Abstract
Lithium-ion batteries have transformed our lives and are now found in everything from mobile phones to laptop computers and electric cars. In lithium-ion batteries, an adequate electrolyte was developed using a winding process nearly related to the progress of electrode chemistries. In this technology, a metal oxide is a cathode, and porous carbon is the anode. The electrochemical interaction of anode material with lithium could produce an intercalation product, which could form the basis of a revolutionary battery system. Structural retention causes this reaction to proceed quickly and with a high degree of reversibility at room temperature. Titanium disulfide is one of the latest solid cathode materials. In this review, the history of intercalation electrodes, electrolytes, and basic principles related to batteries based on intercalation processes and their effect on battery performance is reported.

Introduction
Energy is a key factor in the growth of any society. It is also required for industrial applications. Consequently, the key challenge is to expand the efficient production, storage, and distribution of this energy. 1 Lithium-ion batteries (LIBs) have dominated the market for advanced energy sources in the modern age since they are widely used in various areas, including electronics, electric cars, and energy storage systems. 2 Traditional energy storage systems, such as batteries, have constraints such as sluggish charging and a limited lifespan. Other storage methods, including mechanical, electrochemical, and electromagnetic, which store energy in different forms, are more costly and sophisticated. 3 Battery performance is influenced by many factors, including chemistry, cell design, battery geometry, working conditions (e.g. temperature and cycle current), and the manufacturing process. 4 Hendricks et al. 5 discovered that mechanical damage, chemical reduction, and corrosion can alter the external properties of a battery, resulting in the following failure modes: Thickening of the solid electrolyte interlayer (SEI), particle breakage, and reduction in electrode porosity.
In the 1960s, German scientist R. Schroeder and French scientist J. Rouxel pioneered the chemistry of reversible intercalation of Li+ into transition metal disulfides (MS2). Whittingham published significant work in Science in 1976, identifying TiS2 as an effective Li+ host for energy storage via Li+-intercalation. It was recently reported on the fundamental properties of the Li, TiS., series of intercalation compounds.6–11 Since alkali metals react strongly with water, an LIB would require an organic liquid Li+ electrolyte, which is flammable. 12 The creation of the LIB makes use of the high degree of Li+ and Co (III) order into alternate octahedral-site planes in lithium cobalt oxide (LCO; LiCoO2). 13 Many nations were investigating the reversible intercalation of Li+ into graphitic carbon at the same time. The reversible intercalation of Li+ into graphitic carbon to create lithiated graphite (LiC6) was demonstrated in this study. 14 The sale of the battery-powered mobile phone, which started the wireless revolution, was made possible by technological improvement. 15 The LIB is used in hybrid and electric road cars, but it has drawbacks that are motivating efforts to replace it. 16 Goodenough recently wrote a viewpoint paper titled “A perspective on Li-ion batteries” in Science China Chemical, 17 in which he reviewed the history, present issues, and interesting research paths of LIBs, the ubiquitous energy-storage devices in electronics.
Methodological part
The use of nanoscale powders as the primary active material for high-energy storage devices is not considered sufficient to enhance their performance due to their low bulk density. However, when the primary nanoparticles are combined to form a dense nanostructured secondary particle, such as the nickel-rich nickel manganese cobalt (LiNi0.6Mn0.2C0.2O2) “meatball” cathodes (see Figure 1), a dense electrode coating can be formed. In addition, the selection of a battery electrode with minimal capacity loss due to side reactions should require a reaction selectivity of at least 99.9% to be able to cycle more than a thousand times. 18

A LiNi0.6Mn0.2C0.2O2 particle with the shape of a meatball.
Majority of the crucial parts and difficulties may be nanoscale. All of the protective coatings that form on both the anode and the cathode, collectively known as the SEI, are examples. These are highly difficult to investigate and comprehend because they are living layers that vary constantly. A problem for all nanoscientists is to figure out how to make an artificial SEI layer that could be formed during the battery production process. This would eliminate the 1-week or longer training cycles currently required for all LIBs.
