Abstract
Antimicrobial resistance is on the rise, constituting a public health problem which accounts for millions of global deaths annually. Thus, there is an urgent need for new antimicrobial compounds that can target virulence factors of pathogens. Supramolecules and macromolecules are attractive antimicrobial candidates since they exhibit antibiotic effects sustained for longer periods. This study reports the synthesis of a phthalimide-triazole-based p-tert-butylcalix[4]arene compound (
Graphical abstract

Introduction
Macromolecular chemistry studies fascinating chemical architectures from natural and synthetic origin composed of building blocks (monomers) that are linked together by covalent interactions into complex molecules with a high molecular weight. Antibacterial materials based on macrocycles and macromolecules are effective against drug-resistant fungi and bacteria with sustained antimicrobial effects.1,2 Calix[n]arenes consist of a class of macromolecular cyclic compounds having multiple numbers of phenolic units bonded together through methylene bridges at ortho positions with the most prevalent systems possessing four to eight phenolic units.3,4 Aromatic rings in calixarenes are arranged in three-dimensional form with an ancient calix crater vase shape, to which it owes its name. 5 The calixarene structure is made up of a narrow lower rim and a wider upper rim, while the middle conic section is called the annulus. 6 Depending on the amount of aromatic units, the cavities of calixarenes vary in their molecular dimensions. The calix[4]arenes, for instance, can adopt the 1,3-alternate (partial cone) or 1,2-alternate (cone) conformers which are stable and useful for studying their structural aspects. 7 Derivatives with varying recognition, selectivity, solubility, and hydrophobicity can be created by functionalizing the calixarenes on the upper and/or lower rim using functional groups of different kinds, including imines, amides, sulfur, semicarbazone, azo, and alkyl groups.7,8
Calixarenes are among the most extensively researched supramolecules, as they provide infinite structures through modification at the lower and/or upper rim and application options because of their ease of modification, which enables the establishment of sizable molecular libraries as a material basis for a variety of biomedical applications.9–12 Upper and/or lower rims can be modified or functionalized, and small ions and neutral compounds can be hosted within the cavity or annulus of calix[n]arenes to confer biological activities.13,14 Various derivatives of calix[4]arene continue to attract special attention from researchers due to their unique structural features, synthetic accessibility, and ease of chemical modification for different applications.15,16 Although they have upper and lower rims, functionalization with groups including alcohols, amines, amides, aldehydes, alkyl, amino acids, and ester derivatives opens the door to their potential application as prodrugs or as biochemical process inhibitors, with the release of active substances usually triggered by external stimuli.17–19 Specifically, the calix[4]arene gives researchers the ability to precisely tune the shape of their structures and shows numerous possibilities for regioselective poly-functionalization on both upper and lower rims. 20
Numerous biological activities including antiviral, antifungal, and antibacterial properties have been reported for various calix[4]arene derivatives. 20 – 22 Since they have versatile molecular structures compared to conventional antibacterial agents, calixarene derivatives offer tremendous potential as antibacterial agents and may be able to prevent cross-resistance with them. 23 Resistance is a situation whereby pathogenic microorganisms become non-susceptible to the effects of antibiotics previously used to treat their infections, and it creates a serious health issue that needs urgent and immediate intervention.24,25 Antibiotic resistance occurs through different mechanisms and constitutes a global health problem since it is detected for almost all antibiotics presently used in conventional medicine and only small amounts of new antimicrobial therapies are getting into the market. 26 Resistance is often aided by microbial virulence factors most especially biofilms, a QS (quorum-sensing)-dependent phenomenon in bacteria. QS consists of a bacterial cell-to-cell communication system, while biofilms are self-produced protective polymeric matrix that keeps sessile communities safe from antibiotics, host immune system, starvation, and drought. 27 About eighty percent of chronic and recurring infectious diseases in humans involve microbial biofilms, and the microbial cells that reside within these biofilms may be more resistant to conventional antibiotics about 10–1000 times more than the same bacterial species living within planktonic colonies.28,29 Antimicrobial compounds of natural and synthetic origin which are able to exert biocidal effects on pathogens through mechanisms such as QS and biofilm disruption are necessary since they address the evolution and development of resistance in microbial pathogens.30–32
Various calix[4]arenes and their derivatives are known to be suitable scaffolds for synthesis of antimicrobial agents. 33 Calix[4]arene derivatives are potential therapeutic agents and particularly, azo-based calix[4]arene derivatives are under-exploited, meanwhile they may be prospective antiviral and antibacterial agents. 34 Because calixarene synthesis is inherently challenging, particularly with regard to purification process, there are still few calixarenic platforms available despite the field’s ongoing attention. 35
In this study, phthalimide-based p-tert-butylcalix[4]arene derivative and its analogue are synthesized and their antimicrobial, biofilm, and QS inhibitory effects are investigated. Computational predictions were applied to investigate structural features of the compounds as well as molecular docking to understand their structure–activity relationships.
