Abstract
Severe community-acquired pneumonia (sCAP) is associated with a significant health burden, both in the UK and globally, with intensive care support needed for many patients. The high morbidity and mortality associated with sCAP has led to the exploration of adjunctive therapies that may help reduce disease burden and improve clinical outcomes. One such proposed treatment is corticosteroids, aiming to moderate the disproportionate inflammation caused by sCAP. Despite several studies suggesting potential benefits, the use of corticosteroids in patients with sCAP remains contentious, with recent large trials producing conflicting results. These variations in trial outcomes have resulted in conflicting national and international guidelines. Such discrepancies align with findings from a recent national survey that indicated ongoing clinical uncertainty regarding the use of corticosteroids for sCAP in UK intensive care units. Several factors contribute to these conflicting outcomes, including patient population, the severity classification utilised, the type and duration of interventions provided, and, perhaps most importantly, the lack of pre-phenotyping to identify patients who may benefit most from the treatment. This narrative review aims to examine the recent literature, current guidelines, and evidence for using corticosteroids in sCAP, while exploring the candidate phenotypes of relevance in the design of clinical trials.
Introduction
Community acquired pneumonia (CAP) represents a significant health burden worldwide. While reported incidence varies between countries, in Europe, the annual incidence of adult CAP ranges between 1.07 and 1.2 per 1000 person-years, increasing to 14 per 1000 person-years in adults ⩾65 years age. 1 Moreover, respiratory tract infection is the most common infection site associated with sepsis and septic shock, accounting for around 60% of patients with sepsis requiring intensive care unit (ICU) treatment in the United Kingdom (UK). 2 According to the American Thoracic Society (ATS), severe community-acquired pneumonia (sCAP) is defined as pneumonia presenting within 48 h of hospital admission that requires ICU level care with major criteria including septic shock with vasopressor requirement or respiratory failure requiring mechanical ventilation. 3 sCAP is associated with poor ICU outcomes with mortality exceeding 30%. 4 Despite receiving appropriate antibiotics, respiratory support (invasive or non-invasive) ICU care, some patients with sCAP will continue to deteriorate requiring prolonged organ support measures and experiencing a range of adverse outcomes.
Whilst the pathological processes associated with sCAP are multifactorial and complex, the lack of adequate response to standard treatments in some patient groups has been hypothesised as resulting from an excessive host-mediated inflammatory response. 5 Corticosteroids, particularly those with glucocorticoid properties, exert their effect by modulating the inflammatory response through a variety of mechanisms and have consequently been evaluated in various acute and chronic inflammatory lung conditions for several decades.6,7 The RECOVERY platform trial showed that the use of dexamethasone reduced mortality by a third in ventilated patients with COVID-19, leading to widespread dexamethasone prescription as standard care in this patient group. 8 Although several trials have evaluated the role of corticosteroids in the management of non-COVID-19 sCAP patients, this remains a contentious area with recent large, randomised control trials (RCTs) producing contrasting results.9–11 National and international guidelines for sCAP management are also inconsistent, with some making conditional recommendations for adjunct corticosteroid use in shocked patients, whilst newer guidelines have a strong recommendation for corticosteroid administration to all sCAP patients.12,13 The UK guidelines do not recommend routine use of corticosteroids.4,14 A recent survey conducted amongst UK intensive care clinicians (n = 160) found that 51% routinely use corticosteroids in their management of sCAP patients, highlighting the ongoing concerns about safety and lack of consistent practice. 