Abstract
Introduction:
Continuous renal replacement therapy (CRRT) dose is usually fixed and primarily weight-based. Whilst this is safe, theoretically, underdosing or overdosing may occur in those requiring acute versus maintenance CRRT respectively. We have developed a dynamic dosing protocol for CRRT which individualises and updates dosing according to biochemistry. Here we describe the protocol and compare it to a fixed dose protocol to evaluate its safety and effectiveness.
Methods:
We conducted a service evaluation of this novel protocol using data from consecutive non-COVID-19 admissions receiving CRRT within Barts Health NHS trust, United Kingdom (UK). Fifty admissions using the dynamic protocol were compared to historically collected data from 108 admissions who used the fixed protocol. Acute and maintenance CRRT subgroups were analysed.
Results:
For the dynamic protocol 49 patients were treated with 135 CRRT circuits. One patient had two admissions. Protocol compliance (compared in one ICU) was 76% (dynamic) vs 61% (fixed) (p < 0.05). For the dynamic versus fixed protocol, median CRRT lifespan censored for reasons other than clotting was: 56 h vs 58 h RCA (ns), 27 h versus 20 h heparin (ns) and 79 h versus 22 h no anticoagulation (p < 0.05). The dynamic vs fixed protocol average CRRT dose was: 30 ml/kg/h (14–57) vs 26 ml/kg/h (20–62) (p < 0.05). The dynamic protocol generated a similar rate of increase of bicarbonate in an acute phase (RCA: 0.2 mmol/l/h vs 0.21 mmol/l/h, ns) and maintained a more stable level in a maintenance phase (RCA: −0.01 mmol/l/h vs 0.07 mmol/l/h, p < 0.05).
Discussion:
Dynamic dosing for CRRT in this institution was safe and may lead to more tailored maintenance of biochemical homoeostasis.
Keywords
Introduction
Several large multi-centre randomised controlled trials have demonstrated that there is no mortality benefit to initiating early CRRT based on acute kidney injury (AKI) staging.1 –3 Their results support reserving CRRT for conventional indications including refractory acidaemia or hyperkalaemia. However, whilst initiation of CRRT is based on the presence of biochemical abnormalities, dosing is generally weight-based in most intensive care units (ICUs). Evidence has shown that targeting effluent flow rates of 20–25 ml/kg/h offers no benefit, in terms of short term mortality, than higher dose regimens.4 –7 However weight-based dosing is potentially counterintuitive if the goal of treatment is the normalisation and maintenance of acid-base and electrolyte parameters. AKI can present with a wide range of biochemical derangements and progress over several different trajectories. 8 Theoretically weight-based dosing, in this heterogeneous and dynamic population, could lead to underdosing in the acutely ill and delivering higher doses than required for stable patients respectively. The latter group of patients would include patients with enduring critical illness and renal dysfunction, necessitating ongoing CRRT on the ICU, but at a point in their trajectory where their acid base and electrolyte requirements are stable. Excessive dose CRRT can cause deleterious biochemical derangements, such as alkalosis and hypophosphatemia, and may delay renal recovery.4 –6,9 To better individualise and rationalise our delivery of CRRT to a patient’s biochemistry and renal injury trajectory, we have developed a conceptually different dosing protocol. In this article we describe this novel CRRT protocol and describe data on its service implementation. Our goal is to enable more individualised therapy, while retaining a simple to use protocol that allows delivery of safe and effective anticoagulation. In our service evaluation we aimed to examine the safety and efficacy of the dynamic dose protocol in achieving biochemical goals.
Methods
Aims
This is a service evaluation aimed at providing a descriptive comparison use of two different CRRT protocols. It is an exploratory and hypothesis generating report which would require future studies to confirm its findings before causative claims can be made. To evaluate the safety and efficacy of the change of protocol we primarily analysed data on CRRT lifespan, machine set dose compared with prescribed target dose 10 and solute control. Respectively, these have been used to ensure the new protocol was non inferior when compared with a historical baseline dataset, that protocol was being used as intended and that unintended consequences did not occur with its use.
The protocol
CRRT in our institution is delivered exclusively using continuous veno-venous haemodialysis (CVVHD). The dynamic protocol prescribes and updates blood and dialysate flow rates according to serial potassium and pH measurements. These prescribed flow rates are tabulated for any combination of pH and potassium values. Two tables are provided, for patients above or below 65 kg ideal body weight. For circuits using heparin or no anticoagulation, blood flow rates are fixed at 150 ml/min. For regional citrate anticoagulation (RCA) circuits, blood flow rates are adjusted in conjunction with dialysate flow rates. As citrate is metabolised into bicarbonate, adjusting the dialysate to blood flow ratio can aid optimisation of acid-base balance. Hence, greater blood flow rates are prescribed for more acidaemic patients and lesser rates for alkalaemic patients (80–150 ml/min). As it can take 6–12 h to reach new equilibrium, dose changes are only recommended at six hourly intervals. Calcium levels are monitored more frequently to adjust calcium replacement. Specifically, it is recommended that calcium is checked before starting CRRT, within 1 h of starting CRRT and after any parameter change (particularly those designed to adjust calcium levels). Comparative historical data was used from patients managed with a fixed dose protocol. Here, effluent dose and blood flow of CRRT were chosen based on weight, with an adjustment made for hyperkalaemia. Once set, changes were only recommended at the extremes of pH or bicarbonate levels, or if hyperkalaemia persisted. For both protocols, CRRT was initiated and discontinued by the treating clinicians, the therapy was implemented using BBraun Omnisets by critical care nursing staff. The Supplemental Appendix and Supplemental Documents 1 and 2 give further details on both protocols, including copies of the prescription tables.
