Abstract
Methods of continuous renal replacement therapy (CRRT) in extracorporeal membrane oxygenation (ECMO) patients include dedicated central venous cannula (CVC) (vCRRT), in-series with filter connected to ECMO circuit (eCRRT) or in-line with haemodiafilter incorporated within ECMO circuit. We assessed the efficacy and safety of eCRRT versus vCRRT in 20 ECMO-CRRT patients. Average filter lifespan was 42 vs 28 hours and filter runs completing 72hours were 40% vs 13.8% (eCRRT vs vCRRT, respectively). One incidence of ECMO circuit air embolus occurred (vCRRT). eCRRT achieved adequate filtration and increased filter lifespan, and has become our default for ECMO-CRRT if a pre-existing dialysis CVC is not present.
Keywords
Introduction
ECMO refers to an external mechanical device providing cardiorespiratory support, permitting time for recovery or bridging to definitive treatment. 1 Critical illness is frequently associated with acute kidney injury (AKI), leading to fluid, electrolyte and acid–base imbalance. As injury progresses, life-threatening complications develop comprising severe metabolic acidosis, resistant hyperkalaemia, pulmonary oedema and uraemia, necessitating CRRT.
CRRT is an established therapy providing fluid, electrolyte and acid–base optimisation. CRRT filter lifespan varies with filter type, membrane, mode, vascular access, flow rates, anticoagulation and individual patient factors. Studies have found varying median filter lifespans, ranging from 14.3 to 21.9 hours, with connections to ECMO circuits trending towards longer lifespans compared to direct vascular access.2,3
Traditionally, a dedicated CVC enables CRRT delivery (vCRRT). Increasingly, alternative configurations include direct introduction of the haemofilter into the ECMO circuit, or integrating a CRRT device into the ECMO circuit (eCRRT). Theoretically, these mitigate some CVC complications, for example, lack of suitable vessels. However, without careful consideration of CRRT placement, large intra-circuit pressure differences may affect filtration. We assessed the efficacy and safety of implementing eCRRT for veno-venous ECMO (VV-ECMO) patients in our Adult Intensive Care Unit.
Methods
A prospective service evaluation of 20 consecutive ECMO patients requiring CRRT was undertaken (May–September 2019). Inclusion criteria included age >18, VV-ECMO and CRRT requirement. The primary outcome was to assess the safety of eCRRT compared to vCRRT, observing adverse outcomes, to deem whether a change of CRRT placement is safe and beneficial. Secondary outcomes included completion of therapy, incidence of complications and filter lifespan.
Data was collected from the Philips Clinical Information System, from the first four filters used per patient (configuration, lifespan, anticoagulation and cessation reason). Additional data included baseline demographics, underlying diagnosis, ECMO information, APACHE scores and adverse events.
