Abstract
Aim:
Levetiracetam is a widely used anti-epileptic in the critical care setting that is almost exclusively (>90%) renally excreted. The altered pharmacokinetics of levetiracetam have been widely studied in intermittent haemodialysis but the evidence and guidance on dosage in continuous renal replacement therapy is varied and poorly defined. Understanding this is critical as a significant number of critically unwell patients develop renal failure requiring continuous renal replacement therapy. The aim of this systematic review is to investigate the pharmacokinetics of levetiracetam in such patients and to understand the implications on dosing strategies.
Methods:
A systematic review of the available literature from 2000 to November 2022 was conducted. Seven articles were identified for inclusion from 54 records. A novel hybrid model was developed to evaluate the quality of pharmacokinetic and haemofiltration data. This data was used to develop a one-compartment model that simulated dosing strategies in 10,000 patients based on an assumed steady state of 72 hr and target trough concentrations of 12–46 mcg/mL.
Results:
From the seven articles included, pharmacokinetic data was retrieved for 24 individual patients. Total clearance was 3.49–4.63 L/hr (mean 3.55, S.D. 0.52). Elimination half-life was 5.66–12.88 hr (mean 9.41, S.D. 2.86). Volume of distribution was 0.45–0.73 L/kg. The proportion of total clearance attributable to continuous renal replacement therapy was 52%–73% (mean 54.7%, S.D. 13.5). Our simulations demonstrate that more than half of patients who received twice daily doses of 750 mg or greater without a loading dose achieved therapeutic drug concentrations. The time to achievement of therapeutic drug concentrations was greatly reduced by the addition of a 60 mg/kg loading dose (up to a maximum of 4.5 g). The use of a reduced loading dose or twice daily doses of 500 mg or less without loading were more likely to result in prolonged sub-therapeutic drug concentrations.
Conclusion:
Levetiracetam clearance in haemofiltration is similar to healthy adults with normal renal function (GFR > 90 mL/min). The use of reduced doses due to renal failure in critically ill patients may result in sub-therapeutic drug concentrations in a high number of patients. A twice daily dosing of 750–1000 mg with an initial loading dose of 60 mg/kg should be considered in such patients alongside therapeutic drug monitoring.
Keywords
Introduction
Over the past decade, the use of levetiracetam in critical care settings both in the treatment and prevention of various seizure disorders has significantly increased.1,2 This increase is owed in large part to several studies that have showed it to be a safe, broad-spectrum and highly effective anti-epileptic drug with minimal drug interactions and a wide therapeutic index.3 –5 Therapeutic drug monitoring for levetiracetam is not typically recommended in clinical practice unless there is suspicion of toxicity and as such there is no agreed target range. However, a large scale dose-ranging study conducted among epileptic patients identified a desired therapeutic target of 12–46 mcg/mL at trough level. 6
Levetiracetam is a small, hydrophilic molecule that is weakly bound to proteins (<10%) with linear kinetics.5,7 –9 In a healthy adult (GFR > 90 mL/min), the total clearance of levetiracetam is approximately 4.03 L/hr, half-life is 6–8 hr and volume of distribution is 0.5–0.7 L/kg. 10 It is almost exclusively (>90%) excreted via the kidneys and so dose adjustments are recommended in patients with impaired renal function.6,8,11,12 Even mild-to-moderate renal impairment has been shown to double plasma concentration and significantly increase the half-life. 13
Renal impairment is common in critical care settings with around half of people admitted to intensive care developing acute kidney injury.14,15 Continuous renal replacement therapy (CRRT) is used in severe acute kidney injury to support renal function and minimise the risk of multi-organ failure and fluid overload. Additionally, people with end-stage renal disease who require intermittent haemodialysis often benefit from a temporary transition to continuous renal replacement therapy in the event of critical illness due to the enhanced haemodynamic and metabolic control it provides. The introduction of renal replacement therapy creates challenges for pharmacological management as pharmacokinetics are altered through both extrinsic (e.g. filtration mode, filter type, exchange rate) and intrinsic factors (e.g. residual renal function, fluid volume status, protein binding). For medications that are renally excreted, such as levetiracetam, the effects on drug clearance are significant. 16
Intermittent haemodialysis (IHD) has already been shown to eliminate 50% of levetiracetam within 4 hr. 13 As a result, the current dosing recommendation for patients receiving IHD is a 750 mg loading dose followed by 500–1000 mg once daily dosing. 11 However, there are no formalised dosing recommendations for patients on CRRT. Indeed, in the UK the product licence provides no dosing guidance outside of IHD. Instead, a range of resources based on limited evidence and expert opinion are used in clinical practice.17 –21 Until recently, the evidence base that underpinned dosing suggestions was limited to three case reports alone.22 –25 Suggestions range from 250 to 750 mg BD dosing without a loading dose, to 750–1250 mg BD with or without a loading dose, to no adjustments to normal dosing at all. In the last few years, there has been a renewed focus on broadening the evidence base using pharmacokinetic studies in this patient population.26 –28 Studies by Kalaria et al. 29 and Chaijamorn et al. 25 have used such data in an attempt to elucidate a safe and effective dose in patients on CRRT.
In this study, we evaluate the latest available evidence on the pharmacokinetics of levetiracetam in critically ill patients undergoing CRRT. We undertake a hybrid study that combines a systematic review and quality assessment of the latest evidence in addition to computational modelling to simulate the effects of different dosing strategies on plasma concentration. This hybrid approach was adopted to overcome the paucity of data on levetiracetam pharmacokinetics in this understudied population, bringing together the disparate data into a single cohesive review to enable a more robust population analysis than a single, stand-alone study. Our computational model is therefore based on a larger sample size than previous pharmacokinetic models and was used to simulate the predicted plasma concentrations of different dosing strategies to determine the optimum regimen for patients on CRRT. This is in direct response to several case reports and case studies that suggest the clearance of levetiracetam in vivo may be significantly higher than previously thought for patients on CRRT – raising the risk of sub-therapeutic plasma concentrations in such patients.22,28,30,31
Methods
Data source and searches
The protocol for this systematic review was developed using the PRISMA-P guideline and was registered on PROSPERO (CRD42022300754). Our search strategy is available in Supplemental Appendix 1.
A literature search was conducted in EMBASE, MEDLINE, PubMed and Google Scholar from 2000 to 01 November 2022. The reference lists of selected articles were also checked to identify additional relevant studies for inclusion. An independent search was performed by two authors with a third investigator to resolve any discrepancies.
Study selection
All case reports, cohort studies and randomised control trials were included and screened to assess eligibility for inclusion.