In addition, nanomaterials can play a critical role in improving the response and/or protection of the active material due to their unique behavior and properties. For example, graphene, with its extremely high aspect ratio, can outperform carbon black as a conducting agent, as proved by its usage to improve the electrochemistry of lithium vanadium phosphate (Li x VOPO4). 19 Nanomaterials will be important in the next generation of LIB anode materials. 20
It is also interesting to note how the first cathodes for lithium batteries, the layered transition metal dichalcogenides that emerged from a study of the effects of intercalating electron donors in, for example, tantalum (IV) sulfide (TaS2) on their superconductivity, are receiving renewed interest. The lightweight, semi-metallic, conductive TiS2 was used for the first LIB. 21
At the start, the evolution of electrolytes in LIBs was limited by cathode chemistry. Lithium’s low standard reduction potential of −3.05 V makes it particularly appealing for high-density and high-voltage battery cells. However, because of its strong reactivity, lithium must be kept away from both air and water to prevent harmful reactions. Whittingham solved the problem by employing non-aqueous electrolyte solutions carefully prepared and validated by other researchers in lithium electrochemistry work a few years earlier. Whittingham’s LIB has a larger cell potential than the lead-acid battery—2.5 V against 2 V.
In 1972, Whittingham 22 developed the first Li insertion cathode using TiS2, and ether-based electrolytes were employed to augment the potential voltage of TiS2, which was inferior to 3.0 V inside the stability potential range of ethers. Nevertheless, esters are not stable at potentials beyond 4.0 V. Furthermore, Whittingham utilized a TiS2 cathode with a lithium metal anode to build a high-energy battery in 1976. Figure 2 illustrates the LIB designed by Whittingham.
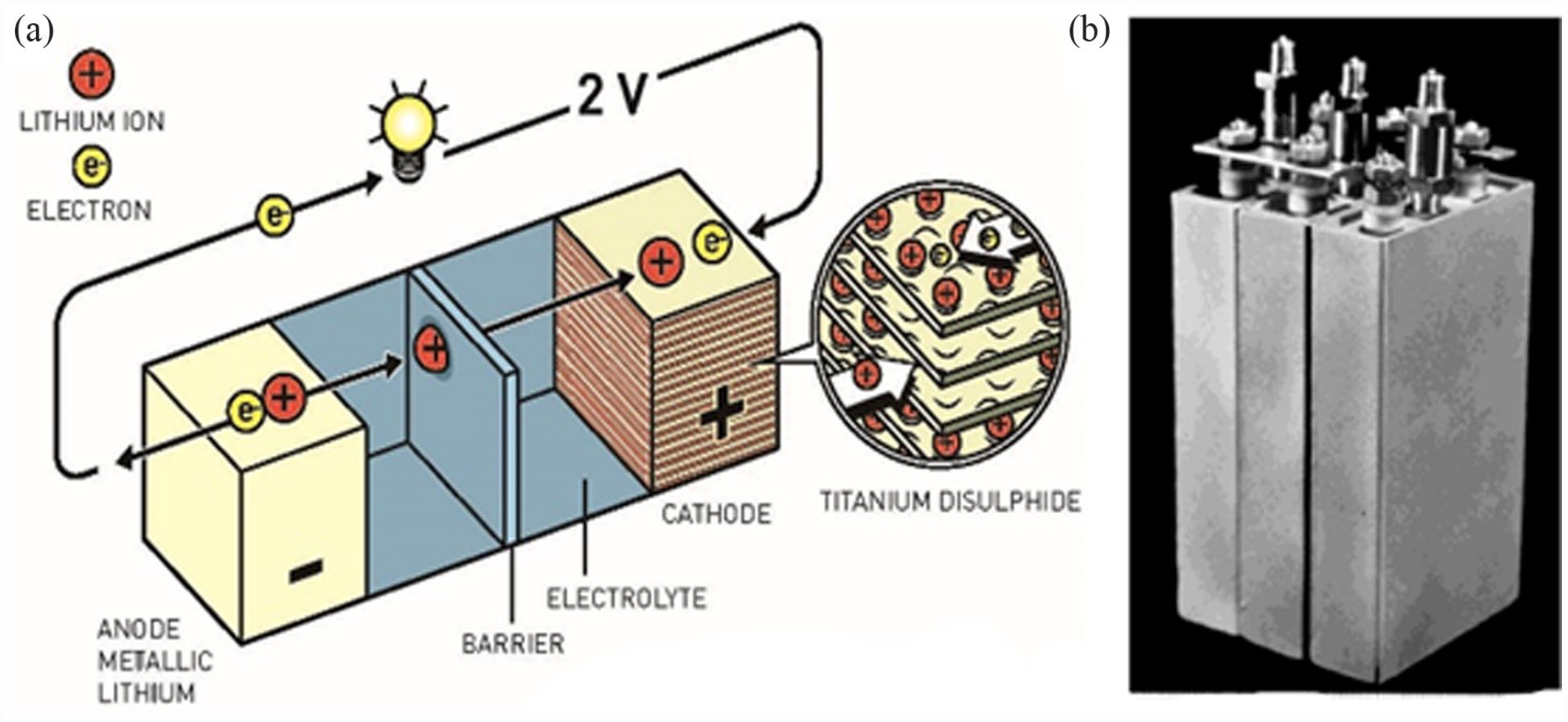
(a) The Whittingham battery scheme. The Royal Swedish Academy of Sciences provided the image. (b) A Whittingham battery pack on display at the 1977 Chicago Car Show in the United States. Chemical Reviews, The American Chemical Society.
