Abstract
Background
To investigate the impact of physical activity interventions, including early mobilisation, on delirium outcomes in critically ill patients.
Methods
Electronic database literature searches were conducted, and studies were selected based on pre-specified eligibility criteria. Cochrane Risk of Bias-2 and Risk Of Bias In Non-randomised Studies-of Interventions quality assessment tools were utilised. Grading of Recommendations, Assessment, Development and Evaluations was used to assess levels of evidence for delirium outcomes. The study was prospectively registered on PROSPERO (CRD42020210872).
Results
Twelve studies were included; ten randomised controlled trials one observational case-matched study and one before-after quality improvement study. Only five of the included randomised controlled trial studies were judged to be at low risk of bias, with all others, including both non-randomised controlled trials deemed to be at high or moderate risk. The pooled relative risk for incidence was 0.85 (0.62–1.17) which was not statistically significant in favour of physical activity interventions. Narrative synthesis for effect on duration of delirium found favour towards physical activity interventions reducing delirium duration with median differences ranging from 0 to 2 days in three comparative studies. Studies comparing varying intervention intensities showed positive outcomes in favour of greater intensity. Overall levels of evidence were low quality.
Conclusions
Currently there is insufficient evidence to recommend physical activity as a stand-alone intervention to reduce delirium in Intensive Care Units. Physical activity intervention intensity may impact on delirium outcomes, but a lack of high-quality studies limits the current evidence base.
Introduction
Delirium is a syndrome characterised by acute fluctuating changes in attention and cognition affecting up to 80% of patients undergoing mechanical ventilation. 1 Intensive Care Unit (ICU) delirium is independently associated with increased mortality, ICU and hospital length of stay.2,3 ICU delirium results in long-term cognitive impairments in up to 70% of ICU survivors.1,4
Delirium pathophysiology remains poorly understood, however both iatrogenic (drugs) and environmental factors (e.g. immobilisation) may contribute. 5 Numerous hypotheses including neuro-inflammation and neurotransmitter imbalance have been proposed to inform potential treatment strategies.6,7
Delirium and physical activity (PA) are closely linked as immobility and functional decline have been identified as potential risk factors. 5 PA encompasses a variety of interventions such as exercise, rehabilitation and mobilisation that can improve functional outcomes and minimise critical illness morbidity. 8 In non-ICU patients, PA has been shown to provide neuroprotective effects by increasing neurotransmitter and anti-inflammatory mediator release and synaptic transmission facilitation. 9 In older adults’ PA can also increase cerebral blood flow and oxygen extraction efficiency, resulting in improved cognition. 10 Alongside physical effects, it is hypothesised that the psychosocial aspects of mobility may also aid preserving cognitive function during critical illness. 10
Early mobilisation (EM) currently has no defined consensus but is thought to be the initiation of a form of PA within the first 2–5 days of critical illness. 11 PA encompassing EM is recommended in International guidelines to reduce negative critical illness outcomes by coordinating better ICU care.12,13 Within these guidelines the ABCDE/F (A-E/F) bundle has been created to minimise negative consequences such as delirium during ICU stay. 14 In particular, the ‘E’ bundle aspect recommending EM to reduce delirium and improve physical outcomes.
The effectiveness of the A-E/F bundle has previously been evaluated but there are few reviews investigating the contribution of the individual bundle elements on delirium.15–17 A 2020 systematic review investigated delirium outcome reporting in prevention or treatment studies but did not specifically focus on EM trials outside of an A-E/F bundle. 18 A systematic review investigating multi-component non-pharmacologic strategies by Rossom and colleagues in 2011 highlighted the need to investigate individual bundle elements, to establish which components are most successful in delivering positive outcomes. 19 This could enable more cost-effective treatment delivery. 19 While it is thought that all the A-E/F bundle components interlink to provide an effective treatment, evaluating individual components will also inform intervention prioritisation.
Within ICU literature numerous questions remain regarding the optimal type, timing, and intensity of PA interventions. The majority of A-E/F bundle recommendation papers tend to utilise only two studies as evidence for the impact PA has on delirium.20,21 As awareness of long-term delirium consequences on ICU survivors has grown, further studies investigating ICU PA interventions on delirium have recently been published.22,23 These studies have yet to be synthesised together to establish any correlation between PA and delirium. Evaluating PA interventions will also help inform the optimum type and delivery to positively impact delirium.
This systematic review aimed to investigate the impact of PA interventions, including EM, on delirium outcomes in ICU patients.
Methods
A protocol for this review was registered prospectively on PROSPERO in November 2020: (CRD 42020210872) (https://www.crd.york.ac.uk/prospero/). The methodology is reported according to the updated 2020 PRISMA guidelines. 24 See Supplementary Material for checklist.