Experimental
Chemicals and reagents
Chemical reagents from Sigma-Aldrich including anhydrous sodium hydroxide (NaOH, ⩾99.95%), formaldehyde (37 wt. % in H2O), potassium carbonate (K2CO3, ⩾99%), sodium chloride (NaCl, ⩾99%), cupper(II)sulfate pentahydrate (CuSO4.5H2O, ⩾99.99%), and anhydrous magnesium sulfate (MgSO4, ⩾99.5%) were obtained. Solvents without further purification, such as dichloromethane (DCM, ⩾99.8%, Merck), N-N-dimethylformamide (DMF, ⩾99.8%, Merck), hexane (⩾99.8%, Merck), ethyl acetate (EtOAc, ⩾99.5%, Merck), diphenyl ether (⩾99%, Sigma-Aldrich), acetone (⩾99.8%, Merck), toluene (⩾99.8%, Merck), butanol (⩾99.8%, Merck), anhydrous methanol (MeOH, ⩾99.8%, Sigma-Aldrich) of analytical grade together with propargyl bromide (80 wt.% solution in toluene, Thermo Scientific Chemicals) and p-tert-butyl phenol (99%, Sigma-Aldrich) were used. TLC (thin-layer chromatography) cards (Merck) and silica gel (Merck) were used for chromatography. Luria-Bertani Broth, Tryptic Soy Broth, Nutrient Broth, Mueller Hinton Broth, Sabouraud Dextrose Broth, and corresponding agars were procured from Merck. For antibiofilm assays, crystal violet (Merck), ethanol (Merck), D-(+)-glucose (Merck), and glacial acetic acid (Merck) were used. N-hexanoyl-DL-homoserine lactone (C6-HSL, ⩾97%, Sigma-Aldrich), NaCl (Sigma-Aldrich), tryptone (Sigma-Aldrich), D-(+)-glucose (⩾99.5%, Sigma-Aldrich), and kanamycin sulfate (Sigma-Aldrich) were used for anti-QS and antimotility assays.
Synthesis of compound 2
Compound
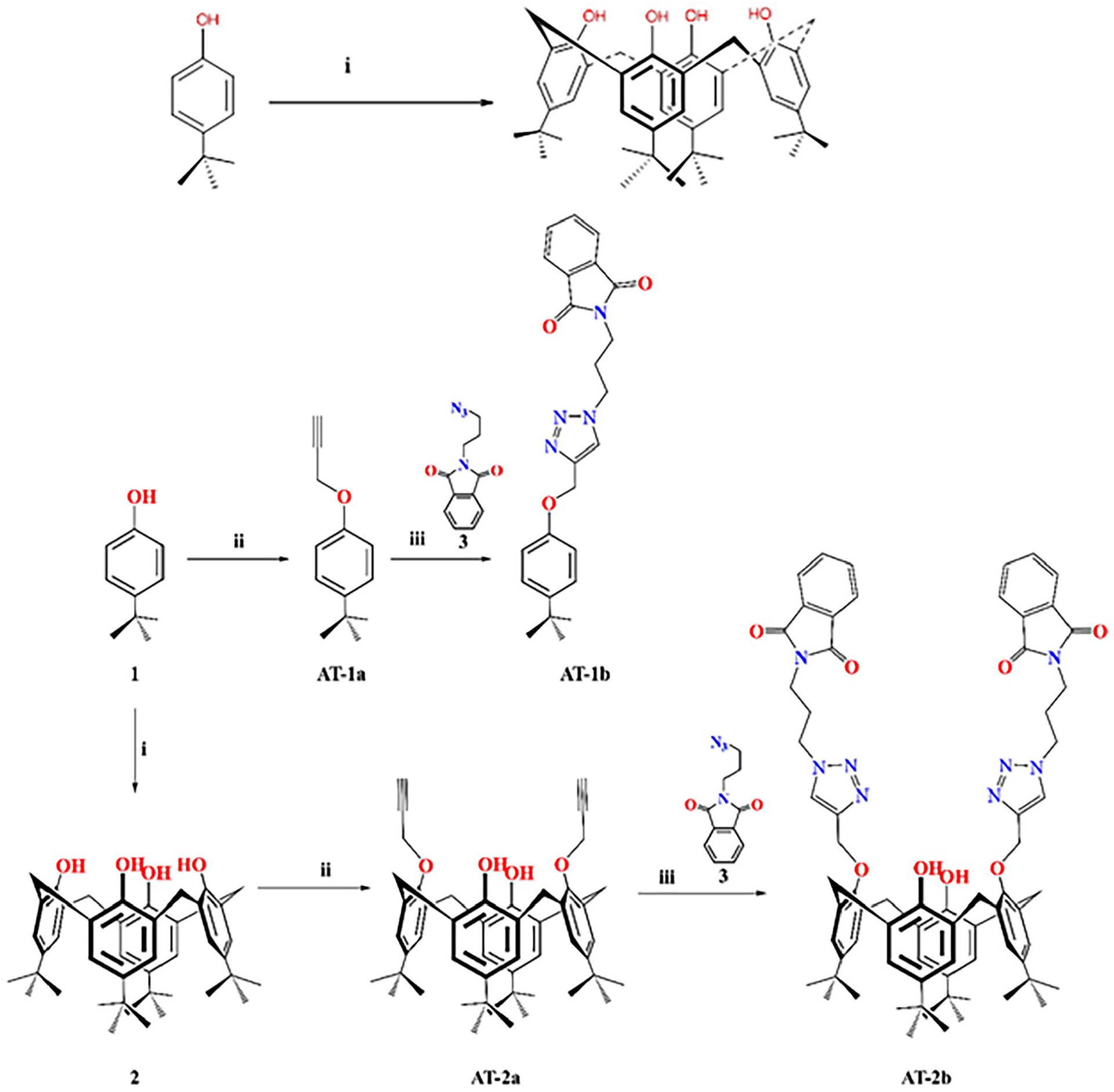
Synthesis of phthalimide-triazole based p-tert-butylcalix[4]arene and its p-tert-phenol derivative.
