Abstract
Stable silver nanoparticles of various sizes are synthesised using the aqueous extract of Tulsi (

Introduction
Silver nanoparticles (AgNPs) are the most widely used nanoparticles due to the ease of their preparation in the pure form. 1 AgNPs share more than 50% of the global market of nanomaterial-based consumer products 2 and have already been approved by the regulatory agencies for use in various applications requiring direct human contact.3,4 Their unique antimicrobial, electronic, optical and magnetic properties have resulted in their application in several diverse fields such as biomedical sensors,1,5 antimicrobial products, 6 food packaging materials, 7 fabrics, bandages, 8 catalysis, 9 optoelectronics and photonics 10 and pharmaceuticals. 11
The unique properties of AgNPs depend mainly on their shape and size. However, the variation in their size and size distribution is the greatest hurdle in the synthesis of AgNPs. 12 Several attempts have been made to control the particle size using different biological, 13 chemical and physical methods, 14 but these are still a matter of ongoing research. The chemical methods are neither eco-friendly nor economical as they use hazardous and expensive reagents and require high-energy input. 15 Thus, there is a need to explore new eco-friendly methods for the synthesis of AgNPs having the potential for large-scale production.
The role of the size of AgNPs in their antimicrobial activity has been reported by several researchers. 11 AgNPs as such or embedded within different polymeric materials have been used for developing antimicrobial films using different materials for applications such as antifouling membranes, 16 packaging 17 and wound dressing. 18 The current focus is on the development of active biocompatible polymer–AgNPs films with antimicrobial properties that permit the diffusion of an active ingredient to the applied site. Such attempts have been made for developing active food packaging materials, which not only protect the food material from bacterial infection but also protect their flavours and nutritional value. 19
A large volume of published information is available on the green synthesis of AgNPs using a variety of plant extracts,
20
but the relationship between the size of the AgNPs formed and the wavelength of the light spectrum has not been reported for
In this work, an effort has been made to study the effect of different wavelengths of the solar light spectrum (UV and visible) on the size of AgNPs synthesised using the extract of
Results and discussion
Analysis of the UV–Visible spectra
The rapid change in the colour of the leaf extract–AgNO3 colloidal solution mixture from light yellow to brownish-yellow due to the reduction of silver nitrate confirmed the formation of AgNPs. The colour intensified with time. The change in colour can be attributed to the reduction of Ag+ to Ag0 by various active biomolecules present in the Tulsi extract in the presence of light. 22 The spectrophotometric measurements were performed by UV–Visible spectrophotometry to study the formation and growth of AgNPs (Figure 1). As can be seen peaks due to AgNPs are observed within the range of 420–430 nm. Figure 1 shows the spectral behaviour due to AgNP biosynthesis after 2 and 6 h of exposure to light. The absorption spectra of Tulsi extract and silver nitrate did not show any peaks in the visible region, but Tulsi extract colloidal solutions with AgNO3 showed a characteristic peak at 420–430 nm due to surface plasmon resonance, a characteristic phenomenon of AgNPs. The observed results are in agreement with the findings of Prathna et al. 23 who used lemon extract for the synthesis of AgNPs and reported that the single absorption peak observed in the wavelength range 400–420 nm corresponds to the spherical shape of the formed AgNPs. The stability of the nanoparticles was monitored as a function of time. The spectrophotometric data confirmed that, with the passage of time, the synthesised AgNPs aggregated, leading to a red shift and broadening of the peak of the spectrum after 6 h of exposure (Figure 1). It was also observed that the broadened peak was prominent in the case of the sunlight-exposed reaction mixture. The UV light–treated solution did not show any peak confirming negligible AgNP formation at wavelengths less than 380 nm.

Effect of the irradiating light wavelength on the spectra of AgNPs after 2 and 6 h of exposure.
Scanning electron microscopy–energy-dispersive X-ray analysis
Analysis of the SEM results clearly showed the presence of spherical AgNPs without any agglomeration (Figure 2). The average particle size obtained through SEM analysis was 85 ± 37 nm. The peak present in the energy-dispersive X-ray analysis (EDX) analysis confirmed the presence of AgNPs. The peak due to silica can be seen in the EDX analysis. This may be due to the inclusion of the base material of the coverslip on which the sample has been prepared for SEM.