Eligibility criteria
Studies that recruited participants aged over 18 admitted to ICU were eligible for inclusion. Studies including participants with pre-existing cognitive impairments prior to admission and non-ICU patients were excluded.
The target intervention was PA which could be delivered independently but not as part of an established A-E/F bundle or deemed, by the authors, to be part of a delirium prevention bundle. Studies including PA as part of interventions listed in Supplementary Material were excluded. For the comparator group, studies delivering no intervention or usual care as defined in the protocol were considered.
Eligible study designs included randomised controlled trials (RCT), cohort, retrospective and cross-sectional studies assessing PA for prevention or management of ICU delirium. Non-English studies were excluded. Studies with interventions beginning outside ICU were not included. Supplementary Material details further inclusion and exclusion information.
The primary outcome of interest was delirium incidence, defined as a positive measurement using a validated delirium screening tool such as Confusion Assessment Method for the ICU (CAM-ICU) or Intensive Care Delirium Screening Checklist (ICDSC) as recommended in the Pain, Agitation, Delirium, Immobility and Sleep disruption guidelines. 12 Studies using non-validated delirium assessment tools were excluded. Secondary outcomes included delirium duration, prevalence, severity, and number of delirium-coma free days.
Information sources
The following databases were searched through Healthcare Databases Advanced Search (AMED, BNI, CINAHL, EMBASE, MEDLINE, PSYCHinfo, PubMed). Other databases were searched through their respective individual platforms, Physiotherapy Evidence Database (PEDro) and Cochrane Library (CENTRAL). Searches included all study types published until 30th November 2020 and were updated in November 2021 with no limits placed.
Search strategy
Key terms and subject headings were used to search the above databases with assistance from a specialist healthcare librarian. The Boolean operators of ‘AND’ and ‘OR’ were employed to narrow and broaden the search, respectively. Asterisks were used where there could be multiple spellings or endings to a search term. Full search strategies are in Supplementary Material. Included studies references were manually screened to identify studies not identified via the electronic database search. Other literature was identified via open grey (https://www.opengrey.eu). Relevant unpublished literature, ongoing studies or pre-print papers were identified from https://ClinicalTrials.gov, International Clinical Trials Registry and ResearchGate.
Selection process
Titles and abstracts of retrieved studies were exported into Rayyan where duplicates were automatically removed. 25 Rayyan is a free web tool designed to assist screening and selection processes. 25 Two independent reviewers screened study titles and abstracts of the retrieved studies (AJ+KC) prior to meeting, with each reviewer blinded to the other’s decision. Any eligibility discrepancies were first discussed between the two reviewers, if no agreement was reached a third reviewer was consulted (OG). The full text of any citation considered potentially relevant by either reviewer was retrieved and screened to confirm eligibility.
Data extraction
Data extracted from eligible studies included study design, setting, intervention and control details, participant demographics, delirium measurements and results (Supplementary Material).
Risk of bias
Tools specific to study design were used to assess risk of bias (ROB). To improve inter-rater reliability the two reviewers (AJ+KC) undertook calibration exercises of the tools before independently appraising each study then discussing discrepancies that arose. The third reviewer (OG) was available if no consensus could be achieved via discussion.
For RCTs both the Cochrane Risk of Bias 2 (ROB2) and Physiotherapy Evidence Database (PEDro) tools were used.26,27 Due to its physiotherapy relevance and having demonstrated good reliability, 28 the PEDro tool was initially utilised. However, whilst trialling the tool, the reviewers decided that it did not investigate each bias component as thoroughly as the ROB2. Therefore, ROB2 results were utilised for the main results and PEDro scores are available in Supplementary Material.
For non-RCTs the Cochrane “Risk Of Bias In Non-randomised Studies-of Interventions” (ROBINS-I) was used. 29 The ROBINS-I tool allowed for some guidance on weighting of non-RCTs, in the overall results of a review, with those deemed as having a low ROB possibly being comparable to an RCT.
Synthesis methods
A meta-analysis was completed using Review Manager (RevMan 5.4, London, UK) for the dichotomous outcome of delirium incidence with a meta-analysis for delirium duration also being considered. 30 A random-effects model was used to calculate pooled estimates. Heterogeneity between studies was determined through the I2 statistic. Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach was used to assess the quality of evidence gained from the meta-analysis and classified into 4 possible ratings: very low, low, moderate or high. 31 Narrative synthesis was undertaken for other outcome variables of interest.