Data collection
All data was collected from hospitals within Barts Health NHS Trust. For the dynamic protocol, data was collected retrospectively from 50 consecutive admissions to a single adult mixed medical-surgical ICU from 01/05/2021 to 30/09/2021. The dynamic protocol had been used for several months prior to the start of data collection, no specific problems or significant adjustments were required in this period. This was compared to historically collected data from 108 consecutive admissions, across three ICUs, who were treated with the fixed protocol from 01/09/2019 to 31/01/2020. The time gap between datasets occurred due to the COVID-19 pandemic. The inclusion criteria were patients >16 years old who received CRRT. The only exclusion was COVID-19. The first data collection occurred prior to the COVID-19 pandemic affecting the UK. Therefore, to maintain intergroup homogeneity, we collected data on the dynamic protocol outside a COVID-19 pandemic peak and excluded patients with a COVID-19 diagnosis. We recorded demographic, clinical, biochemical and dosing data for the duration of CRRT on the ICU. For each CRRT run the following was recorded: total number of hours, blood gas at start, end and at any setting change along with machine set doses. Consecutive data collection was a pragmatic choice performed to reduce selection bias and aid generalisability. For this reason and to ensure like-for-like comparison with the historical dataset, other special case CRRT indications, such as for toxicology treatment, were not excluded. Retrospective analysis of anonymised routine data by the direct care team did not require direct patient consent. This was a service development project registered as two audits. No power calculation was used as we aimed to include all patients who were put onto the protocols.
Data analysis
Categorical and continuous variables were analysed using Fisher’s exact test and a two tailed Mann-Whitney U test respectively. Subgroup analyses were performed for differing anticoagulation strategies and those in an acute phase and maintenance phase of their CRRT need. These were defined as, acute phase group: first CRRT session, pH < 7.35 or potassium >5 mmol/l, maintenance phase group: pH > 7.35 and potassium <6 mmol/l. Protocol compliance was assessed semi-qualitatively by identifying if CRRT dosing deviated significantly or persistently from what was recommended. At initiation of CRRT, and after each setting change, portions of the CRRT run were coded as compliant or non-compliant. This depended on whether the set machine dose 10 matched the prescribed target dose. 10 The whole CRRT run was then judged by two researchers performing data extraction to be compliant or not using the codes from each timepoint. The total run was coded as non-compliant if there were proportionally large and or prolonged deviation(s). In the instance of no deviations or proportionally small and or short deviation(s), the total run was coded as compliant. Clinical judgement was used to determine what counted as insignificant deviations; an example would include machine set errors where an incorrect weight was used that was corrected within a few hours. Numerical cut-offs were not used, where a CRRT run contained a mixture of non-compliant and compliant segments these were discussed between coders. A conclusion was reached over whether the deviations were significant enough to constitute non-compliance. An overall percentage was then taken from this. Whilst this is a blunt measurement it was intended to give a gross impression of whether the greater demands of the dynamic protocol would adversely affect the ability of staff to correctly implement it. The historical fixed dose dataset was used as a comparative baseline. Finally, median filter lifespan was analysed using a Kaplan-Meier survival curve and censored for reasons other than clotting.
