Abstract
Digital health refers to the field of using and developing technology to improve health outcomes. Digital health and digital health interventions (DHIs) within the area of intensive care and critical illness survivorship are rapidly evolving. Digital health interventions refer to technologies in clinical interventional format. A DHI could support clinicians with increasing clinical demands to have improved oversight of their patients’ recovery trajectory or potential for deterioration, improve efficiency of healthcare delivery, and/or predict patient outcomes. In this narrative review, DHIs are explored across the continuum from in the ICU (recognising and managing clinical deterioration, identifying individuals at risk of poor recovery outcomes, tailoring care of the ICU patient and supporting the emotional needs of their family) through to integration in the primary care setting (adjuncts to ICU follow-up clinics and tracking, coaching and remote monitoring). Some of the DHIs discussed in this narrative review (to name a few) include interventions delivered via: Telehealth, artificial intelligence, wearable devices, virtual reality, and mobile phone applications (apps). Additionally, exploration of DHIs used successfully in other health fields are discussed to highlight potential opportunities for adaptation to the ICU context. Finally, the review provides an overview of considerations needed in the development of new DHIs. Development should consider the intended user, barriers to technology engagement and design. In the implementation of a new DHI, the World Health Organization (WHO) Global Strategy on Digital Health and appropriate evaluation should be considered prior to scaling up. Optimal implementation of DHIs could help address the key challenges of the ICU field.
Introduction
Digital health, as defined by the World Health Organization (WHO) is “the field of knowledge and practice associated with the development and use of digital technologies to improve health”. 1 Digital health interventions (DHIs) are a subset under the digital health umbrella and refer to technologies in clinical interventional format designed to facilitate healthy behaviours, enhance health outcomes, and deliver healthcare provision remotely. 2 Delivery of DHIs can occur in a variety of settings including within healthcare facilities, through government initiatives, in the community, workplaces, schools or an individual’s home. 2 Examples of DHIs include (but are not limited to) those delivered via: Telehealth, mobile phone applications (apps), virtual reality (VR), and instant messaging. 2 DHIs have burgeoned within the healthcare sector over the past decade, with the goal of improving patient care, and efficiency of health systems. 2 The proliferation and adoption of DHIs occurred rapidly during the COVID-19 pandemic where healthcare pivoted to alternative platforms. 3
Since the intensive care field first began in the 1950s, we have seen rapid advancements in medical care leading to growing population of survivors of critical illness alongside increasing demand for high acuity intensive care unit (ICU) level care. 4,5 However, survivors of critical illness report fragmentation in care when they leave hospital, 6 with many suffering from Post-Intensive Care Syndrome (PICS) impairments. 7 Patients desire support targeted to their individualised needs to help support them through the recovery journey given the heterogeneity of symptoms experienced. 6 Their families, who are also at-risk of being affected by PICS-family (PICS-F), 8 desire emotional support and to be engaged in their loved one’s care throughout the ICU 9 and recovery journey.10,11 Whilst not all DHIs are have validated or proven to improve outcomes, there are many promising DHIs which could optimise survivorship care.
The purpose of this narrative review is to discuss recent DHIs that have been implemented in ICU and/or recovery after critical illness as part of survivorship care. Digital health interventions are highlighted according to key topics along the continuum of care. Other DHIs within different healthcare settings or health conditions are also presented as considerations for new opportunities for the ICU field.
Table 1 provides a summary of the key definitions in relation to the digital health field. Figure 1 summarises the potential and currently used DHIs across the continuum of ICU survivorship care.