Effect of sunlight on the AgNP shape and composition: (a) SEM image and (b) EDX image.
Effect of the refractive index
The optical properties of AgNPs are also affected by the refractive index of the surrounding medium used for their synthesis and stabilisation. As the refractive index of the medium increases, it tends to shift the extinction spectrum to a higher wavelength (red shift) and vice versa. The peak of the spectrum becomes broadened, and due to the formation of larger particles, secondary peaks may appear. Mock et al. 24 also reported similar observations using oils of different refractive indices. The refractive index of the plant leaf extract was found to be 1.36, which is slightly higher than that of water. In colloidal solution, a layer of plant extract with active functional molecules is formed around the AgNPs, so the observed peak is slightly shifted towards the red wavelength of light (Figure 1). This may be one of the prominent reasons why there is an apparent red shift in the spectral behaviour with Tulsi extract. This effect appears more prominent in the samples treated with a wavelength in the range of 520–740 nm.
Particle size and zeta (ζ) potential
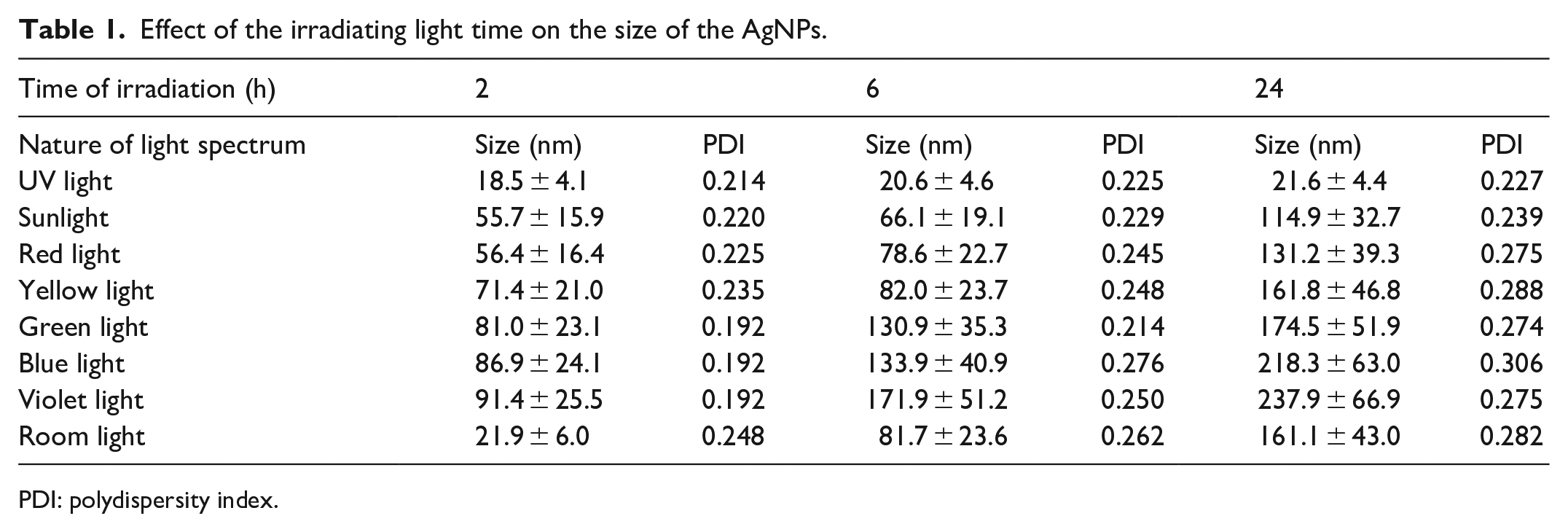
The particle size analysis of the AgNPs was carried out by the dynamic light scattering (DLS) technique. The particle size distribution curves (Figure 3) show that the size of the particles formed due to the photo-reaction varies between 50 and 240 nm. The average size of the AgNPs formed at different wavelengths of visible light spectrum is listed in Table 1. The particle size distribution for AgNPs varies with the wavelength of irradiating light. As the size of the AgNPs lies in the wavelength range of

Effect of the light wavelength on the particle size distribution: (a) diffused light, (b) sunlight, (c) red light, (d) yellow light, (e) green light, (f) blue light and (g) violet light.