15 In summary, there is no established consensus for the use of corticosteroids in sCAP and inconsistency between guidelines. This narrative review of recent literature and guidelines seeks to provide more clarity on the role of corticosteroid prescriptions in bacterial and non-COVID-19 viral sCAP. We screened for relevant articles on PubMed, Ovid MEDLINE, Embase and Google Scholar using medical subject headings, keywords, and synonyms related to sCAP (e.g. pneumonia, community acquired pneumonia, CAP, sCAP), corticosteroids (e.g. glucocorticoid, corticosteroid, hydrocortisone, methylprednisolone, prednisolone), and intensive care (e.g. intensive care unit, critical care, critical illness, ICU) until April 2025. We particularly focussed on research published after 2022 which is when the first large trial was completed. We have not included studies which had COVID-19 as a primary focus.
What is severe community-acquired pneumonia?
While definitions vary, sCAP is a high severity lung parenchymal infection that is acquired outside of hospital setting, without recent history of inpatient hospital treatment, that requires ICU admission for organ support measures.3,4 The common causative organisms have classically been split into ‘typical’ and ‘atypical’ subtypes based upon clinical presentation. Typical pneumonia presents as pleuritic chest pain, fever and a productive cough. This is associated with a high leucocyte count, C-reactive protein (CRP) and homogenous opacities on chest radiograph (CXR). In contrast, atypical pneumonias may present with non-specific symptoms such as myalgia, fever and headache, without the associated significant rise in inflammatory markers. In these patients, CXR imaging may show diffuse patchy opacification or ground glass shadowing. 16 The most common cause worldwide is Streptococcus pneumoniae. 4 This is followed by Haemophilus influenzae, Staphylococcus aureus and Klebsiella spp. Atypical organisms include Legionella, Mycoplasma and Chlamydia spp, accounting for around 8% of pneumonia cases worldwide. 17 With a large variety of causative organisms, pneumonia microbiology is a spectrum of disease with varying susceptibility in different population subgroups. Risk factors for sCAP development include immunosuppression, old age, frailty, multiple co-morbidities, alcohol dependence and diabetes mellitus. 18
The pathophysiology of sCAP is a complex balance between local and systemic defence mechanisms which differ depending upon the causative organism. 19 Two common features to all lung parenchymal infection are an alveolar and systemic inflammatory response. Pathogens within the alveolar space create a localised inflammatory response characterised by a complex production of pro and anti-inflammatory cytokines, interleukins and acute phase proteins (Figure 1).20,21 The destruction of microorganisms is then coordinated via the inward migration of immune cells such as neutrophils, lymphocytes and alveolar macrophages, triggered by the cytokine response. Although beneficial in terms of localised control of infection, overt or disproportionate alveolar inflammation can cause airway collapse and pulmonary oedema. This can lead to significant ventilation-perfusion mismatch and acute hypoxaemic respiratory failure.22,23 In sCAP, this dysregulated hyperinflammatory response may also cause widespread systemic pathology such as sepsis and septic shock. 16 This combination of acute respiratory failure with/without systemic hypoperfusion syndrome forms part of the severity assessment for CAP, enabling identification and prediction of severe disease. 24
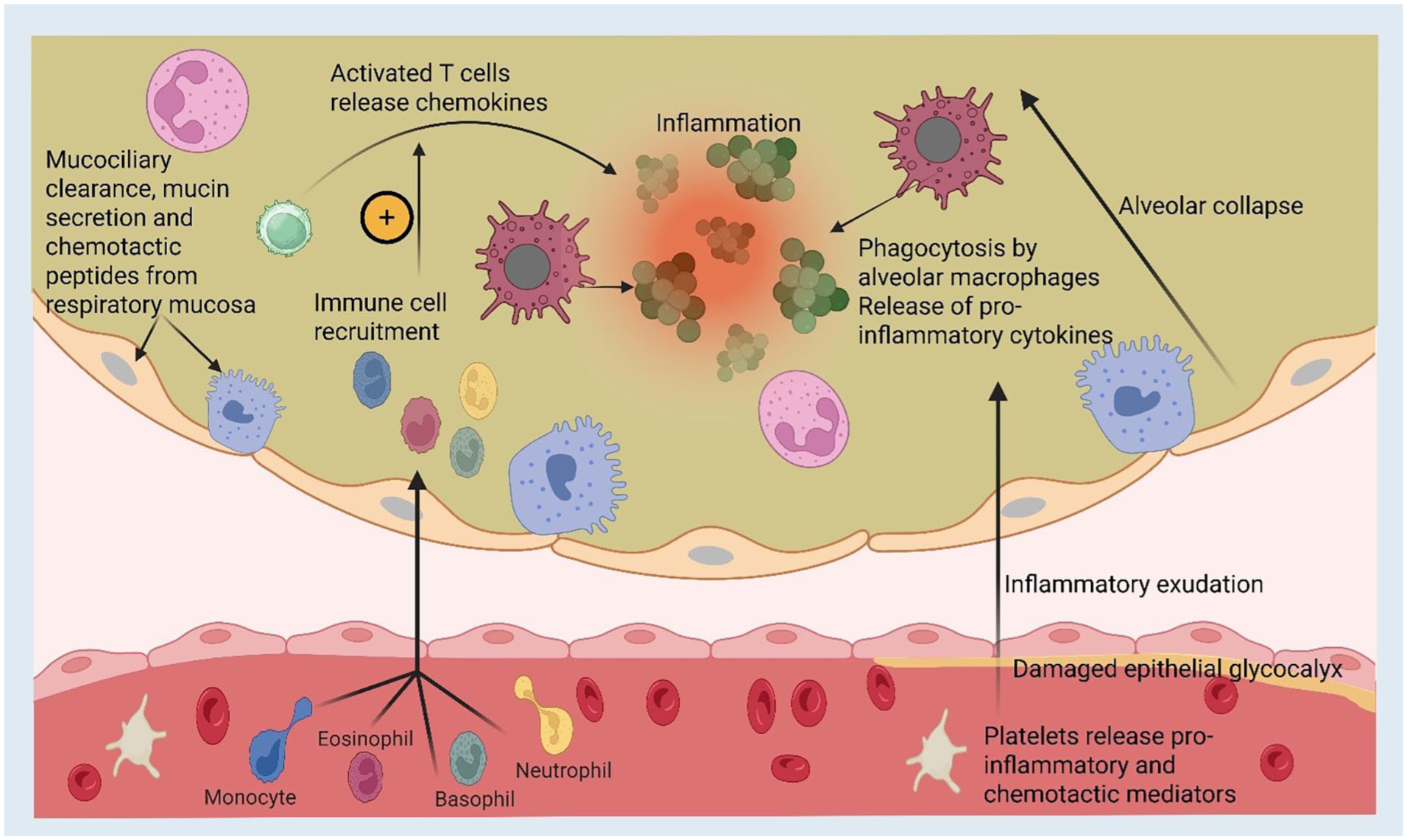
Diagram representing the acute inflammatory processes and the pathological consequences that occur in sCAP. Immune cell recruitment triggered by the inflammatory chemokines from respiratory mucosa leads to phagocytosis of pathogens and further pro-inflammatory states. Within the blood vessels, the epithelial glycocalyx may become damaged leading to increased exudation and alveolar oedema. If the infection is not controlled, this state acts as a positive feedback loop, worsening inflammatory and infective states.
