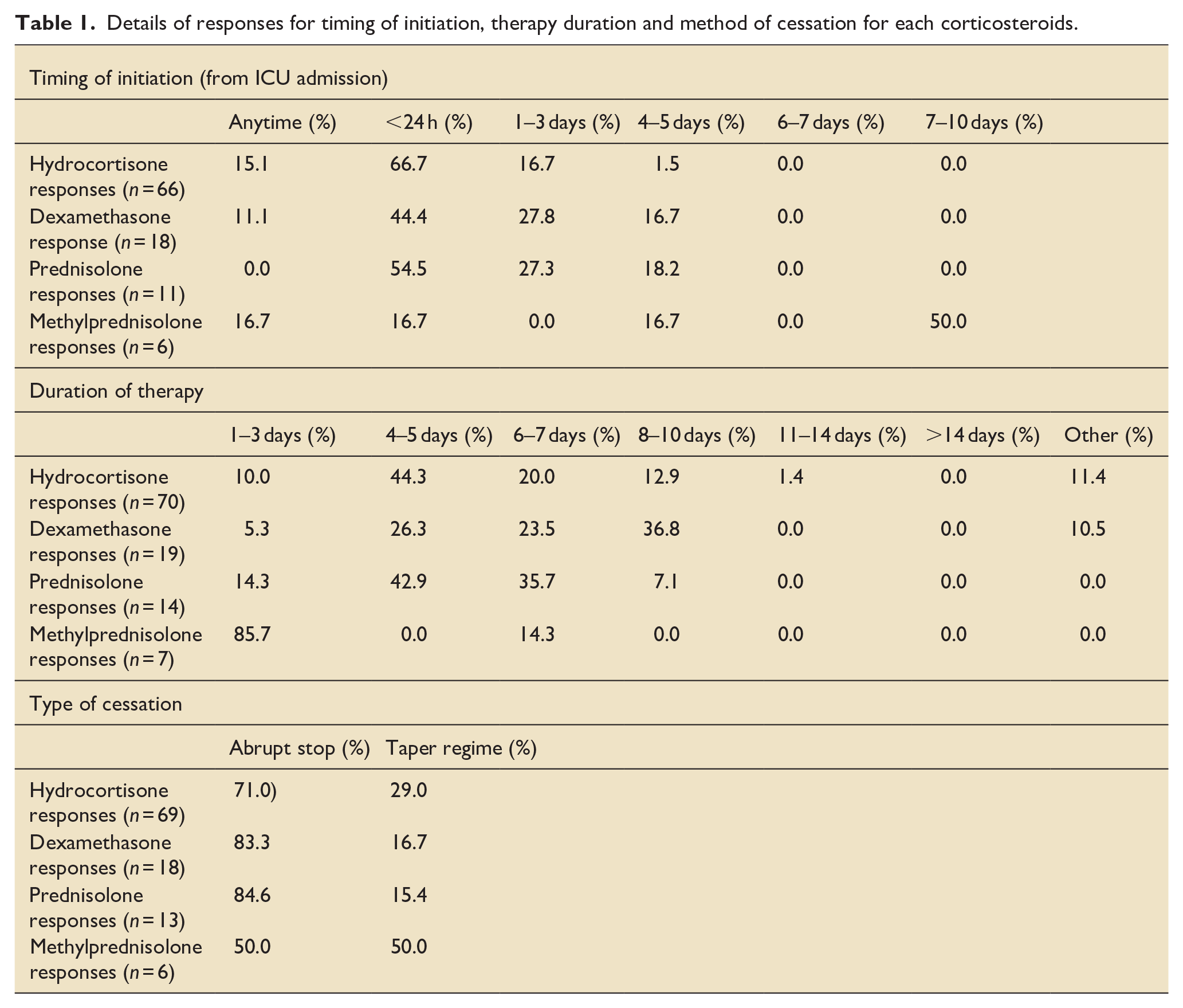
Hydrocortisone was the most common choice of corticosteroid (67.9%) followed by dexamethasone (14.3%) and prednisolone (10.7%). The most prevalent regime used was hydrocortisone 50 mg four times a day for 4–5 days starting within 24 h of ICU admission. An exception to early initiation was the use of methylprednisolone, where initiation at 7–10 days was favoured (50.0%). For all steroids except methylprednisolone, an abrupt stop was favoured over a tapering regime (Table 1).
