Abstract
A 76-year-old lady was found on the floor following a fall at home. She was uninjured, but unable to get up, and had been lying on the floor for roughly 18 hours before her son arrived. She had been unwell for the past 3 days with a cough and shortness of breath. She had a past medical history of diabetes, hypertension, hypercholesterolaemia and atrial fibrillation (AF). On examination, she was alert but distressed, clinically dehydrated, febrile and tachycardic. She was treated for community acquired pneumonia with co-amoxiclav and was fluid resuscitated with Hartmann’s solution. Her hyperkalaemia was treated with 50 mL of 50% glucose containing 10 units of rapid-acting insulin. Her creatinine kinase (CK) on admission was 200,000, and she had an acute kidney injury (AKI). Urine dipstick was positive for blood. However, her renal function continued to deteriorate over the succeeding 48 h, when she required renal replacement therapy (RRT) due to fluid overload and anuria.
Keywords
Rhabdomyolysis
Rhabdomyolysis is characterised by skeletal muscle injury and release of intracellular contents into the systemic circulation – namely, potassium, phosphate, myoglobin, creatinine kinase (CK) and lactate dehydrogenase (LDH). The plasma protein binding capacity for myoglobin is exceeded when 100 g of muscle is lost. This results in free myoglobin being present in the urine and acute kidney injury Figure 1.
1
Observations and investigation results.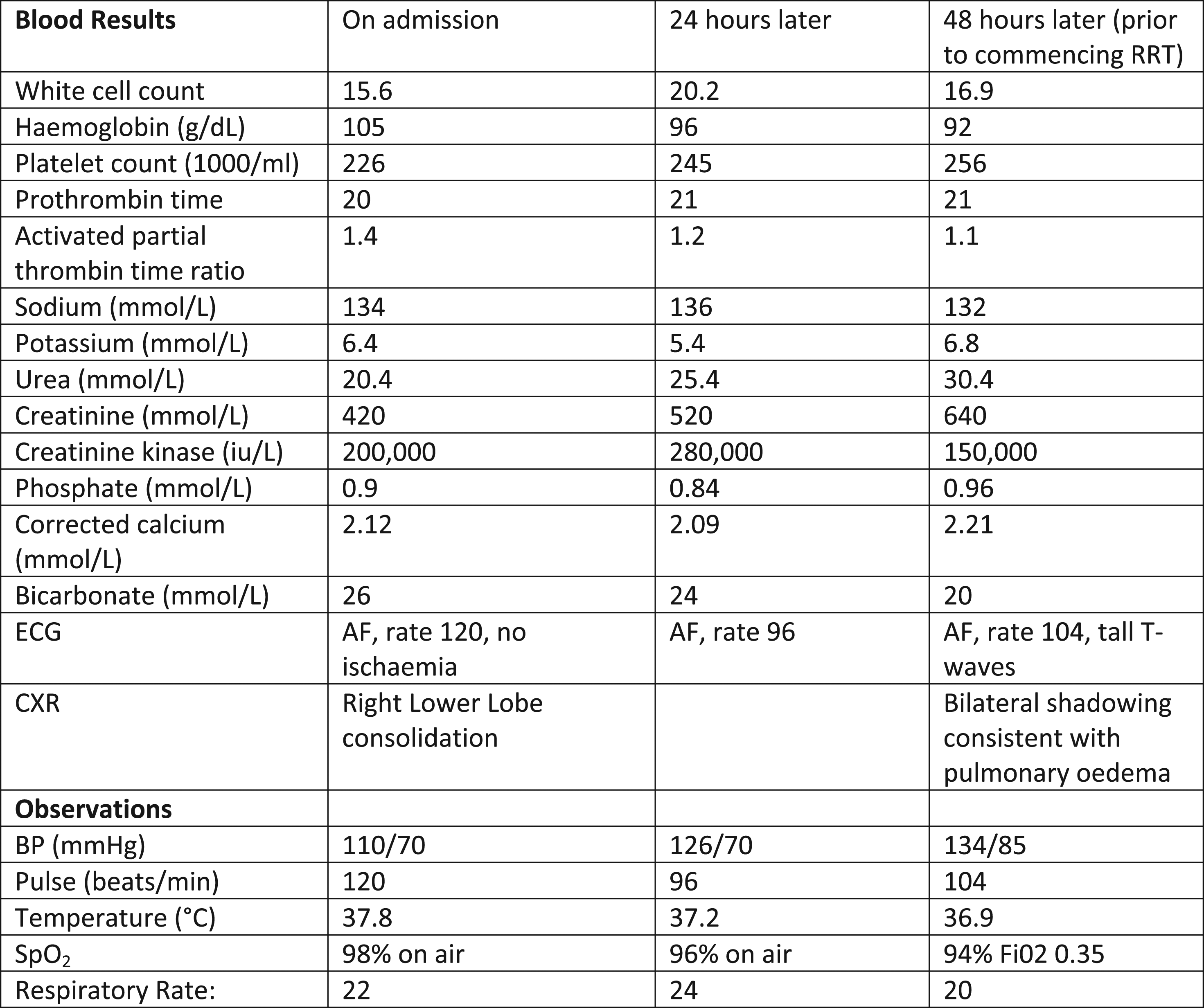
Rhabdomyolysis may complicate various disease states and is frequently multifactorial (Figure 2). The diagnosis is suggested by a congruent history and confirmed with an elevated CK, which may frequently number in the hundreds of thousands. Other clinical features include myalgia, acute kidney injury (AKI) and myoglobinuria (which causes a urine dipstick to be positive for blood). The cause for rhabdomyolysis is frequently multifactorial – in a case review of 475 hospitalised patients, 60% had greater than one risk factor for rhabdomyolysis. The commonest individual contributing factor was drugs, implicated in 46% of cases. The drugs involved were diverse and involved both prescribed medication and recreational drugs. 10% had rhabdomyolysis secondary to neuromuscular diseasei. In the patient mentioned above, the potential causative factors were prolonged immobility, sepsis and medications. Causes of rhabdomyolysis.