A study was eligible for inclusion if it met the following criteria: (1) patients were treated with levetiracetam (regardless of its formulation); (2) patients were undergoing CRRT via continuous veno-venous haemofiltration (CVVHF), continuous veno-venous haemodialysis (CVVHD) or continuous veno-venous haemodiafiltration (CVVHDF). Studies were excluded for the following reasons: (1) it was published in a non-English language; (2) the study was a computational or model-based study not using human patient data; (3) there were insufficient pharmacokinetic data; (4) review article with no additional new pharmacokinetic data.
Data extraction and quality assessment
Data were independently extracted from the included studies by two authors (J.S., S.A-M.) using a standardised database. Data extracted included: study design, patient demographics, reason for admission, indications for levetiracetam and CRRT, residual renal function, dosing regimen, CRRT characteristics and settings, and pharmacokinetic parameters (e.g. half-life, clearance). Any discrepancies on data extraction were arbitrated by a third investigator.
Our quality assessment was performed using a hybrid model that combined the Quality of Evidence (QoE) and Acute Dialysis Quality Initiative (ADQI) scoring systems.32 –34 Each study was assessed independently by two authors (J.S., S.A-M.) who allocated separate QoE and ADQI scores as per the protocol detailed in Supplemental Appendix 2. To eliminate discrepancies in ADQI scores between authors, a mean ADQI score was calculated for each study. Overall quality assessment scores were stratified into low, medium and high-quality equivalents based on the average quality of evidence across the QoE and ADQI assessments. For example, a study was ranked medium quality if it achieved a strong QoE score but a weak ADQI score. Any disputes were resolved by a third investigator.
Data synthesis and simulation of dosing scenarios
Data were collated using Microsoft Excel. Our primary analysis was a mixed narrative and statistical analysis. Descriptive analytics and multiple regression modelling were performed using IBM SPSS Statistics 28. In cases where specific values were not reported (e.g. ultrafiltration rate, filtration fraction, volume of distribution) these were calculated using the available data and standard formulae where possible. Statistical heterogeneity was not assessed as the Cochrane Q Test and I2 statistic are not applicable to our dependent variable which is continuous rather than dichotomous.
Simulations were undertaken in R using tidyverse and linpk packages.35 –37 Elimination rate constant and volume of distribution was derived directly from the patient level data in the included studies. Volume of distribution was not available for 8 patients (Chappell et al.) and was instead calculated from the elimination rate constant and clearance rate using standard pharmacokinetic equations. Simulated patients (n = 10,000) were then created from these values using the MASS package and assuming a multivariate log-normal distribution of parameters. 38 For pharmacokinetic profiles, a one compartment model was assumed as this has been demonstrated in previous population pharmacokinetic studies as the most accurate description of in vivo pharmacokinetics.39 –42 Profiles were simulated to 72-hr, on the assumption that steady state would be achieved by then and that this early period of treatment was likely to be the most important in achieving therapeutic concentrations. We utilised a target trough concentration range of 12–46 mcg/mL as this is the most widely used therapeutic index.6,22,43,44
Results
Our search found a total of 58 articles (Figure 1) of which four were unable to be reviewed in full text. Of 54 articles reviewed in full text, seven fulfilled our eligibility criteria for inclusion. Of the included studies, five were case reports22–24,43,45 and two were prospective pharmacokinetic studies.29,44 In all studies except one (where clinical indication was not recorded) patients were treated with levetiracetam either for known seizures or as prophylaxis (e.g. following traumatic brain injury). In one study, 44 four patients were treated with SLED and so were excluded from our data extraction. The total number of patient cases extracted from the included studies was 24.
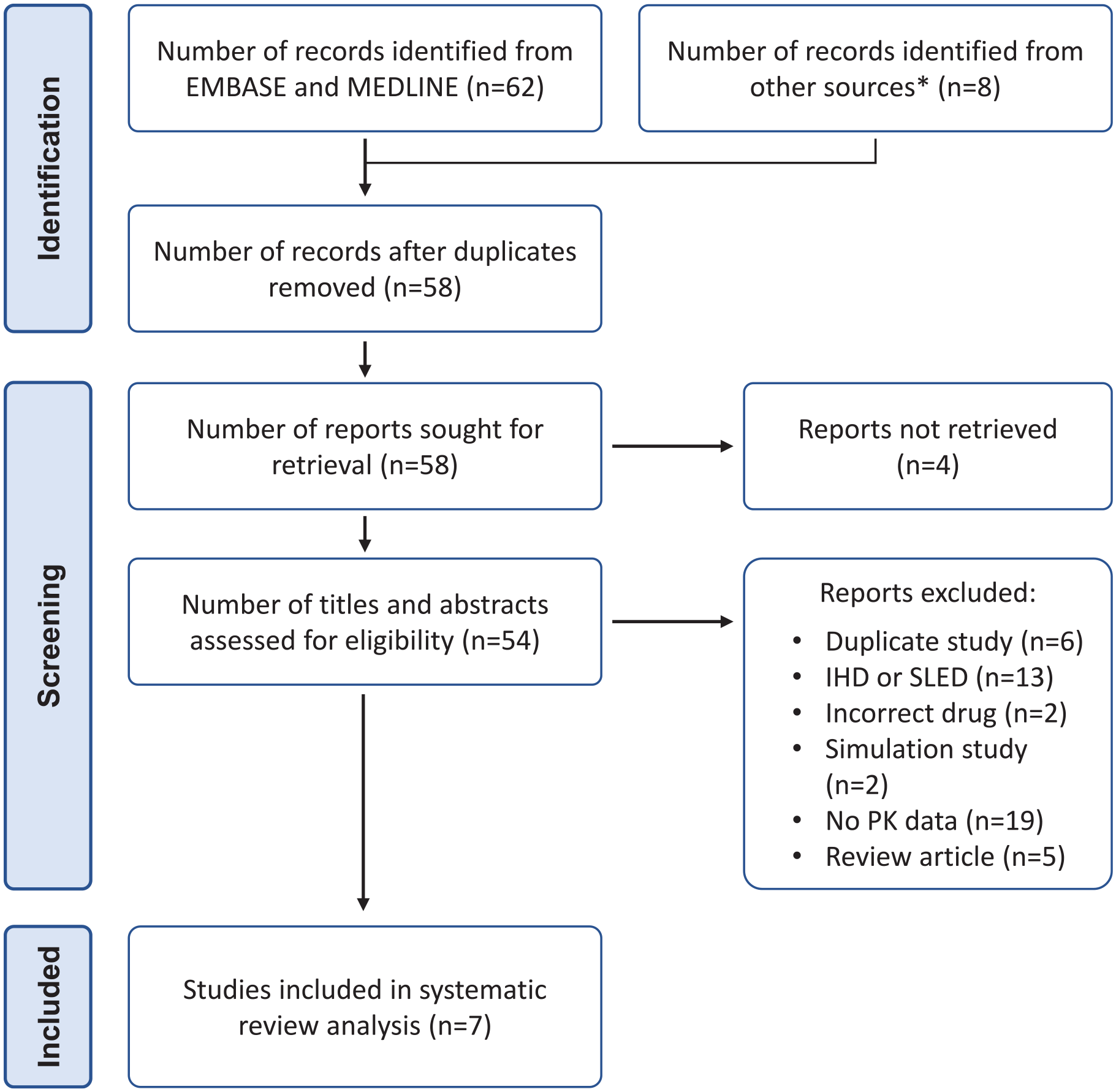
PRISMA flow chart for study inclusion.