Intercalation reactions include the integration of lithium into TiS2 layers [Li x TiS2 (0 ⩽ x ⩽ 1)], lithium into vanadium oxide (V4O10), and lithium into graphite layers, resulting in LiC6. Two instances of lithium intercalation into host lattices are shown in Figure 3.

Two intercalation compounds: (a) C6Li and (b) Li x V4O10. The Li ions are depicted in green. Li ions are shuttled between these two chemicals in an LIB.
This Li x TiS2 intercalation molecule is now the ideal example of intercalation because it has complete lithium solubility for all values of x ranging from 0 to 1, implying that no energy is lost in the formation of a new phase. Figure 4 depicts how the cell voltage constantly swings during such single-phase activities. 23

Cell potential versus composition for a single-phase electrode reaction for such Li x TiS2.
In this technology, the lithium insertion reacted with TiS2 to form a single-phase (Li x TiS2) at the electrode. This caused the continual variation in cell voltage with composition as shown in Figure 4. The cell voltage reached up to 2.5 V after 76 cycles at 10 mA cm−2 after an intercalation reaction without using any carbon conductive diluent. It is the former reaction that Whittingham and his team 23 discovered with a reaction energy of roughly 2.5 V, and this laid the groundwork for the first rechargeable LIBs. However, the Li/TiS2 cell gets still charged and discharged but showed a working voltage of 1.5 V after consuming 90% of its compound. The formation of a single phase (Li x TiS2) in the first LIB occurs with a complete solubility of Li+ in the host material but was found to be thermodynamically unstable.
Though Whittingham’s LIB had a cell potential of 2.5 V as opposed to the lead-acid battery’s 2 V. The lower potential of lithium titanium disulfide (LiTiS2) indicates that it may react with oxygen and water under lithium deprivation. To avoid this, a conductive diluent, such as carbon black, must be added, which reduces the volumetric capacity and rate capability because the reaction can only take place at the intersection of three phases: electrolyte, carbon, and LiTiS2 particles. To address this issue, a new second generation of electrodes with improved properties such as cell voltage, cycle life, stability, and safety had to be defined.
For this reason, an ineluctable substitution of ether to esters occurred after the discovery of transition metal oxides (e.g. LiCoO2) as high-potential cathode materials by Goodenough and Mizushima. 24 LiCoO2 has the advantage of a higher voltage (3.8 V), is easier to handle (less sensitive to air), and the cells can easily be built in a discharged state since the cell potential is excessive for the lithium to react with water. Goodenough and Yoshino improved the technology by replacing the cathode and anode with LiCoO2 and graphite (C6), respectively. LIBs (LiCoO2/Li+ electrolyte/ graphite) are depicted schematically as shown in Figure 5. In the early 1970s, Stanley Whittingham harnessed lithium’s tremendous urge to liberate its outer electron when he created the first practical lithium battery.

The first Li-ion battery (LiCoO2/Li+ electrolyte/graphite) was depicted schematically.
It should be noted, change in LiCoO2 structure relies on lithium content. Removal of a high amount of lithium from LiCoO2 will cause structural disarray and capacity loss. LiCoO2 possesses a three-layer structure, in which the CoO2 layers are each offset by one-third from the adjacent layers, resulting in a single-layer repeating structure. However, in CoO2 itself, the layers are stacked directly on top of each other, just as in Li x TiS2 for all x values. In structural terms, the oxygen atoms in LiCoO2 are tightly packed cubically, whereas the anions in CoO2 and LixTiS2 are densely packed hexagonally. Another reason that not all lithium is removed from cobalt oxide is the intrinsic instability of Co4+ in CoO2. SONY 26 used large particles (15–20 μm) of LiCoO2 to avoid surface reactions that could lead to thermal runaway and had an excess of lithium in the combination Li1+xCo1−xO2. Because of the thermal stability, safety, and cost issues, lithium iron (II) phosphate (LiFePO4) is used as a popular cathode material for rechargeable lithium batteries to substitute LiCoO2. Its high-energy density, wide-operating temperature range, thermal stability, long cycle and application life, environmental friendliness, and safety make it a promising electrode material for greener and safer LIBs.27–29
In most cases, LiFePO4 batteries outperform equivalent LIBs. They are less susceptible to burns and thermal runaways, making them safer for home use. In addition, LiFePO4 batteries last up to five times longer than LIBs. A summary of the characteristics of three cathode materials LiTiS2, LiCoO2, and LiFePO4 studied are given in Table 1.
