Abstract
Nebulisers are devices that reduce a body of liquid into a fine aerosol suitable for inhalation. Utilising the efficiency of pulmonary drug absorption, they offer a safe and powerful modality for local and systemic drug delivery in the treatment of critical illness. In comparison to conventional jet (JN) and ultrasonic nebulisers (USN), the advent of vibrating mesh nebulisers (VMN) has significantly improved the therapeutic potential of modern devices. This review article aims to summarise the history and evolution of nebulisers from first inception through to the modern vibrating mesh technology. It provides an overview on the basic science of nebulisation and pulmonary drug delivery, and the current use of nebulised therapies in critical care.
Introduction
Nebulisers are devices that reduce a body of liquid into a fine aerosol suitable for inhalation. 1 Utilising the efficiency of pulmonary drug absorption, 2 they offer a safe and powerful modality for local and systemic drug delivery in the treatment of critical illness. This includes inhaled bronchodilators for exacerbations of acute asthma, 3 inhaled heparin for airway burns and inhaled antimicrobials for drug-resistant ventilator-acquired pneumonia (VAP).4,5 Nebuliser use outside of critical care is also widespread, with multiple guidelines recommending their use for inpatient and community management of chronic respiratory disease.3,6 In comparison to conventional jet (JN) and ultrasonic nebulisers (USN), the advent of vibrating mesh nebulisers (VMN) has significantly improved the therapeutic potential of modern devices. 7 VMN benefit from shorter treatment times, reduced drug waste and improved pulmonary drug delivery, making them ideal devices for expensive inhaled novel therapeutics. 8 Their ability for in-line usage during mechanical ventilation also decreases fugitive emissions and risk of nosocomial infection in the pandemic era. 9 These performance benefits are however associated with higher up-front device costs. 10 VMN can also struggle to nebulise highly viscous and crystallising solutions due to clogging of micro-apertures, requiring regular cleaning and maintenance to ensure continued performance with time.8,11 Despite this, highly efficient VMN have the potential to expand the use of inhaled therapeutics in critical illness and the non-invasive treatment of respiratory and systemic disease. This review article aims to summarise the history and evolution of nebulisers from first inception through to the modern vibrating mesh technology. It provides an overview on the basic science of nebulisation and pulmonary drug delivery, and the current use of nebulised therapies in critical care.
The history and evolution of nebulisers
Although the word nebuliser was first coined in 1872, 1 the inhalation of aerosolised substances for medicinal purposes was first documented in approximately 2000 BC. Those practicing traditional ayurvedic medicine in India smoked pipes containing spices and the plant genous datura – which contain powerful tropane alkaloids with anticholinergic and bronchodilating effects. 12 Around this time the Ancient Egyptians (1500BC) are also known to have smoked Black Henbane (Hyoscyamus niger) for its similar effects secondary to atropine. 13 Hipocrates (460–370BC) is credited with the design of one of the earliest inhalation devices, fashioned from a simple pot and a long wooden reed, however primitive designs such as his saw little innovation until the dawn of the industrial revolution. 14 John Mudge first used the word ‘inhaler’ in 1778 to describe his device made from a pewter tankard through which air was drawn through heated water creating a steam-based aerosol prior to inhalation (Figure 1a). 13 This device was the first commercially available inhalation device delivering a medicated vapour and was initially intended for the inhalation of opium for chronic cough. 13 Over the next century various commercially available devices delivering medicated vapours were developed, including the famous Nelson inhaler (Figure 1b). 14 As the sophistication of these designs progressed so did their therapeutic uses. The British Pharmacopedia (1867) lists multiple compounds that could be inhaled for the treatment of cough (hydrocyanic acid, hemlock) tuberculosis (chlorine, iodine) and bronchitis (creasote), indicating widespread use of inhaled therapies at this time. 15

The advent of nebulisers in the mid-1800s was a major advancement for aerosolised drug delivery, starting a period of rapid and significant technological growth. In contrast to previously described inhaler devices (which delivered a steam-base aerosol via fully saturated water molecules), nebulisers reduce a body of medicated liquid into a fine spray and contain a contain a baffle system to catch coarser droplets unsuitable for inhalation. The fine aerosol generated by these early devices was much more likely to be deposited in terminal airway branches than their predecessors, increasing drug absorption and therapeutic effect. Bergson’s adaptations of initial nebuliser designs resulted in the Hydrokonium (1862), the first example of jet nebulisation. 13 Utilising the Venturi principle that high velocity air running directly over a tube results in suction (Figure 2), medicated solutions could be atomised using high-velocity airflow powered by mechanical bellows or steam. The discovery of rubber towards the end of the 19th century further enhanced their utility, allowing the development of the hand-bulb nebuliser (Figure 1c). 15 Rather than relying on steam or mechanical methods, hand-powered nebulisers drastically improving portable symptom control for asthmatics.