Results
Study selection and characteristics
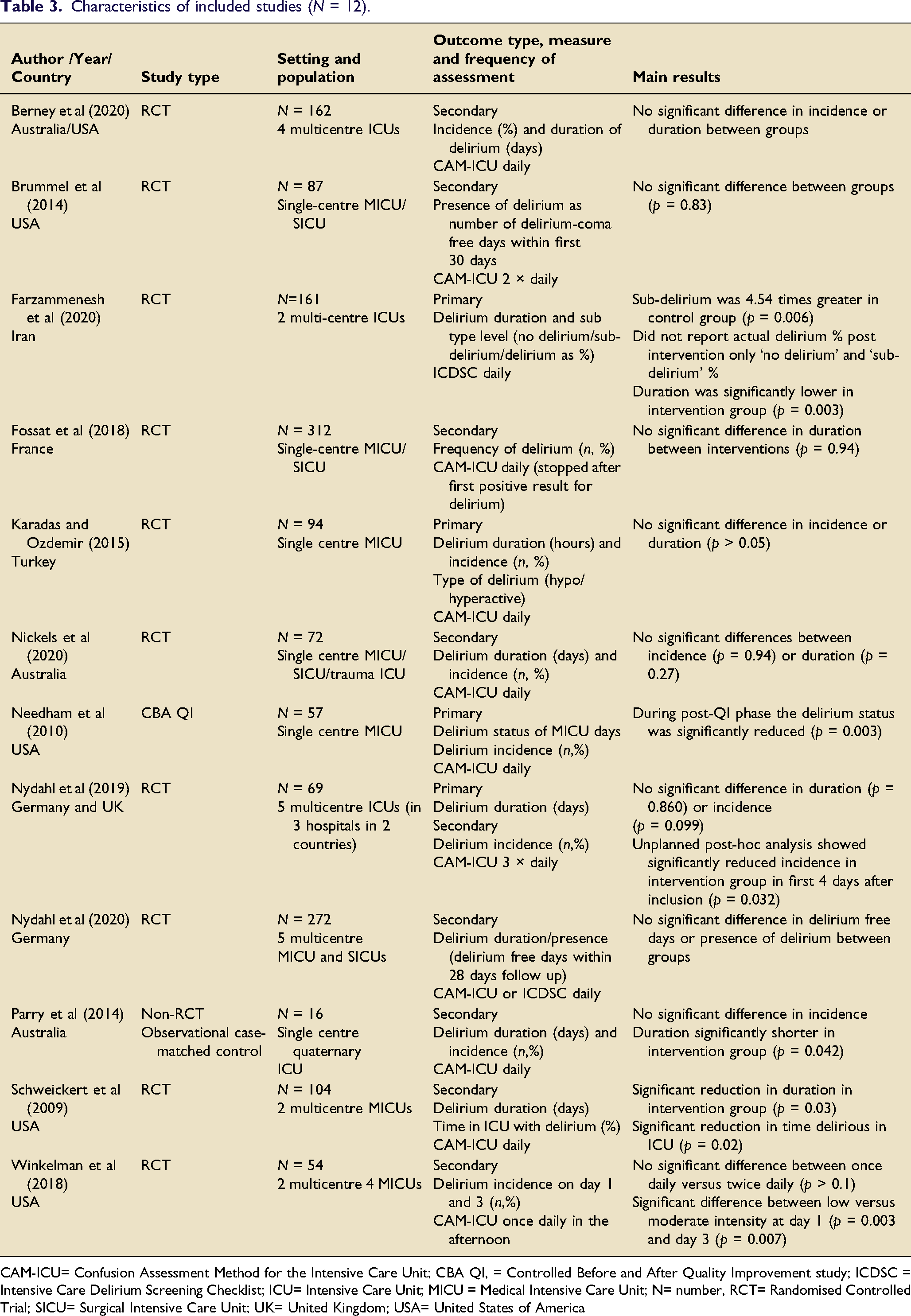
Study selection is detailed in the PRISMA flow sheet (Figure 1). After excluding duplicates, 3014 papers were screened for eligibility. Subsequently 27 full texts were retrieved with 11 meeting the inclusion criteria. One further study was included after reference screening. The 12 included studies (Table 1) comprised of 10 RCTs20,22,23,32–38 one observational case-matched control
39
and one before-after quality improvement study.