Results
Patient and CRRT session demographics
Data was generated from 135 and 320 CRRT sessions for the dynamic protocol cohort and the fixed dose comparator cohort respectively (Table 1). Patient demographics, gender and proportion of patients with end stage renal failure were similar between groups. Patients in the dynamic cohort were younger and more often had trauma or surgery as reason for admission. This is likely because data for the dynamic cohort was collected from one site, the Royal London Hospital, which is a major trauma centre and a high-volume surgical centre. Similarly, the dynamic protocol used more RCA because this was the preferred modality at the Royal London Hospital.
Patient demographics and continuous renal replacement therapy session breakdown.
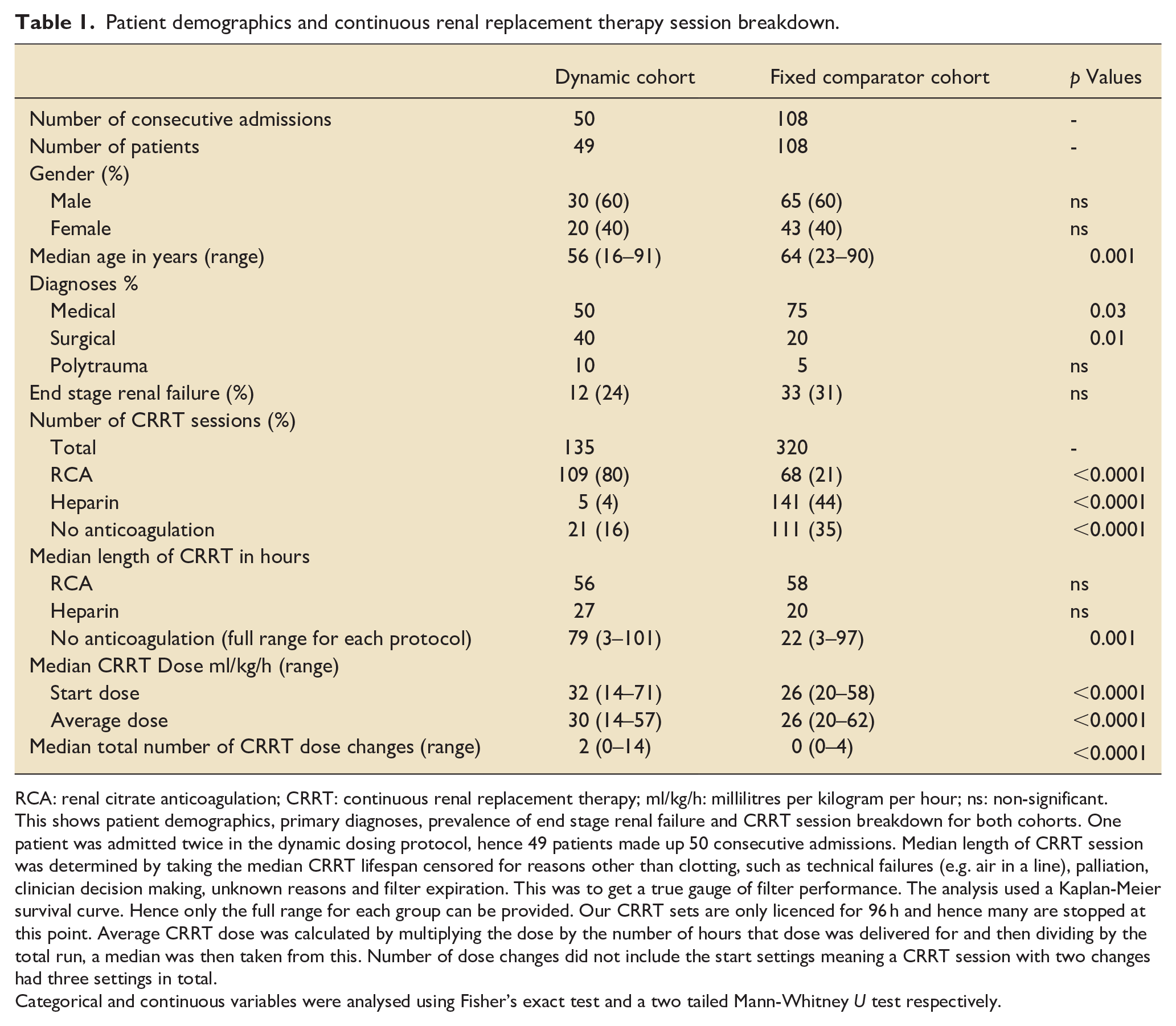
RCA: renal citrate anticoagulation; CRRT: continuous renal replacement therapy; ml/kg/h: millilitres per kilogram per hour; ns: non-significant.
This shows patient demographics, primary diagnoses, prevalence of end stage renal failure and CRRT session breakdown for both cohorts. One patient was admitted twice in the dynamic dosing protocol, hence 49 patients made up 50 consecutive admissions. Median length of CRRT session was determined by taking the median CRRT lifespan censored for reasons other than clotting, such as technical failures (e.g. air in a line), palliation, clinician decision making, unknown reasons and filter expiration. This was to get a true gauge of filter performance. The analysis used a Kaplan-Meier survival curve. Hence only the full range for each group can be provided. Our CRRT sets are only licenced for 96 h and hence many are stopped at this point. Average CRRT dose was calculated by multiplying the dose by the number of hours that dose was delivered for and then dividing by the total run, a median was then taken from this. Number of dose changes did not include the start settings meaning a CRRT session with two changes had three settings in total.
Categorical and continuous variables were analysed using Fisher’s exact test and a two tailed Mann-Whitney U test respectively.
Safety
There were no adverse events for either cohort. Apart from those using no anticoagulation, where the numbers for the dynamic protocol are small (n = 21), CRRT circuit lifespan was similar between groups (Table 1). The number of patients switching from RCA to heparin anticoagulation because of alkalosis was 3 (3%) versus 1 (1.5%) (ns) for the dynamic versus fixed protocol. Dynamic protocol compliance was 76% (total n = 135) versus 61% (total n = 109) (p < 0.05) for the fixed protocol at the Royal London site. Compliance data was compared only for this site because RCA is used as first line there, which is often a more complex therapy to deliver, and this ICU has a significantly different case mix to the others. Therefore, to control for these inter-site differences and ensure like-for-like comparison, the compliance data from other sites was excluded.
Dosing
The dynamic protocol delivered higher start and average effluent doses. However, compared to the start dose the average dose was lower, suggesting a decrease over time. In contrast the fixed protocol delivered a static dose. Additionally, as intended, a greater number of CRRT setting changes were made in the dynamic protocol group (Median CRRT changes: 2 vs 0, p < 0.05).
The relationships shown in Figure 1 reflect the characteristics of the dynamic protocol for RCA. Hyperkalaemia is addressed purely by increased dialysate flow. Acidosis is addressed by an increase in blood flow and buffer delivery as metabolised citrate, while dialysate flow (with a lower bicarbonate calcium free dialysate) is increased to address pH extremes of acidosis or alkalosis.
