Abstract
Objective
We aimed to summarize the characteristics and outcomes of patients undergoing left ventricular assist device (LVAD) implantation, and to determine the incidence, clinical implications, and in-hospital risk factors of acute kidney injury (AKI) requiring continuous renal replacement therapy (CRRT).
Methods
Patients who underwent LVAD implantation from October 2022 to July 2025 were included in this retrospective analysis. Data collection included preoperative and intraoperative baseline characteristics, in-hospital complications, and coronary intensive care unit and total hospital stays. AKI was defined according to the KDIGO criteria. Univariate and multivariate logistic regression were used to identify independent risk factors for CRRT. Receiver operating characteristic curve analysis was used to assess the impact and predictive efficacy of significant variables.
Results
Of the 27 patients (median age: 61 years; male: 88.9%), 24 (88.9%) developed AKI and 5 required CRRT postoperatively. Based on CRRT requirement, patients were stratified into two groups: CRRT (n=5) and non-CRRT (n=22). Univariate analysis associated CRRT occurrence to cystatin C levels, duration of vasoactive drug use, ventricular arrhythmias, and fluid balance on the second postoperative day; the duration of vasoactive drug use was revealed as an independent risk factor. The vasoactive-inotropic score (VIS) strongly predicted CRRT occurrence when VIS exceeded 54.5.
Conclusion
This study found a high incidence of AKI requiring CRRT in patients receiving LVAD implantation. VIS and duration of vasoactive drug use were significant factors linked to CRRT, suggesting the potential of vasoactive drug usage as a predictive indicator for CRRT and further validation through larger-scale studies is still required.
Keywords
Introduction
Heart failure (HF) is a major public health concern affecting approximately 1%-2% of the adult population, with an incidence of 100-900 cases per 100,000 persons each year. 1 Advanced HF is resistant to optimal medical treatment and unsuitable for conventional surgery. It poses a substantial burden on patients and the healthcare system due to its high short-term morbidity and mortality. 2 Consequently, left ventricular assist devices (LVADs) play a significant role as a salvage therapy, serving as a bridge to transplantation or permanent destination treatment in patients with acute or chronic end-stage heart failure (ESHF). Despite the establishment that cardiac output increases following LVAD implantation3,4; it is still a life threatening and high-risk surgical procedure.
Acute kidney injury (AKI) is a common complication after cardiac surgery, with an incidence of 15%-45% after LVAD implantation. AKI leads to high mortality, right heart failure and arrhythmia, and shortened life expectancy.5–9 However, there are conflicting reports regarding the impact of AKI on renal function after LVAD implantation. Despite studies reporting an increase in the estimated glomerular filtration rate (eGFR) in the early postoperative, period 1 month after LVAD implantation, 10 other studies have shown contrasting results. 11 Moreover, the development of AKI after LVAD implantation has generally been reported at 1 month, 3 months, and 1 year postoperatively; hence, data from the first week are scarce. 10
Therefore, in this exploratory study, we aimed to summarize the characteristics and outcomes of patients who underwent LVAD implantation at our center, evaluate the early incidence of AKI within the first week post-implantation, and identify the in-hospital risk factors for continuous renal replacement therapy (CRRT), providing important insights for future large-scale studies.
Materials and methods
Study design and population
This single-center, retrospective analysis included 27 consecutive patients with ESHF who received LVAD implantation as destination therapy between October 2022 and July 2025 at our center. The inclusion criteria for this cohort were defined as: (1) aged ≥ 18 years, (2) meeting the diagnostic criteria for ESHF and intolerance to conventional anti-heart failure drug therapy, and (3) provision of signed informed consent for LVAD implantation and data use. The exclusion criteria were as follows: (1) aged < 18 years; (2) death within 48 hours after LVAD implantation; (3) intolerance to conventional anticoagulation or surgery; (4) uncontrolled infections; (5) end-stage diseases of other organ systems, such as malignancies or uremia; and (6) psychological disorders, mental illnesses, or cognitive impairments preventing compliance with treatment instructions. The study flowchart is shown in Figure 1. The flowchart of this study.