2
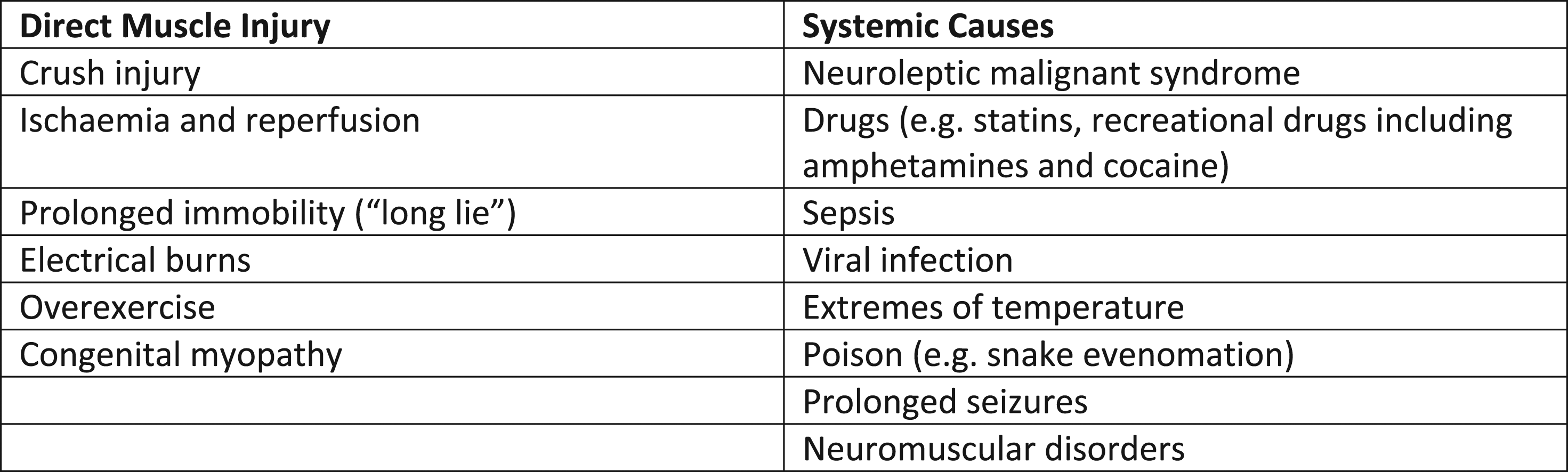
Diagnosing rhabdomyolysis
While a thorough medical history and examination may raise suspicion of rhabdomyolysis, the diagnosis is usually confirmed by the finding of an elevated creatinine kinase (CK). Features suggestive of rhabdomyolysis in the history include an episode of limb ischaemia, a fall followed by an extended period of immobility, and concomitant drug use. Clinical features suggestive of rhabdomyolysis include aching muscles, an unusual colour to the urine (classically described as “tea coloured”), and oligo-anuria. 3 Presenting patients may have a tachycardia secondary to pain, dehydration or fluid shifts into the muscles. Muscle swelling can be present on admission or become apparent after the patient has received fluid resuscitation. Skin changes such as bruising, and evidence of pressure necrosis can point to a compression injury. In critically ill patients, clinical signs may be masked or blunted, and therefore a high clinical index of suspicion is warranted. A rare, but important cause of rhabdomyolysis in critical illness is propofol infusion syndrome (PRIS). Various criteria have been used for diagnosing rhabdomyolysis, but the commonest recommended criterion in use is that a CK >1000 or 5 times the upper limit of normal is considered diagnostic of rhabdomyolysis. 4 CK typically continues to rise for the first 12–24 h following injury before beginning to declineii. Serial CK measurement at 12 hourly intervals until it begins to fall is useful for prognostication in terms of renal dysfunction. Dipstick urinalysis may show positive for blood, but urine microscopy typically reveals tubular casts (from the precipitation of myoglobin with Tamm–Horsfall protein in the urine), but no blood.
Patients with neuromuscular disease frequently have a chronically elevated CK. In these patients, reference to the baseline CK is important, and in diagnostic uncertainty, serial urinary myoglobin measurement may be useful.