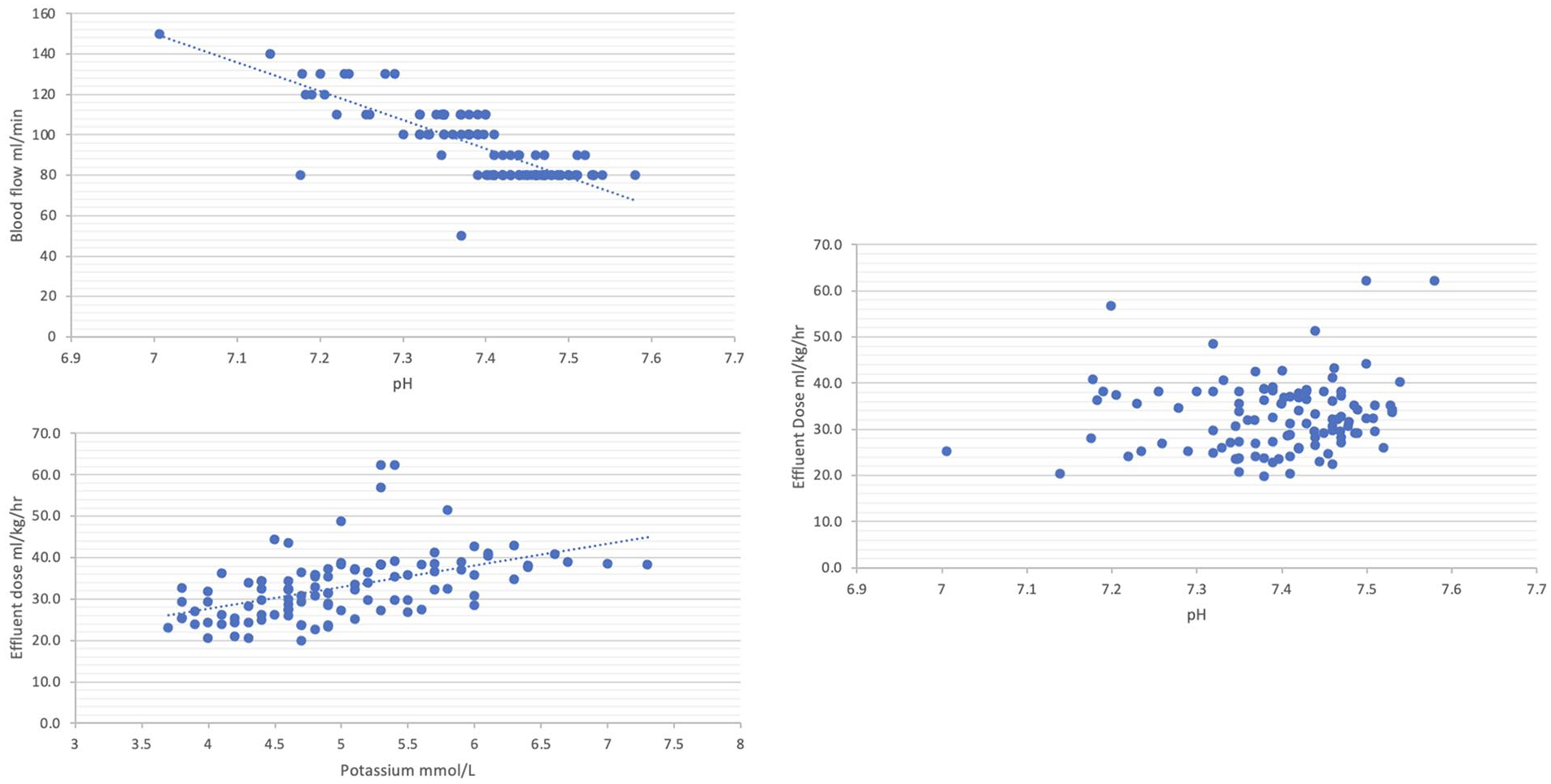
Dynamic cohort blood flow and effluent dose settings plotted against biochemistry for CRRT circuits using RCA: (a) upper left: starting blood flow settings against starting pH (n = 109), (b) bottom left: Starting effluent dose delivery against starting potassium (n = 108), and (c) right: starting effluent dose delivery against pH (n = 108).
Biochemistry
For the subgroup analysis starting CRRT settings are shown (Table 2) as this corresponds to the patient’s starting biochemical state, which defines the phasic groups. The average dose may have been different; particularly for the dynamic group where dose in general reduces over time (Table 1).
Biochemistry breakdown for Acute and Maintenance groups per cohort and anticoagulation strategy.
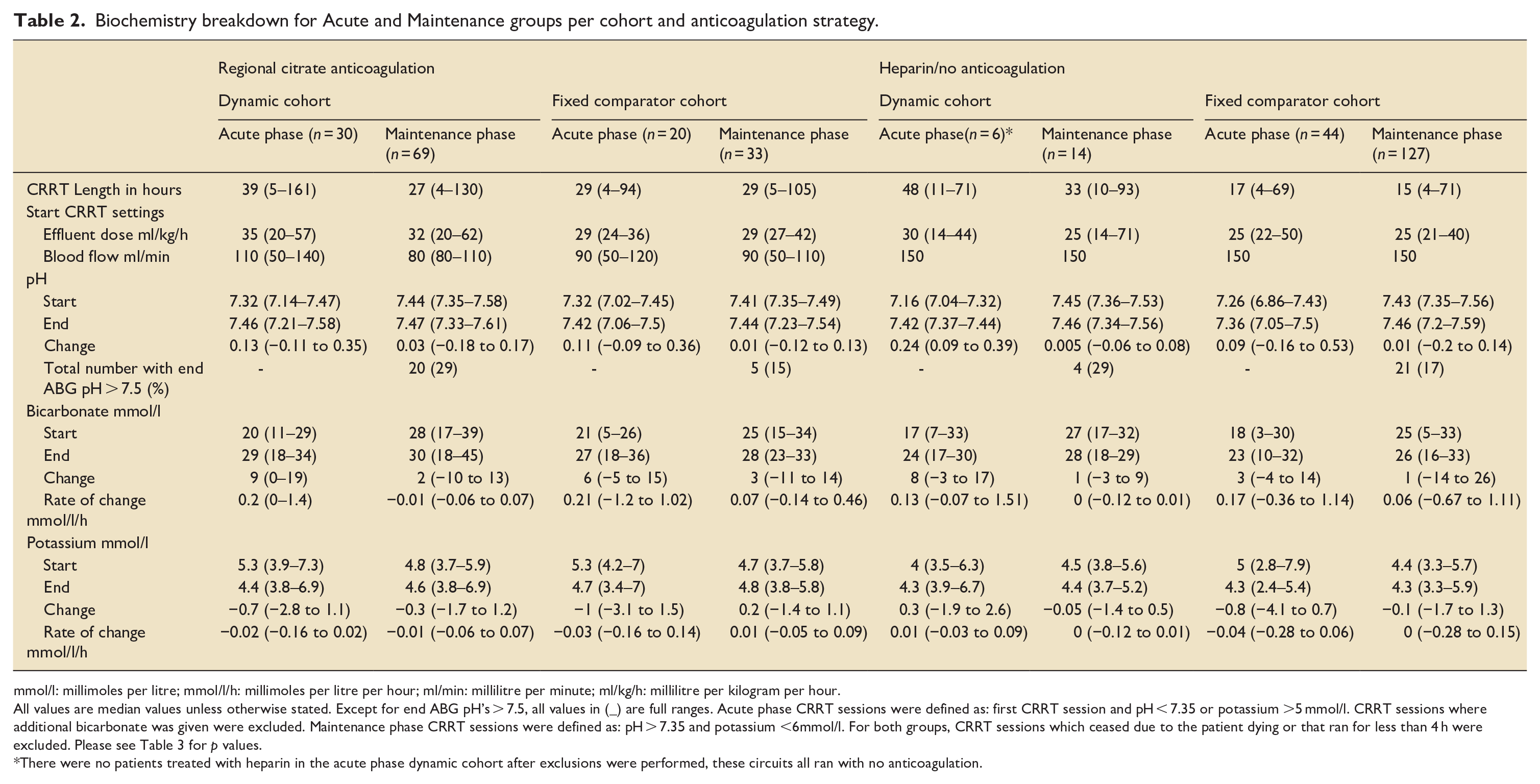
mmol/l: millimoles per litre; mmol/l/h: millimoles per litre per hour; ml/min: millilitre per minute; ml/kg/h: millilitre per kilogram per hour.