Comparison of the three cathode materials LiTiS2, LiCoO2, and LiFePO4.
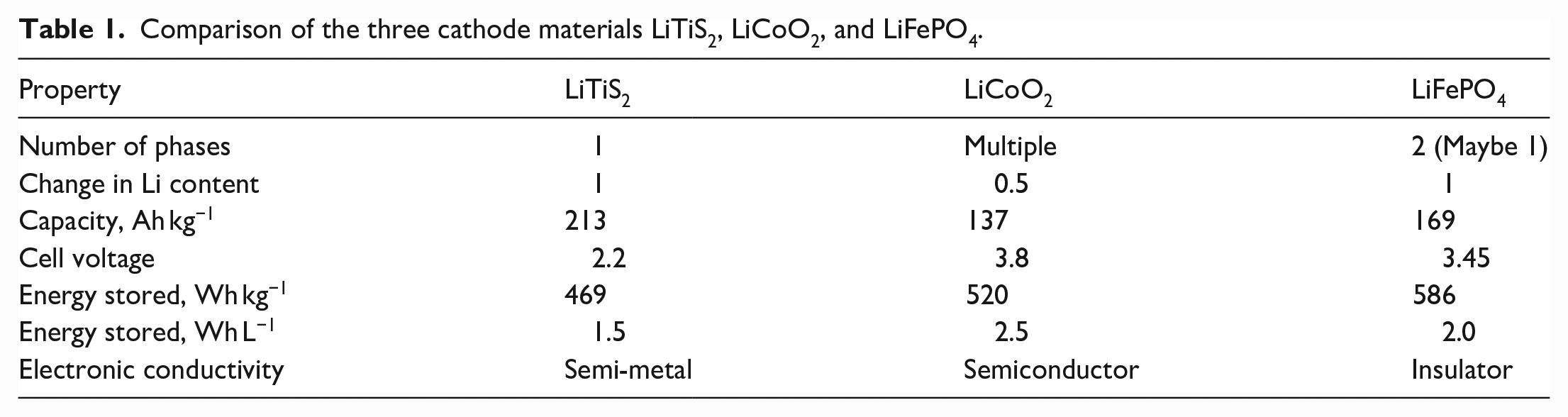
All values only consider the active material; the conductive carbon additive and binder are not included. The LiTiS2 does not require any carbon additive; this will increase its storage capacity by 10%–20% relative to the others. The anode receives no weight. Table was adapted from the study by Whittingham. 30
In the early 1950s, 31 ethylene carbonate (EC) and propylene carbonate (PC) are the two ultimate remarkable elements of the family of esters including carboxylate or carbonate esters, known as non-aqueous solvents. PC was greatly preferred due to its strong solvation capacity and high voltage stability (> 4.0 V vs Li/Li), while EC was seen as disadvantageous due to its high melting point (~37°C). Yoshino of Japan was the first to investigate the possibility of organic conductors such as polyacetylene as potential negative electrodes. By analyzing several carbonaceous materials, he was able to extend the notion of intercalation. The secondary battery made from this unique combination permitted reliable charging and discharging through numerous cycles over a long period. 32 This discovery (1985) resulted in the development of a novel secondary battery that used Goodenough’s LiCoO2 as the positive electrode and lithium intercalated petroleum coke (a carbon substance) as the negative electrode.
From the 1950s to the 1990s, typical non-aqueous electrolytes were consistently made up of diverse lithium salts dispersed in a combination of PC-including solvents. 33 After complete cloaking, EC was recognized as the miraculous solvent that remarkably empowered nearly all the theoretical capacity of LiC6. In 1990, Fujimoto and his co-workers at Sanyo replaced amorphous carbon with graphite but confronted the issue of discovering an electrolyte that does not desquamate the structure of C6. They also attempted to reduce EC’s high viscosity and melting point by blending it with dimethyl carbonate and diethylene carbonate, both linear carbonate esters like EC. 34
In the patent of the first LIB model, graphite was mainly ruled out using the quantification of crystallinity by the X-ray diffraction tool in a determined attempt to supply PC-centric electrolytes. 34 Finally, these attempts gave a lead to the original LIB, which was commercialized by Sony in 1990. The first commercial LIB was introduced in 1991, leaving just a short interval between laboratory development and industrial manufacture. Asahi Kasei invented and produced the first LIB in response to the demand for improved batteries. Sony was the initial organization to commercialize the technology, followed by A&T Battery Co. (a joint venture of Toshiba Batteries and Asahi Kasei Co.) in 1992.