Synthesis of AT-1a and AT-2a
Propargyl bromide and p-tert-butylcalix[4]arene (
Synthesis of AT-1b and AT-2b
Azidopropyl phthalimide
Strains of microbes
Staphylococcus aureus ATCC 25923, Candida albicans ATCC 10239, Escherichia coli ATCC 25922, Chromo-bacterium violaceum CV026, C. violaceum CV12472, and Pseudomonas aeruginosa PA01 were used in the different microbial studies.
Measurement of antimicrobial activity
A microtiter broth dilution method as described previously was used to measure the minimal inhibitory concentration (MIC). 40 MIC was the lowest concentration of compound at which no visible growth was observed. The Mueller–Hinton broth and a bacteria concentration of 5 × 105 colony-forming units (CFU)/mL were used. Into wells of 96-well microtiter plates, 100 μL of fresh overnight microbial suspensions were inoculated with or without compounds at serial final concentrations (5, 2.5, 1.25, 0.625, 0.3125, 0.15625, 0.078125, 0.0390625, 0.01953, 0.009765 mg/mL) and incubated for 24 hours at 37 °C after which MIC values were measured.
Biofilm inhibition assay
Biofilm-forming inhibition of the compounds at sub-MIC and MIC concentrations was evaluated using the microplate antibiofilm assay. 41 Summarily, 1% of overnight cultures of bacteria were seeded in 200 μL of Tryptose-Soy Broth (TSB) supplemented with 0.25% glucose and inoculated in the presence or absence of the compounds at 37 °C for 48 h. The wells were rinsed with distilled water to remove planktonic cells, and biofilm cells were stained with crystal violet (0.1% solution) at room temperature for 10 min. Unabsorbed crystal violet solution was removed, and 200 μL ethanol (for Gram-negative and Candida) or 33% glacial acetic acid (for Gram-positive) was added to the wells. In total, 125 μL were pipetted from each well after shaking into sterile tubes, and the volume was adjusted with distilled water to 1 mL. A Thermo Scientific Multiskan FC spectrophotometer at 550 nm was used to read optical density (OD) which was used in calculating biofilm inhibition percentages using the.
Quorum-sensing inhibition assay against C. violaceum CV026
Quorum-sensing inhibition (QSI) was determined as explained previously 42 with little modifications. 5 mm of lukewarm molten Soft Top Agar (2.0 g tryptone, 200 mL deionized H2O, 1.3 g agar, 1.0 g NaCl) was mixed with an overnight culture of CV026 (100 µL) in LB (Luria-Bertani) broth and treated with 20 µL of external acylhomoserine lactone (AHL), that is, C6HSL (100 µg/mL) and kanamycin solution (10 µL). The mixture was immediately poured and spread as an overlay on the surface of solidified LB agar plates. In total, 5 mm diameter wells were carved after solidification and filled with 50 µL MIC and sub-MIC concentrations of compounds and incubated at 30 °C for 3 days. Cream or white halo zones around each well over the purple lawn of growing CV026 bacteria indicated areas of QS inhibition, and their diameters were measured in mm. Each assay was repeated three times.