Incidence
Given the varying definitions used for the diagnosis of rhabdomyolysis, the exact incidence is unknown. However, the Intensive Care Network Audit and Research Centre (ICNARC) reported 733 admissions to critical care where rhabdomyolysis was reported as the primary diagnosis in the time period 2006–2010. 5 This constituted 0.2% of all admissions to non-specialised adult critical care units. 75% of patients were male and the median age was 54.5. The in-hospital mortality was 31.1%. However, ICNARC only requires the reporting of the primary diagnosis, and given that rhabdomyolysis frequently complicates other disease states, this is likely to be an underestimate of the true incidence on the intensive care unit.
Complications of rhabdomyolysis
The commonest life-threatening complication of rhabdomyolysis is acute kidney injury, which complicates 13–60% of all casesv. Electrolyte disturbance is also common, due to the release of intracellular ions into the plasma. Disseminated Intravascular Coagulation (DIC), and circulatory shock can also complicate rhabdomyolysis regardless of underlying aetiology.
Acute kidney injury
AKI requiring renal replacement therapy complicates 13–60% of patients presenting with rhabdomyolysis. Acute kidney injury in rhabdomyolysis is multifactorial. 6 Muscle injury results in capillary leak and hence hypovolaemia. This causes renal arteriolar vasoconstriction via activation of the renin-aldosterone-angiotensin system. This is compounded by precipitation of myoglobin in the renal tubule, where it forms a compound with Tamm–Horsfall protein (also known as uromodulin). Tamm–Horsfall protein is the protein most frequently found in normal human urine. Precipitation of myoglobin is accelerated at lower urinary pH. 7 This causes direct tubular injury. Haem itself; contained within myoglobin; also causes oxidative cell damage to the renal tubule. 8
The McMahon score is a validated scoring system to predict those at higher risk of requiring renal replacement therapy, with a score greater than 6 conferring a risk of renal replacement therapy of 52% Figure 3.