Patients received ECMO using a CentriMag™ system with a Medos Hilite oxygenator® or Maquet Cardiohelp™. Pigtail catheters were used to access the ECMO circuit with eCRRT (Figure 1). This configuration minimises the potential impact of an air embolism via air entrapment in the oxygenator, whilst being cognisant of the negligible recirculation occurring. Systemic anticoagulation for ECMO was heparin (first-line) or argatroban, with monitoring by serum anti-Xa and APTT respectively. CRRT was via the Gambro Prismaflex system, with effectiveness defined as the first four filters lifespan. Schematic representing how we, at our institution, combine CRRT within the VV-ECMO circuit, with CRRT access occurring post-oxygenator and return pre-oxygenator via pigtails. CRRT, continuous renal replacement therapy; VV-ECMO, veno-venous extracorporeal membrane oxygenation.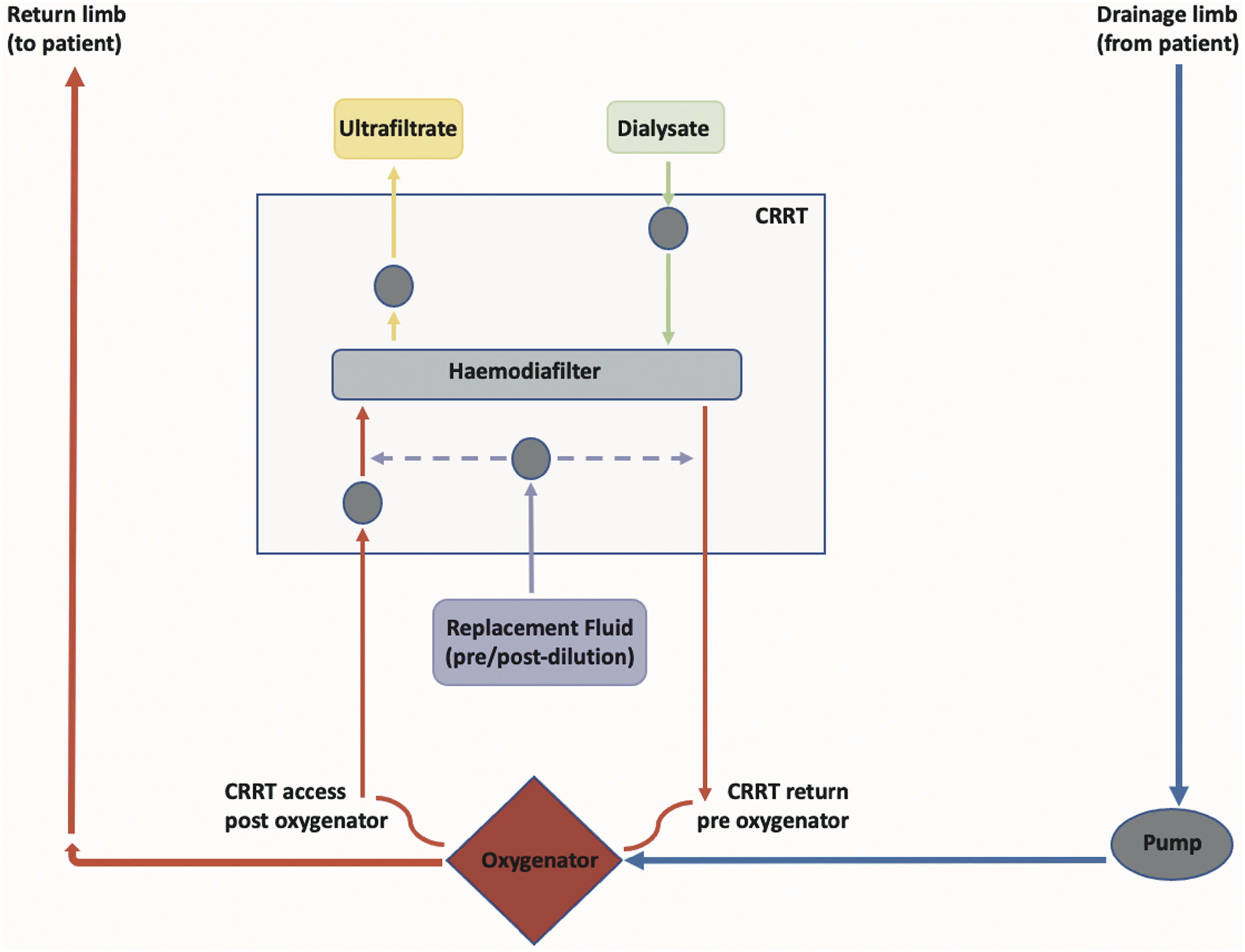
Results
Results of our service evaluation project comparing eCRRT vs vCRRT, including patient demographics, APACHE scores, ECMO data and complications, CRRT filter data and anticoagulation strategies.

Discussion
Our small project demonstrated that eCRRT is safe and effective. There was one significant ECMO related adverse event; an air embolism in the vCRRT cohort. Whilst the exact cause was not ascertained, the possibility of air entrainment during CVC-RRT attachment is not to be overlooked. eCRRT was not associated with an increased incidence of adverse events, nor was filter lifespan affected.
Instigating CRRT whilst on ECMO via a pre-existing dialysis CVC is practical. However, if insertion of a CVC is necessary, this poses increased risks secondary to systemic anticoagulation. 1 Furthermore, access sites are limited due to pre-existing CVC/ECMO cannula. Regardless of the eCRRT configuration, the need for CRRT anticoagulation will be reduced/eliminated due to ECMO-related systemic anticoagulation.
Incorporation of a CRRT device into the ECMO circuit is an increasingly used approach. A multi-centre international survey reported 21.5% centres exclusively using an in-line haemodiafilter, with further 50.8% centres exclusively using a CRRT machine connected to the circuit. 4 This in-series approach increases the duration of efficient CRRT therapy, compared with independent vascular access 5 and increases fluid balance accuracy during ECMO. 6 One study demonstrated an increase in CRRT lifespan in ECMO patients when CRRT was connected in-series rather than an independent CVC, due to better flow rates rather than anticoagulation regimens. 7
The disadvantage of our eCRRT configuration is the potential for recirculation and reduced efficacy. Additionally, regional citrate anticoagulation is not possible due to the lack of safety studies and inaccuracy/practicality of calcium compensation via a syringe driver to the high-pressure system. Future endeavours could consider systemic calcium supplementation. Cost and staff training are further issues.
ECMO-CRRT combined can increase complications. Increased haemolysis from erythrocyte fragmentation secondary to shear stress, positive pressure, wall impact forces, and non-endothelialised surfaces can occur. 8 Higher peak levels of plasma-free haemoglobin can arise compared to ECMO alone, 6 with potential adverse effects on multi-organ function.
Combining CRRT within the ECMO circuit is safe and achieves adequate CRRT. This approach has become our default for patients without a dialysis CVC in situ. We would stress that this approach is not standardised and depends on ECMO centres experience and expert opinion. We welcome communication from other centres on their experience.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