Violacein inhibition assay against C. violaceum CV12472
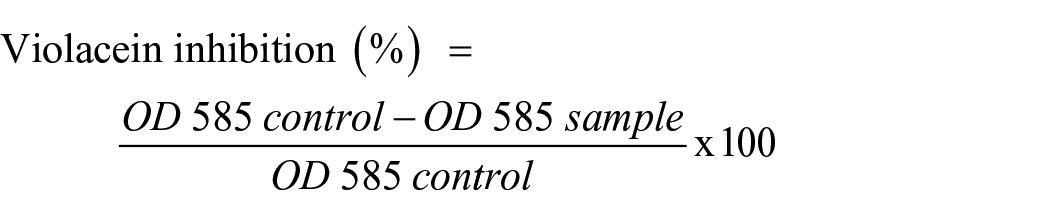
Qualitative analyses of violacein inhibition by the compounds were measured against C. violaceum ATCC 12472. 43 In total, 180 µL of Luria–Bertani broth and 10 µL of overnight culture (0.4 OD at 600 nm) of C. violaceum were filled into each well of sterilized 96-well microtiter plates. In total, 10 µL of MIC and sub-MIC amounts of compounds were added in order of decreasing concentration, and well without compounds served as controls. Plates were incubated for 24 h at 30 °C and monitored for the prevention of violacein production. The absorbance of each well was measured at 585 nm, and the percentage inhibition of violacein production is calculated as shown as follows.
Swimming and swarming inhibitions against P. aeruginosa PA01
Swarming inhibition was performed as described elsewhere with slight modifications. 44 Swarming plates were prepared by mixing 0.5% agar, 0.5% of D-glucose, 1% peptone, and 0.5% NaCl in distilled water together with test compounds (MIC, ½ MIC, ¼ MIC) and allowing them to solidify. Overnight cultures of P. aeruginosa PA01 were point inoculated at the center of each plate. Plates without compounds served as controls. The plates were incubated for 18 h in an upright position, after which the swarm migration zone diameters were measured and used to calculate percentage inhibition in relation to the control plates. Swimming agar consisted of 1.5% agar, 0.5% D-glucose, 1% peptone, and 0.5% NaCl, and the process was similar as for swarming inhibition assay. Saran papers were used in wrapping the plates to avoid dehydration. The plates were incubated in an upright position for 18 h at 37 °C. The percentage reduction in swimming movement by compounds was calculated.
Density functional theory calculations
The frequency calculations and geometry optimization of the ground state geometries of
Molecular docking details
The predicted binding affinities of
Statistical analyses
Analysis of variance (ANOVA) was used in determining the differences in activities among the compounds. Experiments were done in triplicates, and the values represent mean ± standard error of the mean of three measurements. The data were analyzed using the Microsoft Excel, and differences were considered to be statistically significant at p < 0.05.
Results and discussion
Inhibition of violacein production, QS, and motilities
The results of violacein inhibition against C. violaceum CV12472 are presented in Figure 2. MIC was 0.156 mg/mL for both

Violacein inhibition against C. violaceum CV12472 by compounds.
The results of QS inhibition against C. violaceum CV026 are presented in Figure 3. MICs against C. violaceum CV12472 were 0.312 mg/mL for both

Quorum-sensing inhibition against C. violaceum CV026 by compounds.
Inhibition of swimming and swarming motilities against flagellated P. aeruginosa PA01 was evaluated at 100, 75, and 50 µg/mL and presented in Figure 4. In the swarming motility assay, at the highest test concentration (100 µg/mL), inhibitions were 47.1 ± 0.55% (

Motility inhibition against P. aeruginosa PA01.
The compounds were investigated for their ability to disrupt virulence factors including violacein production, motilities, biofilm, and QS against the microorganisms. Antivirulence compounds are a class of drugs that can prevent pathogens from attacking the host by targeting virulence factors instead of killing or stopping their growth.
61
Antivirulence therapy is a suitable strategy for weakening pathogens and blocking their pathogenicity, thereby reducing the administration of broad-spectrum antimicrobials and the prevalence of resistant strains.
62
The goal of any given antimicrobial technique is to control virulence factors and microbial survival, and these chemicals accomplish this via disrupting signaling pathways known as QS. Violacein produced by C. violaceum is an easily measurable means for evaluating QS. The compounds inhibited the production of C. violaceum CV12472, which produces this pigment in a QS-mediated process while growing normally.
Antimicrobial and antibiofilm activities
The antimicrobial activity of the compounds is presented in Figure 5. MIC was determined for fungus (C. albicans), Gram-negative (E. coli), and Gram-positive (S. aureus) bacteria. MIC values against S. aureus (Gram-positive) were 39.06 µg/mL (

Antimicrobial activity of the compounds.
The percentage inhibition of biofilm formation for each bacterium was determined at sub-MIC and MIC, and the results are presented in Figure 6. Biofilm inhibition against S. aureus, varied from 45.15 ± 0.55% (MIC) and 4.22 ± 0.06% (MIC/4) for

Biofilm inhibition activity of the compounds.