Study characteristics
The characteristics of our included studies are summarised in Table 1. In most cases, patients were commenced on CRRT for acute kidney injury or known end-stage renal disease with multi-organ failure. Sixteen out of twenty-four patients were treated with CVVHF with eight other patients treated with CVVHDF. Two patients22,23 were receiving simultaneous extracorporeal membrane oxygenation (ECMO). In most patients (17 out of 24) a 1000 mg 12-hourly dose of levetiracetam was administered as intermittent intravenous infusions. One patient received enteral levetiracetam, however as the oral bioavailability of levetiracetam is 100% the impact of this is likely to have been minimal.
Characteristics of studies included within systematic review.
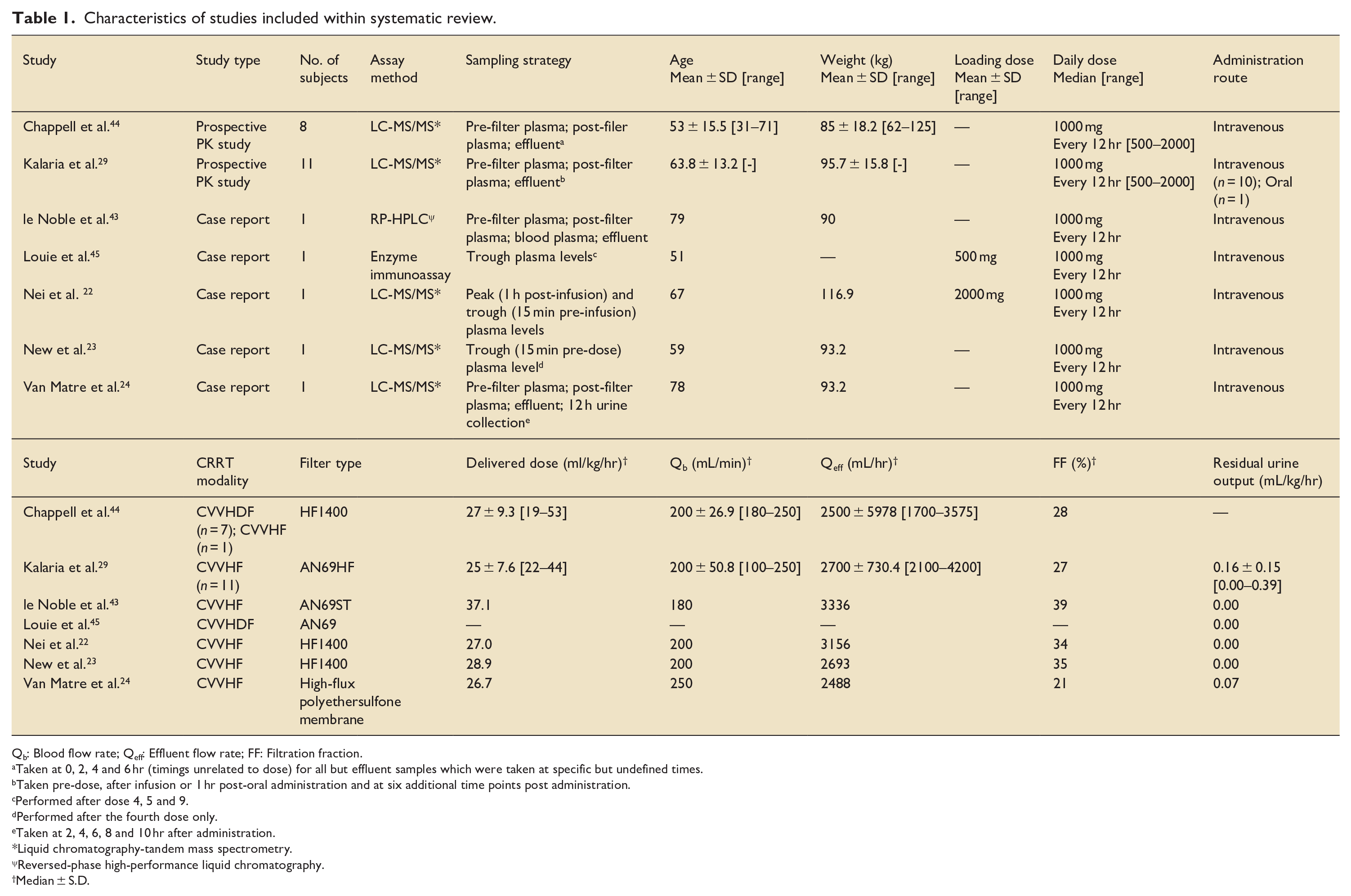
Qb: Blood flow rate; Qeff: Effluent flow rate; FF: Filtration fraction.
Taken at 0, 2, 4 and 6 hr (timings unrelated to dose) for all but effluent samples which were taken at specific but undefined times.
Taken pre-dose, after infusion or 1 hr post-oral administration and at six additional time points post administration.
Performed after dose 4, 5 and 9.
Performed after the fourth dose only.
Taken at 2, 4, 6, 8 and 10 hr after administration.
Liquid chromatography-tandem mass spectrometry.
Reversed-phase high-performance liquid chromatography.
Median ± S.D.
Sampling strategies across studies varied significantly. Pre-filter, post-filter and effluent samples were collected in four studies while in three studies only plasma samples (either peak or trough levels) were taken. In most studies, levetiracetam levels were not taken until steady state had been achieved. One compartmental modelling for pharmacokinetic analysis was used in three studies,22 –24 while Kalaria et al. used a non-compartmental approach.
The recommended delivered dose in CRRT varies but is considered to be between 20 and 35 mL/kg/hr. The median delivered dose in these studies ranged from 26.7 to 37.1 mL/kg/hr with significant variation seen in Chappell et al. and Kalaria et al. Blood flow rates ranged from 180 to 250 mL/min and were broadly consistent across studies. The majority of patients were either anuric or oliguric (defined as urine output <400 mL in 24 hr) but it should be noted that these data were not collected by Chappell et al.