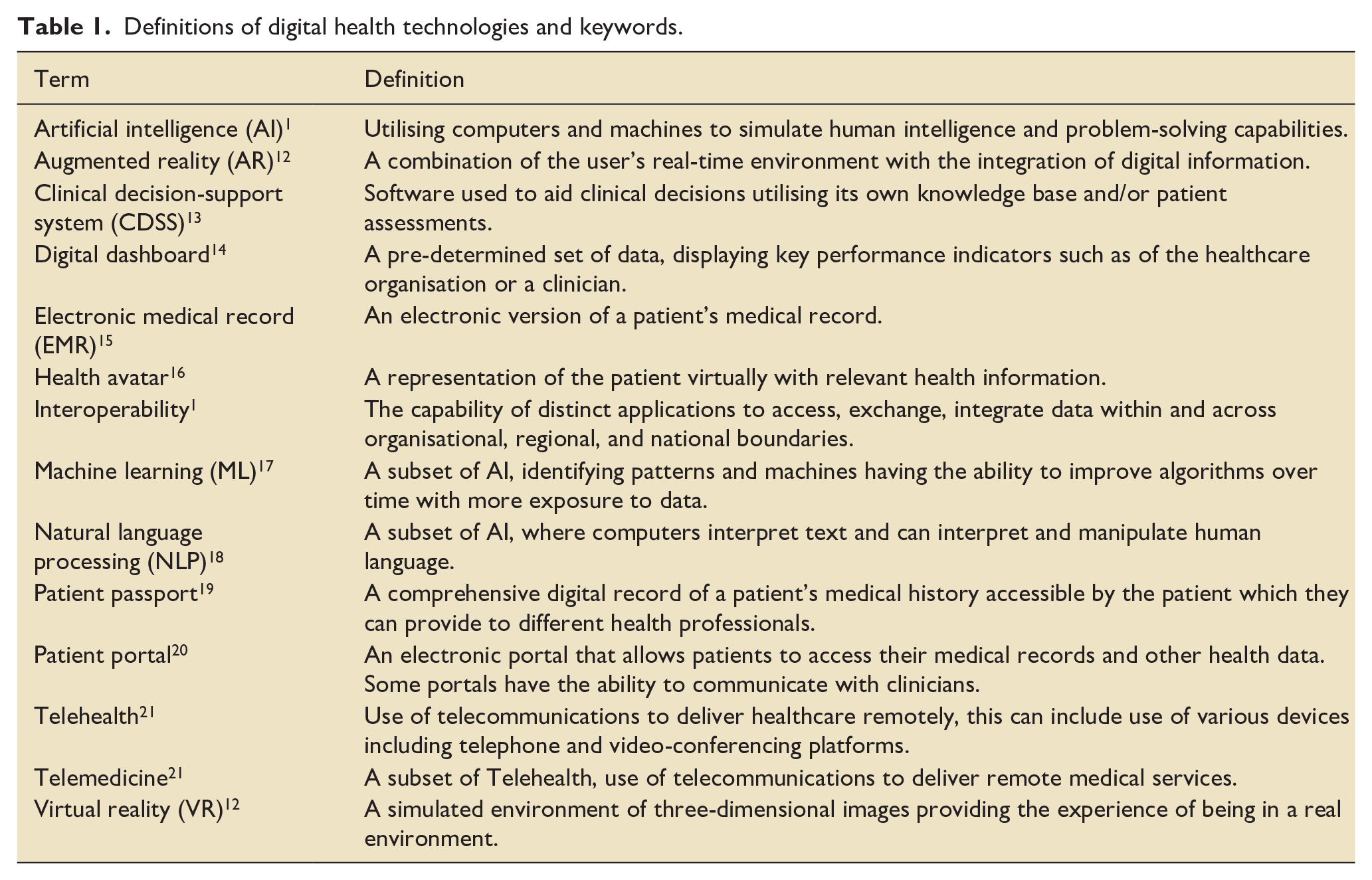
Definitions of digital health technologies and keywords.