9
The patient above would have a McMahon score of 11, demonstrating a high risk of needing renal replacement therapy. McMahon score for assessing risk of AKI in patients with rhabdomyolysis.
9
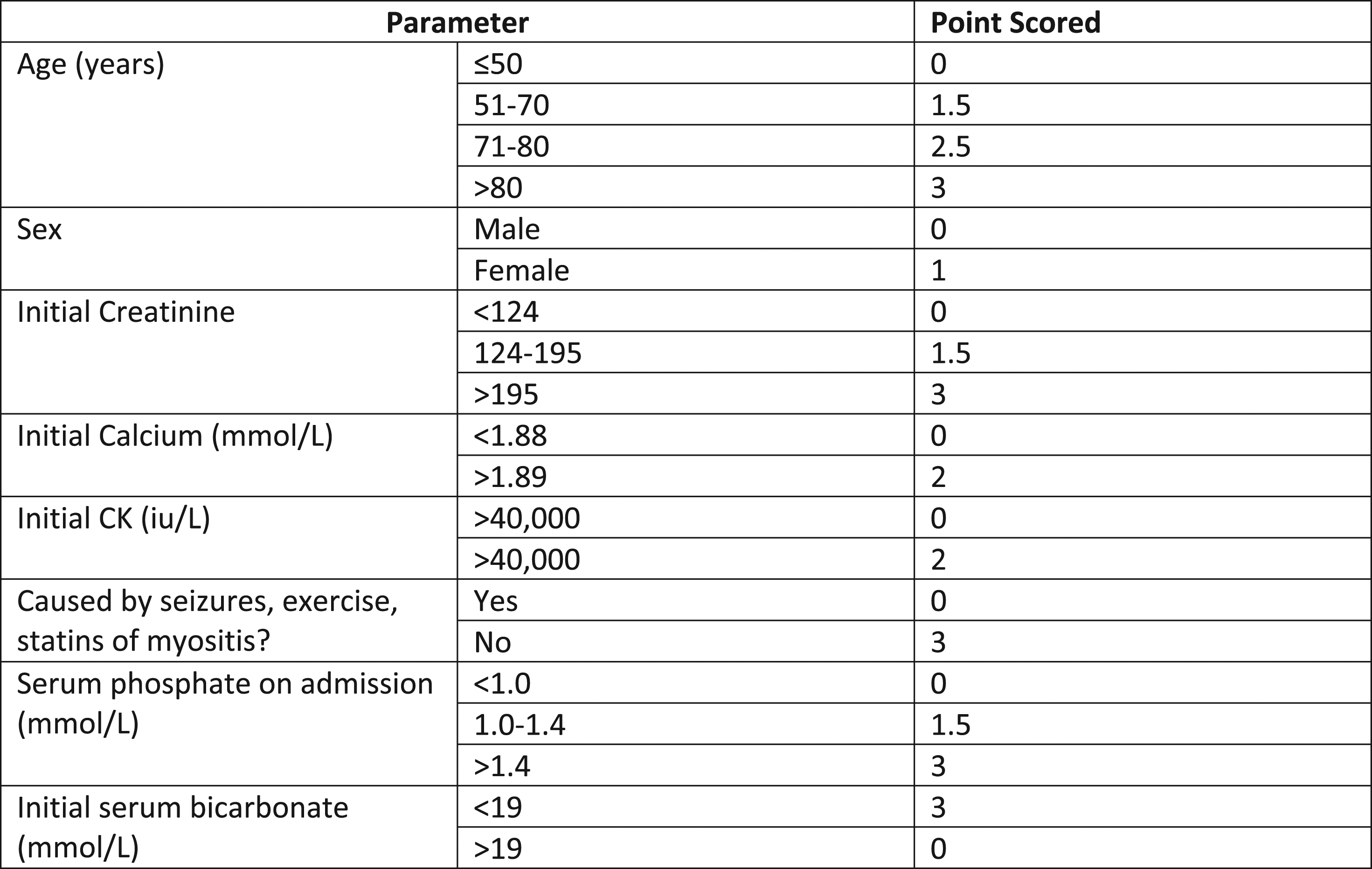
Electrolyte disturbance
Electrolye disturbances, including life-threatening hyperkalaemia are common due to the release of intracellular ions into the systemic circulation. Ion release can result in transient or prolonged hyperkalaemia, hyperphosphataemia and hypocalcaemia, which can overwhelm homoeostatic regulatory processes, and be exacerbated by AKI. 10 The management of these electrolyte disturbances differs little from the management in other settings, as in the patient above.