Pharmacokinetic data
Reported levetiracetam clearance from CRRT was between 1.80 and 2.98 L/h (Table 2). Where total clearance was calculated, the clearance attributed to CRRT alone was between 52% and 73% (Mean 54.7%, S.D. 13.5). Reported total clearance of levetiracetam was between 3.49 and 4.63 L/hr (Mean 3.55, S.D. 0.52). Elimination half-life varied significantly with a range of 5.66–12.88 hr and a mean 9.41 hr (S.D. 2.86, S.E. 0.60). Volume of distribution ranged from 0.45 to 0.73 L/kg. In the majority of patients, peak and trough plasma concentrations were within the therapeutic target range of 12–46 mcg/mL. The exception to this was in Kalaria et al.’s study in which six patients demonstrated trough plasma concentrations below 12 mcg/mL (1.93–9.94 mcg/mL).
Levetiracetam pharmacokinetic data from included studies.
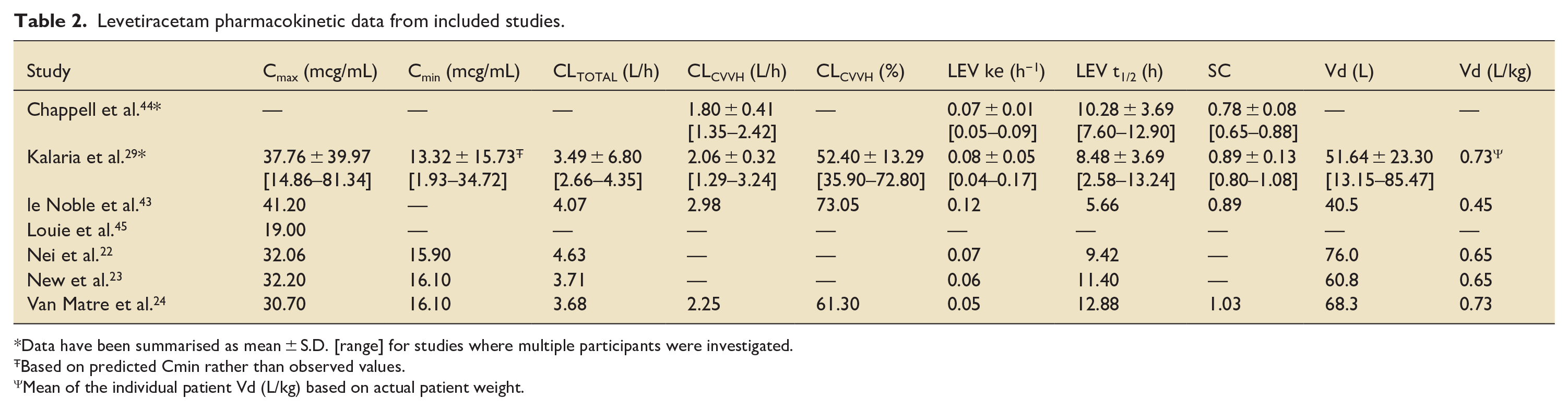
Data have been summarised as mean ± S.D. [range] for studies where multiple participants were investigated.
Based on predicted Cmin rather than observed values.
Mean of the individual patient Vd (L/kg) based on actual patient weight.
Strength of evidence
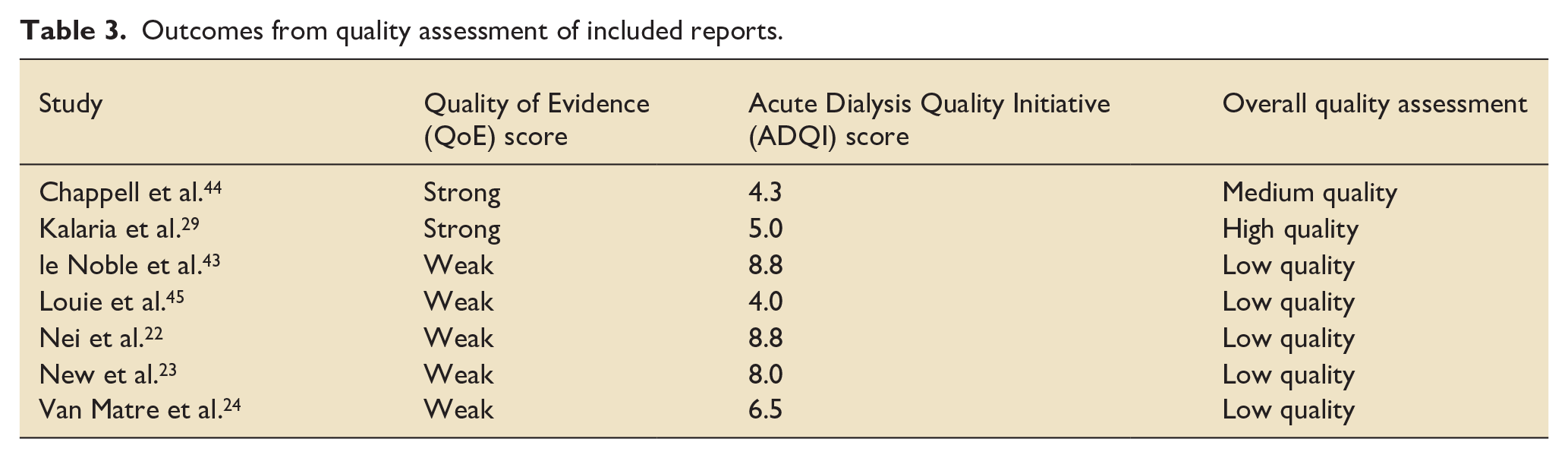
The outcome of our quality assessment is shown in Table 3. In general, we determined that the strength of evidence was overall of low quality. The level of information provided about CRRT techniques was broadly inconsistent with few studies providing the level of information required to enable comprehensive interrogation of the data. In one study, 29 demographics were only provided at aggregate level which prevented individualised pharmacokinetic analysis. Time spent on filter was only mentioned in one study 23 and while it could be assumed that haemofiltration was continuous, it was clear that at least some patients experienced filter clotting and pauses to treatment. The two prospective studies in our data set29,44 were judged to be of higher quality predominantly due to the more robust and comprehensive nature of their pharmacokinetic data.
Outcomes from quality assessment of included reports.
Multiple regression
Due to a limited base size and various missing data discussed above, it was not possible for us to perform multiple regression analysis with any meaningfully significant results.
Simulation of dosing scenarios
Figure 2 illustrates the proportion of patients who achieved our therapeutic target level (12–46 mcg/mL) across a range of simulated doses without a loading dose. At a target level of 12 mcg/mL, 65% of simulated patients who received a dose of 1000 mg BD achieved target level in comparison to 53% of patients who received 750 mg BD, 34% of patients who received 500 mg BD and 7.5% of patients who received 250 mg BD.