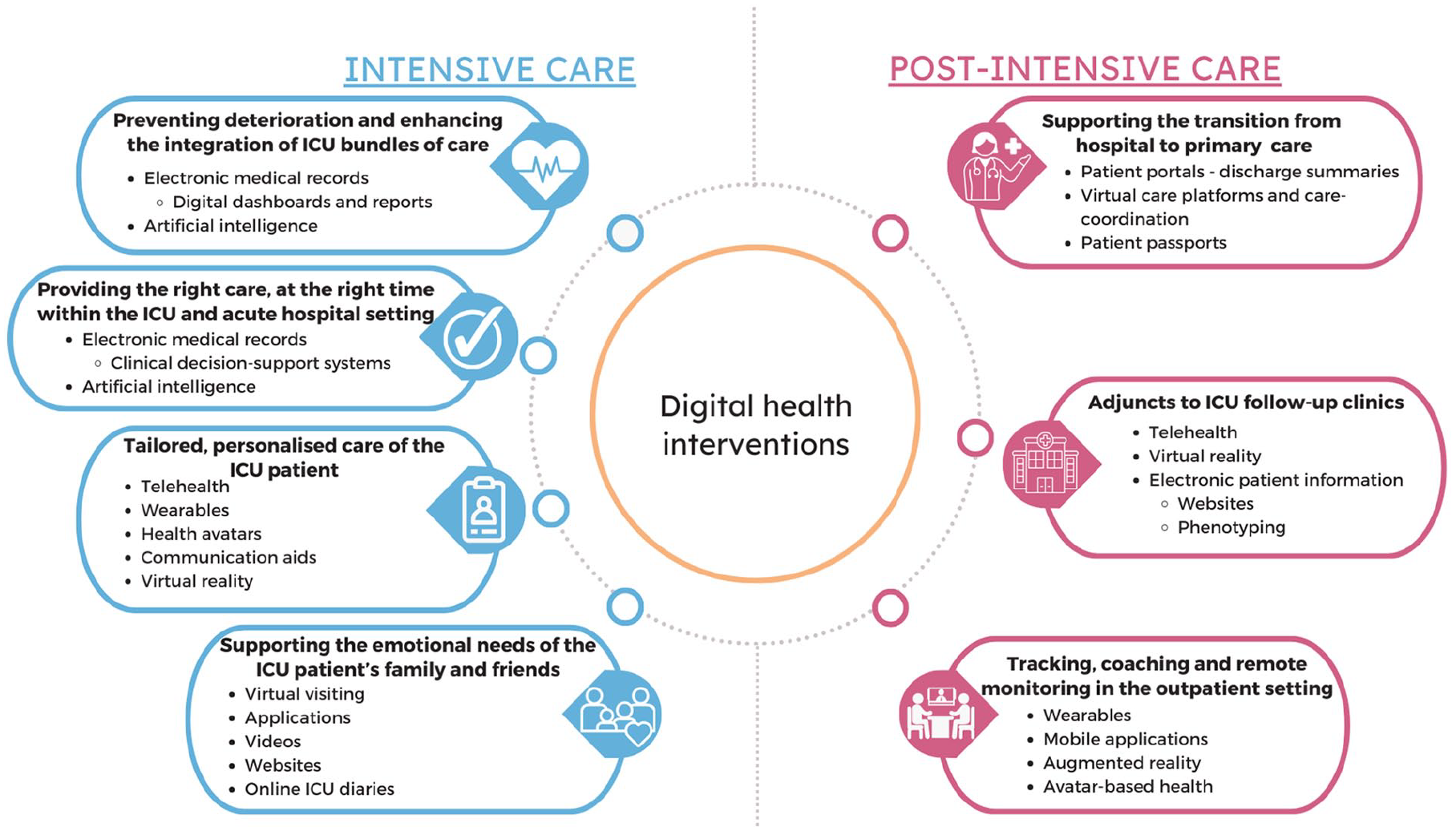
Digital health intervention applications across the continuum of care.
Preventing deterioration and enhancing the integration of ICU bundles of care
Electronic medical records (EMR) enable the storing of patient admission data electronically. 15 The use of EMR has improved patient care, including reducing laboratory and radiological tests performed and medication errors in hospital settings. 15 Thereby also presenting economic benefits. 15 The EMR presents the opportunity to create custom digital dashboards and performance metrics reports. 14 A digital dashboard can relay a pre-determined set of data, such displaying data trends determine if a patient is deteriorating. 14 However, challenges with effective dashboard and report use include the reliance on accurate data entry on the system and clinician’s capability to utilise data dashboards to their full potential. 14 In the implementation of the ICU Liberation (ABCDEF) bundle, which is recommended to improve outcomes of critically ill patients, a combination intervention including training clinicians in data literacy and access to digital dashboards has been trialled. 22 Increased compliance with the ABCDEF bundle and reduced likelihood of next-day ICU patient mortality were found with the intervention, noting that once clinicians understand the functionality of the EMR, it can prevent patient deterioration. 22
The collection of patient data electronically through EMR as well as in national registries and portals provides large datasets which can also be used to predict deterioration and risk. 23 These large datasets enable the ability to learn from past patient encounters to improve real-time service delivery by determining trends. 17 Artificial intelligence (AI) has the capacity to distil these big datasets. Using AI, means to employ computers and/or machines to simulate human intelligence and problem-solving capabilities. 1 Artificial intelligence has been used in various ICU studies to create risk prediction models of deterioration including prediction of sepsis and pressure injuries.24,25 The ability to determine factors that could lead to deterioration is then useful in identifying at-risk ICU patients in real-time.