Circulatory failure
Extensive muscle cell death may result in a dysregulated inflammatory response, in the same way that any major injury can. This causes capillary leak, intravascular depletion and vasodilatation. Third spacing of fluids at the site of muscle injury can lead to compartment syndrome. This results in circulatory failure and hypotension. Release of pro-inflammatory cytokines from necrotic cells also decreases myocardial contractility. 11 Treatment of circulatory failure in rhabdomyolysis is the same as in non-rhabdomyolysis induced inflammation-mediated circulatory failure, that is, isotonic crystalloid, vasopressors and inotropes.
Disseminated intravascular coagulation
Disseminated intravascular coagulation (DIC) is a consumptive coagulopathy which can occur from a variety of aetiologies including severe systemic inflammation. It results from widespread activation of the coagulation cascade which causes microthrombus deposition in small vessels. This causes organ dysfunction due to microcirculatory abnormalities and a bleeding diathesis due to consumption of platelets and clotting factors. 12 The diagnosis of DIC can be suspected in patients who have thrombocytopenia (98% of DIC sufferers), elevated D-dimers, prolonged prothrombin time (PT) and activated partial thrombin time (APTT) (50–60%), and low fibrinogen (28%). Fragmented red cells may also be seen on blood film. 13 Treatment of DIC is primarily supportive; namely proactive treatment of the underlying cause. In the event of bleeding, clotting factors and platelets can be replaced under the guidance of a haematologist. Tranexamic acid should not be given to patients in DIC unless they have active bleeding. It is recommended that prophylactic dose low molecular weight heparin is continued in non-bleeding patients with DIC who do not require surgical intervention. 11
Compartment syndrome
Compartment syndrome occurs when pressure in a fascial compartment containing muscle exceeds first venous haemostatic pressure then arterial blood pressure due to muscle swelling. The cardinal clinical finding is pain on passive stretch of the muscle, which does not respond to conventional analgesia. These signs may be masked in critically ill patients and therefore a high index of suspicion is required. Symptoms and signs of ischaemia a (i.e. pale colour, loss of distal pulses and paraesthesia) are late findings and should not be relied upon for diagnosis. Compartment syndrome can initially be treated conservatively by elevation and removal of tight dressings, but can only definitively be treated by fasciotomy to relieve pressure in the offending compartment. 14 There are several case reports for the treatment of rhabdomyolysis-induced compartment syndrome by fasciotomy. 15 A retrospective case study of 164 cases (113 with a fracture and 38 without) in 2014 demonstrated that, compared to adults with fracture-associated compartment syndrome, adults without fractures experienced a longer time to surgery, and were more likely to require muscle debridement (20% vs 8%). 16 Delay to fasciotomy is associated with worse outcome in terms of limb function. 17
Renal protective therapy in rhabdomyolysis
Given the high incidence of AKI in patients with rhabdomyolysis, there has been considerable interest in therapies which may prevent the onset of AKI requiring RRT. 18 However, high quality evidence is lacking, with most coming from crush injuries sustained in earthquakes. Strategies advocated include aggressive fluid resuscitation, promoting alkalinisation of urine and use of diuretics.