The results suggest that C. albicans-established biofilms are less susceptible to the compounds than S. aureus-established biofilms, although planktonic C. albicans is more susceptible than planktonic S. aureus. This may depend on the biofilm architecture of both pathogens. The rising antibiotic resistance to available antibiotics requires the synthesis and evaluation of the effects of new antimicrobial.69,70 The compounds showed antimicrobial activity against C. albicans, S. aureus, P. aeruginosa, E. coli, and C. violaceum. The compounds showed biofilm inhibition effects against C. albicans, S. aureus, and E. coli. As shown in Figure 6, S. aureus biofilm was most susceptible to the compounds, while E. coli biofilms were least susceptible. Compound
DFT calculation results
The optimized geometries and electrostatic potentials (ESPs) of

Optimized geometries (left) and ESPs (right) of
Tables 1 and 2 summarize the predicted and experimental 13C and 1H NMR chemical shifts of
Experimental and predicted 13C-NMR chemical shifts (ppm) in DMSO-d6 of
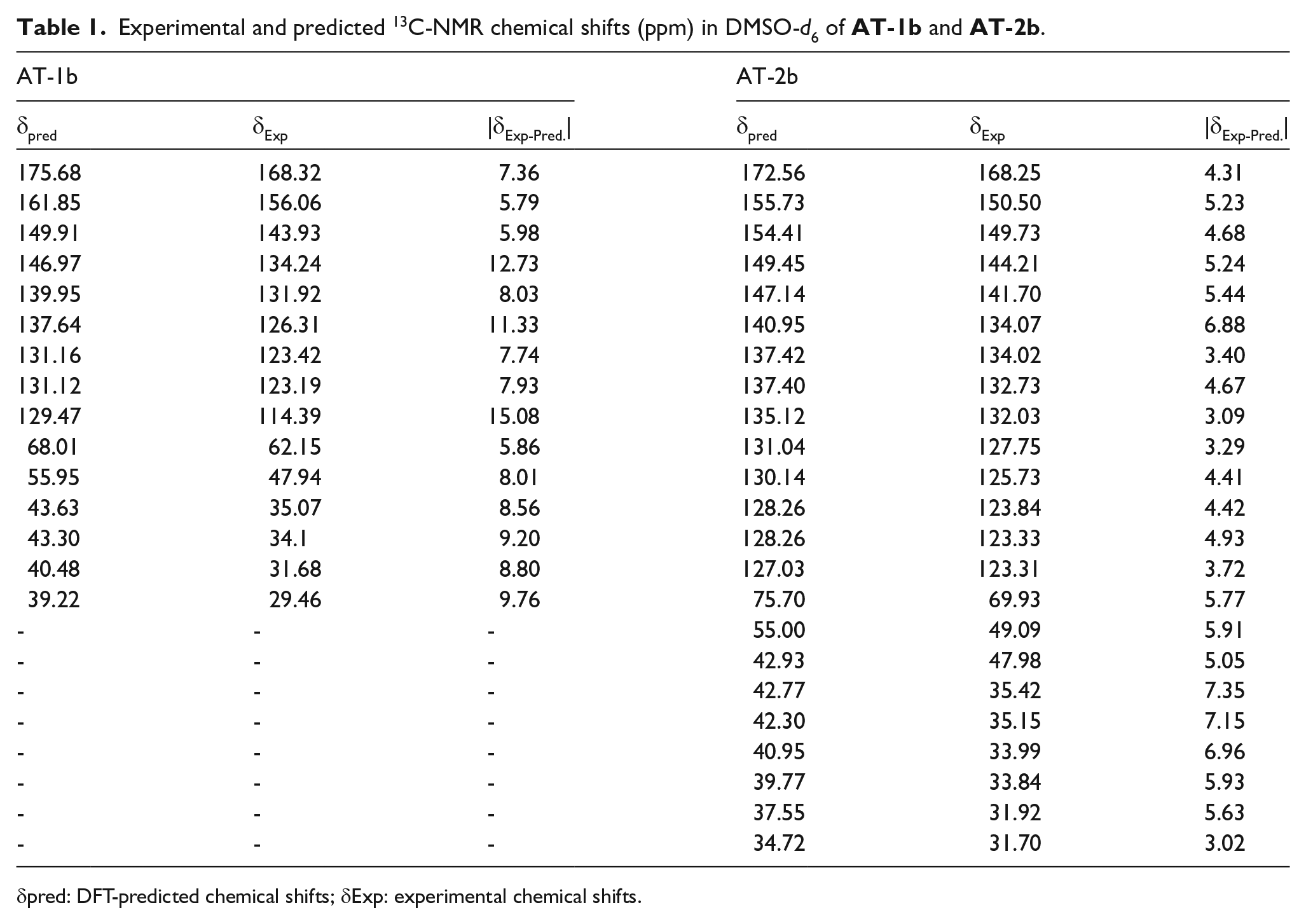
δpred: DFT-predicted chemical shifts; δExp: experimental chemical shifts.