Risk of deterioration can be further evaluated through previous patient data incorporated with new data as they are available. This form of analysis is called machine learning (ML), which refers to machines identifying patterns in data, creating algorithms, and improving over time with more exposure to data. 17 Machine learning has already been used to detect clinical deterioration in both ICU and hospital ward settings, 26 predict risk of ICU-acquired weakness (ICU-AW), 27 ICU readmissions, 28 and discharge destination needs. 29 Delirium is another potentially preventable risk in ICU which is usually determined by clinical assessment such as the Confusion Assessment Method-ICU (CAM-ICU). 30 Patient’s behaviour and cognition may also be recorded in progress notes however does not usually form part of formal delirium assessment. 30 A study by Young et al. reviewed the content of progress notes to diagnose delirium via computers. 30 This method is called natural language processing (NLP) and refers to a branch of ML where computers interpret text and manipulate human language. 18 Incorporating demographics, prescribed antipsychotic medications and progress notes, NLP identified 54% of patients as having a behavioural disturbance compared with 25% being assessed clinically as CAM-ICU positive by clinicians. 30 Furthermore, the patients identified as having a behavioural disturbance by NLP required significantly longer ICU and hospital stays, and had greater in-hospital mortality compared with those not identified. 30 Natural language processing can analyse many datapoints to detect a behavioural disturbance compared with the clinical measure of the CAM-ICU which assesses an isolated timepoint. Digital health interventions may be able to identify at-risk patients in a more efficient manner to enable more targeted interventions to support clinical care.
Providing the right care, at the right time within the ICU and acute hospital setting
The use of DHIs can assist in ensuring that care is provided at the right time. The EMR can help facilitate this with programmed alerts and tailored reminders. Clinical decision-support systems (CDSS) are software that utilise data entered into an EMR with its own knowledge base to assist clinicians in point-of-care decision making. 13 A key area where CDSS has been used in ICU care is preventing medication errors for example, alerting a clinician at the point-of-care of a patient’s allergies. 31 Furthermore, in the case of delirium, a bedside anti-delirium CDSS-assisted tool is being compared to usual care to identify and develop delirium management plans. 32 The tool completes the CAM-ICU and a delirium risk factor assessment, then generates a nursing care plan and activity checklist in the hopes of reducing delirium frequency. 32 Despite the positives of CDSS implementation, some criticisms and shortfalls include disrupting clinician workflow, alerts appearing at inappropriate times, excessive alerts, increased reliance on alerts affecting clinical practice, clinician digital literacy and CDSS maintenance requirements. 15 Nevertheless, CDSS presents the opportunity to program alerts for patients if they meet a certain criteria to optimise care delivery.
Delivering of the right care at the right time can further be assisted by the application of ML into clinical practice. Medical imaging is a key area where ML has been used in ICU to improve clinician bedside interpretation by facilitating quick analysis including lung and muscle ultrasound.33,34 Such DHIs that assist with medical imaging, could be essential to monitor changes in muscle characteristics and presence of ICU-AW. This is particularly beneficial for non-experts of image analysis. 34 Another ML model that has been implemented in clinical practice assists in determining optimal extubation time. 35 Models such as this one, could not only optimise patient care through shorter intubation times but could also result in cost savings from an ICU operational perspective. As some models have already been proven efficacious at the patient’s bedside, there is a window of opportunity for future ML ideas to improve clinical care directly.