All values are median values unless otherwise stated. Except for end ABG pH’s > 7.5, all values in (_) are full ranges. Acute phase CRRT sessions were defined as: first CRRT session and pH < 7.35 or potassium >5 mmol/l. CRRT sessions where additional bicarbonate was given were excluded. Maintenance phase CRRT sessions were defined as: pH > 7.35 and potassium <6mmol/l. For both groups, CRRT sessions which ceased due to the patient dying or that ran for less than 4 h were excluded. Please see Table 3 for p values.
There were no patients treated with heparin in the acute phase dynamic cohort after exclusions were performed, these circuits all ran with no anticoagulation.
Dynamic cohort: Acute versus maintenance phase
The pH and bicarbonate in the acute phase CRRT sessions increases whilst in the maintenance patients it is kept relatively static (RCA Change in pH: 0.13 vs 0.03, p < 0.05 and bicarbonate rate mmol/l/h: 0.2 vs −0.01, p < 0.05). A similar pattern is seen for potassium reductions (Table 2).
Acute phase: Dynamic cohort versus fixed cohort
The rate of increase of bicarbonate was similar in the dynamic cohort compared to the fixed cohort (RCA: 0.2 mmol/l/h vs 0.21 mmol/l/h, ns, Heparin/No anticoagulation: 0.13 mmol/l/h versus 0.17 mmol/l/h, ns) (Table 2).
Maintenance phase: Dynamic cohort versus fixed cohort
Higher starting pHs are seen for the dynamic cohort. This may have resulted in the higher median end pH, or the greater number of end ABG pHs > 7.5, seen in the dynamic versus fixed cohort (RCA: 20 (29%) vs 5 (15%), ns). Of note, three of the CRRT sessions with a pH > 7.5 in the dynamic cohort were noncompliant or given additional bicarbonate. The bicarbonate rate of change was near zero for the dynamic cohort and slightly positive for the fixed cohort (RCA −0.01 mmol/l/h vs 0.07 mmol/l/h p < 0.05, Heparin/No anticoagulation −0 mmol/l/h vs 0.06 mmol/l/h, p < 0.05). There was also a greater rate of potassium reduction in the dynamic RCA cohort than the fixed cohort (−0.01 mmol/l/h vs 0.01 mmol/l/h, p < 0.05) (Table 2).
Dynamic group pH over time
The dynamic cohort corrects pH in a curvilinear fashion with steeper changes initially, when patients are typically more acidaemic, which become more gradual as treatment enters a maintenance phase. It also shows pH is maintained in a safe range after therapy is established. Comparison with the fixed cohort was not possible, as most data for CRRT sessions only had a start and finish ABG recorded (Figure 2).
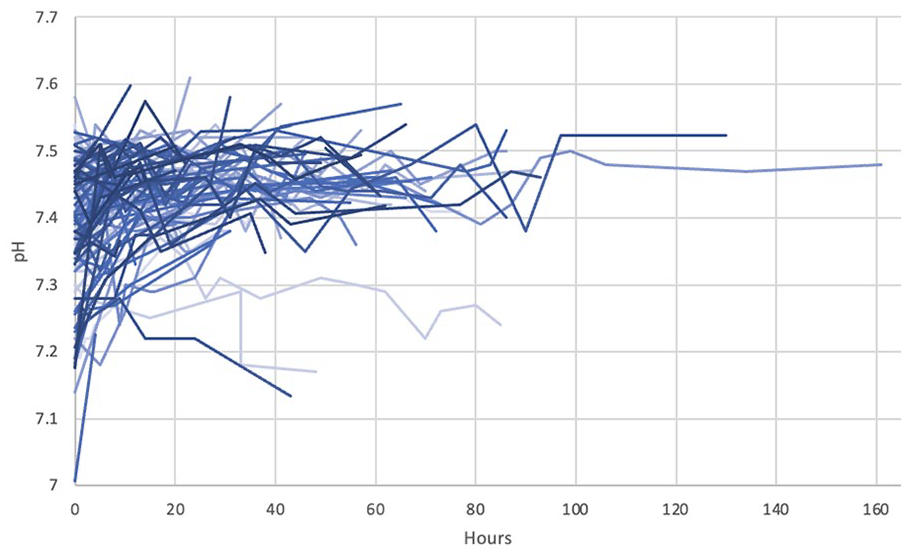
Dynamic cohort pH results plotted against time for CRRT circuits which used RCA (n = 109). Each line refers to a different CRRT run, these have differing darkness or lightness to aid delineation.