Experimental and predicted 1H-NMR chemical shifts (ppm) in DMSO-d6 of
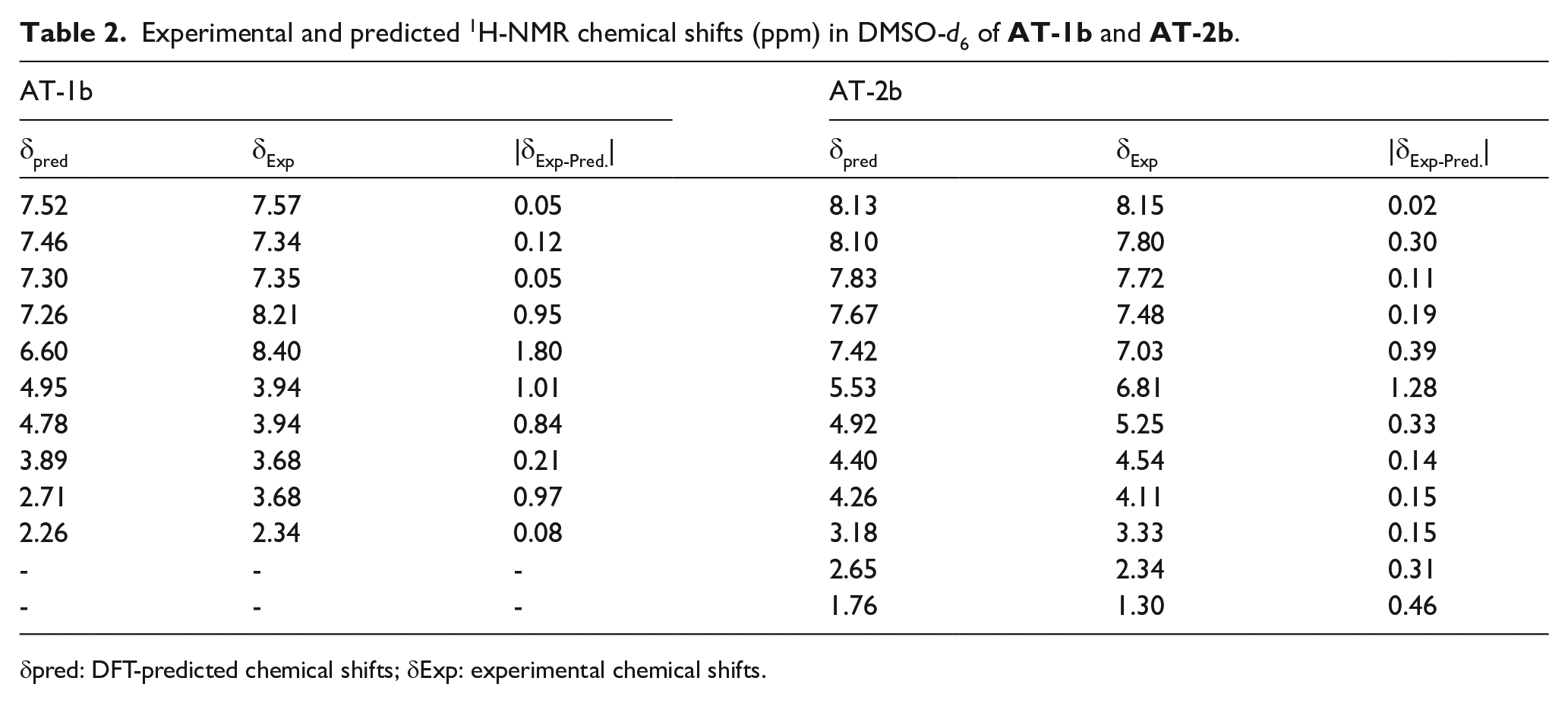
δpred: DFT-predicted chemical shifts; δExp: experimental chemical shifts.