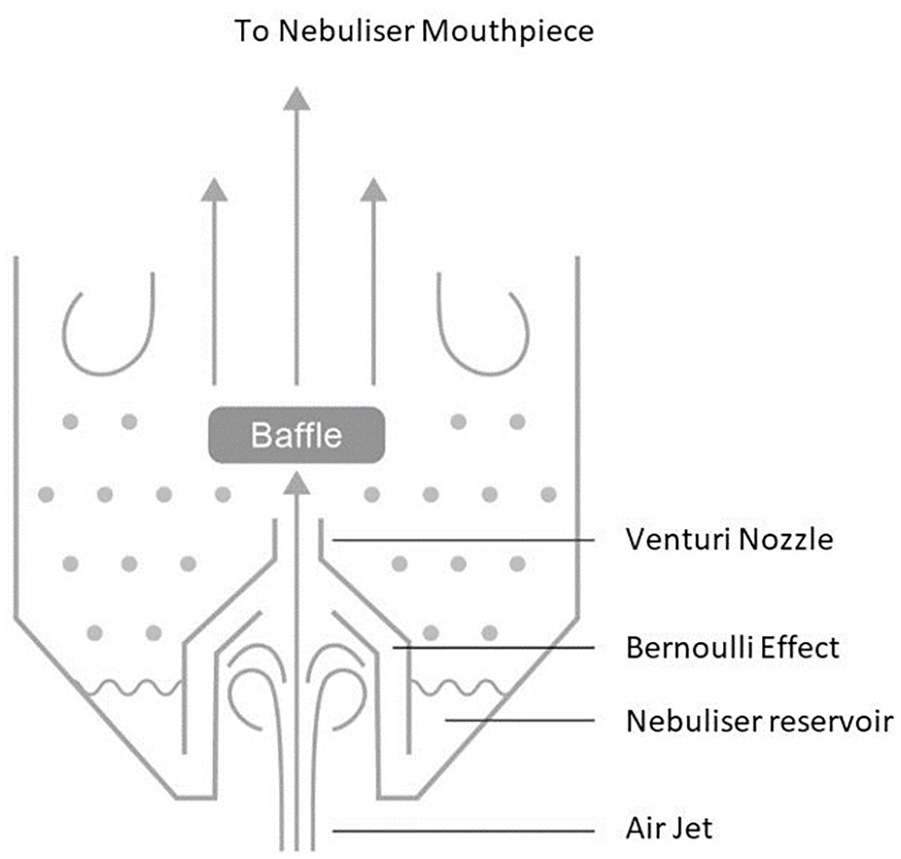
Venturi principle in jet nebuliser.
The technological innovation of nebulisers accelerated throughout the 20th century with further scientific advances. The Pneumostat (1930) was the first electric nebuliser (Figure 1d), although its popularity was limited by its price and poor portability as patients had to travel to central locations (e.g. pharmacies) to access it. 16 The Wright nebuliser in the 1950s pioneered precision moulded plastic venturi orifices, enabling the generation of much finer sprays (1–5 μm) more reflective of modern devices. 15 Shortly afterwards Robert Lang (1962) invented the first USN, using piezoelectric crystals to atomise solutions through ultrasound waves. The USN never rivalled the commercial success of the JN, partially due to poor reliability of early models. 13 Coupled with the discovery of new drugs (epinephrine, corticosteroids and beta-agonists) 17 these design improvements enabled the more effective treatment of a variety of respiratory diseases. The pivotal invention of the metered dose inhaler (MDI) (1956) (Figure 1e) and subsequent breath-actuated MDI (1970) further revolutionised portable respiratory therapies, particularly for asthmatic and COPD patient populations, 14 with modern iterations of these devices still widely used today.