21
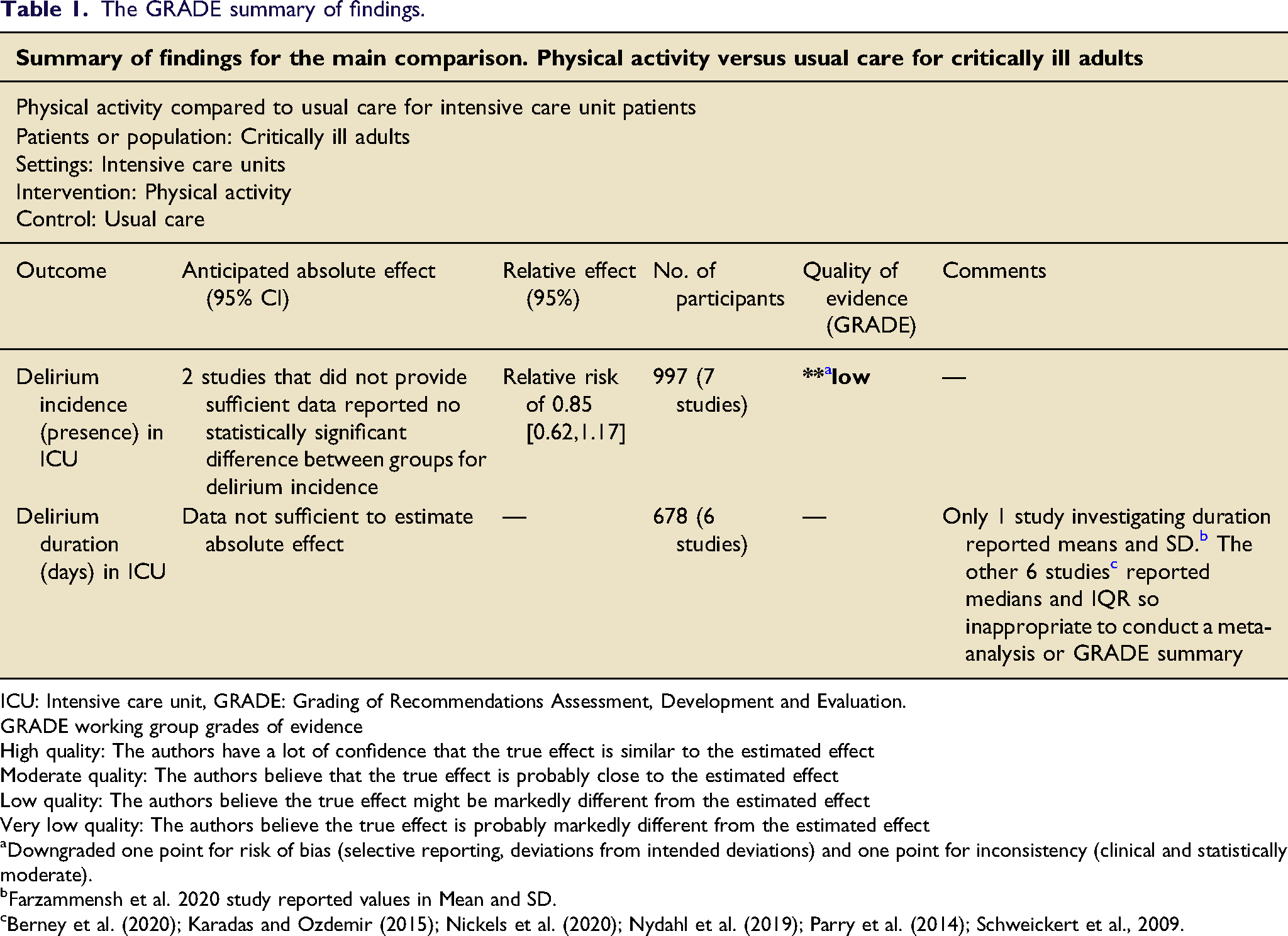
The two reviewers reached consensus regarding study inclusion, data extraction, quality, and ROB assessments (100% agreement), without recourse to a third reviewer. Preferred reporting items for systematic reviews and meta-analyses flow diagram, n = number. The GRADE summary of findings. ICU: Intensive care unit, GRADE: Grading of Recommendations Assessment, Development and Evaluation.GRADE working group grades of evidenceHigh quality: The authors have a lot of confidence that the true effect is similar to the estimated effectModerate quality: The authors believe that the true effect is probably close to the estimated effectLow quality: The authors believe the true effect might be markedly different from the estimated effectVery low quality: The authors believe the true effect is probably markedly different from the estimated effect aDowngraded one point for risk of bias (selective reporting, deviations from intended deviations) and one point for inconsistency (clinical and statistically moderate). bFarzammensh et al. 2020 study reported values in Mean and SD. cBerney et al. (2020); Karadas and Ozdemir (2015); Nickels et al. (2020); Nydahl et al. (2019); Parry et al. (2014); Schweickert et al., 2009.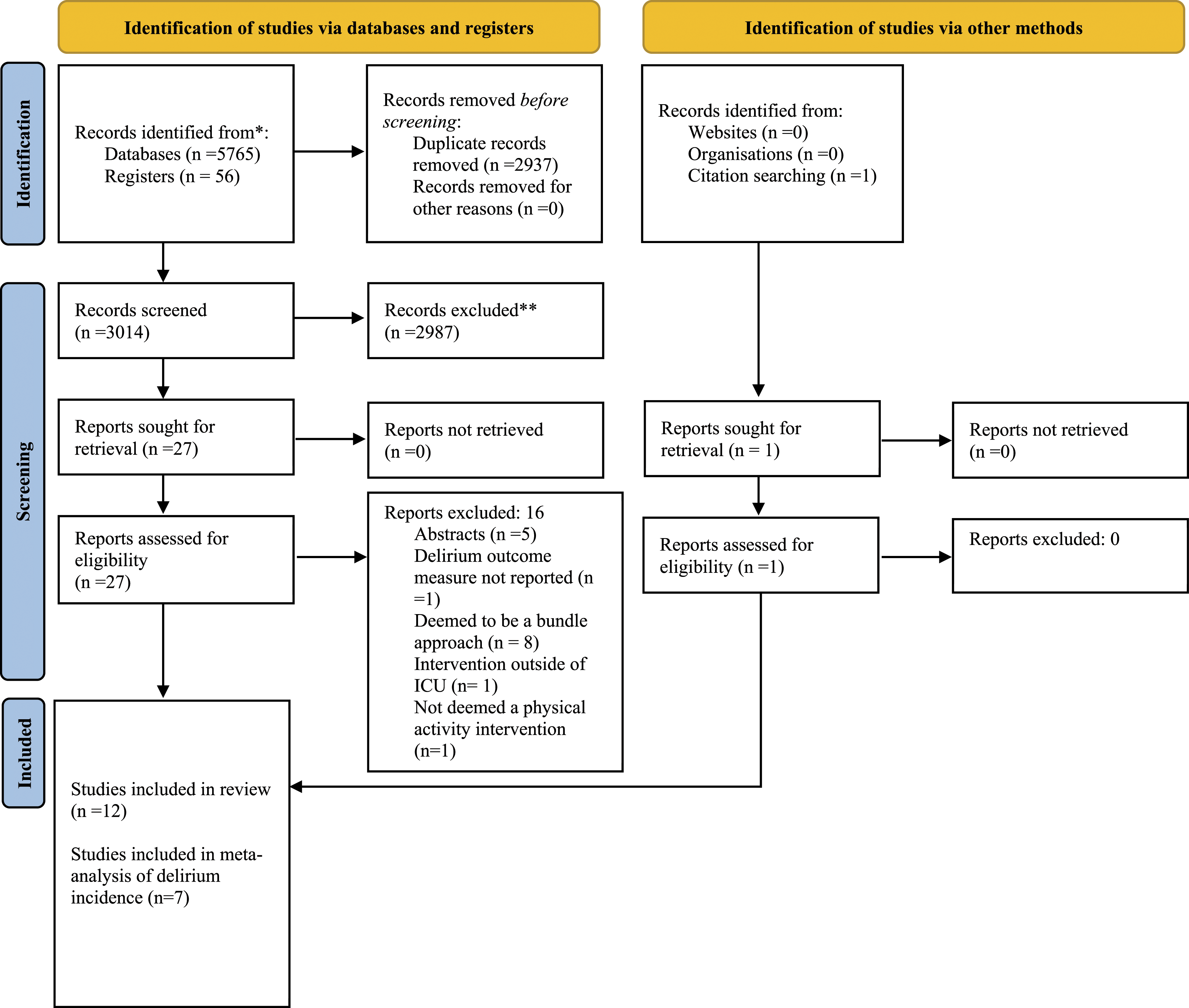
Overall, 1460 patients were included across the studies (Table 1). Sample sizes varied from 16 39 to 312 participants. 32 One study had an age requirement of 65 years and above. 33 All other studies included participants over 18 years with varying intervention start times from minimum of 24 h33,37 to maximum 96 h post admission 22 (Supplementary Material).