Alongside this inflammation, there is evidence of lipopolysaccharide-mediated reduced leucocyte capacity to release Tumour necrosis factor-a (TNF-a), interleukin (IL)-1B and other pro-inflammatory cytokines. This immune suppression is implicated in the development of secondary complications such as sepsis, infection seeding and increased microbial pathogenesis leading to multiorgan failure. 23 An important consideration in the pathophysiology of sCAP is alterations to the native lung microbiome. Within healthy lungs there is a diverse ecosystem that dynamically adapts to environmental changes and maintains important interactions with respiratory mucosa. 25 In sCAP, this microbiome is dominated by the infective agent leading to reduced biodiversity in association with high microbial mass. Moreover, pathogens that cause pneumonia further thrive on mucosal and environmental changes associated with hyperinflammation, perpetuating their microbial dominance and limiting resolution. 26 When combined with critical-illness related immunosuppression, this may lead to emergence of secondary bacterial growth promotion with nosocomial infections, worsening illness severity. 27
Given the pro-inflammatory response and in some, dysregulated inflammation occurring in sCAP, corticosteroids have been proposed as having potential therapeutic benefit, particularly in patients with concurrent septic shock. 28
Mechanisms of corticosteroids in sCAP
The immunomodulatory properties of corticosteroids have long been assessed in acute inflammatory critically ill patients with sepsis, acute respiratory distress syndrome (ARDS) and pneumonia. 29 Of note, the pathophysiological rationale for corticosteroid response may be variable between patients and characterising of such phenotypes merits investigation. The mechanism of action of corticosteroids in severe pneumonia, although not fully defined, is a complex process involving pathogen virulence and host immune response with several self-defence mechanisms, including increased expression of pro-inflammatory cytokines (TNF)-α, IL -1β, IL-6 and IL-8), and anti-inflammatory cytokines (IL-10, IL-1 receptor antagonist (IL-1Ra)) alongside defence cell migration that provide vital protection against the pathogenic process. 30 Glucocorticoids act by inhibiting the action of many cytokines through binding to the cytoplasmic glucocorticoid receptor (GRα). Activation of GRα leads to multiple consequences such as transactivation (induced transcription of target genes), transrepression (inhibition of gene expression), and complex formation with pro-inflammatory transcription factors resulting in gene inhibition. This in turn leads to a reduction in products that are central to pro-inflammatory states. 31 Glucocorticoids can modulate endothelial nitric oxide synthase (eNOS) expression and vascular tone through several mechanisms. Studies have shown that corticosteroids downregulate eNOS expression through transcriptional and post-transcriptional effects leading to reduced nitric oxide (NO) production, improved vascular tone and vasoconstriction. Paradoxically, glucocorticoids can also increase eNOS activity by non-genomic pathways via receptor and messenger signalling through Phosphatidylinositol-3 kinase/Akt kinase causing contextual vasodilation during acute inflammation (Figure 2).32,33
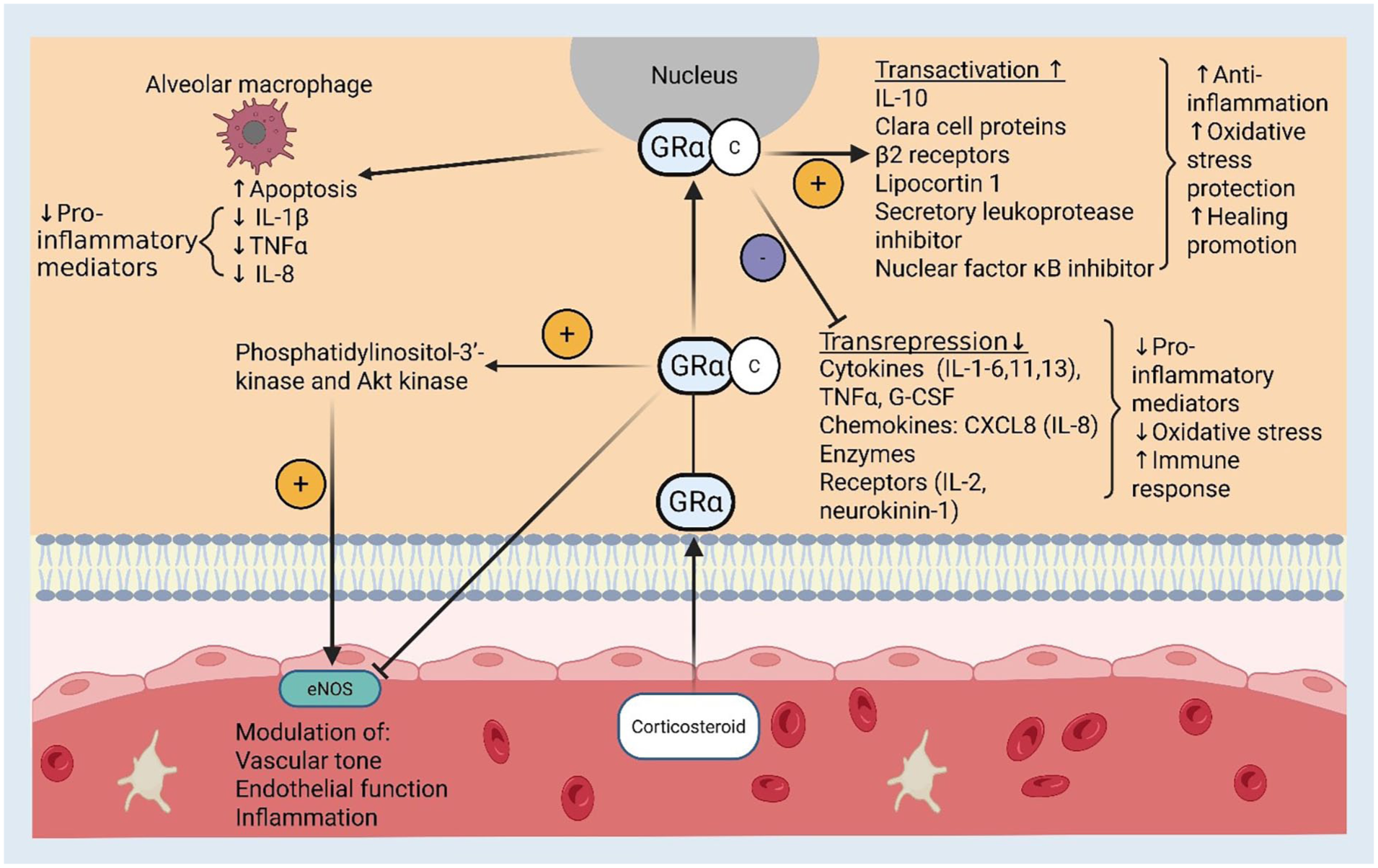
Proposed actions of corticosteroids in severe community acquired pneumonia pictured within respiratory alveolar epithelium. Delivery of the corticosteroid drug binds to the GRa receptor triggering various intracellular mechanisms (as pictured). Inhibition of alveolar macrophages, gene transactivation, gene transrepression and nitric oxide pathways cumulate in a dampened inflammatory response, reduced oxidative injury from free radical species and increased endothelial function.
Corticosteroid properties vary depending on the medication type (Table 1). High potency, long-acting glucocorticoids, such as methylprednisolone and dexamethasone, can lead to prolonged immunosuppression when compared to short acting, lower potency glucocorticoids such as hydrocortisone. 34 Although longer immunosuppression may be advantageous in the management of chronic conditions such as autoimmune diseases, in sCAP, short acting immunosuppression is likely to be preferred as it may allow for an acute reduction in hyperinflammation without excessive down-modulation of host immune response which could predispose to complications such as secondary opportunistic infections. 35 Hydrocortisone has additional mineralocorticoid properties when compared with dexamethasone and methylprednisolone, which have predominantly glucocorticoid actions. This may support the natural adrenal response to severe infection and assist with fluid and electrolyte shifts associated with a systemic stress response and critical illness.36,37
Pharmacological and physiological properties of common corticosteroids used in sCAP research.