In November 1991, Fujimoto et al. 35 filled up their patent that bounded the frame method for contemporary LIB electrolytes: LiPF6 dispersed in a combination of EC and a linear carbonate chosen out of dimethyl carbonate, diethylene carbonate, or ethyl methyl carbonate. The next restriction on electrolytes was thus caused by graphite-based anode materials. In hindsight, the same electrolyte design (EC/diethylene carbonate) had come out before in a patent by Okuno and Koshina, 36 but they do not principally assert graphite as a host material for the anode nor show any proof for LiC6 formed in such EC-centric electrolytes.
The LIB has significantly impacted the contemporary world with its wide application in electronic devices such as mobiles and computers. Besides, the current growth of smartphones and tablets has been subject to improved user experience, but this growth continues to demand electronic devices with more working cycles that incorporate skinnier and lighter LIBs. The name of the LIB type is derived from the cathode composition. Lithium transition metal oxides (LTMOs) such as lithium cobalt oxide (LCO), nickel cobalt aluminum oxide (NCA), lithium cobalt phosphate (LCP), nickel cobalt manganese oxide (NCM), lithium manganese oxide (LMO), lithium iron phosphate (LFP), lithium iron fluoro sulfate (LFSF), and lithium titanium sulfide (LTS) have been employed as cathode materials due to their high capacity for lithium intercalation and suitable physical and chemical properties for Li-ion transport. All three materials—LCO, NCA, and NCM have layered structures with high energy density at the expense of reduced thermal stability and high-priced cobalt. 37 Despite its modest capacity, LMO features a spinel-like structure, excellent thermal stability, and high voltage. Because of its low cost, high average voltage, and reduced sensitivity to thermal runaway compared to NCM and LCO, LFP has gained substantial interest38-41 and offers a fair balance of performance and safety.42-44 It also has a solid olivine structure.
The first Li1−xCoO2/C cell was developed by Yoshino et al. 34 at Asahi Kasei Corporation and was commercialized by the SONY Corporation in a cellular phone and a video camera. At present, the energy stored in the 18650 LIB cell has now been effectively enhanced by more than 3.0 Ah. Nevertheless, advanced cell engineering has been founded to improve the volumetric energy density, in addition to the capability of synthetic chemistry to adjust the size and morphology of the active particles as well as the architecture of the current collectors, which has almost attained its maximum. The original LCO has been enhanced by the addition of chemicals to stabilize the crystal structure and boost capacity. Figure 6 highlights the substantial contributions made by Goodenough and others in the chemical tuning of several cathode materials, such as LiCo1−xNi x O2, LiMnO2, and olivine-based LiFePO4.

Voltage versus capacity of various cathode materials about the organic electrolyte window (e.g. 1 M lithium hexafluorophosphate (LiPF6) in an organic carbonate).
The history of LIBs is not very old, unlike fuel cells, which have been around since Grove described the first battery in 1839. 45 Under 40 years, two new battery systems, Li-ion and nickel metal hydride (NiMH), have not only dominated portable devices but also the automotive industry. These advancements have all been founded on the concept of intercalation reactions. How far may intercalation chemistry be taken? Energy densities of some LIB cells developed based on intercalation are summarized. The statistics presented in Table 2 show that there is still much potential for improvement in the storing capabilities of Li-ion cells. These findings beg the question, “Where has all the energy gone?”
Energy densities of some lithium battery cells.

The theoretical values in the table solely consider the active components; no volume or weight for lithium other than that in the cathode is anticipated.