The phthalimide p-tert-butylcalix[4]arene derivative (
Molecular docking results
The antimicrobial activity of
Figures 8 and 9 show the binding modes of
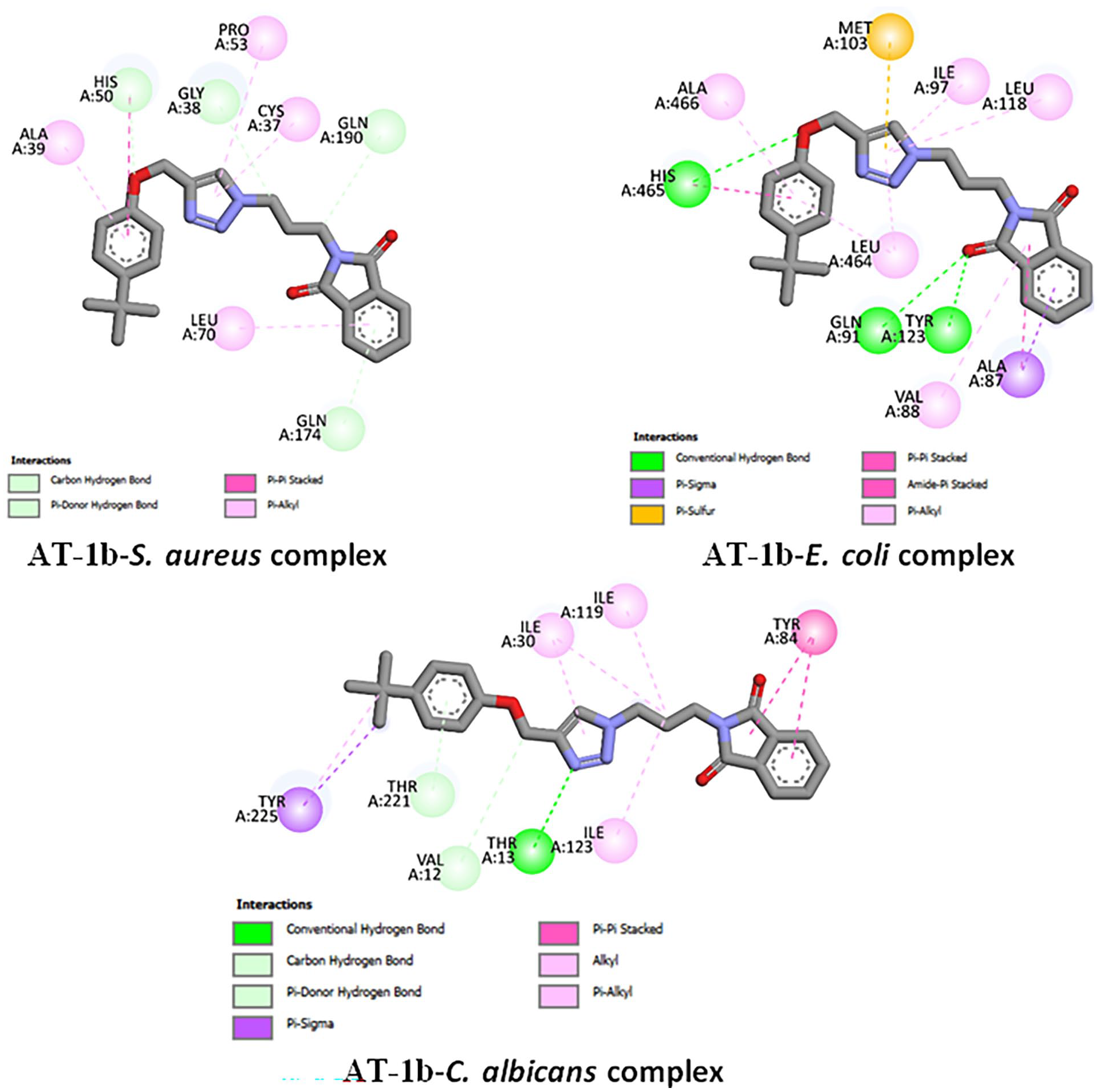
2D binding of
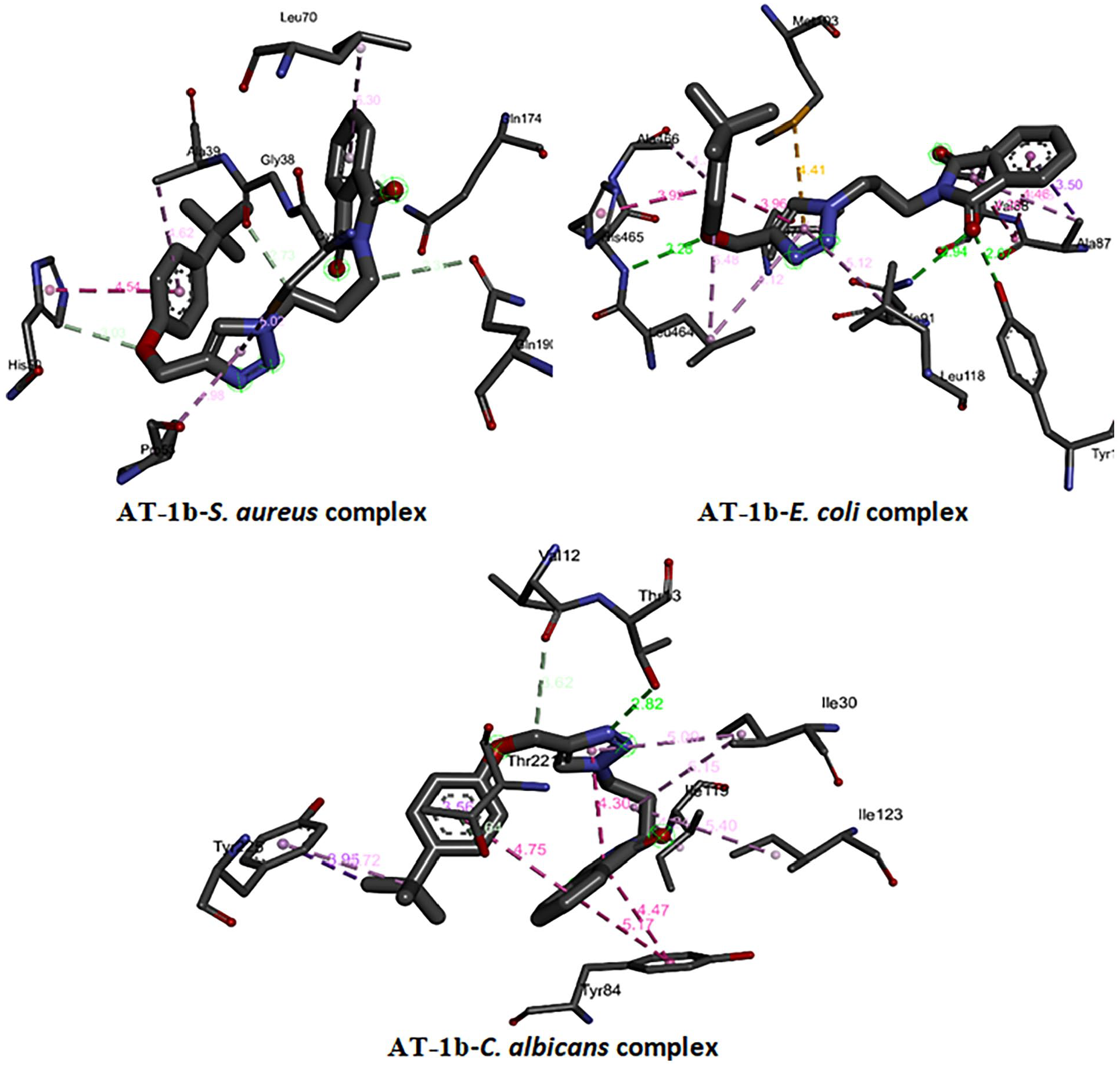
3D binding of
Free binding energies, hydrogen bonding, and number of closest residues to the docked

Molecular docking was used to predict the interactions between the synthesized compounds and receptor proteins of the S. aureus, E. coli, and C. albicans to identify the most effective method of bonding to create stable complexes. Docking studies substantiated antimicrobial studies, as the compounds had negative free binding energies, an appreciable number of H-bondings, as well as several interactions of the closest residues to the docked ligand in the bonding site. Due to its large size,
Conclusion
The present study focused on the synthesis of phthalimide-triazol-based p-tert-butyl calix[4]arene derivative and its p-tert-butylphenol analogue and the evaluation of their antimicrobial, antibiotilm, and QS inhibitions. DFT calculations were used to predict optimized geometries and MEPs as well as 1H NMR and 13C NMR chemical shifts. The compounds (
Supplemental Material
sj-docx-1-chl-10.1177_17475198251368414 – Supplemental material for Synthesis, DFT calculations, and molecular docking of phthalimide-triazole based p-tert-butylcalix[4]arene derivative and its analogue with antimicrobial, antiquorum-sensing, and antibiofilm properties