After inducing general anesthesia, cardiopulmonary bypass was initiated through cannulation of the ascending aorta and superior/inferior vena cava. A median sternotomy was performed, the LVAD was implanted at the left ventricular apex, and the outflow artificial graft was anastomosed to the ascending aorta. All the surgical procedures were performed by the same highly-skilled and experienced surgical team throughout the study. Goal-directed fluid therapy (GDFT) was implemented in surgical fluid management. Advanced hemodynamic monitoring tools, such as pulmonary artery catheters or transesophageal echocardiography, were employed to obtain real-time and precise measurements of key indicators including cardiac output, stroke volume, heart chamber sizes, and inferior vena cava width. Based on these indicators and the specific conditions of each patient, fluid infusion was dynamically adjusted to prevent fluid overload or insufficiency. Moreover, autologous blood transfusion devices were prepared preoperatively for surgeries expected to have significant blood loss, and the autologous blood transfusion technique was used intraoperatively. This approach reduced the demand for allogeneic blood. Additionally, corresponding allogeneic blood products were precisely transfused according to the patient’s coagulation function and blood loss status to maintain normal coagulation function and minimize bleeding risks. Postoperatively, a standardized and consistent management strategy was carried out by the same coronary intensive care unit (CICU) team, strictly adhering to established clinical guidelines. 12
This study was approved by the Ethics Committee of Nanjing Drum Tower Hospital (Approval Number: 2025-1290-01; Date: 2026-01-29), and written informed consent was obtained from patients and their families before surgery.
Study endpoints and data collection
Broadly, we collected and evaluated the following patient data: preoperative characteristics, perioperative information, duration of hospital and CICU stay, and postoperative complications. Specifically, preoperative traits included patients’ characteristics and demographics (age, sex, comorbidities, cause of heart failure, and body surface area [BSA]), baseline hemodynamics parameters (cardiac index, pulmonary artery wedge pressure [PAWP], mean pulmonary artery pressure [mPAP], central venous pressure [CVP], and left ventricular ejection fraction [EF]), use of an intra-aortic balloon pump (IABP) or ventilator support, laboratory data (blood urea nitrogen [BUN], creatinine, transaminase, cystatin C [Cys-C], total bilirubin [TB], white blood cell count [WBC], platelet count, and international normalized ratio [INR]); perioperative information included intraoperative bleeding volume and transfusion requirements, cardiopulmonary bypass duration and aortic cross-clamp time; postoperative hemodynamic parameters, results of transesophageal echocardiography on postoperative day 1 (the ratio of the left and right heart and the width of the inferior vena cava [IVC]), LVAD pump-related metrics, and fluid balance records for 3 consecutive postoperative days in the CICU.
We used the Kidney Disease Improving Global Outcome (KDIGO) criteria to define AKI, 13 during the first week after LVAD implantation. Serum creatinine, eGFR and BUN values were evaluated each day within the first postoperative week. CRRT was defined as the need for treatment with either continuous venovenous hemodiafiltration. Patients were assigned to the CRRT group if they required CRRT within the first week after LVAD implantation. Other adverse complications included re-thoracotomy, sepsis, stroke, and HF. Data were obtained from a computerized database and electronic patient records, which were systematically collected during hospitalization.
Statistical analysis
Continuous variables are presented as medians and interquartile ranges or means and standard deviations (SDs), depending on the distribution, and were compared using the Welch two-sample t-test or Wilcoxon rank-sum (Exact) test. Categorical variables are presented as counts and percentages, and were compared using Pearson’s chi-squared test for trends. Univariate and multivariate ordinal logistic regression analyses were performed for the identification of risk factors associated with AKI for CRRT. Given the relatively small sample size of our study and the aim to comprehensively explore potential risk factors, variables with a p-value of less than 0.10 in the univariate analysis were included in the multivariate models. All multivariate models were constructed using the forward selection method. Analyses were conducted using SPSS version 25.0 (IBM SPSS, Inc., Armonk, NY) and GraphPad Prism version 10 (GraphPad Software, Inc., La Jolla, CA). A p-value of < 0.05 was considered statistically significant.