Effect of the irradiating light time on the size of the AgNPs.
PDI: polydispersity index.
The zeta potential calculated using the Smoluchowski equation based on the electrophoretic mobility,
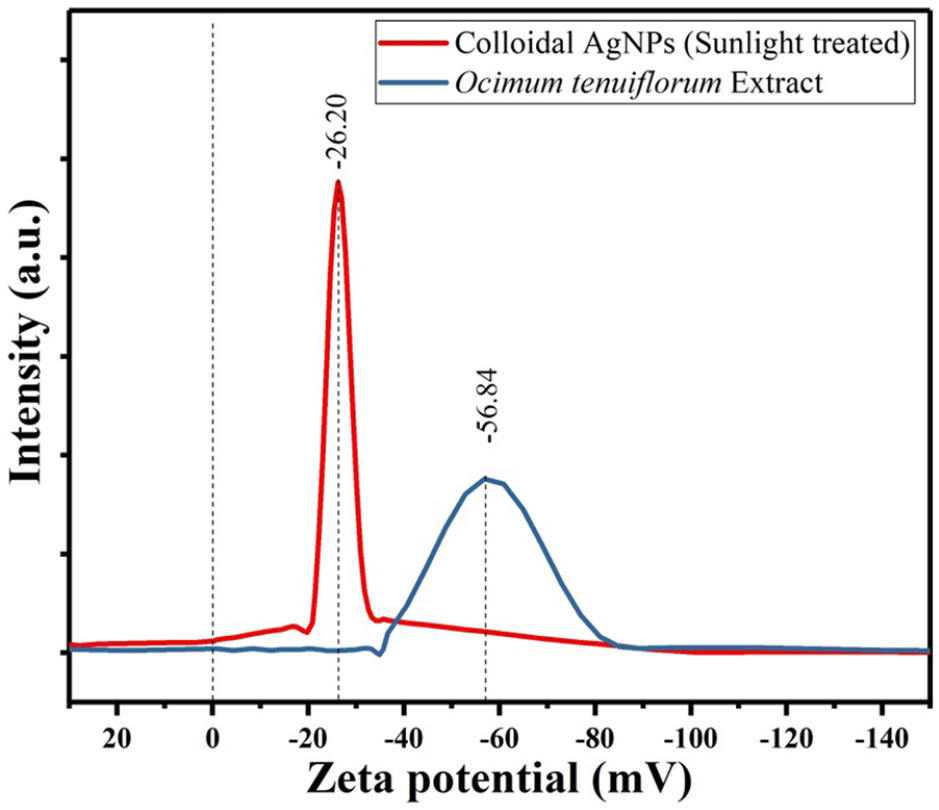
Zeta potentials of
Effect of light wavelength on AgNPs
The formation of AgNPs takes place through a photo-catalysed reaction. The results clearly show that the wavelength of the incident light not only governs the rate of biosynthesis of AgNPs but also affects their size distribution (Figure 3; Table 1). The size of the AgNPs was found to be inversely proportional to the wavelength of visible light, that is., red light led to the formation of the smallest AgNPs and violet light the largest (Table 1). It was also observed that exposure to sunlight resulted in the formation of the smallest nanoparticles. Direct sunlight contains a wide range of incident radiations (200–900 nm). Possibly, this may be the reason that sunlight readily catalyses the biosynthesis process. On the contrary, in the case of diffused room light which generally contains less intense visible light and near-infrared radiation, the process gets delayed, and no AgNPs were seen to be form until 2 h had passed. In the presence of UV light, negligible biosynthesis of AgNPs took place as analysed spectrophotometrically, and no peak was observed at 427 nm for the UV-treated colloidal solution (Figure 1). The smaller particle size shown in the range of 18–22 nm may be due to the macromolecules present in the plant extract (Table 1). So it can be inferred that visible light catalyses the formation of AgNPs.