The lungs as a portal of entry
The lungs provide an excellent portal for local and systemic non-invasive drug administration. The large surface area (commonly quoted at ~80 m²) and a thin air-blood barrier allow rapid absorption of local therapies coupled with high target-tissue concentrations and reduced systemic side effects. 18 The enhanced permeability of the terminal airways in combination with avoidance of the liver’s first pass metabolism also make the lungs an attractive route for systemic drug delivery. 19
The purpose of a nebuliser is to deliver finely aerosolised medications to the terminal alveoli. The overall efficiency of nebulisers is determined as a composite of the emitted dose (ED) of the drug and the fine particle fraction (FPF). 5 In a nebulised solution, the average size and shape of the particles is referred to as the mass median aerodynamic diameter (MMAD) and dictates the airway level at which drug deposition occurs. Large particles (>10 µm) are unable to pass further than the proximal airways (orophaynx and mouth), whilst medium sized particles (5–10 µm) are deposited in the central airways. Only fine particles (<5 µm) reach the distal alveoli where the most effective drug absorption occurs. 2 Airway deposition at different airways levels occurs through three different mechanisms: initial impaction; gravitational sedimentation; and diffusion through Brownian motion. Absorption in the proximal airways predominantly occurs through impaction (molecules colliding with tissue) due to high velocity turbulent airflow. In the distal airways only accessible by fine particles, low velocity airflow means that drug absorption occurs primarily through diffusion with some contribution from gravitational sedimentation. 2 As a result of the significant surface area occupied by the terminal alveoli, the FPF achieved during nebulisation disproportionately influences tissue drug bioavailability.
The theoretical potential for pulmonary drug absorption is however limited by a variety of real-life factors. Drug absorption is heavily influenced by patient-factors such as airway physiology and anatomy. Newman describes mechanical, immunological and behavioural features as patient-specific barriers to pulmonary drug delivery. 18 In diseased lung tissue, ventilation perfusion mismatch (due to pulmonary consolidation, atelectasis, bronchoconstriction etc.) and increased mucociliary clearance act as mechanical barriers to absorption. 