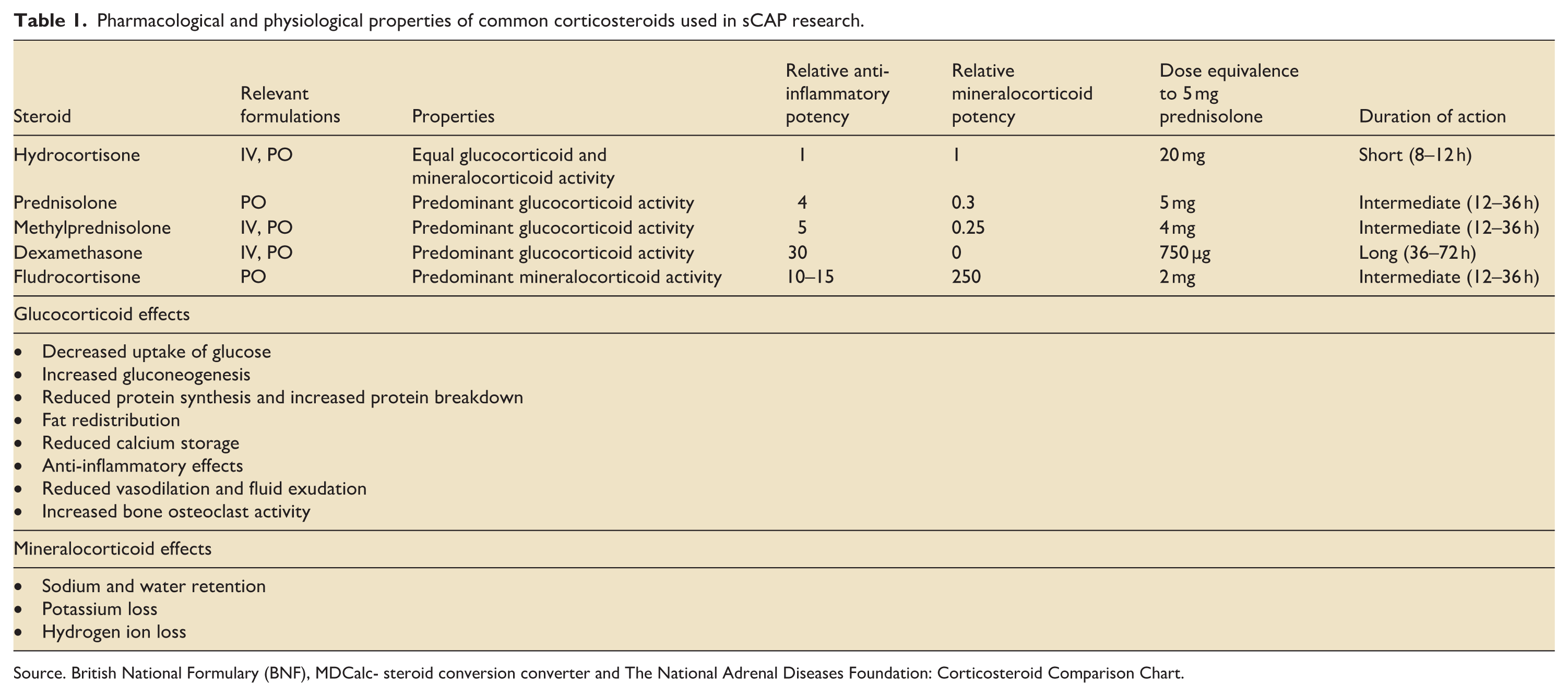
Source. British National Formulary (BNF), MDCalc- steroid conversion converter and The National Adrenal Diseases Foundation: Corticosteroid Comparison Chart.
What is the current evidence for corticosteroids in sCAP?
Studies assessing the role of corticosteroids in sCAP management have produced mixed results.6,35,38,39 A number of small studies published prior to 2020 suggested potential benefits of corticosteroids in sCAP.40–45 Recently, the three largest trials to date have produced differing results. The ESCAPe (Extended Steroid Use in Community-Acquired Pneumonia) trial (n = 584) found no difference in 60-day mortality between patients administered low-dose methylprednisolone versus placebo. Although the direction of signal was to benefit, the confidence interval overlapped equivalence meaning harm could not be excluded. In addition, there was no difference in secondary outcomes including in-hospital complications or ICU length of stay with low dose corticosteroid. 9 In this study, the methylprednisolone regime consisted of an intravenous bolus dose (40 mg) followed by a maintenance continuous infusion of 40 mg/day for 7 days tapered slowly (20 mg/day for 8–14 days, 12 mg/day for 15–17 days, and 4 mg/day on days 18–20) for a 20-day course. In contrast, the CAPE COD (Community-Acquired Pneumonia: Evaluation of Corticosteroids) trial (n = 795) found a 5.6% reduction in 28-day mortality in sCAP patients treated with an equivalent starting dose of hydrocortisone. 10 In this study, 200 mg hydrocortisone a day for 4 days was given before an adaptive weaning regime. Along with a reduction in all-cause mortality (6.2% intervention group vs 11.9% placebo) at day 28, they reported reduced ICU length of stay, mechanical ventilation and vasopressor requirements associated with corticosteroid use. However, the most recent multi-national REMAP-CAP multi-platform trial investigating the use of hydrocortisone 50 mg four times a day for 7 days in sCAP patients was stopped for futility due to a low probability that hydrocortisone improved 90-day mortality and possible harm unable to be excluded. 11 For all three studies, control group mortality was low (ESCAPe: 16% at day 60, CAPE COD: 11.9% at day 28, REMAP-CAP 9.8% at day 90) compared to previously reported population-level ICU data. In 2006, the UK Intensive Care National Audit & Research Centre (ICNARC) reported, over the course of a 9.5 year study period, an average ICU mortality of 34.9% and ultimate hospital mortality 49.4% for those admitted to ICU with CAP. 46 Moreover, the low mortality rate may reflect study inclusion/exclusion criteria. Some studies excluded patients with septic shock, where there may be a lack of equipoise for this intervention.9,10
One potential explanation for these differences in outcomes may be the timing of corticosteroid administration. In CAPE-COD, hydrocortisone was started within 24 h of hospital admission, whereas in ESCAPe, methylprednisolone was initiated within 72–96 h of admission and REMAP-CAP within 48 h of hospital admission. Previous evidence has suggested benefits for early corticosteroid administration in patients with sepsis or shock, however, this itself remains controversial. 38 Furthermore, there were differences in the characteristics of study participants. The ESCAPe trial was based in 42 military veteran hospitals across the United States leading to a 97% male study population with an average Sequential Organ Failure Assessment (SOFA) score of 6.6. In comparison, the CAPE COD population was 70.2% male with a SOFA score of 4.0, indicating a possible lower mortality risk and disease severity for those on the CAPE COD trial. As REMAP-CAP was a multi-centre international trial, the study population was comparative to the general adult ICU population.