Results
Baseline characteristics
During the study period, 28 patients received continuous-flow LVADs, with one patient excluded owing to intraoperative death for severe cardiogenic shock. The median age was 61 years, and 88.9% of patients were male. The mean BMI was 24.57 ± 3.94 kg/m2, and the median BSA was 1.82 (1.67-1.89) m2. History of smoking and alcohol consumption were noted in 14 and 5 patients, respectively. Five patients had a history of implantable cardioverter-defibrillator implantation. Coronary artery disease was present in 55.6% of patients; however, only 14.8% underwent percutaneous coronary intervention. Hypertension and diabetes were common comorbidities (50% and 60%, respectively), with chronic kidney disease (CKD; 11.1%) and atrial fibrillation (29.6%) being less frequent. One patient had stroke-related hemiplegia, and three had ventricular arrhythmias. One patient had a history of prior cardiac surgery. HF was caused by idiopathic cardiomyopathy in 66.7% of patients, the rest being caused by ischemia.
Most patients (77.8%) were classified as Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) levels 1-4, and 48.1% were New York Heart Association (NYHA) functional class IV. Continuous catecholaminergic support was needed in 22.2% of the patients, with one requiring extracorporeal membrane oxygenation assistance. Biological markers showed elevated brain natriuretic peptide (BNP) or NT-proBNP levels. Echocardiography showed an enlarged left ventricular end-diastolic diameter (LVDd) of 7.24 ± 0.78 cm and borderline right ventricular function, as indicated by a tricuspid annular plane systolic excursion of 1.57 ± 0.28 cm, an S′ velocity of 8.5 (7.3, 10) cm/s, and a fractional area change of 35.25% ± 6.55%. Approximately 44.4% of the patients had severe mitral regurgitation, with one patient each having severe aortic regurgitation and tricuspid regurgitation.
Demographics, preoperative factors, and laboratory data of the patients.

Note. The continuous variables and categorical variables were presented as mean ± SD, median and IQR, numbers and percentages, respectively.
1Wilcoxon rank sum test.
2Welch Two Sample t-test.
3Wilcoxon rank sum exact test.
4Pearson’s Chi-squared test.
Bold values represented P < 0.05.
Operative and post-operative outcomes of patients with LVAD
All patients underwent LVAD as destination therapy. A Corheart 6 (Shenzhen Core Medical Technology Co., Ltd., Shenzhen, China) was implanted in 20 patients, an EVAHEART (Chongqing Evaheart Medical Device Co., Ltd., Chongqing, China) was implanted in 2 patients, and a HeartCon (Rocor Medical Technology Co., Ltd., Tianjin, China) was implanted in 5 patients. Different concomitant procedures were performed in 20 patients during the index operation, and one patient received a right ventricular assist device. The median operative time was 325 (300, 409) min, with average cardiopulmonary bypass and aortic cross-clamp times of 157.31 ± 30.87 and 106.73 ± 23.8 min, respectively. The median blood loss was 1200 (900, 1400) mL, and the average blood transfusion volume was 1380.63 ± 618.84 mL.
Postoperatively, patients were transferred to the CICU, where the median rotational speed of the LVAD was 2599 (2498, 2797) rpm, with an average flow rate of 3.98 ± 0.8 L/min. The median vasoactive inotropic score (VIS) was 53 (33, 85), mean arterial pressure was 78.33 ± 10.02 mmHg, CVP was 13 (12, 16) mmHg, and PAWP was 15 (11, 18) mmHg. Transoesophageal echocardiography showed balanced left and right ventricular sizes, with an IVC diameter of 21.3 ± 1.45 mm and a variability of 9 (8, 15) %. The patients received respiratory and circulatory support therapy, with a median duration of inotropic drug use for 5 (3, 7) days and mechanical ventilation time of 1066 (880, 1218) min.
Interoperative and postoperative factors of the patients.