Fluid resuscitation
Proactive crystalloid fluid resuscitation is the mainstay of treatment in the non-anuric patient with rhabdomyolysis. The aim of fluid resuscitation is to reduce renal vasoconstriction and produce dilute urine, thereby reducing myoglobin precipitation. There is limited anecdotal evidence suggesting an increase in the incidence of AKI in those in whom treatment is started >40 h after injury. 19 There is little evidence to support the use of one isotonic crystalloid solution over another. That said, 0.9% sodium chloride solutions are associated with hyperchloraemic metabolic acidosis, and worsening acidaemia may worsen intratubular myoglobin precipitation. 20 Many protocols recommend a target urine output of 200 mL/hr. Caution should be exercised in those with cardiac failure, or in patients with oligo-anuria which does not improve with adequate blood pressure management as overzealous fluid administration can result in pulmonary oedema.
Forced alkaline diuresis
Administration of Sodium Bicarbonate to increase urine pH has been an historic standard of care for rhabdomyolysis. The rationale for this is that Tamm–Horsfall protein precipitates more readily at lower pH. Hence, urine alkalisation may reduce tubular obstruction and be reno-protective. However, this is controversial in current practice. An observational study of patients with post-traumatic rhabdomyolysis showed no difference in the incidence of renal failure, renal replacement therapy or death in those receiving sodium bicarbonate and mannitol over those receiving fluid resuscitation only. 21 To date, no trials have compared administration of sodium bicarbonate alone to administration of other isotonic crystalloid. A systematic review of strategies to prevent acute kidney injury in rhabdomyolysis demonstrated no benefit in the use of any pharmacological agent beyond isotonic crystalloid. 22
Diuretic therapy may be reasonable to treat overload; however, there is insufficient evidence to support their routine use in patients with rhabdomyolysis. In particular, the use for furosemide to drive a target urine output of up to 200 mL/hr does not reduce the need for renal replacement therapy (RRT). The use of loop diuretics promotes aciduria, accelerating precipitation of myoglobin in the renal tubule. 23
Renal replacement therapy
There is a rationale for early renal replacement therapy in rhabdomyolysis, that is, enhancing myoglobin clearance may reduce tubular precipitation and promote renal recovery. However, a Cochrane review conducted in 2014 concluded there was insufficient evidence of benefit over conventional therapy to recommend RRT without an additional indication. 24 Even so, early RRT may be beneficial in patients who have contraindications to aggressive fluid resuscitation. The renal glomerulus remains the highest efficiency filter for myoglobin. The molecular weight of myoglobin is 17 kDa, and hence high flux, high permeability membrane haemofiltration or haemodiafiltration is needed to reduce plasma myoglobin concentration.25,26 RRT use has not been correlated to a reduction in the risk of needing long-term RRT, and the only descriptions of its use are from case series and case reports. 27
Development of AKI requiring RRT confers a mortality of 59%, compared to 22% without in the context of critical illness. However, survivors of rhabdomyolysis complicated by renal failure showed renal recovery in all cases of an observational series of 28 patients. 28
Conclusion
Rhabdomyolysis is an underdiagnosed, life-threatening complication of many critical illness states. Diagnosis is made on the basis of an elevated CK. The major complication is AKI requiring RRT, and the McMahon score may be useful for risk-stratifying patients. There is as yet little evidence to support any therapy beyond proactive fluid resuscitation. As with many clinical syndromes, addressing the underlying cause is critical in preventing further muscle injury. The major complications are compartment syndrome, renal dysfunction, electrolyte disturbance and DIC. Critically ill patients with rhabdomyolysis have a high mortality; however, those who survive AKI and RRT are likely to recover to non-dialysis dependent renal function.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