Table 3 lists the specific p values for Table 2. This are given for transparency and should be used descriptively. The reader is cautioned in over interpreting these and reminded that this paper is primary exploratory and hypothesis generating. All results need further ratification in studies that are suitably designed, powered and prospective.
All p values for acute and maintenance group comparisons.
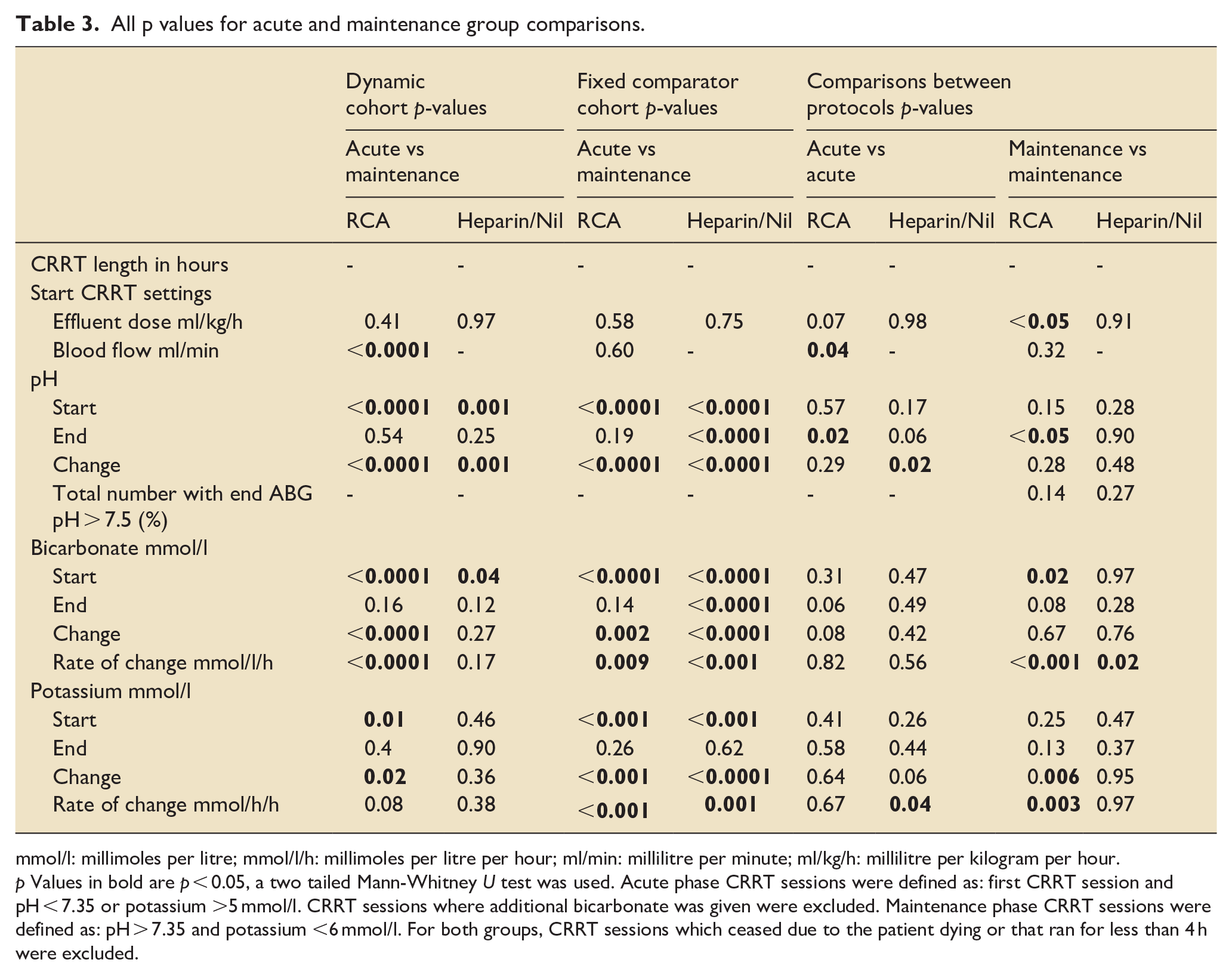
mmol/l: millimoles per litre; mmol/l/h: millimoles per litre per hour; ml/min: millilitre per minute; ml/kg/h: millilitre per kilogram per hour.
p Values in bold are p < 0.05, a two tailed Mann-Whitney U test was used. Acute phase CRRT sessions were defined as: first CRRT session and pH < 7.35 or potassium >5 mmol/l. CRRT sessions where additional bicarbonate was given were excluded. Maintenance phase CRRT sessions were defined as: pH > 7.35 and potassium <6 mmol/l. For both groups, CRRT sessions which ceased due to the patient dying or that ran for less than 4 h were excluded.
Discussion
In this service evaluation we examined the safety and effectiveness of implementing a dynamic dosing CRRT protocol. Our descriptive results suggest that the dynamic protocol has comparable safety to a fixed dose protocol, in terms of dose delivery, CRRT lifespan and solute control.