PA interventions
Description of included studies physical activity interventions in ICU (N = 12).
ETM= Early Therapeutic Mobility; FES= Functional Electrical Stimulation; ICU=Intensive Care Unit; LL= Lower Limb; MV= Mechanical Ventilation; OT= Occupational therapist; PROM= Passive Range of Movement; PT= Physiotherapist; QI= Quality Improvement; RASS= Richmond Agitation Assessment Score; ROM= Range of Movement; RN= Registered Nurse; SOEOB= Sit on edge of bed; UL = Upper Limb
Of the twelve included studies, six interventions were delivered specifically by Physiotherapists (PT) or Occupational Therapists (OT) (Table 2).20–22,32,38,39 In three studies, the interventions were delivered by either registered nurses (RN), physicians or a primary researcher whose primary role was undefined. Both in-bed cycling studies utilised a RN for the in-bed cycling and PT for the functional activity.22,39 In one study, sessions delivered by PTs or OTs lasted 23 minutes on average compared to fifteen when the same intervention was delivered by physicians or nurses. 36
Delirium measurement
Most of the studies20–22,32–34,36–39 used CAM-ICU to measure delirium (Table 1). One study 23 used the ICDSC, and one multi-centre study utilised both tools . 35 Delirium measurements varied in assessment frequency from once to three times daily, at differing times of the day.