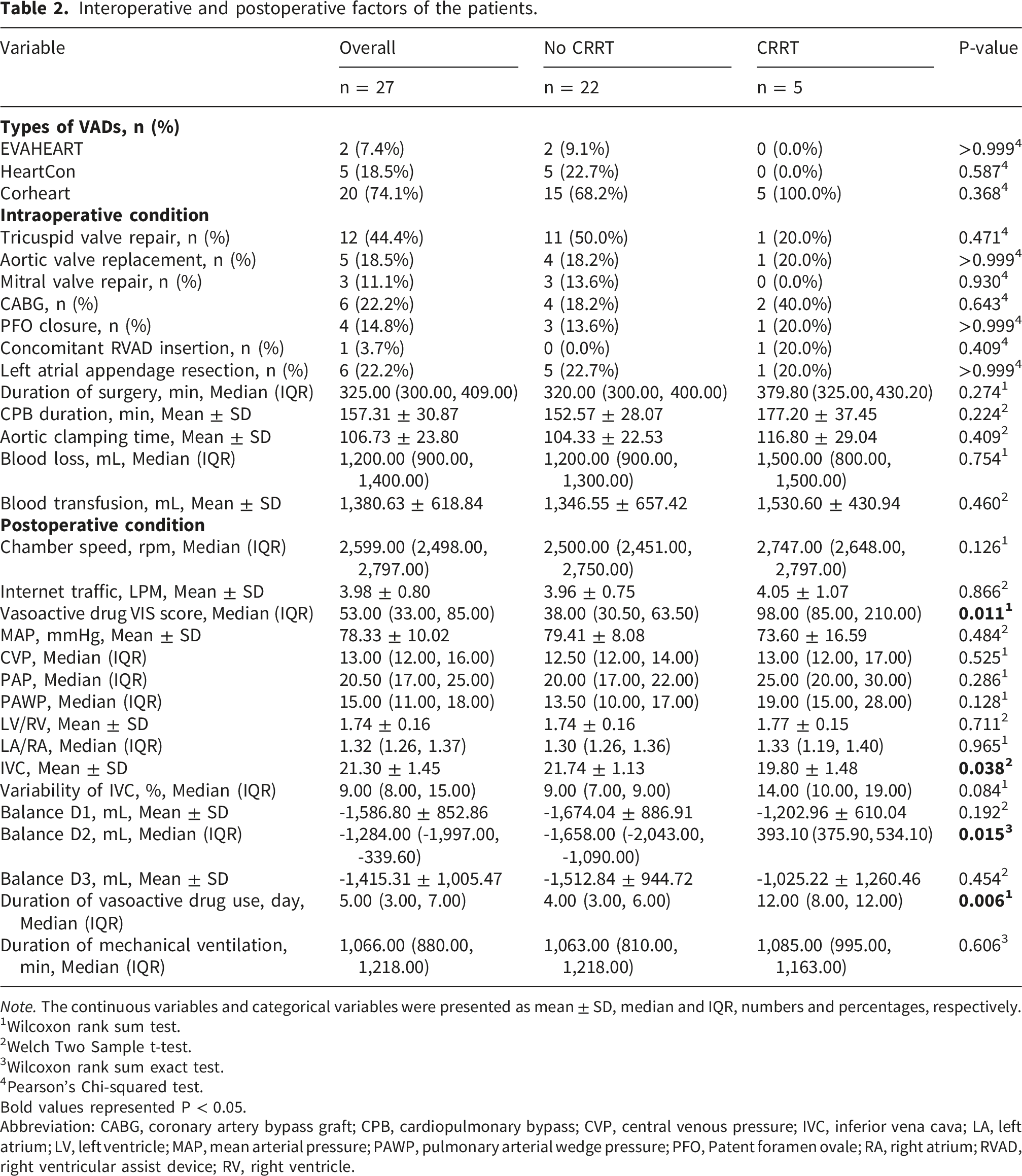
Note. The continuous variables and categorical variables were presented as mean ± SD, median and IQR, numbers and percentages, respectively.
1Wilcoxon rank sum test.
2Welch Two Sample t-test.
3Wilcoxon rank sum exact test.
4Pearson’s Chi-squared test.
Bold values represented P < 0.05.
Abbreviation: CABG, coronary artery bypass graft; CPB, cardiopulmonary bypass; CVP, central venous pressure; IVC, inferior vena cava; LA, left atrium; LV, left ventricle; MAP, mean arterial pressure; PAWP, pulmonary arterial wedge pressure; PFO, Patent foramen ovale; RA, right atrium; RVAD, right ventricular assist device; RV, right ventricle.