CRRT lifespan is a key quality indicator for CRRT delivery. 11 Other centres have reported continuous veno-venous haemodialysis (CVVHD) with RCA lifespans to be around 45 h.12 –14 Our median of 56 h for this modality exceeds this benchmark. Other evidence shows CRRT doses of 20–40 ml/kg/h are safe,4,7,15 we can see the machine set doses (Table 1, Figure 1) are almost always within this range, which correspond to the prescribed target doses from dynamic protocol (see Supplemental Appendix).
Solute clearance is another key quality indicator of CRRT safety and efficacy.11,16 Dosing in the dynamic protocol vary logically with biochemical state during the acute and maintenance phases. In the acute phase of illness, the dynamic protocol had a similar increase of bicarbonate, but importantly, this was near zero in the maintenance phase patients where treatment induced metabolic alkalosis may be a hazard (Table 2, Figure 2). Many institutions may in some instances deliver a de facto dynamic dosing regimen through individual clinician’s practice. However, as far as we are aware this is the first systematic application of dynamic dosing for CRRT in a protocolised fashion. KDIGO guidelines and international consensus statements suggest that CRRT dosing should be frequently assessed and patient specific.10,17 The dynamic protocol adheres to this moving CRRT in the direction of precision medicine with more systematic individualised solute control.18,19
Our institution is a major trauma centre, here a significant proportion of patients have a renal injury trajectory characterised by a high need for CRRT early post injury, which can become chronic necessitating prolonged periods of CRRT on the ICU. This trauma population across an ICU stay can need substantially varied CRRT dosing. We feel the data described here helps illustrate how a dynamic dosing strategy could offer systematically individualised dosing and solute control, better managing groups with this kind of trajectory. This is particularly important in a maintenance phase where higher dosing than necessary can cause hypophosphataemia, alkalosis, CRRT dependence and may potentiate harmful inflammatory mediators.4 –6,9,20 These biochemical complications can cause downstream clinical effects limiting the rehabilitation potential of patients.20,21 Separate to the clinical issues, with higher dosing CRRT, there are additional resource and economic consequences. An economic analysis from the United States found that CRRT costs can increase by 15%–25% when using 35–40 ml/kg/h versus 20 ml/kg/h. 22 Overall CRRT dosing in the dynamic group was similar when compared to historical controls, solute control however was notably different in the maintenance phase. This could suggest at an individual level dose is being tapered when maintenance is required and that the overall averages are being affected by higher initial dosing in the acute period, when metabolic and biochemical derangements are more severe. Further data, perhaps compared in a head-to-head fashion, is needed to better characterise the machine set dose in these maintenance patients.
Rates of alkalosis were higher in the dynamic group (Table 2). However very few patients needed to switch anticoagulation and differences here were also non-significant. Rates of bicarbonate change are near zero in the maintenance phase in the dynamic protocol, but still positive in the fixed protocol. Therefore, these differences may be due to differences in respiratory effort and baseline pH, rather than the effects of the dynamic protocol which is appropriately combating bicarbonate accumulation. Nonetheless this divergence warrants further study and there may be further optimisations that can be made to the dynamic protocol.
Recorded compliance was greater with the dynamic cohort than with the fixed cohort (76%vs 61%, p < 0.05). This finding is also supported by the fact that the machine set doses recorded elsewhere (Figure 1) correspond well to the prescribed target doses recommended by the protocol (see Supplemental Appendix). Whilst our compliance assessment is a blunt and semi-qualitative metric it can serve to generate new hypotheses and can give some suggestion of critical care staff’s intent to follow the protocols. A fixed protocol may appear simple, however troubleshooting worsening acidosis or alkalosis is complex and requires a deep understanding of how RCA-CRRT parameters affect acid-base parameters. The dynamic protocol simplifies this, as it recommends blood flow and dialysate rates for any given range of potassium and pH. Although the dynamic protocol involves more variation in treatment, we believe it is more logical to follow and clinical teams may have had more confidence in its individualised recommendations. This would need further investigation to confirm and data here is insufficient to suggest this as a causative finding. An improved way of assessing of compliance in future studies could be achieved by measuring total hours during which machine set dose matched the prescribed target dose. 10 This would increase robustness and data granularity.
Overall, our results suggest that the dynamic protocol rationalises therapy to biochemistry. It tapers its dose as time goes on while maintaining dose in a best-practice range. Tailored outcomes were recorded for different subgroups. Finally, an excellent filter lifespan was maintained along with at least non-inferior, if not improved, bedside protocol compliance.
Limitations
This study has several limitations given its exploratory design as a preliminary safety and efficacy study. A prospective and controlled study would be needed to confirm the observed differences, seen in the rates of maintenance-phase change in bicarbonate, between the protocols.
Studies with larger populations may need to account for CRRT being used in rarer situations such as severe hyponatraemia or for toxicology treatment. These patients were not specifically excluded in our dataset but did not appear in significant numbers. One patient in our dynamic dataset was recorded as having a high machine set dose as part of toxicology treatment, the relevant CRRT run was coded as non-compliant. Future studies may wish to consider strategies for managing analysis of these special cases, for example excluding them altogether a priori or separating them into subgroup analyses.
The definitions used for the maintenance phase (pH > 7.35 and potassium <6 mmol/l) and acute phase (first CRRT session, pH < 7.35 or potassium >5 mmol/l) are arbitrary, but were chosen in context with the dosing specifications for each protocol. They overlap and so would tend towards making the populations more alike each other and therefore bias towards the null hypothesis. Despite this underestimation there were still notable biochemical differences recorded for within protocol comparisons. Similarly, many patients were not severely acidaemic (Figure 2) or severely hyperkalaemic on commensal of CRRT (Table 2). CRRT was started as per the clinician’s discretion; many of these patients would be being filtered for fluid overload or uraemia, in addition there will also be a reasonable number of critically ill chronic dialysis patients (25%–30% Table 1) who may have CRRT commenced on the basis of predicted rather than present metabolic disturbance. Using CRRT for these other indications not recorded in our outcomes dilutes our sample of outcomes of interest. However again, this would bias towards the null hypothesis and the biochemical differences, such as those seen in rates of change of bicarbonate in maintenance phase groups, would be likely be an underestimate. Given this, a genuine difference is still suggested by the data.