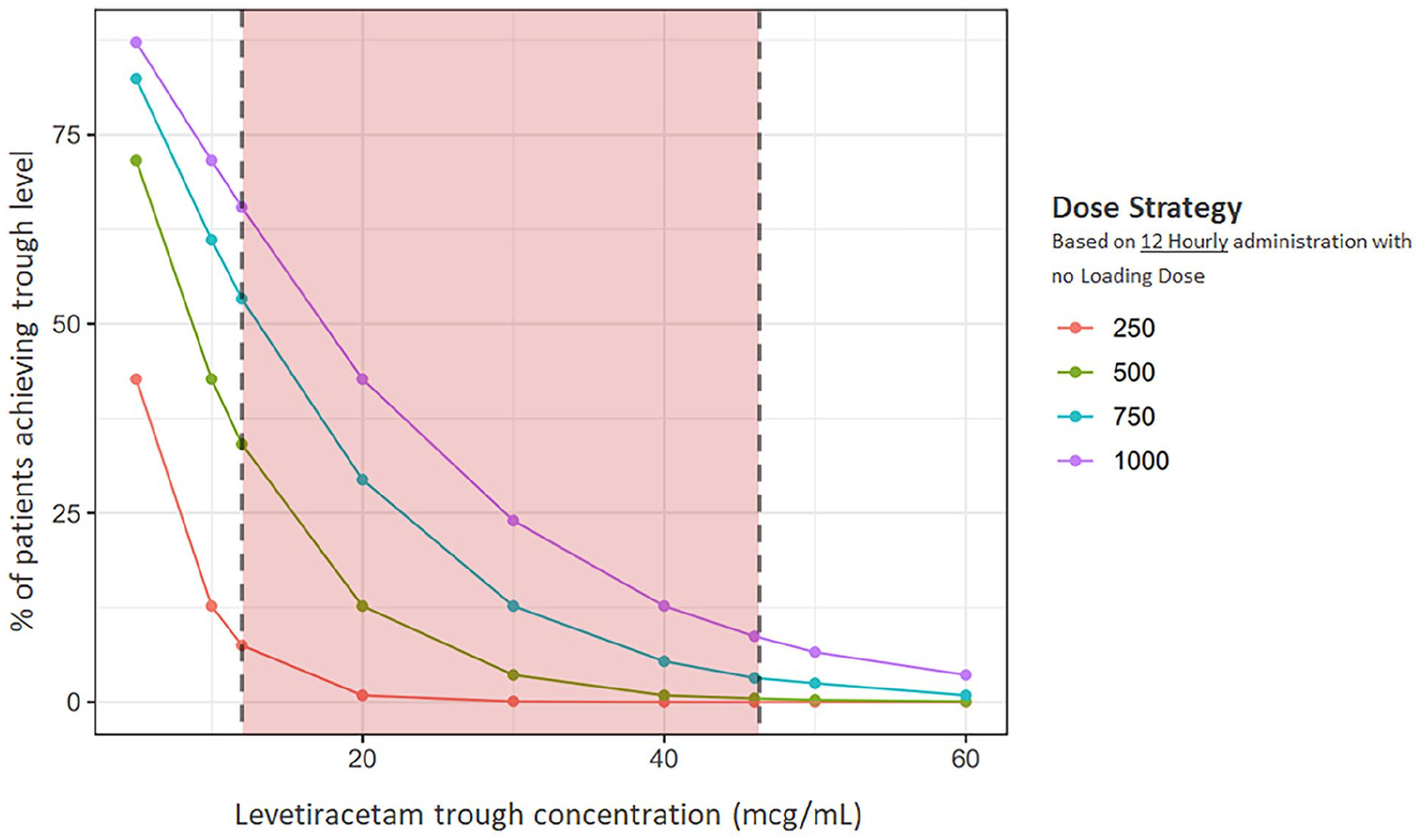
Percentage of simulated patients achieving trough concentrations based on 12-hourly dosing strategies.
Table 4 demonstrates the trough concentration profiles at these various doses without a loading dose. At doses <750 mg BD the median trough concentration was below the target range, although a minority did achieve therapeutic drug concentrations. Almost all patients who received 250 mg BD experienced trough concentrations below the target level. Those patients who received higher doses (⩾750 mg BD) were more likely to achieve therapeutic drug concentrations.
Trough concentration profile for a range of simulated levetiracetam doses during continuous renal replacement therapy.

Dosing strategies are based on 12 hourly administration of levetiracetam with no loading dose.
Median (10%–90%).
Our simulations also evaluated levetiracetam concentrations in patients on CRRT receiving a range of doses over a 72-hr period. In Figure 3, patients received a standard loading dose of 60 mg/kg (up to a maximum of 4.5 g) as recommended for the treatment of neurological emergencies.45,46 In Figure 4, patients received a 750 mg loading dose as recommended for patients on IHD. In Figure 5, patients received no loading dose.