Postoperative comorbidities and outcomes of the patients.
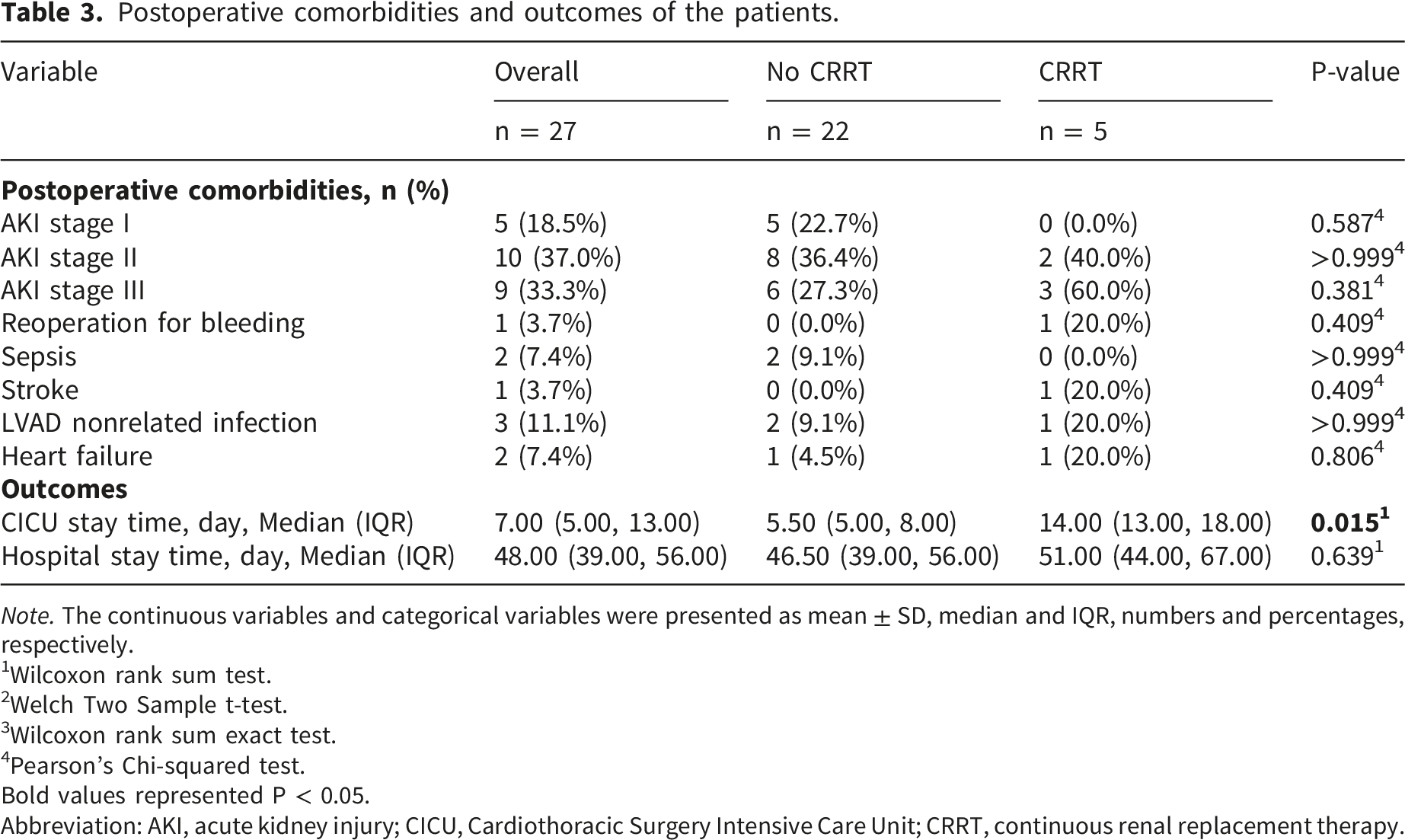
Note. The continuous variables and categorical variables were presented as mean ± SD, median and IQR, numbers and percentages, respectively.