5 Immunological factors such as drug engulfment by pulmonary macrophages are also patient-specific, and can also be influenced by the presence of other chemicals such as surfactant. 20 Behavioural features such as inhaler technique and adherence to nebuliser therapy also affect drug delivery, a factor that is particularly important in paediatric and elderly populations. Similarly, suboptimal prescription regimens and poor patient education by health-care professionals have a negative impact on therapeutic drug delivery. In addition to patient specific factors, drug-specific pharmacokinetics such as solubility, stability, membrane permeability and metabolic elimination are also essential components determining end-tissue bioavailability via the pulmonary route. 5
Inhaled therapies in critical care
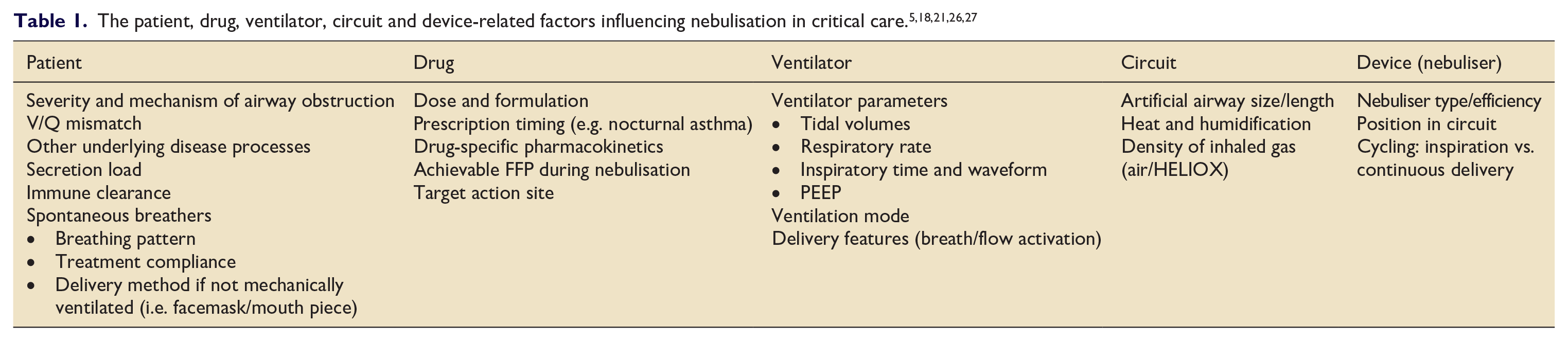
In critically ill and mechanically ventilated patients there are further mechanistic considerations that need to be taken into account for drug deposition during nebulisation. 21 These are listed in Table 1. Circuit specific factors such as heat and humidity have been demonstrated in-vitro to reduce nebuliser efficiency by causing aerosolised medication to precipitate in the circuit. 22 Despite this, recent in-vivo analysis suggests that these detrimental effects may be over-estimated. 23 The position of the nebuliser in the circuit also affects delivery. In-vitro paediatric and adult studies demonstrate optimal nebuliser efficiency when placed at the ventilator/humidifier, with proximity to the Y-piece associated with worsening performance.24,25
Artificial airway size is also an important feature affecting airflow and thus drug absorption. Poiseuille’s law states that turbulent airflow increases with reducing airway diameter, 28 meaning that smaller artificial airways are associated with increased non-therapeutic deposition via initial impaction. Conversely a larger artificial airway is more likely to result in laminar airflow and increase distal drug deposition. This is particularly important in smaller airways (e.g. paediatric) where delivered dose via MDI has been shown to decrease from a half to a third when downsizing from a 5 to 4 mm endotracheal tube. 29 Similarly, dyssynchrony and abnormal breathing patterns in mechanically ventilated and spontaneously breathing patients with respiratory distress is associated with turbulence. Longer low flow inspiration, adequate tidal volumes and breath actuated aerosol release are optimal for enhanced aerosol delivery. 