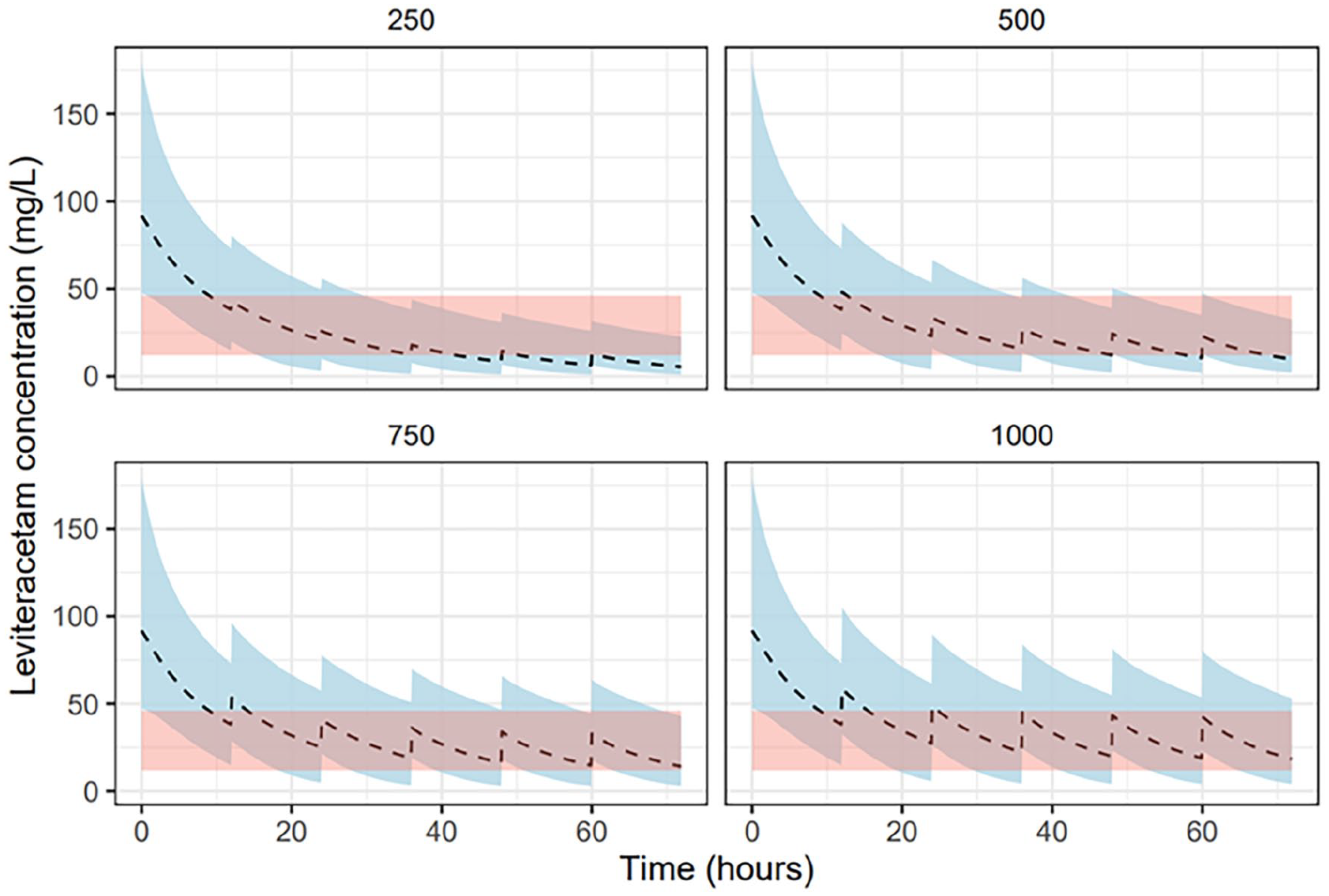
Pharmacokinetic profile for a range of levetiracetam doses with 60 mg/kg loading dose in simulated patients receiving renal replacement therapy.
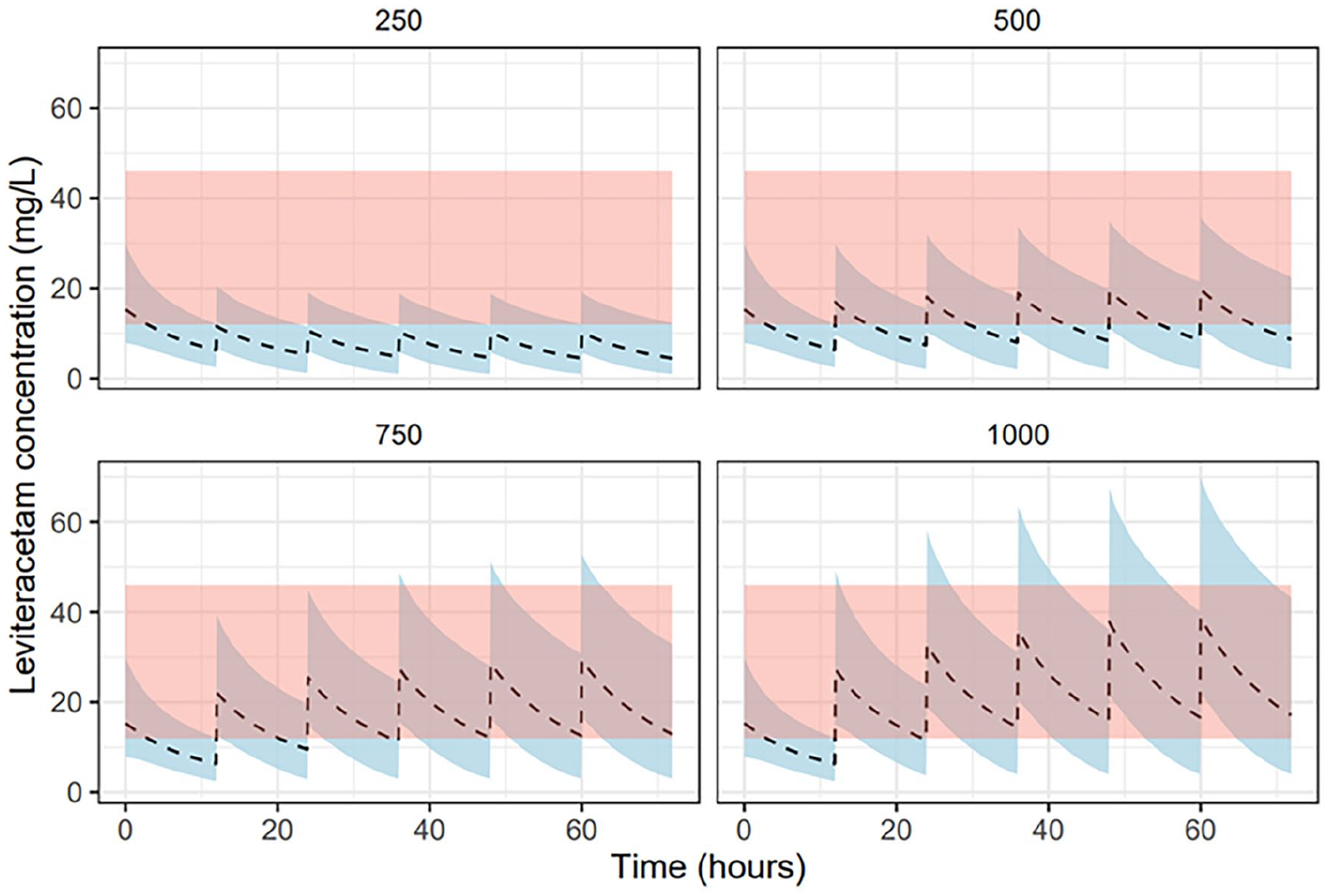
Pharmacokinetic profile for a range of levetiracetam doses with 750 mg loading dose in simulated patients receiving renal replacement therapy.