There is a considerable opportunity for improvement. Today’s batteries and supercapacitors have a lot of dead weight and volume, and the full capability of the active materials has not been realized yet. As is observed, the volumetric energy density of LIBs has grown from 250 to 570 Wh L−1 in the recent decade. Panasonic hopes that their new 18650 cells will improve it even more when they enter the market. There is no reason to suppose that the energy density cannot be raised further, maybe by 50% on both a volumetric and gravimetric basis, within the next 10 years. The volumetric energy density (Wh L−1) is far more relevant than the gravimetric energy density (Wh k−1g) in almost all applications. Further advancements will necessitate considerable changes in the materials utilized. In the preceding computations, it was believed that only one lithium could be intercalated into the structure; nevertheless, in some circumstances, two lithium could be inserted. Some examples include Li2MO2 and Li2VOPO4, although there are some considerable problems.
Liquid and traditional polymer electrolytes are dual-ion conducting systems, where both anions and cations are mobile. Since cations (Li-ions) are transmitted by electrodes across the electrolyte, ambipolar conductivity results in a salt concentration gradient, with depletion on the intercalation or coating side and over-concentration on the co-intercalation or dissolving side. This will increase electrode polarization and provokes the creation of dendrites in the case of Li-metal plating. 46 In 1982, Yazami reported the world’s first successful experimentation demonstrating the electrochemical intercalation and de-intercalation of lithium in graphite.47,48 Even though Yazami used a solid electrolyte, this experimentation offered the technical base for using graphite as an anode material—as is the average in LIBs currently. A solvent combination, such as dimethyl carbonate or diethyl carbonate, designed to enhance battery performance often contains lithium salts as the electrolyte in an LIB cell. The presence of lithium salts dissolved in the electrolyte indicates that the solution includes lithium ions. This implies that individual lithium ions do not have to travel from the anode to the cathode to complete the circuit. Others that are already present in the electrolyte near the electrode surface and being ejected from the anode can easily be absorbed (intercalated) into the cathode. The opposite occurs when recharging. Lithium quickly intercalates into graphite, either chemically or electrochemically. The first stage LiC6 binary chemical is formed as a result of this well-known reaction. Nevertheless, preparing a ternary compound by the intercalation of a lithium alloy is more complex. This is mostly due to thermodynamic reasons. Figure 7 depicts the intercalation principle of LIBs.

Intercalation principle of lithium-ion batteries. 49
The movement of lithium ions between electrodes takes place at a significantly higher voltage than in other battery types, and because they must be balanced by an equivalent amount of electrons, a single lithium-ion cell can create a voltage of 3.6 V or higher, depending on the cathode materials. Typically, an alkaline battery only generates 1.5 V. A conventional lead-acid automotive battery requires six 2-V cells stacked together to provide 12 V.
The reactions listed below demonstrate how chemistry works in moles. The chemical reaction of the cathode in the study by Bao et al. 50 is depicted as follows

where M= transition metal,
During discharge, the cathode is reduced as a result of the electrode receiving electrons from the circuit and absorbing lithium ions by the lithium dioxide.
The chemical reaction of the anode is given in the study by Zhang et al. 51 as follows

The lithium ions enter the anode (graphitic carbon powder) when the battery charges and leave the anode when the battery discharges.
In LIBs, the cathode and anode materials are LTMO and LiC6, which are structured in layers on aluminum and copper current collectors, respectively. 52
The overall reaction of the LIB is given in Zhang et al. 53 as follows

Although LIBs have many positive traits, the primary distinctions between various LIB types are found in the substance that serves as the electrode.
The performance and durability of an LIB are mostly determined by its charging and discharging cycles. The optimized and high-quality charging and discharging of the LIB improve the system’s efficiency and life cycle. Only one Li may intercalate with the six carbon atoms in C6, leading to the stoichiometry of LiC6, which has an equivalent reversible specific capacity of 372 mA g−1. As a result, replacing graphite anodes with materials having better capacity, energy, and power density is essential. 54
In 2019, Goodenough (University of Texas at Austin), Whittingham (Binghamton University in New York), and Yoshino (Asahi Kasei Corp and Meijo University in Japan) were given the Nobel Prize in Chemistry for their work on LIBs. The technology was made with a Cobalt oxide cathode and a petroleum coke anode. The 2019 Chemistry Laureate, John Goodenough, quadrupled the potential of lithium batteries (4 V), laying the groundwork for a far more powerful and practical battery. Yoshino successfully removed pure lithium from the battery and replaced it entirely with lithium ions, which are safer than pure lithium. This allowed the battery to be employed in practice. Their discovery enabled the commercial-scale production of LIBs and accelerated research into a wide range of new technologies, including wind and solar power. Figure 8 illustrates the design of the recent LIB technology developed by the three scientists.