Alongside these large trials, there have been several smaller studies assessing steroids in sCAP. There is a high level of variability in the choice, dose, duration and weaning of corticosteroids used in these trials with limiting comparability. Recent systematic reviews and meta-analysis have conflicting outcomes.6,35,38,39 See et al. found 30% lower all-cause mortality, 50% rate reduction of mechanical ventilation and 60% lower rates of shock associated with corticosteroid use. Among the corticosteroids, only hydrocortisone had significant impact. 35 These outcomes were supported three other systematic reviews who also found mortality benefit.47–49 In contrast, Briel et al. analysed individual patient data finding no mortality benefit and increased risk of CAP-related rehospitalisation. 50 Saleem et al. also found no mortality benefit however corticosteroid use did lower incidence of progression to mechanical ventilation requirements. 51 The most recent systematic review included 30 RCTs with 7519 patients. They concluded that corticosteroids probably reduce short-term mortality, need for invasive mechanical ventilation and length of ICU stay. This review included ARDS and Sepsis trial subgroups as well as a range of pneumonia severity, with marked variation in the definition of ‘severe’ CAP. Within the chosen studies, only 51% were admitted to ICU and there was significant variability in the reported mortalities of study populations from <1% to 58%. 52
Recent data from the APROCCHSS phase 3 trial (Hydrocortisone plus Fludrocortisone for Adults with Septic Shock) subgroup analysis of CAP related septic shock also found a 12% of absolute risk reduction in 90 day mortality compared to placebo in patients given a combination of hydrocortisone (50 mg four times a day) and fludrocortisone (50 µg once a day) for 7 days. 53 However, an individual patient level meta-analysis for the use of hydrocortisone in septic shock including 7 large trials with 1701 patients with pulmonary infection, suggested no 90-day mortality benefits with hydrocortisone. 54 In summary, the role of corticosteroids in sCAP management is unclear, although use of hydrocortisone, particularly in those with shock, seems to suggest potential benefits and has been implemented into ICU guidelines aligned with sepsis guidelines detailed below. 13
What do the current guidelines say?
Variations in the interpretation of this complicated evidence base has led to disparities in current national/international guidelines (Table 2). The 2024 Society of Critical Care Medicine (SCCM) guideline update strongly recommends for the use of corticosteroids for hospitalised patients with sCAP. This was based on pooled analysis of 10 randomised control trials with sCAP subgroups, in which corticosteroids reduced mortality, requirements for invasive mechanical ventilation and length of stay (ICU and hospital). 13 This view is partly supported by the joint European Respiratory Society, European Society of Intensive Care Medicine, European Society of Clinical Microbiology and Infectious Diseases, and Latin American Thoracic Association (ERS/ESICM/ESCMID/ALAT) guideline provides a conditional recommendation for corticosteroid use in patients sCAP and concurrent shock. 12 In comparison, the UK guidelines from the British Thoracic Society (BTS) and the National Institute of Health and Care Excellence (NICE) do not recommend the use of corticosteroids.4,14 However, it should be noted that BTS guideline is likely outdated as it was published in 2008 and updated in 2015.14,55 The 2019 Infectious Diseases Society of America (IDSA)/ATS consensus guidelines also advise against the routine use of corticosteroids in adults with severe CAP or influenza pneumonia, based on limited evidence and the risk of corticosteroid therapy. 3 It is possible that as large RCT data emerges, updated guidelines may produce a more uniform or consensus view. This disparity also highlights the importance to move towards a unified approach to align different international guidelines, with the current situation confusing and unhelpful to ICU clinicians.
Current national and international guideline suggestions for the use of corticosteroids in the management of sCAP.