1Wilcoxon rank sum test.
2Welch Two Sample t-test.
3Wilcoxon rank sum exact test.
4Pearson’s Chi-squared test.
Bold values represented P < 0.05.
Abbreviation: AKI, acute kidney injury; CICU, Cardiothoracic Surgery Intensive Care Unit; CRRT, continuous renal replacement therapy.
Risk factors for postoperative AKI with CRRT in patients with LVAD
AKI was a common complication (24/27), with five patients requiring CRRT. Patients were divided into two groups: CRRT (n = 5) and non-CRRT (n = 22). No significant differences were found in preoperative comorbidities and cardiac function between the two groups. However, compared with the non-CRRT group, the CRRT group had lower BMIs and worse renal function (as evidenced by higher Cys-C levels) preoperatively; and narrower IVC diameters, higher IVS, longer durations of vasoactive drug use, greater positive fluid balance on the second day (Balance D2), and extended CICU stays (all p < 0.05) postoperatively. Baseline data are summarized in Tables 1–3.
Univariate and multivariable analysis.

Receiver operating characteristic (ROC) curve analysis was performed by including factors with a p-value of < 0.1 in the univariate analysis. Compared with the case where there is no real predictive link between the variables and the requirement for CRRT due to AKI (the null hypothesis scenario), the results demonstrated that Cys-C level, fluid balance on the second day post-surgery, duration of vasoactive drug use, ventricular arrhythmia, VIS, and IVC width were all predictive of the need for CRRT owing to AKI in patients following LVAD implantation (p < 0.05). Notably, VIS had the highest area under the curve (AUC: 0.9; p=0.008), with 100% sensitivity and 81.2% specificity when exceeding 54.5. Moreover, vasoactive drug use duration over 7.5 days predicted CRRT with 80% sensitivity, 93.7% specificity, and an AUC of 0.88 (Figure 2, Table 5). Receiver Operating Curve (ROC) for postoperative AKI with CRRT in patients with LVAD. Receiver operating characteristic curve. Abbreviation: AUC, the area under the curve; BMI, body mass index; CKD, chronic kidney disease; Cys-C, cystatin C; CRRT, continuous renal replacement therapy; IVC, inferior vena cava. Bold values represented P < 0.05.

Discussion
Recently, China has achieved breakthroughs in the research and development of LVADs, with domestic devices proving effective and safe in trials, greatly benefiting patients with ESHF. 14 However, this progress raises the risk of complications, spurring demand for more perioperative management and postoperative complication prevention/treatment experience. AKI is a frequent complication of LVAD implantation, severely impacting patients who may subsequently require CRRT. This study summarizes our single-center experience in the perioperative management and in-hospital outcomes for patients undergoing LVAD surgery, and explores the possible risk factors for AKI and subsequent CRRT.
AKI is a common complication seen in 15%-45% after LVAD implantation,6–9 while the incidence of RRT ranges from 11%-33%,15,16 which is mainly closely related to factors such as surgical duration, blood transfusion, and cardiopulmonary bypass. Although studies have demonstrated that the majority of patients have renal improvement after LVAD support. However, this improvement only lasts a month, after which eGFR values decrease again. 17 In our study, all the patients received LVAD implantation were in end-stage heart failure. Patients with ESHF often have long-term excessive use of diuretics and repeated hospitalizations for the administration of nephrotoxic medications such as antibiotics. 18 Moreover, given the decreased renal perfusion due to low cardiac output and use of vasoconstrictors such as positive inotropic agents, it is not surprising that patients with HF undergoing LVAD implantation often have impaired baseline renal function. 19 In our study, the proportion of patients with CKD was 3/27 (11.1%), which undoubtedly significantly increased the risk of postoperative AKI.
According to the KDIGO diagnostic criteria, 24 patients developed AKI, with an incidence rate of 88.9%, which is much higher than the incidence rates reported in previous studies. This may be because most of those previous studies generally focused on the development of AKI after LVAD implantation at 1, 3 months, and 1 year, 10 while our study paid attention to the changes in renal function and urine output within 1 week after surgery. Nevertheless, after volume adjustment and drug support within one-week, renal function in 19 patients returned to the preoperative level, and ultimately, only 5 (18.5%) patients required CRRT, which is consistent with previous research data.15,16 Moreover, with the advancement of ventricular assist device technology and improvements in perioperative management, the incidence of AKI is expected to further decrease in the future.