Risk of bias
Figure 2 presents the included RCTs ROB2 assessments. Five studies were judged to be at high ROB either through having high risk in one domain or some concerns in multiple domains.23,33–35,37 Cochrane Risk of Bias 2 judgements for included randomised control trials. Red= High risk of bias, Yellow = medium risk of bias, green =low risk of bias.
Characteristics of included studies (N = 12).

CAM-ICU= Confusion Assessment Method for the Intensive Care Unit; CBA QI, = Controlled Before and After Quality Improvement study; ICDSC = Intensive Care Delirium Screening Checklist; ICU= Intensive Care Unit; MICU = Medical Intensive Care Unit; N= number, RCT= Randomised Controlled Trial; SICU= Surgical Intensive Care Unit; UK= United Kingdom; USA= United States of America
Delirium Incidence
Nine studies investigated delirium incidence21–23,32,33,35,37,39 with variation in how incidence was reported or categorised. Seven of the nine studies were included in a meta-analysis22,32,33,35,37–39 (Figure 3). The other two papers were excluded as one totalled delirious days across patients meaning incidence was unable to be calculated.
21
The other study did not report actual delirium post intervention but rather sub-delirium or no-delirium incidence.
23
The pooled relative risk for incidence was 0.85 (0.62–1.17) which was not statistically significant in favour of physical activity interventions. Heterogeneity may have been moderate (I2 = 51%, p0.06). This could be explained by the inconsistency in individual study effect estimates and validates the choice to use a random effects model. Meta-analysis for delirium incidence with physical activity versus usual care.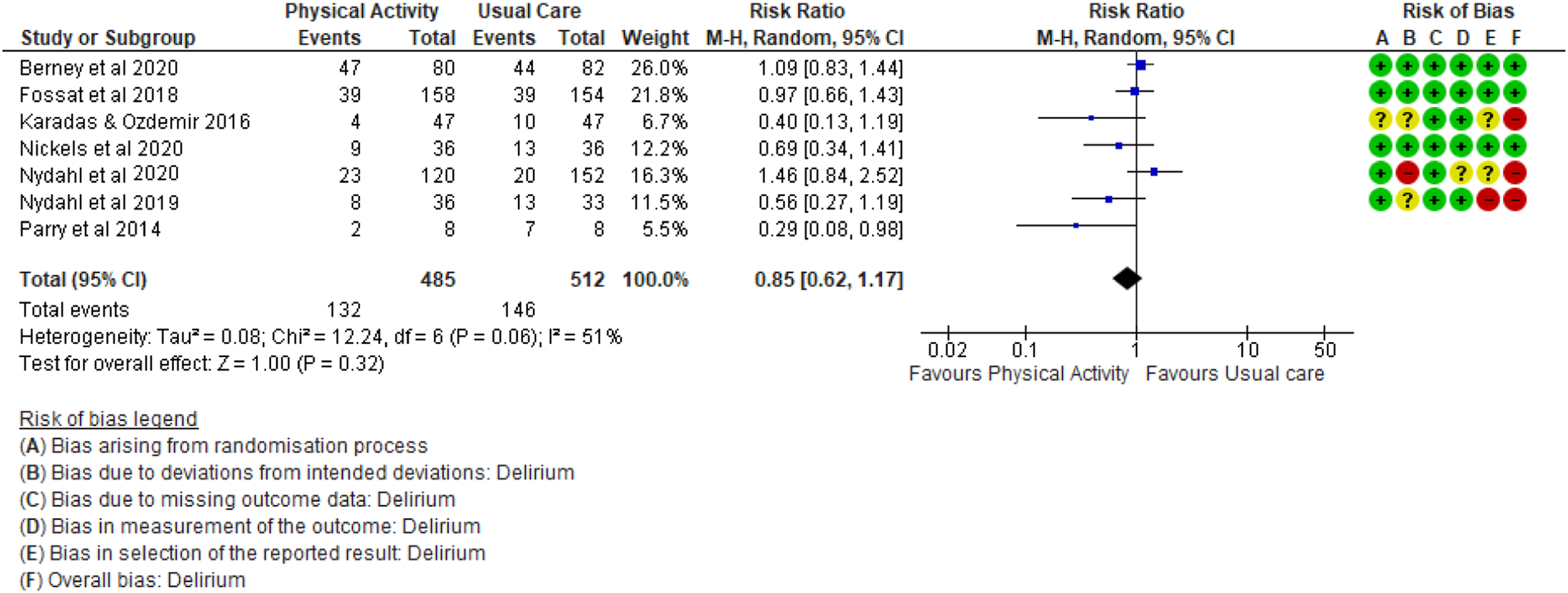
Delirium duration, severity, prevalence, and delirium-coma free days
Six studies investigated delirium duration in days20,22,23,37–39 with one study examining it in hours 33 (Table 1). Meta-analysis was not possible for this outcome due to insufficient summary statistics, so a narrative synthesis was performed. One study no longer assessed for delirium once a positive CAM-ICU score was detected meaning it wasn’t possible to calculate duration or compare to other studies. 32 Three comparative studies found statistically significant improvements in favour of PA interventions for delirium duration ranging from a median difference of 0–2 days.20,23,33 One study found no statistically significant difference. 32 Two studies reported their delirium outcome as the number of delirium-coma free days within the first 30 or 28 days of a patient’s ICU stay.35,36 None of the studies reported any outcomes on delirium severity or prevalence.