AgNPs are well known for their strong interaction with a visible portion of the light spectrum due to surface plasmon resonance. 25 The conduction electrons on the metal surface oscillate collectively under the influence of light of a specific wavelength. The absorption and scattering properties of AgNPs depend upon various factors like particle size, shape and surrounding medium refractive index. Tuning these properties could give AgNPs of the desired shape and size depending upon the application. 28 Mono-dispersity of size and uniformity of shape is the reason behind the sharpness of the peak. 29 The low value of PDI (Table 1) indicates reasonably high mono-dispersity (Figure 3). The observed PDI values of the samples are in the range required for analysis as per the ISO 22412:2017.
The peaks of the formed AgNPs when viewed from the nano-confinement point of view, which states that when the particle size is in the comparable range of the wavelength of electrons, then due to the quantum confinement effect, transition from continuous to discrete energy levels takes place increasing the bandgap. 30 This increase ultimately results in a shift of absorbance to the lower wavelength region of higher energy or a blue shift. 31 In this study, a red shift has been observed. The findings of this study are contrary to nano-confinement, which is evident from the UV-visible absorbance curve (Figure 1). The possible reason for the deviation may be due to the size of the formed nanoparticles, which are greater than 50 nm and the nano-confinement effect applies only to smaller particles in the range of wavelength of electrons. 32
Antimicrobial activity of AgNPs
The zones of inhibition shown in Table 2 indicate the antimicrobial properties of the formed AgNPs against gram-positive and gram-negative pathogenic bacteria. Based on the results obtained (Table 2), it can be inferred that the UV treatment does not catalyse the AgNP biosynthesis; hence, mere plant extract and silver nitrate solution showed limited antibacterial activity. The maximum zone of inhibition occurs with the sunlight-treated mixture as the formed AgNPs are the smallest in this case (see Table 1). The antibacterial activity or zone of inhibition decreases as the size of the AgNPs increases with a decreasing wavelength of irradiating light, as shown in Table 1. The high antimicrobial activity of smaller AgNPs is in agreement with the findings of Kumari et al. 11 and others. Some antimicrobial activity has been observed in the case of AgNO3 due to its inherent nature. Silver nitrate solution has been previously reported for antimicrobial activity against a wide range of bacterial species. 33 Tulsi plant aqueous extract also showed a little antimicrobial activity as reported by Hanna et al. 21 The zone of inhibition of Tulsi extract seems least probably due to dilution.
Antibacterial activity of AgNPs against gram-positive and gram-negative bacteria based on their size.

Size after 2 h of specific light exposure.
Solution casting and diffusion of active constituents
A clear film of poly(vinyl alcohol) (PVA) containing AgNPs and Tulsi extract formed after 24 h over the Petri plate kept in an incubator at 45°C (Figure 5(a)). The average thickness of the formed film was found to be 0.15 ± 0.03 mm. The film obtained has good strength. This film, when tested for antimicrobial properties using the overnight grown bacterial culture, exhibited no growth over the film, and a zone of inhibition all around the film was also observed indicating its antibacterial activity. The peripheral zone of inhibition is small due to the slow diffusion of active constituents from the film towards the solid medium.