30 Increased lung deposition has been observed during pressure support (15.1%) in comparison to volume control ventilation (10.5%), 31 however further research is required to investigate the significance of this finding. Achieving the described ideal ventilator parameters for nebuliser delivery is not always feasible in intubated patients. Increased sedation and neuromuscular blockade may be required to maximise lung compliance and drug absorption, 5 risking prolonged mechanical ventilation and difficulty weaning sedation.
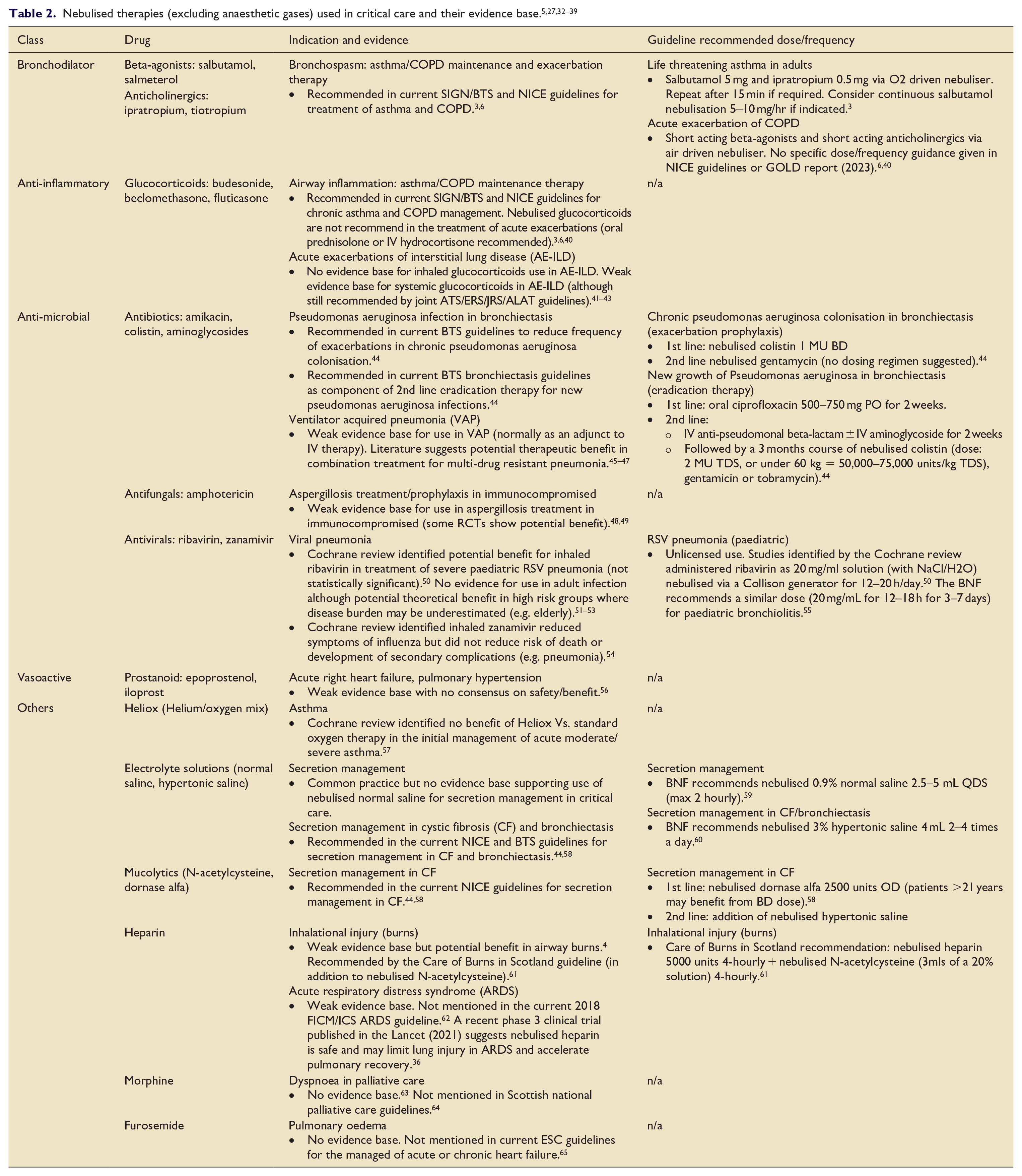
The overall use of aerosolised therapies in critical care is extensive. Ehrmann et al. conducted an international 2-week cross-sectional study capturing over 2800 patients across 81 critical care departments in 2016, finding that 24% of patients received nebulised therapies during their stay. 32 Importantly only 10% of these patients used inhaled medications pre-admission, demonstrating the importance of inhaled therapies outside the remit of chronic respiratory disease. The incidence of nebulised therapy varied between mechanically ventilated patients (22%) and those requiring NIV (50%), as did the modality of nebulisation: JN (56%); hand-held devices (in-line usage) (e.g. MDI) (23%); USN (10%); and VMN (10%). The main nebulised therapies were bronchodilators (82%), corticosteroids (13%), antimicrobials (5%), electrolyte solutions (5%) and mucolytics (3%). A similar more recent study conducted by Lyu et al. across 28 Chinese ICUs in 2021 found that 38% of patients received nebulised therapies. 33 One major difference between these studies was the utilisation of mucolytic therapies for secretion management in the Chinese population, which was eightfold higher and the most common indication for nebuliser therapy (23% of all patients). A likely contributing factor for this difference is the timing of the studies, as the Lyu et al. took place during the COVID-19 pandemic and Ehrmann et al. before it. Reports of side effects associated with nebulisation were infrequent in both studies (1% and 8% respectively) and mostly mild (e.g. cough, tachycardia). In total there were only three reports of bronchospasm, all of which were associated with nebulised colistin, 32 indicating that inhaled therapies are safe in the critically ill population. A summary of nebulised therapies used in critical care from the literature and their evidence base is given in Table 2.