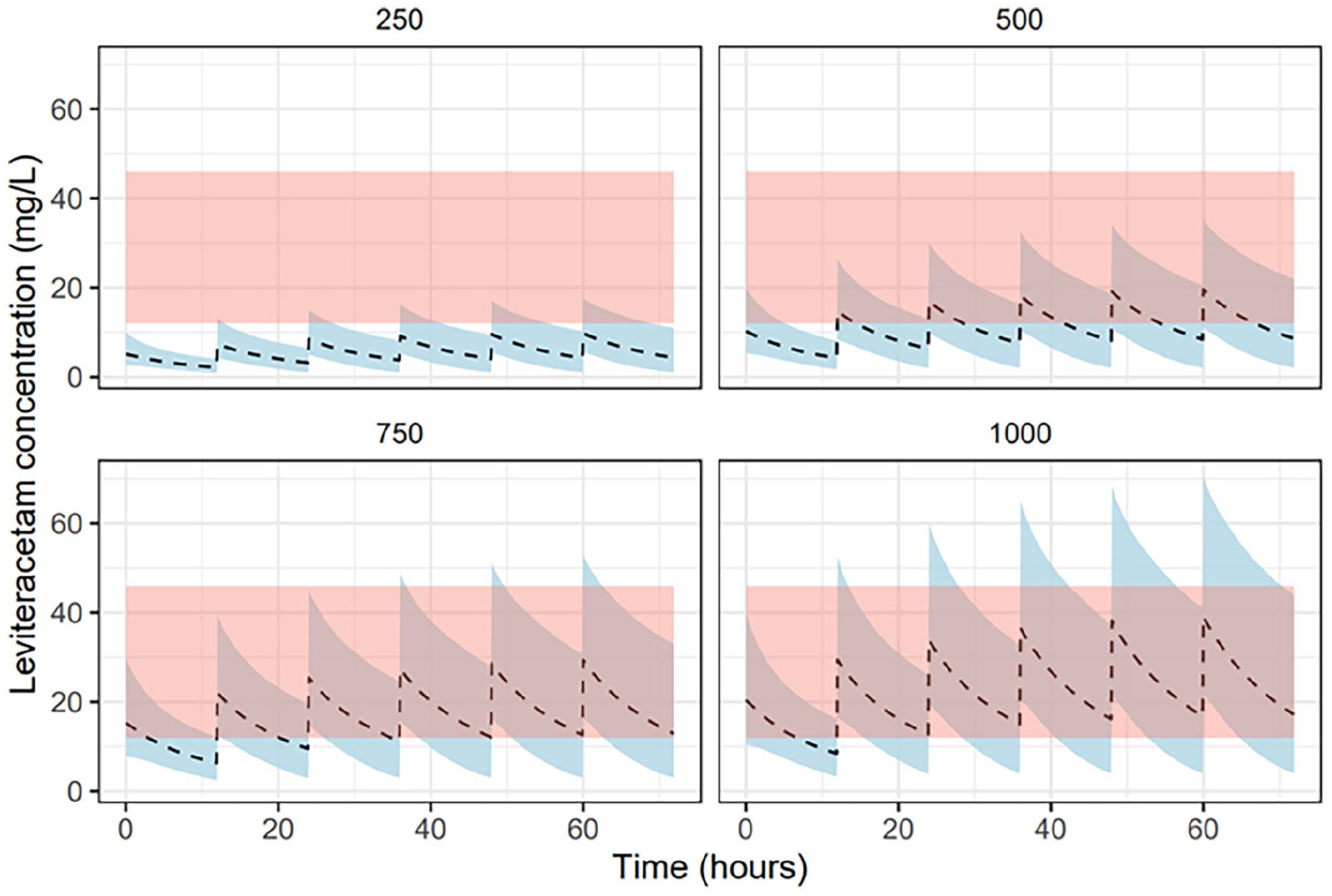
Pharmacokinetic profile for a range of levetiracetam doses with no loading dose in simulated patients receiving renal replacement therapy.
As shown in Figure 3, the standard loading dose achieved therapeutic drug concentrations within the first 24 hr for almost all simulated patients. Conversely, a significant number of patients who received a reduced (Figure 4) or no loading dose (Figure 5) experienced sub-therapeutic drug concentrations for an extended period – with the majority of patients receiving a 250 mg BD dosing spending the entire 72 hr below target concentration. Only at higher doses (⩾750 mg BD) did median concentrations remain within target range at 72 hr.
Discussion
Our findings suggest that the clearance of levetiracetam in critically ill patients undergoing CRRT is similar to that seen in healthy adult patients. Both mean total clearance (3.55 L/hr) and elimination half-life (9.41 hr) are broadly equivalent to that of healthy adults (4.03 L/hr and 6–8 hr respectively). The mean clearance attributable to CRRT was over 50% confirming that CRRT is responsible for the majority of drug clearance. Moreover, it demonstrates that IHD and CRRT have similar pharmacokinetic effects in terms of their ability to eliminate levetiracetam. Taken together, these findings suggest that reduced doses in critically patients on CRRT may pose a serious risk of sub-therapeutic drug concentrations.
In the UK, current guidance is based on limited data and expert opinion and is variable. The drug monograph in Medicines Complete for Critical Illness recommends no dose reduction on the basis that levetiracetam is freely cleared by CRRT. 20 The Lexi-Comp Drug Information Handbook recommends doses of 750–1250 mg BD in patients undergoing CRRT with consideration of doses up to 4 g per day for patients on higher effluent rates. 21 These recommendations are based on Kalaria et al.’s influential PADRE-01 study which is the most extensive pharmacokinetic study to date but remains limited by its base size (n = 11). A recent study by Chaijamorn et al. used published pharmacokinetic data to identify an appropriate dose for patients with liver cirrhosis on CRRT and recommended a dosing regimen of 750–1000 mg BD. 25
Our simulation data support recommendations for a 750–1000 mg BD dosing strategy with a standard loading dose of 60 mg/kg where rapid cessation of seizure activity or neuroprotective control is required. Dosing at lower levels (250–500 mg BD) was unlikely to achieve therapeutic levels by steady state even with the addition of a loading dose (Figures 3–5). At higher doses (750–1000 mg BD) the proportion of patients achieving therapeutic drug concentrations at steady state was significantly higher. The addition of a standard loading dose reduced the time to attainment by up to 36 hr (Figure 3). A significant proportion of patients at this loading dose, however, had trough concentrations over 80 mcg/mL for the first 24 hr and may benefit from close monitoring for potential drug toxicity.
46
In contrast, the use of a reduced 750 mg loading dose as recommended for patients on IHD resulted in delayed attainment of therapeutic trough concentrations up to 24–36 hr into treatment (Figure 4). As demonstrated in Figures 2 and 5, the majority of patients who received
This study is not without limitations. A major limitation in our analysis is the variation in study protocols and data collection within the current literature. Differences in dose administration and sampling strategies undoubtedly introduce elements of confounding. Moreover, the lack of a standardised approach to reporting pharmacokinetic data (for instance, volume of distribution was unavailable in one study) and CRRT treatment parameters limit the ability to analyse amalgamated study data. Our novel approach to evaluating the strength of evidence, while based on established methods, remains untested outside of this study. Finally, while several studies have demonstrated that a one compartment model is consistent with in vivo pharmacokinetics of levetiracetam, the majority of these have been undertaken in a non-critically ill population. The altered pharmacokinetics of critically ill patients owing to fluid shifts, multi-organ failure and altered protein binding remain unclear and require further study.