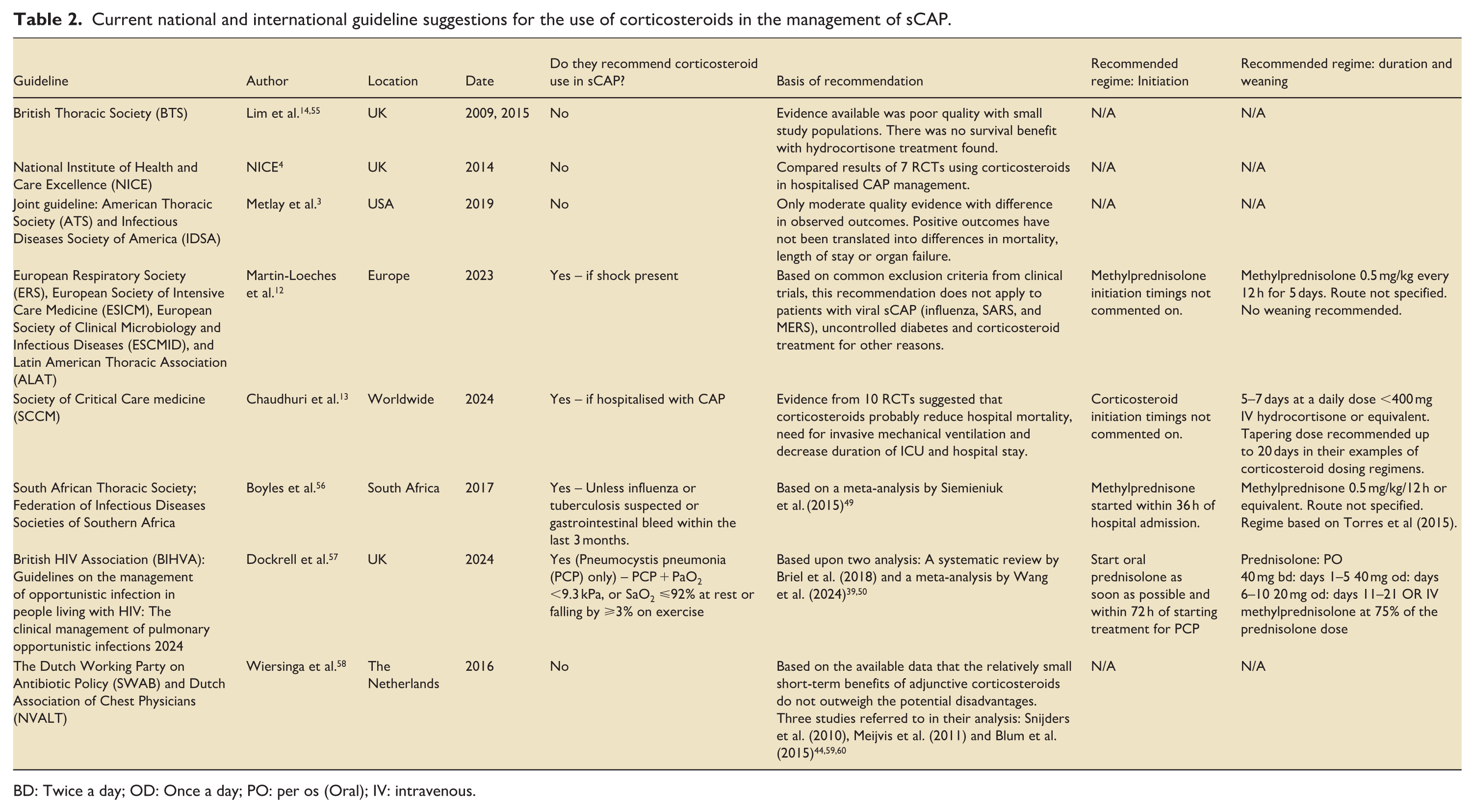
BD: Twice a day; OD: Once a day; PO: per os (Oral); IV: intravenous.
What is the current practice?
In 2024, a UK-wide survey amongst intensive care physicians (n = 160) found only 51% of the respondents use corticosteroids routinely as part of in their management of sCAP patients. 15 The most commonly used regime was hydrocortisone 50 mg intravenously four times a day for 4–5 days started within 24 h of hospital admission and stopped without a weaning regime. Over half (53.4%) did not use any guidelines to inform decisions about corticosteroids administration in sCAP. A survey conducted in North America in 2016 related to the study centres in the OSCILLATE (High-Frequency Oscillation in Early Acute Respiratory Distress Syndrome) trial, reported only 3% of 103 clinicians using corticosteroids “sometimes” for sCAP, while most (81%) ‘almost never’ prescribe corticosteroids for sCAP. 61 With emerging evidence from COVID-19 and recent trials suggesting potential benefits, it is likely that clinical practice has evolved since this 2016 survey was conducted. However, the results of both these studies reflect the inconsistencies in current evidence and show that clinician preference remains one of the key factors for decisions around the use of corticosteroids in sCAP.
The limitations of current evidence
Variations in categorising sCAP patients
The categorisation of CAP and sCAP is also variable between studies with different risk stratification tools used in trials (Table 3). By using different risk stratification tools, studies may increase or decrease the sCAP severity estimates leading to potential differences in clinical outcomes including mortality, therefore reducing the validity of comparisons between studies and conclusions drawn from pooled data.
Different sCAP scoring systems used clinically and in research.

Variability of corticosteroid dose and duration
The variability of corticosteroid dose used in clinical trials is also challenging in this context. Regimes suggested by current national/international guidelines allow for differences in drug choice and dosage (Table 1). These mostly vary between the use of methylprednisolone and hydrocortisone at a dose of 175–400 mg hydrocortisone equivalence a day (based on a 70 kg patient). Without consistency in regime, it is challenging to ascertain whether different study outcomes were produced by the difference in steroid choice/dosage, or other factors. 71 Further, the lack of personalisation in current recommendation is a limitation: the current surviving sepsis ‘one size fits all’ in the hydrocortisone regimes means that a 120 kg younger man with augmented drug clearance receives same dose as a 50 kg, frail older woman with much more limited drug clearance. This strategy takes no account of altered pharmacokinetics and pharmacodynamics in intensive care. 28
Heterogeneity of study populations
They are large amounts of variation seen between study population characteristics. As previously mentioned, there was a significant male predominance seen in the ESCAPe trial compared to that of CAPE COD and possible differences in illness severity between the two studies.9,10 A larger proportion of the ESCAPe trial participants had diabetes mellitus (48.3% compared to 22.8%) which may account for the increased rate of hyperglycaemic adverse events, even with consideration of the higher total glucocorticoid dose (ESCAPe: 468 mg total, CAPE-COD: 220 mg total) administered. It is also unclear how the adaptive weaning regime used in CAPE-COD impacted study outcome as shorter corticosteroid administration may have limited excessive immunosuppression. It is noted that studies assessing sCAP in older populations report higher mortality rates compared to those with younger cohorts. 72 This is supported by evidence that a high frailty score and poor functional status at baseline are strong predictors of sCAP mortality. 73
Lack of defined immunological phenotypes and stratification of suitable candidates
Beyond the pharmacokinetic variability, and differences in study population inclusion outlined above, existing studies of corticosteroids have not adequately addressed the issue of heterogeneity in immune responses. The understanding that sepsis is not a single disease process has developed over the past 30 years, in part as a consequence of repeated failures of candidate immunomodulatory therapies. 74 However, even when restricted to specific syndromes such as pneumonia, or even sub-syndromes such as ‘community-acquired pneumonia’, the range of immune responses varies. This arises from three key factors: (1) the infecting pathogen – where different organisms, and classes of organism such as respiratory viruses versus gram-positive bacteria evoke distinct patterns of immune response; (2) the intrinsic host response – where inherited and acquired factors, such as comorbid disease, acquired somatic mutations and intercurrent drug therapy interact with the pathogen to produce distinct patterns of inflammation and (3) the interaction between body compartments, where tissue-dependent inflammatory patterns combine with organ cross-talk and extra-pulmonary organ failures to influence the whole organism response.75–77 To add further complexity, immune responses are not simply ‘hyperinflammatory’ or ‘hypoinflammatory’, but may include features of both immune hyperactivation and impaired antimicrobial functions, as pro-inflammatory molecules may drive an apparently paradoxical dysfunction. 78 Care must also be taken to distinguish ‘impaired immune states’ from ‘immunoquiescence’ with the latter being closer to a homeostatic set point developing during disease resolution. Secondary infections developing whilst in ICU can further disrupt these processes.