Vibrating mesh nebulisers
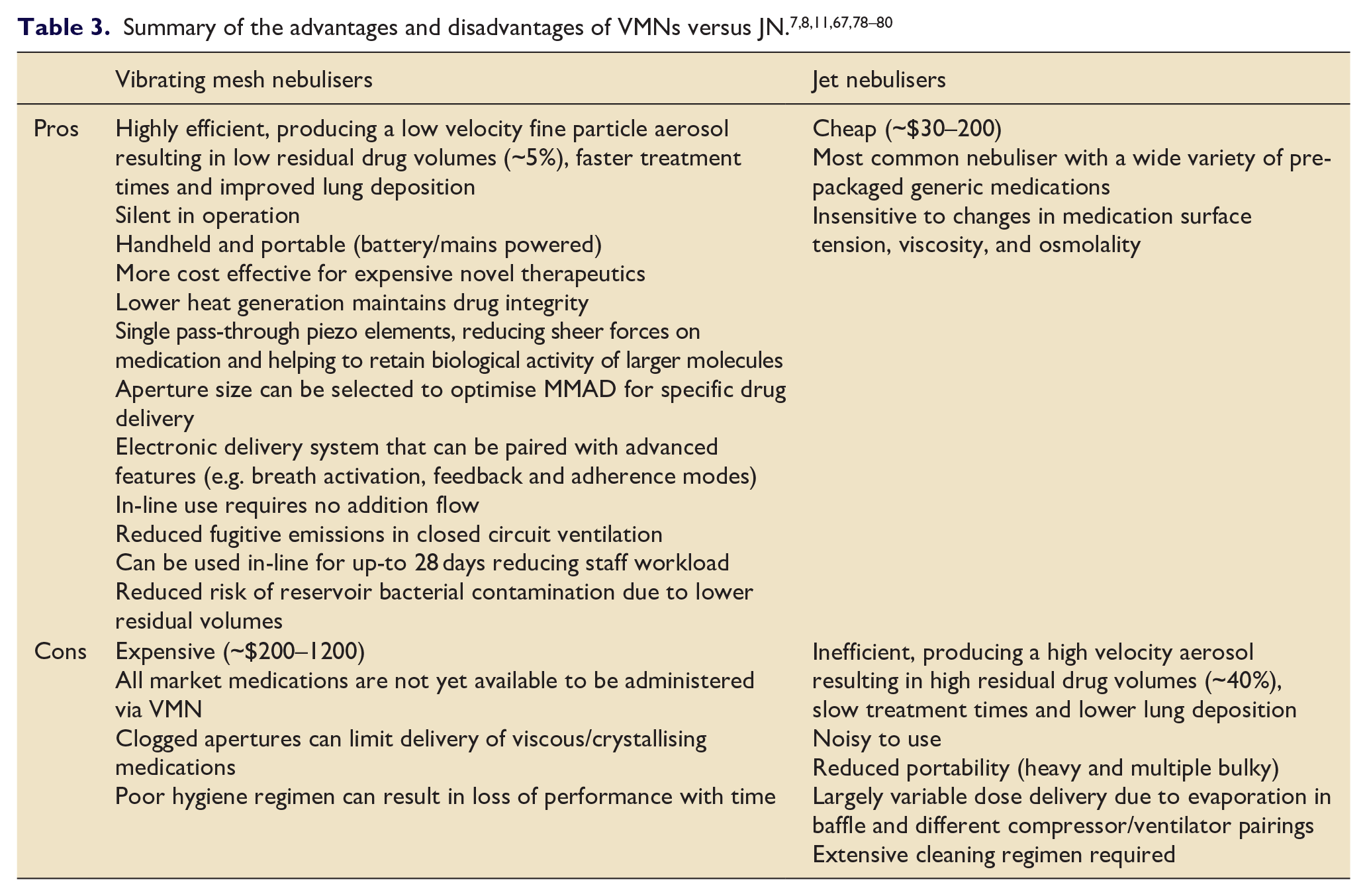
Highly efficient nebulisers are imperative to overcome the myriad of factors affecting drug delivery to the lungs in critical illness. Continual development of the JN has drastically improved their efficiency and ease of use from first inception, but the superior VMN is likely to replace them as the clinical standard for nebuliser delivery. 7 Introduced in 1993, VMNs generate an aerosol via a rapidly vibrating mesh or plate (also known as a piezo element) with thousands of tiny apertures (Figure 3). As the piezo element vibrates, each aperture acts as a tiny micro-pump nebulising the liquid drug solution it is in contact with. 66 The piezo element itself is typically manufactured from inert metal (e.g. palladium) which undergoes precision laser cutting to create apertures of a desired size, allowing a specific MMAD to be achieved in the nebulised solution. 8 Through this mechanism, VMNs produce a precisely sized low velocity aerosol which reduces circuit condensation and dramatically increases drug delivery to the lungs. 67 Highly efficient first-pass nebulisation and lack of a buffer system has the added benefit of reducing heat and shear forces to the medication, helping to retain the biological activity of larger macro-molecules and preserve drug integrity. 11 The VMN is also able to nebulise much smaller drug volumes than JN and significantly lower residual drug volumes are widely reported in the literature (~5% of the nebulised solution in VMN vs ~40% in JN).68–70 In combination these factors allow administration of small volume medications with minimal drug waste, making the VMN an attractive and potentially cost-effective delivery device for expensive novel therapeutics.8,71
Vibrating mesh nebuliser.
Increased lung deposition with VMN has been demonstrated across multiple studies and modes of oxygen delivery. 67 Radio-aerosol studies in healthy individuals receiving NIV have found VMN to double airway deposition in comparison to conventional JN, 68 with a further small randomised control trial showing a threefold increased deposition in patients with moderate/severe COPD. 69 Other imaging studies using low-dose computed tomography have also shown increased deposition when using VMN, reporting six-times greater deposition in spontaneously breathing healthy adults compared to JN with similar total lung penetration. 72 Ari & Fink demonstrated that VMN significantly increased airway drug deposition threefold compared to JN. 73 This was found to be the case during mechanical ventilation (23.2%vs 6.8%), non-invasive ventilation (18.4%vs 6.1%) and spontaneous breathing (17.3%vs 6.1%). Importantly VMN has been shown to outperform JN and USN regardless of position in the circuit in both adult and paediatric models.24,25