Tailored personalised care of the ICU patient
The heterogenous nature of the ICU patient population necessitates assessments and interventions to be individually tailored. Most healthcare services are familiar with Telehealth, which is the use of telecommunications to deliver healthcare remotely utilising various devices including telephone and video-conferencing platforms.21,22 Telehealth provides the opportunity to tailor patient care remotely as it can facilitate remote communication with the patient as well as within the clinical team about the patient. 36 Telehealth also can to improve access to clinical expertise through virtual rounding, collaborative reviews, and remote review of patient assessments. 36 Virtual ICUs existed prior to the COVID-19 pandemic mostly for remote consultations for example, with rural hospital services. 37 However, there was an increase in usage of Telehealth in ICU during the COVID-19 pandemic, predominantly to facilitate communication with the ICU patient’s family and the healthcare team with the patient in the ICU. 38 Telehealth continues to be a valuable DHI in the ICU to conduct tailored, personalised consults, assessments and rounds remotely. 36
Care of the ICU patient can be further tailored by monitoring of their physiological parameters such as physical activity, vital signs, and sleep via wearables. Wearable technology is rapidly evolving in healthcare.39,40 Sensors can be placed in various sites including but not limited to the arm, chest, foot, hand, head, waist and wrist. 39 An example of a wearable device that measures rest and activity non-invasively is actigraphy.41,42 A sensor records gross movement data, then software interprets it into activity counts based on algorithms. A challenge with wearable devices including actigraphy is that many are not clinically validated, particularly for a hospitalised or critically ill patient.41 –43 Depending on the device and placement of the sensor, some wearables are not sensitive in capturing low activity levels, particularly activities that are bed or sitting-based which are relevant to critically ill patients.41,42 Some devices may also misestimate energy expenditure which may put the patient at risk of malnutrition if relied upon for nutritional intake. 44 A review of wearable technology for hospitalised patients found that heart rate and sleep parameters had the most compelling evidence for being valid in an inpatient population. 43 Wearable technology still requires validation, there is an opportunity for development but often unfortunately, many devices are superseded before they are validated in a clinical population. 45 Despite this, clinically validated wearable devices to record early mobilisation in ICU, sleep and vital signs could be useful to improve patient care for example, assess exercise response, determine personalised exercise prescription, and dose.
Tailoring of healthcare can also be supported via the use of health avatars. Health avatars are a representation of a patient virtually with health information and can be used to display data more visually. 16 An application of health avatar-based monitoring trialled in the ICU featured the patient’s vital signs conveyed as an avatar on monitoring screens. 46 Clinicians found this beneficial, citing the avatar’s holistic representation of the patient’s vital signs made interpretation easier and lowered perceived workloads. 47 Visual-based methods such as patient depiction via avatar may assist clinicians with quickly recognising abnormalities in vital signs and prompt them to investigate further.
Due to the ICU care interventions and effects of being critically ill, the patient may experience impacts such as communication difficulties, anxiety, and pain. 48 To improve communication for the patient, high-technology augmentative and alternative communication methods are often used. 49 These methods are mostly software applications through which the patient can communicate their feelings via pictures and options to draw or type. 49 Patients are also generally accepting of the technology to improve their communication challenges, ability to describe symptoms and express their thoughts. 49 Virtual reality enables a simulated environment of three-dimensional images providing the experience of being in a real environment, and has been used in short sessions in the ICU. 12 This includes to promote early mobilisation through gamification, and to prevent delirium through neurocognitive stimulation where patients are orientated, provided information, and reassured to relax through simulation. 12 Whilst promising as a DHI, VR technologies have only been trialled in small studies, with limited acceptability and implementation challenges in time taken to set up/run and clean/maintain. 12
Supporting the emotional needs of the ICU patient’s family and friends
During the COVID-19 pandemic, visiting restrictions prevented family from being at the bedside of their loved one. As an avenue for families to connect with the patient remotely, virtual visiting was introduced. Family reported barriers to virtual visiting included improper camera setup, lack of staff resources including technical skills, inadequate connectivity and often the lack of call closure. 50 Despite this, family perspectives of the virtual visiting were generally positive including enhancing family involvement in care. 50