Supplemental material, sj-docx-1-chl-10.1177_17475198251368414 for Synthesis, DFT calculations, and molecular docking of phthalimide-triazole based p-tert-butylcalix[4]arene derivative and its analogue with antimicrobial, antiquorum-sensing, and antibiofilm properties by Alfred Ngenge Tamfu, Selahattin Bozkurt, Ozgur Ceylan and El Hassane Anouar in Journal of Chemical Research
Footnotes
Acknowledgements
The Scientific and Technological Research Institution of Turkey (TUBITAK) is greatly acknowledged. The authors are grateful to the participating institutions, which are Usak University, Mugla Sitki Kocman University, Prince Sattam bin Abdulaziz University, and the University of Ngaoundere.
Ethical considerations
Ethical approval is not applicable for the article.
Author contributions
A.N.T.: Conceptualization; Methodology; Investigation; Writing – original draft; Writing – review and editing; Formal analysis; Data curation; Software; Funding acquisition; Resources. S.B.: Conceptualization; Methodology; Investigation; Writing – original draft; Formal analysis; Funding acquisition; Resources; Project administration. O.C.: Methodology; Writing – review & editing; Visualization; Resources. E.H.A.: Methodology; Writing – review & editing; Formal analysis; Data curation; Software; Resources.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Scientific and Technological Research Council of Turkey (TUBITAK) under the project 1059B212200092.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data generated in this study are available within the article and its supplementary data files or upon request from the corresponding author.
Statement of informed consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.