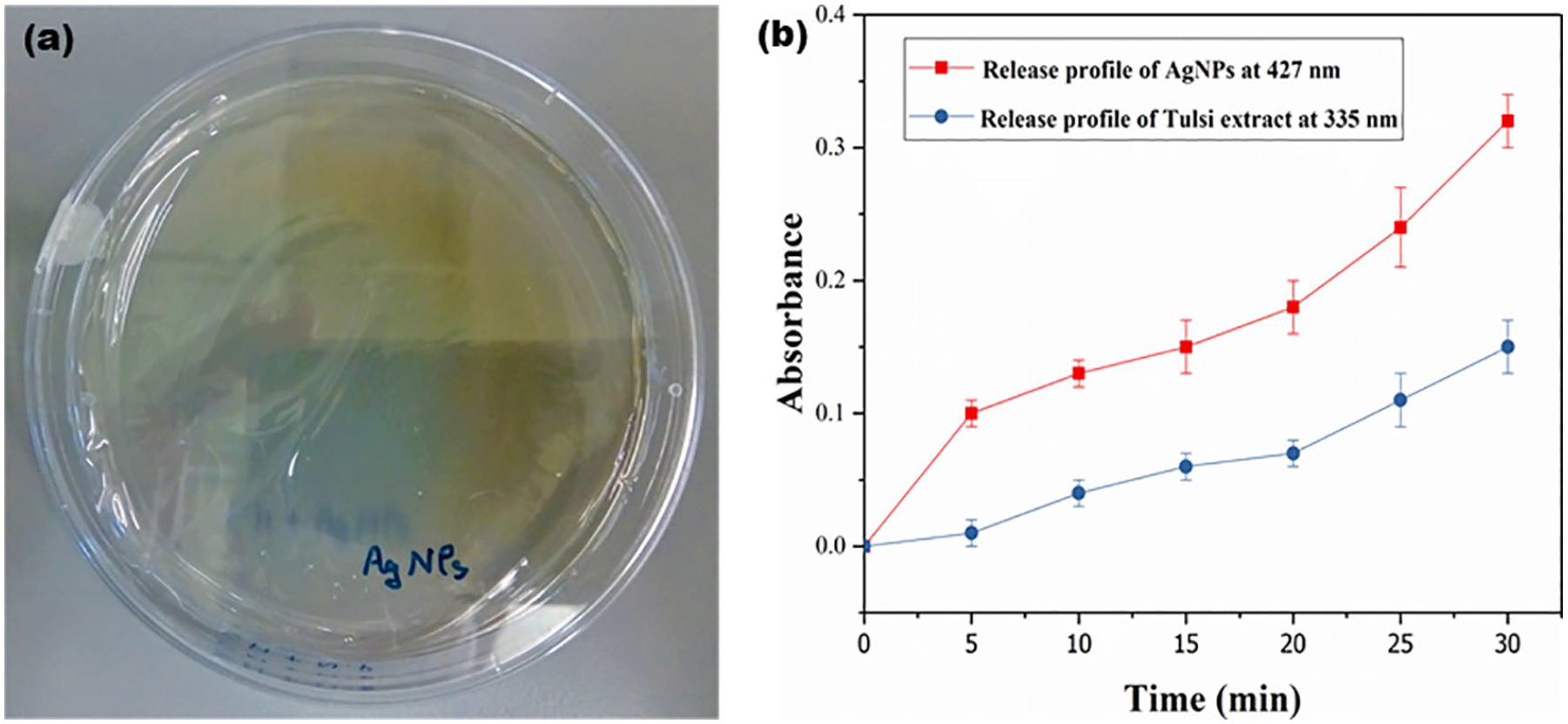
(a) Morphology of the antimicrobial active film and (b) release profile of AgNPs and
The diffusion of AgNPs and plant extract was monitored after 5, 10, 15, 20, 25 and 30 min by measuring the spectrophotometric absorbance values at 427 and 335 nm, respectively. The corresponding increase in absorbance value with time represents the diffusion of AgNPs and plant extract in the surrounding microenvironment (Figure 5(b)). Thus, the prepared film can be considered as an active antimicrobial film, which releases the active constituents, AgNPs and Tulsi extract, simultaneously. The diffusion rate of the active constituents is not constant. Initially, it is slow, but later on increases because the PVA film begins to swell and becomes porous to release the constituents actively.
Conclusion
The biosynthesis of AgNPs has been carried out using
Experiment
Chemicals
Silver nitrate (AR Grade) and PVA (MW 85,000–124,000) were obtained from Sigma-Aldrich, Gillingham, UK. Luria Bertani (LB) agar was procured from Himedia Laboratories Pvt. Ltd., Mumbai, India. The borosilicate glassware used in the experiments was acid-washed, rinsed with double-distilled water (DDW) followed by heat sterilisation before use. The DDW prepared in the laboratory was used for preparing chemical solutions and for washing glassware.