As families desire to be involved in patient care, empowering them through informing them about ICU procedures and interventions has been explored. 11 Several interventions are focused on the family including software applications (accessible on mobile, tablet or computer), education videos and websites. 11 These DHIs intend to improve family engagement and empowerment for example, participation in electronic ICU diaries. 51 Whilst it is promising to see the development of DHIs to support families and involve them in the patient’s care, an area which is not yet evaluated is the impact of these DHIs on PICS-F. It is also noted that in evaluating these interventions, many were not iteratively designed for example, incorporated feedback from families to improve the intervention design. 11 Participatory methods to improve user-design are increasingly recognised as best practice in the development of new digital technology to improve their suitability to the intended user. 11
Supporting the transition from hospital to primary care
After the patient has been discharged home, patients and families have emphasised the importance of continuity of care. 48 Many hospital services now have patient portals or virtual care platforms which allow patients access to their medical records including discharge summaries, see future appointments and/or provide an avenue to communicate with clinicians electronically. 20 One such DHI that is targeted specifically at ICU survivors includes a virtual care platform where they can identify recovery goals to work on after hospital discharge.52,53 The platform was found to be feasible and acceptable to ICU survivors. 54 Despite the intended benefits of portal use and virtual care platforms as mechanisms for improving continuity of care, it has been found that usage is particularly low in older people, ethnic minorities, lower socioeconomic status, lower health literacy, chronic illness, and disabilities. 20 These groups are already most vulnerable to not receiving proper care and therefore, a valid concern is that DHIs may be increasing the health inequality gap. 20
Another concern in ICU recovery is the fragmentation between hospital inpatient care and transition to primary care. 48 General practitioners (GPs) are often provided with inadequate discharge summaries from the treating hospital. However, in many countries, after hospital discharge GPs must undertake the main coordination of a patient’s care. 48 This is where DHIs such as electronic patient passports may have a role. Patient passports enable a patient to have access to their health record information electronically and are most commonly accessed via mobile devices. 19 An example was the accessibility of COVID-19 vaccine certificates via mobile applications. 19 The benefits of patient passports are that they not only allow the transmission of health record data to other clinicians, but importantly they also empower the patient to facilitate this process. 19 A review of the role of patient passports in primary care, found positive themes of ‘empowering the patient, helping with communication, improving relationships, improving the quality of care, maintaining health records, sharing records and saving time’. 19 Patient passports are a potential avenue to improve the transition from acute care, by patients providing GPs with timely information. However, the problem of providing direct feedback loops between acute care health care professionals and the practitioner still exists. 55
Adjuncts to ICU follow-up clinics
As a mechanism to follow-up patients, many hospital services utilise post-ICU clinics. Telehealth can diminish the geographical barrier some patients face between their residence and returning to the treating hospital outpatient service. Many have utilised or are using Telehealth particularly as video-based consults, and patients and caregivers are accepting of this modality in terms of convenience and efficiency. 56 The benefits of peer support for ICU survivors are recognised 57 and some follow up clinics run peer support groups. 58 Both hybrid and fully virtual models were trialled during the pandemic, finding that the amended model was not only feasible and acceptable but also had patient-reported benefits. 58
Follow up clinics have also been trialling the use of VR to facilitate virtual visiting and provide educational information about the ICU. 59 Vlake et al. randomised participants at their 3 month follow up clinic visit, into the VR intervention and had the opportunity to revisit the ICU environment with audio explanations. 59 The intervention was reported to be feasible and improve satisfaction of aftercare however did not change psychological outcomes for these patients. 59 Given these results, it may imply that patients are welcoming of virtually-delivered patient information.
Another potential adjunct to follow-up clinics is the provision of patient information electronically. There are already some ICU and PICS information websites hosted by ICU societies. 60 This provides a great opportunity to deliver targeted information to patients that are endorsed by high quality sources. However, amongst 127 webpages hosted by ICU and critical care societies, alarmingly, all the materials were found to be significantly higher than a 6th grade level reading level (typical 11-year-old student). 60 This highlights that despite good intentions of providing patients with information, many members of the community would not be able to interpret it. Additionally, we suggest considering tailoring web-based patient information, given the heterogeneity of the survivorship population. Whilst information can be easily transferred to online webpages, an area which could be developed is to phenotype survivors according to their health condition. Digital phenotyping is the ability to determine cohorts that have certain similarities. 61 This method has been already used for ICU patients however could be utilised in an outpatient setting to determine various phenotypes of ICU survivors and their common symptoms post-hospital. Phenotypes of cohorts that are most vulnerable are then known, their education needs can be understood by clinicians and information can be targeted.