Increased airflow turbulence during high flow nasal oxygen (HFNO) therapy results in substantially reduced therapeutic aerosol deposition, although the higher efficiency of VMN technology may offset the reduced nebuliser performance during HFNO therapy. Dugernier et al. conducted a radioisotope study in healthy volunteers using JN and VMN during HFNO therapy, demonstrating significantly increased deposition with the use of VMN (1%vs 3.6% of administered dose). 74 Reminiac et al. compared improvements in FEV1 after nebulised albuterol administration via HFNO and conventional face mask oxygenation in patients with obstructive lung disease. 75 Patients receiving nebulised albuterol via VMN and HFNO were found to have similar improvements in FEV1 compared to patients randomised to JN and conventional face mask oxygen. This was shown to be the case even after controlling for the observed bronchodilatory effect of HFNO therapy, making it the first controlled study to demonstrate non-inferior nebuliser performance during HFNO therapy compared to conventional face mask delivery.
The increased efficiency of VMN over JN has translated into quantifiable improvements in patient care. Moody et al. conducted a single-blinded randomised control trial comparing VMN versus JN in over 200 children with moderate/severe asthma exacerbations in the emergency department. 76 Patients who received nebuliser via VMN were found to have significantly fewer bronchodilator treatments, reducing the time to reach a mild asthma score by 28.4%. They also observed a 6.3% absolute reduction in admission in the cohort treated with VMN, although this result did not reach statistical significance. In another similar prospective study, 77 adults presenting to the emergency department requiring bronchodilators were found to require fewer treatments with VMN compared to JN. The use of VMN was also shown to significantly increase likelihood of hospital discharge (OR = 1.5) and reduced the median length of stay in the emergency department by 13% (37 min). In addition to improvements in efficiency, the use of VMN has the potential to improve patient compliance through reduced treatment times, silent operation and enhanced portability. 11
Despite the aforementioned benefits of VMN, several factors have limited their uptake into clinical practice since their inception. The vast majority of market medication is pre-packaged for use with conventional JN, including most of the generic medication for the treatment of common respiratory disorders such as asthma and COPD. 8 VMNs also have issues nebulising highly viscous and crystallising solutions due to clogging of the micro-apertures, requiring regular device hygiene to ensure continued performance with time. 11 Finally VMN are also more expensive than JN (device cost of ~$200–1200 vs $30–200). 78 In a recent literature review by Edge and Butcher, there was found to be insufficient evidence to conclude on the long-term cost effectiveness of VMN in-comparison to other nebuliser devices despite their improved efficency. 10 Although the higher initial cost of VMN technology may be counteracted by future off-patent models and their increased efficiency at delivering expensive novel therapeutics,8,71 more research is required to further explore their cost-effectiveness. A summary of the pros/cons of JMN versus JN is given in Table 3.