This systematic review demonstrates that there is an urgent need for further study in this population and suggests several areas of interest. The specific extrinsic factors related to CRRT, such as effluent flow rate or dialysate rate, that influence levetiracetam clearance remain unclear. Few studies investigated the haemofiltration characteristics that influence clearance with the exception of Kalaria et al. Their analysis identified effluent flow rate and sieving co-efficient as the main influences on clearance. Effluent rates in excess of 3.5 L/h were also associated with increased clearance and so higher doses may be required in these patients, although the potential for toxicity with such doses owing to drug accumulation is unclear.
Similarly, further investigation into the intrinsic patient-specific factors that affect clearance is also required. While mean clearance was consistent between studies, there was a wide variation in individual patient clearance and plasma concentrations which are not easily explained by effluent rate and sieving co-efficient alone. Six patients demonstrated trough drug concentrations below the therapeutic target level but a post-hoc descriptive analysis (Supplemental Appendix 3) found no obvious explanation. Three of these patients had significant residual urine output (>300 mL per day), while three others had high effluent rates. However, other patients received similar doses at higher effluent rates or indeed had higher urine outputs without their plasma concentrations falling outside of target levels. These data suggest that there is a high inter-person variability of drug pharmacokinetics and determining optimum dosing strategies for individuals may require therapeutic drug monitoring.
Finally, the lack of a standardised and consistent approach to pharmacokinetic studies in this population limits the quality of evidence on which to base recommendations. Data are limited to a small number of case reports and two small prospective studies with a lack of consistency between studies, both in terms of methodology and data reporting, which limits the extent to which modelling can be performed on larger data sets. In this paper, we have introduced a novel quality assessment method that utilises a combination of the QoE framework, which assesses the strength of pharmacokinetic studies based on the quality of data and pharmacokinetic modelling provided, and the ADQI minimum reporting criteria, which was developed to standardise comparisons between studies that reported on CRRT techniques. Both methods have been used in similar systematic reviews in this way47,48 but never before in combination. By combining these assessment methods, our quality assessment method allows for equal consideration of both the pharmacokinetic and haemofiltration data. This enables an assessment of both the quality of the data itself and the ability to perform statistical analysis. It also provides a framework for protocol development so that future studies present data in a standardised manner.
Future studies should seek to understand the high inter-person variability in levetiracetam clearance and the in vivo effects of different dosing strategies. The goal of such studies should be to inform pharmacokinetic models that will enable clinicians to determine patient-specific dosing strategies. The collection of standardised data can facilitate meta-analysis and modelling and our recommendation is that this should include the ADQI minimum reporting criteria alongside the following: (1) IBW; (2) urine output; (3) baseline bloods (albumin, creatinine, haematocrit, liver function); (4) dosing strategy; (5) a sampling strategy that at a minimum includes pre-filter, post-filter and effluent levels; (6) information on whether seizures were controlled or uncontrolled.
In addition to in vivo studies, experimental study using lab-based models that simulate critically ill patients undergoing CRRT may be valuable. These studies, such as the latest ex vivo in vivo study by Kalaria et al., 39 would be useful in evaluating the effects on clearance of different CRRT parameters and levetiracetam dosing without the potential risk of exposing patients to possible sub-therapeutic or toxic drug levels.
Conclusions
The current available data on in vivo clearance of levetiracetam in critically ill patients undergoing CRRT does not support dose reduction in this population. Clearance is consistently demonstrated to be similar to healthy adults with normal renal function (GFR > 90 mL/min). Reduced dosing based on creatinine clearance is likely to result in sub-therapeutic drug concentrations in a high number of patients. A lack of consensus on a therapeutic window for levetiracetam makes dosing recommendations challenging and clinicians may find the information in Figure 2 useful in determining dosage based on whether targeting a therapeutic range or likelihood of patient attainment is more desirable. There is a growing consensus that a twice daily dose of 750–1000 mg with an initial loading dose of 60 mg/kg is appropriate in such patients. However, further study is needed to evaluate the potential for drug accumulation and toxicity. Therapeutic drug monitoring may be useful in guiding dosage and identifying those at risk of toxicity.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251320557 – Supplemental material for Levetiracetam dosing in continuous renal replacement therapy: A systematic review and development of a novel pharmacokinetic model to optimise dosing in critically ill patients. Do recommended doses achieve therapeutic drug concentrations?
Supplemental material, sj-docx-1-inc-10.1177_17511437251320557 for Levetiracetam dosing in continuous renal replacement therapy: A systematic review and development of a novel pharmacokinetic model to optimise dosing in critically ill patients. Do recommended doses achieve therapeutic drug concentrations? by James Sweatman, Sarraa Al-Mahdi, Dagan O Lonsdale, Susannah Leaver and Andrew Rhodes in Journal of the Intensive Care Society
Supplemental Material
sj-pptx-2-inc-10.1177_17511437251320557 – Supplemental material for Levetiracetam dosing in continuous renal replacement therapy: A systematic review and development of a novel pharmacokinetic model to optimise dosing in critically ill patients. Do recommended doses achieve therapeutic drug concentrations?
Supplemental material, sj-pptx-2-inc-10.1177_17511437251320557 for Levetiracetam dosing in continuous renal replacement therapy: A systematic review and development of a novel pharmacokinetic model to optimise dosing in critically ill patients. Do recommended doses achieve therapeutic drug concentrations? by James Sweatman, Sarraa Al-Mahdi, Dagan O Lonsdale, Susannah Leaver and Andrew Rhodes in Journal of the Intensive Care Society
Footnotes
Acknowledgements
Our thanks to Stephen Reid at St. George’s University London who provided invaluable support on optimising our search criteria for this under-researched topic area.
Author contribution statement
J.S. and S.A-M. developed the protocol for the presented research. J.S., S.A-M. and D.L. participated in data collection and analysis. D.L. performed the simulated modelling. S.L. and A.R. verified the analytical methods. All authors discussed the results and contributed to the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was received from the St. George’s Hospital Charity and had no impact on the authors’ independence or execution of the research.
Ethics and integrity statement
No ethics or patient consent approval were required for this review article. Our protocol was registered on PROSPERO (CRD42022300754) prior to the commencement of the research. No materials within this paper were reproduced from other works. All figures and diagrams are original to this paper.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