Tracking, coaching and remote monitoring in the outpatient setting
After hospital discharge patients are susceptible to PICS and there is need to support their physical, mental and/or cognitive health. Facets of PICS could be managed through wearable technology and mobile applications in the outpatient setting. To address psychological distress, meditation has been embedded as a mobile phone application in research. 62 Unfortunately, with many technology studies, reliance is on the patient to have the right equipment. In this study, 6% of patients were excluded due to not having adequate technology or internet connection, meaning that certain technologies are inaccessible to some members of the public. 62 Despite this, there are also other avenues where mobile applications could be useful, extrapolating from other fields. Training and problem-solving applications have been used for mild cognitive impairment, Alzheimer’s disease, and dementia. 63 Whilst efficacy is generally reported as high, studies have variable outcomes, and therefore it is recommended that they need further evaluation. 63 Furthermore, to address physical impairments associated with PICS, applications may link to wearable devices to track movement and wirelessly transfer data for clinician review. This enables key parameters to be measured by the clinician to then help gauge how patients are physiologically coping and implement appropriate interventions to improve safety and outcomes. 64 In ICU survivors, most studies utilising wearables have shown that survivors are sedentary. 64 In conjunction with these wearables, an untapped potential in survivorship care is to embed augmented reality to improve physical function. 65 Augmented reality is the combination of the user’s real-time environment with the integration of digital information. 12 Augmented reality can be incorporated into a patients’ training regime whilst monitoring physiological parameters. 65 Whilst unknown if it is being used in the outpatient setting for ICU survivors this could enhance physical activity in the home environment.
Another mode of providing coaching or remote monitoring is avatar-based health. Uses of avatar-based health in other fields include the patient developing a non-interactional avatar based on their physical similarity to interact with health professionals, an avatar that can be used to interact with other patients in a simulated environment and an avatar that is a health coach who guides the patient in managing their illness. 66 Avatar-based health has been used in an outpatient setting for many chronic mental health conditions including cardiovascular disease, cancer and depression. 66 Evaluations of avatar use are also heterogeneous but there are reports of patient satisfaction and improvement in symptoms and pain. 66
Implementation challenges and future directions
When designing and implementing DHIs, many considerations are required to ensure proper engagement and implementation (Figure 2).
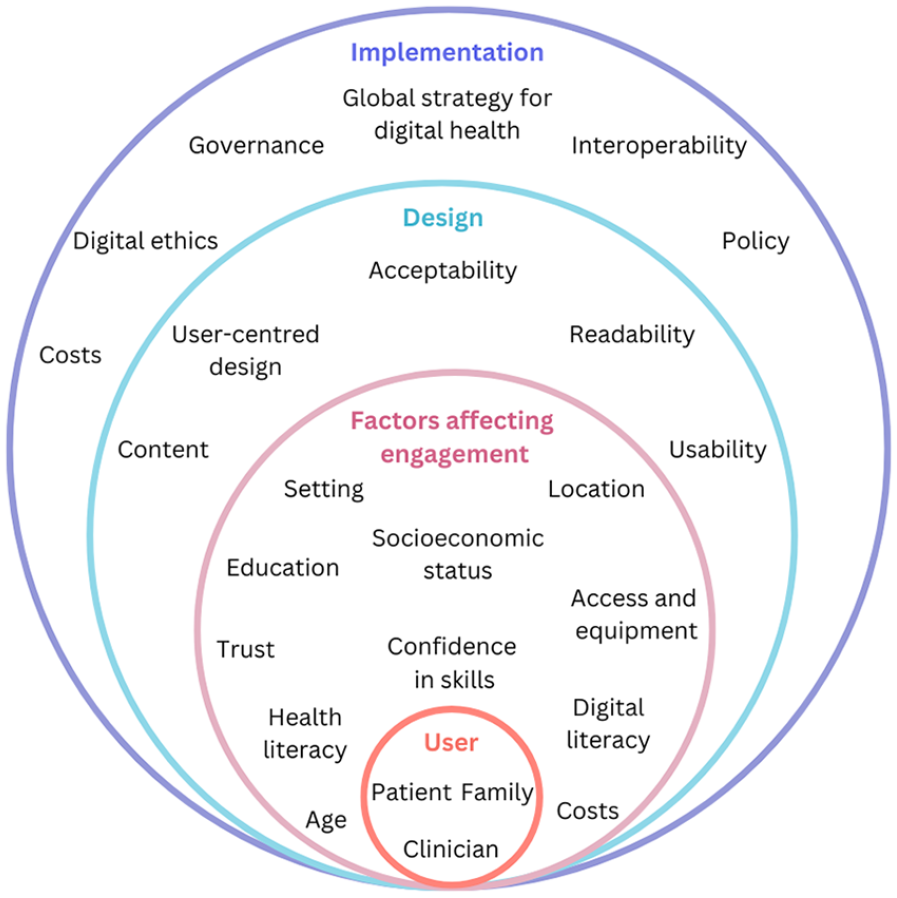
Key considerations in the development of DHIs in ICU and post-ICU care.