Quality of Evidence
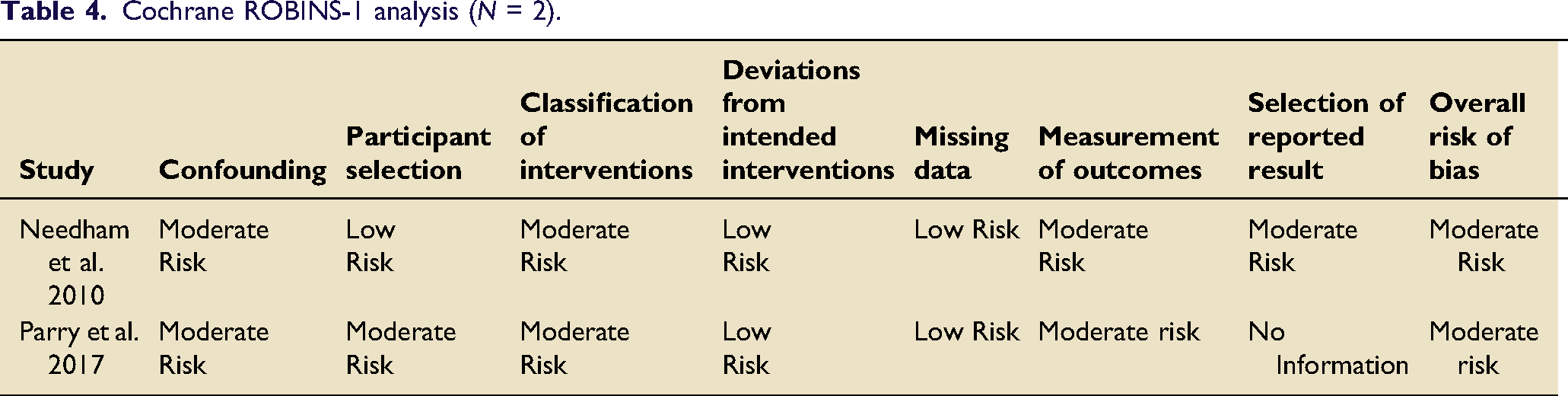
Cochrane ROBINS-1 analysis (N = 2).
Discussion
This review has investigated PA intervention impact on delirium outcomes in ICU patients from 12 eligible studies. While the impact of PA on delirium outcomes was not the primary outcome of interest or focus for the majority of studies, when assessing the five highest quality papers only one demonstrated a significant benefit of PA on delirium duration (p = 0.03). 20 The other four high quality papers all had very small sample sizes limiting the certainty of effect of PA interventions on delirium outcomes in ICU patients.22,32,36 There is weak evidence that PA may shorten delirium duration and lead to reduced risk of becoming delirious, but the quality of evidence is considered low with no statistical significance shown in meta-analysis.
Our review is the first to fully investigate the sole contribution of PA interventions on ICU delirium outcomes. This review demonstrates that there is insufficient evidence to recommend one type or dosage of PA over another. These results agree with a review by Haley and colleagues who investigated PA impact on delirium outcomes in hospitalised patients both in and out of ICU. 40 They also found extensive variation in intervention type, frequency, and intensity, concluding an inability to recommend PA as a sole intervention over a multi-component bundle. PA delivery differences and whether physiotherapy was included in usual service delivery across the different services and countries may have affected the included studies results and generalisability.