The occurrence of AKI significantly prolongs hospital and ICU stays. Moreover, it is strongly associated with increased mortality, particularly among patients requiring CRRT. 11 Moreover, earlier studies that addressed the impact of AKI on mortality after LVAD implantation have reported that AKI is an independent predictor of mortality,6,20 with mortality rates as high as 40%-70%. Patients requiring CRRT are at the highest risk for mortality after LVAD implantation.16,21,22 Preoperative eGFR, use of an IABP, advanced age, elevated LVAD risk scores, prolonged cardiopulmonary bypass time, intraoperative bleeding, and need for reoperation have been validated as predictors of postoperative renal failure.9,16,23 However, studies on AKI incidence in patients with LVADs remain limited by their single-center design and inconsistent criteria for AKI definition. In this study, we reported the overall incidence of AKI according to the KDIGO guidelines. In addition, we stratified patients based on requirement for CRRT due to AKI to evaluate the risk factors for patients requiring CRRT. Moreover, we identified the duration of vasoactive drug use as an independent risk factor, showing 80% sensitivity and 93.7% specificity, and an AUC of 0.88, when the duration of vasoactive drug use exceeded 7.5 days. Additionally, the VIS also emerged as a strongly predictor of CRRT, with an AUC of 0.9. A VIS threshold exceeding 54.5 demonstrated 100% sensitivity and 81.2% specificity in predicting the need for CRRT. Prolonged exposure to vasoactive agents may contribute to inadequate renal perfusion, leading to renal dysfunction and, ultimately, the need for CRRT. Previous studies have also identified inotropic medicine usage as an independent predictor of AKI and its severity. However, they failed to evaluate dosing regimens or treatment durations. 11 This study utilized the VIS and the duration of vasoactive drug use to precisely predict the need for CRRT in patients with AKI, emphasizing the importance of optimizing pharmacotherapy in the perioperative period. It also emphasized the critical role of postoperative hemodynamic status in identifying high-risk individuals and enabling timely prophylactic interventions.
An increasing number of LVADs are being implanted as destination therapy.24,25 This rise is partly attributed to patients who are referred for LVAD as destination therapy because of their ineligibility for heart transplantation due to renal failure.26,27 A worse baseline eGFR is reported to be a predictor of mortality and worse renal function during LVAD support.16,28 Consistent with previous findings,29–31 our study showed that preoperative renal function, as reflected by Cys-C levels and CKD comorbidity, was significantly associated with the risk of postoperative CRRT for AKI. Patients with baseline renal impairment were more prone to developing severe AKI necessitating CRRT. This finding underscores the necessity for thorough preoperative renal assessment and optimization strategies to mitigate postoperative renal complications.
Intraoperative factors such as cardiopulmonary bypass time and aortic cross-clamp time did not emerge as independent predictors of CRRT in our study. This may be attributed to the extensive experience of our center in surgical techniques and postoperative management. Interestingly, postoperative fluid balance, particularly positive fluid balance on the second postoperative day, was associated with AKI necessitating CRRT, emphasizing the importance of early and aggressive fluid management. Patients who receive LVADs often exhibit significant volume overload and tissue oedema postoperatively; hence, strict monitoring of fluid status and timely adjustments can help prevent fluid overload and subsequent renal dysfunction. Additionally, our univariate analysis indicated an association between ventricular arrhythmias and CRRT, which may be related to the hemodynamic instability caused by ventricular arrhythmias, leading to reduced renal perfusion. Therefore, close postoperative surveillance of renal function, haemodynamics, fluid status, and score and time of vasoactive drug administration may enable the prompt identification of patients at risk for CRRT, thereby facilitating timely interventions.