Safety, fugitive emissions and COVID-19
VMN also have added safety benefits as they do not require an external gas flow to deliver nebulised medication. 8 This means that they can be used in-line in contrast to JN which require connection to medical airflow metres to drive the nebulised solution through the circuit. NHS England’s recent patient safety alert details 108 ‘never events’ between April 2018 and 31 March 2021 where patients were inadvertently connected to medical air instead of oxygen due to the proximity of the oxygen and medical air valves. These errors were attributed to nine in-patient cardiac arrests over this period and a total of six deaths, all of which could have been avoided with the use of VMN and removal of medical air valves. The MHRA also issued a warning regarding oxygen consumption during the COVID-19 pandemic, highlighting the potential for hospital-wide failure of oxygen delivery systems if demand exceeded the maximum capacity of central delivery systems. 81 Joint guidance from the Intensive Care Society and the British Thoracic society suggests staggering nebuliser to reduce total oxygen consumption, 82 a measure that is not necessary with the in-line use of VMNs.
VMN are also the preferred choice of nebuliser in ventilated COVID-19 patients.79,83 They have been shown to significantly improve drug deposition during COVID-19 low-tidal volume ventilation strategies, 84 in addition to reducing staff workload and the risk of fugitive emissions.85 –87 This is because they can be used in-line for up to 28-days, making them the only mode of nebulisation compatible with a completely closed circuit. The no-additional flow requirements of the in-line VMN system reduces the potential generation of bioaerosols,9,79,87 and the separation of the medication reservoir from the breathing circuit reduces the likelihood of bacterial contamination of the reservoir between doses.80,88 A closed circuit also has the benefit of not causing a drop in circuit pressure during drug-refills, preventing potential lung de-recruitment.87,89 Joyce et al. demonstrated undetectable fugitive emissions during VMN drug refill in a simulated mechanical ventilation model, whilst JN refill resulted in significant fugitive emissions. 89 Similarly, Eian et al. found no detectable fugitive emissions during aerosol delivery with a closed circuit VMN compared to detectable emissions with both MDI and JN. 90
Conclusions
Nebulised therapies in critical care offer an effective non-invasive route for pulmonary and systemic drug delivery. Novel VMN are more efficient at delivering a therapeutic aerosol in comparison to conventional nebulisers, in addition to other benefits such as decreased residual volume and treatment times. VMN also offers several key safety benefits through in-line usage and lower risk of fugitive emissions in the pandemic era. Nevertheless, an improved understanding and appreciation of the numerous barriers limiting drug absorption during nebulisation in critical care patients is require for them to reach their true therapeutic potential. More research is required regarding the long-term cost-effectiveness of VMN in comparison to pre-existing cheaper alternatives, in addition to large scale randomised control trials evaluating the efficacy of nebulised therapeutics described in the critical care literature. Ultimately, efficient modern nebulisers and novel therapeutic discoveries have the potential to fully exploit the physiological advantages of pulmonary drug delivery, potentially revolutionising the non-invasive treatment of respiratory and systemic disease in critical care.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA has received honoraria from Aerogen Ltd for presenting at a meeting on nebulisers. MS is an external clinical advisor for Aerogen Ltd and has received honoraria for speaking at several meetings on nebulisers. MW has no competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.