One proposed hypothesis of the pathophysiology of delirium is of dysregulated cytokines causing a systemic inflammatory response. 41 Gleeson et al. (2011) reported how moderately intense PA could produce an anti-inflammatory response in healthy individuals. 42 PA intensity has previously been studied in ICU and conclusions drawn that passive leg cycling is of an insufficient intensity to elicit systemic changes to produce an anti-inflammatory response. 43 In contrast FES-cycling can potentially produce sufficiently high intensity to elicit anti-inflammatory effects and therefore could impact delirium. 43 This is in agreement with the significant reduction in delirium duration reported by Parry and colleagues in their FES-based study (p = 0.042). 39 However, while duration was significantly reduced, no difference was found in delirium incidence and the study was judged at moderate ROB. 39 Usual care has evolved significantly since Schweickert et al. (2009) demonstrated the impact early mobilisation can have on outcomes such as delirium. While usual care was often not elaborated on, most studies included some form of PA which may contribute to the lack of significant changes in incidence or duration that were found. Some studies specifically altering PA intensity between groups demonstrated reductions in both delirium duration 20 status 21 and incidence. 34 However further research into how PA intensity may impact delirium is required.
Intervention delivery time may be impacted by professions. Previous studies report greater perceived mobilisation barriers for ICU nurses with safety concerns classed as the main limitation.44,45 Overall, PA delivery by non-PTs may be reduced compared to interventions conducted by PTs as seen in the 8-min difference reported by Brummel and colleagues. 36 With ICU patients, who have a lower physiological reserve than healthy patients, such time differences could result in differing intensities being achieved emphasising this could be a potential factor in delirium prevention.
This review has several strengths, including methodology transparency by registering the study protocol prior to database searching. 46 It is one of few reviews not investigating the A-E/F bundle, but instead exploring the EM aspect as per recommendations from previous reviews. 19 The use of two reviewers independently searching and analysing the extracted data prior to discussion further improves this reviews rigour.
This review was limited by the exclusion of non-English language studies, potentially missing relevant studies in other languages. One of the papers included was identified and found in pre-print as part of the ROB assessments given its relevance to the question of interest. 37 Post ROB completion it has now been published with a change in methodological approach from an RCT to a pilot RCT therefore investigating evening mobilisation feasibility as its primary aim rather than proving the effect of mobilisation on delirium outcomes. 47 This means the primary outcomes also changed from delirium duration to feasibility measures. Therefore, results presented for this study should be interpreted with caution. Wide variation in delirium outcome reporting made data synthesis challenging, as also found in a recent systematic review by Rose et al. (2020). 18 Increased awareness of the variation in delirium assessment and documentation methods has led to the recent creation of an established delirium core outcome set (COS). This aims to improve comparisons regarding effective delirium interventions in future studies. 48
Current evidence is limited as delirium is generally only explored as a secondary outcome alongside poor intervention reporting and a lack of clarity about which PA intervention is most effective at impacting ICU delirium. Overall, there is insufficient high-quality evidence to suggest that PA is beneficial as a stand-alone treatment for delirium. In future, reviews may be better able to compare interventions given the recent focus on delirium and PA COS.48,49 Future, well designed and powered studies should investigate altering PA type, frequency, and intensity. Adequate and transparent reporting of delivered interventions to enable study replication and synthesis to inform best practise is also recommended.
Conclusion
In summary there is limited evidence to support the use of PA as stand-alone intervention to impact delirium in ICU. Delirium is a highly complex condition with poorly understood pathophysiology, suggesting that A-E/F bundles are likely to benefit delirium outcomes in ICU. PA intensity shows promise regarding impact on delirium but requires further research. Future reviews should include papers using specified COS allowing for thorough meta-analyses to be conducted and firmer conclusions drawn to guide best practice.
Supplemental Material
Supplemental Material - Investigating the impact of physical activity interventions on delirium outcomes in intensive care unit patients: A systematic review and meta-analysis
Supplementary material for Investigating the impact of physical activity interventions on delirium outcomes in intensive care unit patients: A systematic review and meta-analysis by Annika Jarman, Keeleigh Chapman, Sarah Vollam, Robyn Stiger, Mark Williams and Owen Gustafson
Footnotes
Acknowledgements
The authors would like to thank the Adult Critical Care Physiotherapy Team at the John Radcliffe Hospital and the Critical Care Research Group at the University of Oxford for their support.
Authors Contributions
AJ, KC, SV, RS, MW and OG all made a significant contribution to the concept and design of the review. AJ and KC undertook searches and data collection. OG supported AJ and KC with risk of bias assessments and interpretation. AJ, KC, SV, RS, MW and OG all contributed to the meta-analysis and GRADE review with MW supporting the interpretation of the results. AJ, KC, SV, RS, MW and OG have all contributed to and reviewed the draft and final versions of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Owen Gustafson, Clinical Doctoral Research Fellow, NIHR301569, is funded by Health Education England (HEE)/National Institute for Health Research (NIHR). Sarah Vollam and Annika Jarman were supported by the NIHR Oxford Biomedical Research Centre. The views expressed are those of the authors and not necessarily those of the NIHR, NHS or the UK Department of Health and Social Care.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