Firstly, from a user-level (patient, family, or clinician), it is pertinent to factor in the intended end-user. Health literacy is a multi-faceted; people need to have the ability to: (1) know where to access information, (2) understand the information, (3) evaluate whether or not it is of use to them, (4) communicate the information to others, and (5) use the information. 67 It is important that information is tailored as many adults have only basic levels of health literacy and education. 60 The effective use of DHIs also relies on digital health literacy. It has been found that less health literate patients are less likely to be technology literate and engage with DHIs. 68 There are also many social factors which may affect technology engagement and digital skills. This includes age, socioeconomic status, location, and trust in technology. 69 A digital divide is traditionally observed between older people compared to younger people in use of technology. 70 This divide also has meant that some older adults receive less benefit from DHIs. 70 Furthermore, access and costs of technology including devices and reliable internet connection may be barriers to uptake of DHIs. 62
To provide guidance on the development and implementation of DHIs, a socio-technical framework, named the non-adoption, abandonment, scale-up, spread and sustainability- complexity assessment tool (NASSS-CAT) could be useful. 71 Whilst this is just one example of an implementation framework, the NASSS-CAT serves to guide developers through the various stages of DHI implementation and scale-up. 71 A focus on implementation challenges also serves to determine if DHIs are going to fail to avoid unnecessary costs. 71 Similarly, a Framework of Implementability of healthcare interventions has been developed. 72 This framework considers the key components of implementation science; acceptability, fidelity, feasibility, and their effects on scalability and sustainability. 72 At the centre of many implementation studies is the emphasis on user-centred design. Involving users at various stages of development is thought to improve content, readability, and acceptability. 72 The framework recommends for this to be prospectively noted to determine what factors denote that an intervention should be scaled up. 72 It may also be beneficial to scope needs. For instance, in a study of older adults using DHIs for respiratory disease, specific needs were found including poor vision and navigation challenges. 73 With use of this information, the DHI developed can make specific considerations to make the technology more accessible. User acceptability is also often not well addressed. The Theoretical Framework of Acceptability contains seven domains: affective attitude, burden, perceived effectiveness, ethicality, intervention coherence, opportunity costs and self-efficacy. 74 Many studies do not look at the entire complexity of acceptability, and therefore this is a key weakness of many implementation studies. 72
To provide more guidance on system-level implementation, in 2020, the WHO first developed a “Global Strategy on Digital Health” outlining guiding principles for use and scale up of technology. 1 In keeping with this strategy, consideration is required when creating new DHIs, as issues are already arising; for example, poor interoperability, which is the ability for computer systems to understand each other. 1 An example of this is when using different electronic medical record (EMR) software and the inability to transfer data easily between hospital networks.
Another consideration included in the WHO’s guiding principles is the security and safety standards on the collection of sensitive personal information. The very nature of DHIs, has brought about the need for policies and regulation on what data can be obtained and its usage. 75 Technology gathering sensitive data is vulnerable and the risk data breaches needs to be mitigated and constantly surveilled. 75 Digital ethics and governance frameworks are also advised. 75
To inhibit or discourage the development of unregulated DHIs, regulation policy does exist. Guidance is published on “software as a medical device.” This includes the United Kingdom’s government-level guidance on ‘Medical devices: software applications (apps) 76 and the United States government Food and Drug Administration page on software as a medical device. 77 Additionally, the International Medical Device Regulators Forum is a collaboration of multiple medical device regulators worldwide that work together to ensure patient safety and the development of safe medical technology. However, some DHIs that are deemed to have a low risk are often unregulated, such as wellness apps. 78 Therefore, users should be wary of the risks and benefits of the technology that they are engaging with.
It is recommended that DHIs are carefully considered through development, and prior to implementation and scale-up to optimise ICU patient care. We are at a critical transition point in healthcare, with increasing adoption and implementation of DHIs. Careful consideration of factors discussed; at user, design and implementation level are needed when introducing new DHIs to ensure success.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Selina M Parry is an Associate Editor for the Journal of Intensive Care Society.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Selina M Parry is a recipient of the Val and Al Rosenstrauss Fellowship. Natalie A Fini is the recipient of a National Health and Medical Research Council Fellowship (grant number: 2026151).