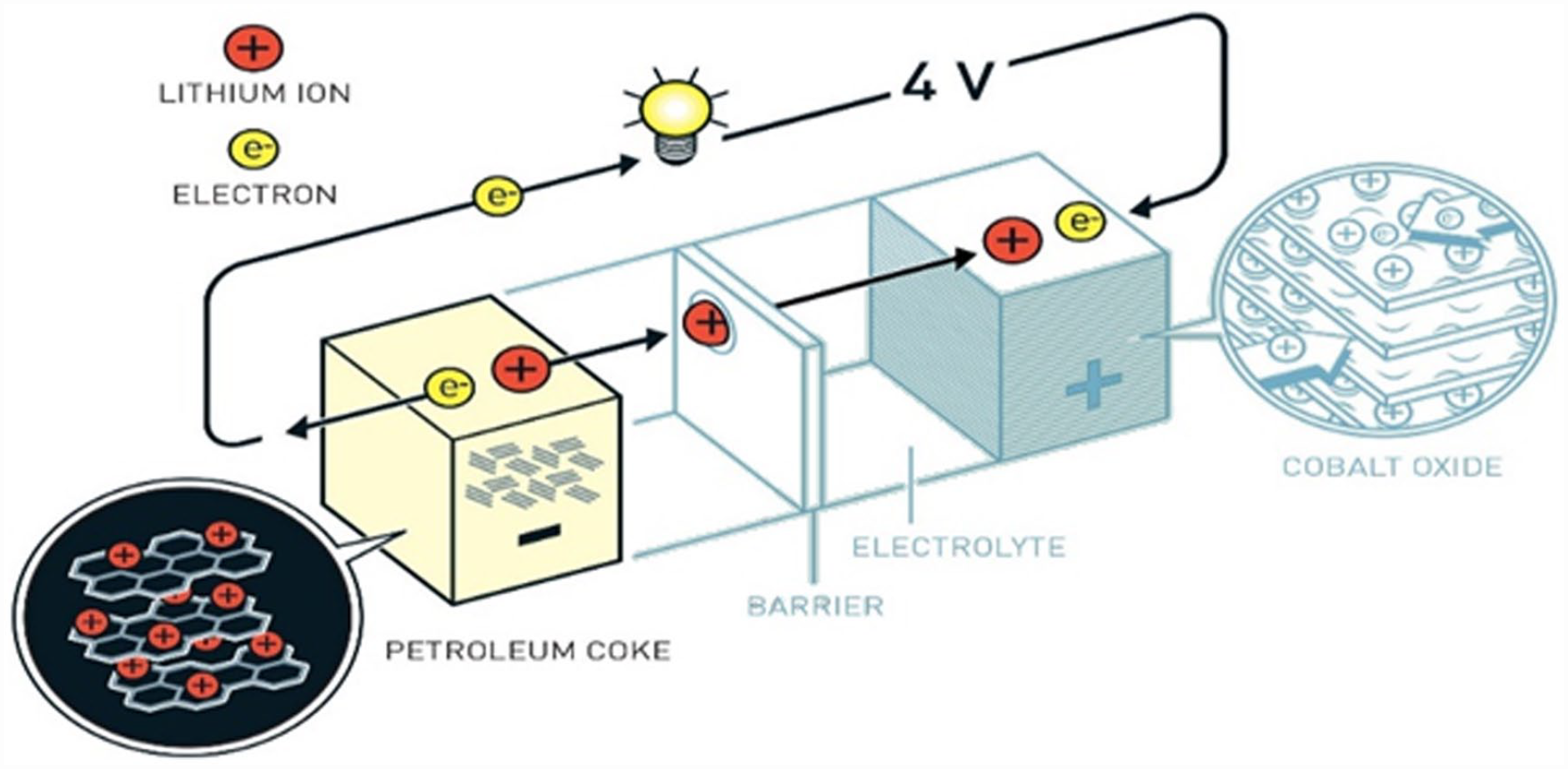
The lithium-ion battery was designed by Goodenough, Whittingham, and Yoshino for their Nobel Prize in Chemistry (2019).
The successful removal of pure lithium from LiCoO2 in this technology caused to double in the working voltage and electrode performance as compared to Whittingham’s technology, owing to the shrinking of the ion transfer pathways within the electrolyte increasing the ion flow current. Petroleum coke as anode material exhibits a wide-area phase that is heterogeneous, mostly independent, and has a few connected micro areas. The capacity of the negative electrode and cycle efficiency improve when the material’s internal structure of the material is more ordered and simpler to graphitize. Nevertheless, although having a high capacity and a steady charging and discharging platform, the cycle performance and low-temperature performance are weak. This is because Li+ is incorporated in the graphite layer, and graphite in the sheets layer generates graphite interlayer compound, causing the graphite layer to expand.