It is possible ‘non-complaint’ fixed dose CRRT runs constitute ad hoc and non-systematic dynamic prescribing to achieve metabolic goals. This might have diminished difference in metabolic control between groups but would on the other hand strengthen an argument towards protocolising dynamic prescription to ensure quality. However, Table 1 still shows significantly different dosing and numbers of CRRT changes between groups.
Heterogeneity exists between populations in terms of primary diagnosis and age. However, this should not majorly affect our primary outcomes of interest, namely biochemistry, dose delivery and filter lifespan. Whilst we used consecutive analysis to reduce selection bias, populations were unmatched and data was collected at different timepoints. Additionally, this protocol may not be easily transferrable to ICU settings outside the UK. In the UK CRRT is delivered by an ICU nurse in a 1:1 fashion in conjunction with dynamic management of other critical care therapies. The dynamic protocols requirements for regular input in changing settings may have different implications in expert versus integrated CRRT service designs. Further investigation would be required for other international environments where external nurses or other clinical teams manage CRRT in the ICU. Similarly, our dataset exclusively examines the use of CVVHD as CRRT, therefore any conclusions drawn may not apply to other modalities of CRRT.
To increase robustness of future studies urea clearance, rates of hypophosphataemia, consumable use, and time to biochemical normalisation should be evaluated.
Conclusion
A dynamic dosing CRRT protocol when compared to a fixed dose protocol is comparable in terms of lifespan, safe dose delivery while potentially better tailoring therapy to the biochemical indications for CRRT. Dynamic dosing, through potentially providing more precise and individualised solute control, could represent a new approach to further optimise CRRT delivery in the ICU.
Supplemental Material
sj-docx-1-inc-10.1177_17511437231202898 – Supplemental material for Dynamic dosing for continuous renal replacement therapy: Service evaluation of the safety and effectiveness of titrating dose to biochemistry
Supplemental material, sj-docx-1-inc-10.1177_17511437231202898 for Dynamic dosing for continuous renal replacement therapy: Service evaluation of the safety and effectiveness of titrating dose to biochemistry by Jack C Eldridge, Aroon Bhardwaj Shah, Susana Lucena-Amaro, Christopher J Kirwan, John R Prowle and Yize I Wan in Journal of the Intensive Care Society
Supplemental Material
sj-jpg-2-inc-10.1177_17511437231202898 – Supplemental material for Dynamic dosing for continuous renal replacement therapy: Service evaluation of the safety and effectiveness of titrating dose to biochemistry
Supplemental material, sj-jpg-2-inc-10.1177_17511437231202898 for Dynamic dosing for continuous renal replacement therapy: Service evaluation of the safety and effectiveness of titrating dose to biochemistry by Jack C Eldridge, Aroon Bhardwaj Shah, Susana Lucena-Amaro, Christopher J Kirwan, John R Prowle and Yize I Wan in Journal of the Intensive Care Society
Supplemental Material
sj-jpg-3-inc-10.1177_17511437231202898 – Supplemental material for Dynamic dosing for continuous renal replacement therapy: Service evaluation of the safety and effectiveness of titrating dose to biochemistry
Supplemental material, sj-jpg-3-inc-10.1177_17511437231202898 for Dynamic dosing for continuous renal replacement therapy: Service evaluation of the safety and effectiveness of titrating dose to biochemistry by Jack C Eldridge, Aroon Bhardwaj Shah, Susana Lucena-Amaro, Christopher J Kirwan, John R Prowle and Yize I Wan in Journal of the Intensive Care Society
Footnotes
Author contributions
Susana Lucena-Amaro, Christopher Kirwan and John Prowle designed and developed the dynamic dosing protocol. Christopher Kirwan led the collection of data for fixed cohort. Jack Eldridge co-led with Aroon Bhardwaj Shah on data collection for the dynamic cohort. Jack Eldridge, assisted by Aroon Bhardwaj Shah and advised by Christopher Kirwan, John Prowle and Yize Wan, synthesised and analysed the data. Multiple discussions were held between all authors regarding overall analysis and write up of the manuscript. Jack Eldridge was the primary author of the manuscript with significant input and edits made by all other listed authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jack Eldridge, Aroon Bhardwaj Shah, Yize Wan and Susana Lucena-Amaro have no conflicts of interest to declare. Christopher Kirwan has received speaker fees from Baxter Inc, B. Braun Medical Ltd, Fresenius Medical Care and Nikkiso. He has received an unrestricted educational grant from Nikkiso and consultancy fees from Nikkiso and B. Braun Medical Ltd. John Prowle has received hospitality from B. Braun Medical Ltd. and speakers fees from Fresenius Medical Care, Nikkiso Europe GmbH and Baxter Inc. He has performed paid consultancy for Nikkiso Europe GmbH, Baxter Inc and Jafron Biomedical Co Ltd and has received an unrestricted research grant from Jafron Biomedical Co Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B. Braun Medical Ltd have offered to fund open access fees on publication of this manuscript. BBraun played no part in the design or analysis of the research. There were no other external funding sources.
Statement of ethics
Ethical approval is not required for this study in accordance with national guidelines. Retrospective analysis of anonymised routine data by members of the direct care team did not require direct patient consent. This was a service development project which was registered as two audits.
Data availability statement
All data generated or analysed during this study are included in this article. Please direct any further enquires to the corresponding author, limited and anonymised datasets may be able to be provided on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