As it may be imagined from the above description, the complexity of immunological phenotypes is hard to unravel. Existing attempts have tended to focus on blood, as a readily accessible biological sample, ignoring the distinct profiles available in the lungs, and have often focussed on a limited range of biological markers. This reductionist approach may have utility and could certainly lead to more readily useable bedside stratification tools. A recent individual patient meta-analysis of steroid studies in CAP confirmed the heterogeneity of responses and identified elevated C-reactive protein as a predictor of response that appeared to be independent of severity of illness. 79 Conversely, viral infection without bacterial co-infection predicted increased mortality when treated with steroids. These relatively ‘simple’ approaches need testing in clinical trials, whilst at the same time efforts continue to understand the more complex reality, seeking to identify novel approaches to immunomodulation in pneumonia (Figure 3) .
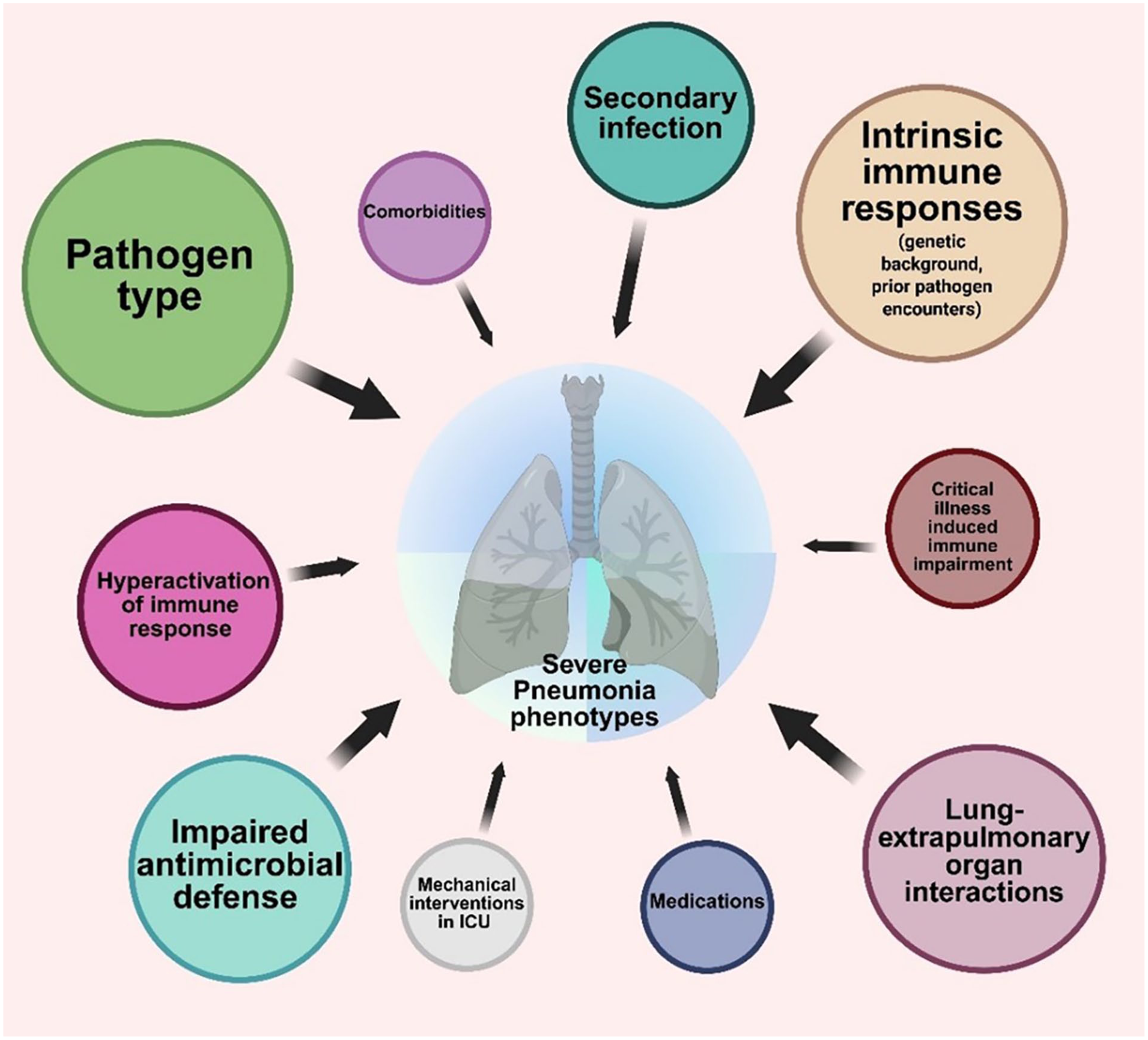
A pictorial representation of major factors that influence the inflammatory responses to pneumonia. These combine to form distinct phenotypes with common molecular mechanisms, which are likely to respond differentially to immunomodulatory interventions such as corticosteroids. Tools to profile these molecular mechanisms are in development and promise a personalised approach to future therapeutic intervention.