It is crucial to pursue research in the area of layered oxides, where commercial cells today reach only 25% of their theoretical capacity. In the field of LIBs, one approach to increasing energy density while reducing costs is to have more than one electron reaction per redox center. 55 This could be achieved, for example, by intercalating two lithium/sodium ions or one magnesium/calcium ion into a host structure. Only a few transition metals have two redox pairs that are stable within existing electrolyte stability constraints.
It should be stressed that a fundamental understanding of the intercalation processes of alkali and alkaline earth ions is crucial to a better understanding of those chosen for energy storage and other uses. Although all rechargeable lithium batteries nowadays employ intercalation reactions (structure retention), conversion reactions should not be overlooked. 56 Conversion reactions have substantially larger capabilities than intercalation reactions. In the latter, the structure is destroyed during the reaction and restored during the charge. The scientific obstacles are somewhat greater here, yet the energy density might be double that of intercalation reactions. However, the energy efficiency is substantially lower than, as low as 75% compared to 90% or higher for intercalation reactions.
LIB as a compact, lightweight rechargeable battery, was successfully developed and commercialized after achieving some key points: (a) The use of a carbonaceous substance for the negative electrode and LiCoO2 for the positive electrode presented an entirely new notion as the fundamental structure of the LIB. (b) It was made possible to employ LiCoO2 as the positive electrode material, leading to a cell voltage of at least 4 V. (c) The utilization of carbonaceous material with a specific crystalline structure for the negative electrode solved the issues of cycle durability and safety that had previously impeded the creation of a workable rechargeable battery utilizing metallic lithium as the negative electrode material. (d) Technology for producing electrodes, such as an aluminum foil current collector that can withstand an electromotive force of 4 V or greater, or higher, and technology for creating thin-film electrodes by coating, allowed for massive current discharge. (e) The use of a separator membrane that melts to shut down activity when temperatures rise over a certain threshold gave great safety qualities. (f) The growth of peripheral technologies such as safety device technology, which includes a positive temperature coefficient (PTC) device, protective circuit technology, and charging and discharging technology. The electrochemistry of LIBs affects their performance, cost, and safety. A lithium-ion cell with an LCO positive and a graphite negative electrode, for example, provides high voltage and energy density but has a larger thermal explosion risk, which can result in cell breakage, venting, electrolyte igniting, and fire. 57
Conclusion
LIBs have the potential to offer higher energy density that can be easily calculated. As stated in the preceding section, we now receive less than 20% of a battery’s potential volumetric capacity, so there is still an opportunity for development in system engineering as well as the application of new materials and processes. Although all rechargeable lithium batteries today use intercalation reactions to store additional energy, conversion reactions have the potential to achieve far larger capacities than intercalation reactions. Conversion reactions allow for substantially higher capacities than intercalation processes. The progress of LIBs has been extensively obstructed by the security problems coming from typical non-aqueous liquid electrolytes. As substitutes for liquid electrolytes, polymer electrolytes have attracted much attention due to their low flammability, flexibility, free oscillations, disturbance, and mechanical deformation. Lately, polymer electrolytes applied in high-voltage LIBs, flexible LIBs, Li-metal batteries, Li-sulfur batteries, Li-oxygen batteries, and smart LIBs have stimulated new investigation interest in both electrochemistry and material science fields.
We briefly discussed the history of intercalation electrodes, electrolytes, and basic principles related to batteries based on intercalation processes. The emergence of LIBs and studies of Li+ intercalation chemistry rely on anode and cathode materials used such as graphite and layered cathode materials, respectively; the type of electrolytes, Li+ interaction with solvent-free polymer electrolytes; and nanomaterials used for electrode activation. Nanomaterials play an important role in improving the response and/or protection of active materials, which may result in higher energy density. The creation of an artificial SEI between the two electrodes during the battery production process would be a solution not only to the existing requirement for week-long or longer formation cycles for all LIBs but also for higher energy density. LIB is a major scientific challenge. Solid-state batteries are gaining popularity among scientists due to their high energy and outstanding safety. They not only hold nearly twice as much energy as a regular LIB, but they also do not catch fire like their commercial equivalents.
Footnotes
Author’s contributions
The authors jointly did this work’s conceptualization, writing, and editing.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author received no financial support for the research, authorship, and/or publication of this paper.