Details of responses for timing of initiation, therapy duration and method of cessation for each corticosteroids.
The most common reason for considering steroids in sCAP was associated sepsis (71.4%). This was followed by acute respiratory distress syndrome (53.9%), biochemical hyperinflammatory state (high C-reactive protein (CRP), ferritin, cytokines, etc.) (45.5%), or a non-resolving pneumonia (38.3%). CRP and computerised tomography were the most frequent markers used to guide the initiation of steroids at 64.8% and 64.7% respectively. Other inflammatory markers such as procalcitonin and cytokine panels were used in 32.9% of responses. 53.4% of respondents report not using any guidelines. For those that use guidelines (46.6%), 11.1% use local, 12.5% use national and 6.8% use international guidelines.
Discussion
This survey found that 56.1% of UK intensivists use corticosteroids for the treatment of sCAP with large variabilities in the choice of corticosteroid regimes used. This is the first survey conducted among the intensive care physicians in the UK to evaluate current practices for the use of corticosteroids in sCAP. A previous survey, conducted in North America, reported 3% of 103 clinicians using corticosteroids ‘sometimes’ for sCAP, while most (81%) ‘almost never’ prescribe corticosteroids for sCAP. 4 Our results show a preference towards corticosteroid use which may reflect awareness of recent randomised control trial evidence. The two largest trials to date, CAPE COD (2023) and ESCAPe (2022), had differing outcomes.5,6 The CAPE COD study used hydrocortisone for 4–7 days finding a reduction in 28-day mortality. 5 In contrast, the ESCAPe trial used 40 mg methylprednisolone for 7 days and found no significant mortality benefits at day 60 or differences in secondary outcomes. 6 These differences in study outcomes are equally reflected in current guidelines. UK guidelines from the British Thoracic Society and the National Institute of Health and Care Excellence do not recommend the use of corticosteroids in sCAP.2,7 This is supported by the American Thoracic Society/Infectious disease society of America. 1 In contrast, other joint committee and Critical Care society guidelines support corticosteroid use in sCAP.8,9
Given the inconsistencies in current research and guidelines, it is therefore understandable that our results reflected variability in UK practice. Considering the limitations of survey research, particularly personal bias and the non-uniformity of UK responses, the nearly 50% response rate of this survey reflects an up to date assessment of UK practice.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251330145 – Supplemental material for Corticosteroids for the management of severe community-acquired pneumonia: A UK-wide survey
Supplemental material, sj-docx-1-inc-10.1177_17511437251330145 for Corticosteroids for the management of severe community-acquired pneumonia: A UK-wide survey by Isis Terrington, Olivia Cox, Elizabeth Webb, Benjamin Eastwood, Patrick Copley, Kordo Saeed, Andrew Conway Morris, Michael PW Grocott and Ahilanandan Dushianthan in Journal of the Intensive Care Society
Footnotes
Acknowledgements
We would like to thank all the study team members who helped to distribute the survey and those who participated in the survey. We would also like to thank all consultants at the University Hospital Southampton General Intensive Care who helped to with the internal validation pilot. We further extend our acknowledgment to Dr. Karthik Iyer, Dr. Mehraab Majeed, Dr. Preetam Upadhyaya and Dr. Nicholas Uren, for their help in distributing the survey.
Author contributions
Conception: IT, AD. Data Collection: IT, OC, BE, EW, PC. Data analysis: IT, BE, OC. Drafting: IT, OC, BE, EW, PC. Figures: IT, PC. Editing/review: AD, MG, KS, ACM. Supervision: AD.
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ACM reports speaking fees from Biomerieux, Thermo-Fisher, Fischer and Paykel and Boston Scientific, he sits on the scientific advisory board of Cambridge Infection Diagnostics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MG holds an NIHR Senior Investigator award and is in part funded by the NIHR Southampton Biomedical Research Centre. AD is supported by an NIHR EME grant. ACM is supported by a Clinician Scientist Fellowship from the Medical Research Council (MR/V006118/1) This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical considerations
Ethical approval was not required. By completing the survey, participants gave consent for their response to be included in the data analysis and subsequent dissemination.
Consent for publication
By completing the survey, participants gave consent for their response to be included in the data analysis and subsequent dissemination.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