The efficacy of different corticosteroids
It is assumed that patients who become critically ill and need organ support measures in the ICU are often affected by dysregulated host immune response when exposed to the infection. The established paradigm also suggests corticosteroids help moderate this heightened immune response in patients with sCAP. However, it is puzzling why commonly used corticosteroids, such as dexamethasone, which possess more potent anti-inflammatory properties than hydrocortisone, have failed to demonstrate any significant survival benefits, even when given intravenously. 9 A recent systematic review concluded that hydrocortisone in the dose range of 200 mg (equivalent to the shock dose) is the only corticosteroid shown to effectively reduce mortality. 35 Interestingly, hydrocortisone’s immunomodulatory properties are relatively weaker than those of dexamethasone, methylprednisolone, or prednisolone. This raises the question: What is the explanation for the potential effectiveness of hydrocortisone demonstrated by CAPE COD trial compared to other steroids when treating sCAP? One possibility is that hydrocortisone provides modest immunosuppression, which does not drastically alter the host’s immune response during an acute severe infection. Another possibility is that its combined glucocorticoid and mineralocorticoid activity may enhance shock reversal and decrease the need for vasopressor support, similar to what is observed in patients with septic shock. However, the possible benefits from hydrocortisone were challenged by the recent REMAP-CAP trial. Further research is required to evaluate the specific mechanisms that underpin the potential beneficial effects of corticosteroids as adjunctive therapy in sCAP. Additionally, studies should focus on a detailed pharmacokinetic and pharmacodynamic (PKPD) analysis to facilitate translational research and to provide personalised corticosteroid therapy.
Adverse events associated with corticosteroids
Despite their potential benefits, there are well known risks and adverse events associated with corticosteroids. Commonly reported side effects include altered glycaemic control, psychiatric reactions, proximal myopathy and osteoporosis, although these can vary with the chronicity of steroid treatment, along with the dose used. 80 In the short term, concerns regarding corticosteroid related immunosuppression and the potential of worsening infection may limit clinicians’ willingness to prescribe corticosteroids. However, in recent years a number of meta-analyses assessing the risks side effects of corticosteroids in pneumonia have not reported significant increase in secondary infections.48,81,82 Similarly, these reviews did not find evidence to suggest causal links between the use of corticosteroids and upper GI haemorrhage, acute kidney injury and neuromuscular weakness.48,81,82 Evidence was more complex with regards to neuropsychiatric disturbance, with some studies indicating associations with methylprednisolone, but not with other steroids, and others not finding significant relationships at all; thus demonstrating the challenges associated with drawing general conclusions regarding adverse reactions to corticosteroids without retaining an understanding of the diversity of different drugs, doses and course integrated under this treatment type.81,82 In addition, there may be further harms associated with high-dose steroids or the use of steroid pulsing which include, for example, alveolar haemorrhage and autoimmune crises, although further analyses is needed in this area. 82
A unifying significant side effect of corticosteroids reported by these analyses was hyperglycaemia, echoed in a recent Cochrane review which found high frequencies of hyperglycaemia adverse events in the groups treated with corticosteroids. 83 Chaudhuri et al. showed a dose-response relationship whereby higher doses of corticosteroids led to increasing risk of hyperglycaemia. 82 This is significant for patients in intensive care, as glycaemic control in these patients is already challenging. In patients admitted to ICU with sepsis, those with the high blood glucose and low glucose variability have the highest all-cause mortality compared to other ICU subgroups. 84 In different patient groups, it is important to highlight that corticosteroid use may be associated with different risks and adverse events, for example amongst diabetic patients, elderly patients and patients with immunosuppression. Given the high prevalence of diabetes in ICU admission populations, these adverse events are an important limitation.
In the longer term, protracted administration of corticosteroids has been associated with the downregulation of cell-mediated immunity, glaucoma, osteoporosis, osteonecrosis and myopathy (amongst others). 84 Current research primarily focuses on short-term follow-up periods, highlighting the need for further study on long-term complications of corticosteroid treatment.
Future research directions and implications for current practice
Given the limitations of current evidence, further robust trials are required to provide more insight into the potential role of corticosteroids in sCAP. To aid this, research into patient factors and phenotypes that lead to sCAP development and treatment failures should also be considered. Variants in the genes involved in corticosteroid signalling or cytokine regulation may underlie inter-individual differences in immune response. Therefore, integrating genomic and inflammatory profiling may enable more precise stratification of patients most likely to benefit from adjunctive corticosteroids. By understanding which patients are at higher risk of sCAP and it’s complications, and develop phenotype classifications, we may gain greater understanding of pneumonia as a general syndrome, characteristics of specific phenotype sub-syndromes, and of the relationship with hyperinflammatory phenotypes associated with sCAP. This could allow for the development of individualised sCAP management regimes. This may include specific criteria for treatment, corticosteroid drug choice and doses, as well as combination of other immune pathway targeting therapies. This was evidenced during COVID-19 with the co-administration of various immunomodulators such as IL-6 antagonists like tocilizumab and Sarilumab. 85 As critical illness can significantly alter drug pharmacokinetics and pharmacodynamics, further research is required to optimise corticosteroid regimes including personalised dosing, administration and weaning. 71 Future trials should also consider further assessment of patient-centred outcomes and long-term monitoring. For example, post-hospital discharge physiotherapy needs, mental health support and return to work timing. Further understanding would support the recognition of the long-term impacts of critical illness and recovery.
Conclusions
Current evidence suggests that corticosteroids, particularly hydrocortisone, may have a potential role in the management of sCAP. However, recent trials have produced conflicting results, and the guidelines differ in their recommendations. Some guidelines endorse their use, while others provide conditional recommendation or advise against it. Given these discrepancies, current practice is formed by individual opinions and personal preferences. Further research is required to allow for more robust conclusions and guidance.
Footnotes
ORCID iDs
Ethical considerations
Ethical approval was not required.
Author contributions
Conception: IT, AD. Information gathering: IT, OC, BE, EW, PC. Analysis of evidence: IT, AD, OC, CM, BE, EW, PC, ACM. Drafting: IT, OC, BE, EW, PC, ACM, MG, CM, KS. Figures: IT, PC, ACM. Editing/review: IT, AD, MG, KS, ACM, CM. Supervision: AD.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MG holds an NIHR Senior Investigator award and is in part funded by the NIHR Southampton Biomedical Research Centre. AD is supported by an NIHR EME grant. ACM is supported by a Clinician Scientist Fellowship from the Medical Research Council (MR/V006118/1). CM is supported by a NIHR Senior Clinical Practitioner Research Award (NIHR 304615). This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ACM reports speaking fees from Biomerieux, Thermo-Fisher, Fischer and Paykel and Boston Scientific, he sits on the scientific advisory board of Cambridge Infection Diagnostics. CM reports an honorarium for her role as Editor in Chief of Critical Illness (![]() ) and speaking fees from Sedana Medical and Biomerieux.
) and speaking fees from Sedana Medical and Biomerieux.