Collection and processing for the preparation of O. tenuiflorum leaf extract
Fresh green leaves of
Synthesis of AgNPs
For the preparation of 10 mL of AgNP suspension, 1 mL of silver nitrate solution (1 mM) and 1 mL of the aqueous leaf extract were mixed with 8 mL of DDW at room temperature (28°C ± 2°C) in separate test tubes wrapped with cellophane films of the desired colour to obtain light of the desired wavelength range. The mixed solutions of each test tube were exposed to red, yellow, green, blue and violet light by exposing them to sunlight for 2–6 h. Suspensions kept in three unwrapped test tubes were placed in direct sunlight, diffused sunlight (room light) and UV light. The UV light source was a laboratory UV lamp (Philips TUV 15W/G15 TB).
Characterisation of AgNPs
UV–Visible spectrophotometry
The change in the spectral behaviour of the synthesised AgNPs was monitored in the wavelength range of 300–900 nm using a UV–Visible Spectrophotometer (Model 2202; Systronics, Ahmedabad, India). The spectrum was observed for each sample in the selected wavelength range after 2 and 6 h of exposure to light.
SEM and EDX
After 2 h of exposure to the light, the unbound moieties from the AgNPs and the colloidal nanoparticle solution were centrifuged at 15,000 r/min for 15 min, and the supernatant was discarded. The pellets of AgNPs thus obtained were re-dispersed in DDW and centrifuged three times. The supernatant in each case was discarded, and the washed pellets were dried in a hot air oven at 80°C for 10 h. The dried AgNPs thus obtained were used for further characterisation.
The size and morphology of the AgNPs were analysed using a scanning electron microscope (Quanta 200F; FEI, Tokyo, Japan) at an acceleration voltage of 10 kV. Simultaneously, EDX analysis was also performed to measure the elemental composition and confirm the presence of AgNPs. The size of the AgNPs was evaluated with the help of ImageJ software.
Zeta (ζ) potential and particle size distribution
A Particle Size/Zeta Potential analyser (Particulate Systems Nanoplus-3; Micromeritics, Norcross, USA) was used to measure the zeta potential of the Tulsi plant extract and the synthesised AgNPs. The average particle size and size distribution of each sample were measured using the DLS method.
Antimicrobial activity of the AgNPs
The green-synthesised AgNPs were tested for their antimicrobial activity against gram-positive (
Preparation of a composite polymeric membrane
The solution-casting method was employed for casting the AgNPs bearing polymeric films. PVA (6% w/v) was dissolved in 25 mL of the prepared colloidal suspension of AgNPs containing the plant extract. The mixture was then heated at 80°C with continuous stirring until a clear solution was formed after the dissolution of PVA. The solution thus prepared was poured onto a Petri plate and kept at 45°C for 24 h to obtain a clear transparent film of uniform thickness. The thickness was measured using an electronic micrometre (Schnut Geometrical Metrology, Groningen, The Netherlands).
Diffusion of the active constituents
The diffusion of AgNPs from the polymeric film was spectrophotometrically analysed by placing a piece of film of known area (0.5 × 0.5 cm2) in a capped Eppendorf tube dipped in 1.5 mL of DDW for 5, 10, 15, 20, 25 and 30 min. After the desired period of incubation, the water sample was spectrophotometrically analysed at 427 nm for the release AgNPs and at 335 nm for the release of plant extract contents. All experiments were performed in triplicate.
Supplemental Material
8_Supplementary_R1 – Supplemental material for Light-induced synthesis of silver nanoparticles using Ocimum tenuiflorum extract: Characterisation and application
Supplemental material, 8_Supplementary_R1 for Light-induced synthesis of silver nanoparticles using Ocimum tenuiflorum extract: Characterisation and application by Vivek Kumar Pandey, Siddh Nath Upadhyay and Pradeep Kumar Mishra in Journal of Chemical Research
Footnotes
Acknowledgements
The authors gratefully acknowledge the MHRD for granting a research fellowship to V.K.P., the Incharge, Sophisticated Instruments Laboratory, IIT (BHU) Varanasi, for SEM and EDX analyses and the Head, Department of Chemical Engineering and Technology, IIT (BHU) Varanasi, for providing necessary laboratory facilities for conducting this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