Interestingly, many of the traditional variables associated with right heart failure (e.g., CVP, PAPi, bilirubin and TAPSE) and resulting in postoperative complications are not seen here as being significant, which may be attributed to the relatively preserved preoperative right ventricular function in our cohort. Additionally, this study focused on early AKI following LVAD implantation, whereas the impact of right heart failure on renal function may primarily manifest in long-term outcomes—an aspect potentially uncaptured by our current analysis.
The strengths of the present study include the consistent strategies in patient selection, operative procedures, and postoperative management in patients undergoing LVAD implantation. Furthermore, the present study included detailed analyses of serial echocardiograms, organ function, and in-hospital outcomes that cannot be addressed in multi-center studies.
However, our study also has a few limitations. First, the retrospective observational study design does not offer the opportunity to establish causality; it is also susceptible to selection bias, and may reflect institution-specific practices. Second, this study lacked prior sample size estimation due to its exploratory nature and insufficient prior data, and future research will expand samples and get more accurate estimates and variability for proper sample size calculation. Third, among the 27 patients, only five required CRRT. This limited sample size may have reduced the statistical power of our analyses and increased the risk of type II errors. Moreover, the multivariate logistic regression may have been overfit due to the small cohort size, limiting the identification of independent predictors. Additionally, collinearity diagnostics were not performed, which means we may fail to evaluate potential correlations among variables, affecting the stability of regression coefficients. The limited sample size also reduces the generalizability of the findings to broader LVAD populations. Forth, this study included three types of LVADs: CorHeart (n = 20), HeartCon (n = 5), and EVAHEART (n = 2). Potentially, each of them has distinct hemodynamic profiles, perioperative management protocols, and complication rates. Pooling patients on different devices into a single analysis without stratification or adjustment for device type introduces a major source of heterogeneity and potential confounding, making it difficult to attribute the observed renal outcomes to common perioperative factors rather than device-specific effects. Hence, a bigger size and more homogeneous patient cohort (by device type) may be necessary to strengthen the validity and clinical applicability of our results in the future. Fifth, the patients’ renal function recovered prior to discharge and they were subsequently weaned off CRRT; however, our statistical analysis was confined solely to the data gathered during their hospitalizations. Given the lack of long-term follow-up, it is necessary that ongoing surveillance be conducted to assess their renal function recovery over an extended period in the future. Finally, although our research first uncovered a potential link between vasoactive drug use and AKI after LVAD implantation, providing initial caution for clinical perioperative care. Due to the small sample size, we couldn’t analyze specific impacts of drug types, doses, or combinations, so we didn’t offer clinical recommendations. In the future, we’ll conduct a multicenter prospective RCT to assess kidney effects of optimized strategies for different vasoactive agents, including dose and timing, to offer high-level evidence for clinical practice.
Notably, although the blood loss and blood transfusion volumes at our center are within the normal range for the types of surgeries involved in this study and are comparable to those of other centers, minimally invasive LVAD implantation surgeries are poised to be a promising development in the future, which are projected to decrease intraoperative bleeding, cut down on unnecessary blood transfusions, and contribute to a speedier recovery for patients.
Conclusion
Our exploratory study revealed a high incidence of AKI necessitating CRRT among patients undergoing LVAD implantation at our center. Notably, the utilization of vasoactive drugs, encompassing both the VIS and the duration of use, significantly linked with CRRT, suggesting the potential of vasoactive drug usage as a predictive indicator for CRRT. This point requires in-depth validation in studies with larger sample sizes in the future owing to the small sample size and retrospective nature of our data.
Footnotes
Acknowledgements
We thank all the researchers for their contribution and support for making this study possible. This work was supported by grants from National Natural Science Foundation of China (No. 82400942).
Ethical consideraions
This study was approved by the Ethics Committee of Nanjing Drum Tower Hospital (Approval Number: 2025-1290-01; Date: 2026-01-29).
Consent to participate
Written informed consent was obtained from the participants/patients for the publication of this manuscript.
Author contributions
Q.D. & Q.Z. & C.C.: Data extraction, data analysis and interpretation and manuscript writing and figure drawing and table preparation. Z.X.: Data provision and data interpretation. J.P. & M.G.: Study design, data interpretation, manuscript writing and drawing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (82400942).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets analyzed and generated during the current study are available from the first author on reasonable request.
