Abstract
Graphite-based anode materials undergo electrochemical reactions, coupling with mechanical degradation during battery operation, can affect or deteriorate the performance of lithium-ion batteries dramatically, and even lead to the battery failure in electric vehicle. First, a single-particle model based on kinetics of electrochemical reactions was built in this article. Then, the lithium-ion concentration and evolution of diffusion induced stresses within the single-particle model under galvanostatic operating conditions were analyzed by utilizing a mathematical method. Next, evolutions of stresses or strains in the single-particle model, together with mechanical degradation of anode materials, were elaborated in detail. Finally, in order to verify the aforementioned hypothesis, surface and morphology of the graphite-based anode dismantled from fresh and degraded cells after galvanostatic charge/discharge cycling were analyzed by X-ray diffraction, field-emission scanning electron microscopy, and transmission electron microscopy. The results show that large volume changes of anode materials caused diffusion-induced stresses during lithium-ion insertion and extraction within the active particles. The continuous accumulations of diffusion-induced stresses brought about mechanical failure of the anode eventually.
Keywords
Introduction
Lithium (Li)-ion cells are very compelling candidates for power supplies with their high-power and energy density and low self-discharge rate. They have been widely used in new energy vehicles such as battery electric vehicles, hybrid electric vehicles (HEVs), and plug-in HEVs in the past few decades. 1 For the health management study of Li-ion batteries in electrical vehicles, knowledge about the mechanical degradation of anode materials under certain operating conditions is of great significance. 2 Mechanical degradation may appear owing to deformations and stress generation as Li-ions intercalate/deintercalate into/from the active anode and cathode materials during charging and discharging operations over time. It is important to understand and mitigate mechanical electrode degradation in Li-ion batteries for EVs since it can accelerate battery aging and further lead to failure. 3
Unfortunately, the mechanical degradation under some special working conditions is one of the critical challenges in improving performance and prolonging life span of the cells at present. The difficulties existing in measurement of the critical properties and phenomena in electrode materials instigated extensive research activities on numerical modeling of Li-ion batteries worldwide in the last few years. 3 –5 Christensen and Newman 6 developed a mathematical model based upon concentrated solution theory and porous electrode theory that calculate the volume changes and simulate the distribution profiles of Li-ion concentration and stress during the repeated Li insertion/extraction processes into/from a spherical electrode particle. Doyle et al. 7 presented numerical models with a finite volume method and porous electrode theory which predict the transport, kinetics, and reaction processes in batteries. These models or methods have shown good agreement with discharge experiments to a certain extent; however, they cannot ideally account for mechanical failure of active materials within the electrodes in batteries applied in EVs. Cheng and Verbrugge 4 recently analyzed the evolution of stress and strain energy due to diffusion-induced stress (DIS) in a spherical insertion electrode particle under either galvanostatic or potentiostatic condition. Deshpande et al. 8,9 established relationships between surface stress, surface energy, and the magnitude of DISs in nanowires and investigated the effects of surface elasticity and surface energy on the stress evolution in spherical electrodes. They also correlated elastic strain energy, DISs, and stress discontinuities at the phase boundaries of the different phases that form during lithiation/delithiation using a core–shell structural model. 10
Literature on modeling of stress and crack formation within active electrode particles is quite extensive. Stresses and fracture in electrodes undergoing volume changes were predicted in a one-dimensional model by Huggins and Nix. 11 Furthermore, a terminal particle size below which particles are not expected to crack was also predicted in their model. Crack formation of electrode particles was modeled with facture mechanics by Aifantis and Dempsey. 12 Other researchers 4,13 –21 noted that the maximum stress within active particles is in proportion to the product of particle size and the concentration gradient. The maximum stress increases with current density, radius of spherical particles, and lower Li-diffusivity in the electrode material, all of which lead to steeper concentration gradients between the surface and the core, and was also verified in literatures. 6,9 –11
Despite significant advances in the theoretical investigation of stress and strain evolution within electrode active materials of Li-ion batteries, some innovative researches are still needed to explore the mechanisms of mechanical failure. In this article, the single particle model (SPM) was established in the “Single-particle electrode model” section. The solutions of the diffusion equations with initial and boundary conditions and analytic expression of evolution of DISs for the galvanostatic control within the SPM were presented in the “Evolution of stresses within an SPM” section. In the “Validation” section, a commercial Li-ion battery was cycled in a constant current (CC) charge/discharge process with a battery cycler in the potential range of 3.0–4.2 V. For comparison, the other fresh battery belonging to the same batch was disassembled in this article. The anodes dismantled from fresh and degraded batteries were analyzed by X-ray diffraction (XRD), scanning electron microscopy (SEM), and transmission electron microscopy (TEM). The measurements from XRD reveal the lattice parameter changes, and SEM and TEM were applied to verify the microstructure differences of the graphitic nanoparticles between the fresh and degraded batteries. These are the main contributions and helpful to understand the DISs in this article. Besides, the complex interplay between the origins and evolution of mechanical degradation and structure changes during electrochemical reaction in electrode materials were also discussed.
Single-particle electrode model
The SPM was first proposed by Haran et al. 22 in determination of the hydrogen diffusion coefficient within a metal hydride electrode and used to calculate the Li-ion diffusion of the insertion electrode during discharging/charging process. 23 A schematic illustration of electrochemical-based Li-ion battery is depicted in Figure 1. The Li-ion battery is considered as three parts: negative electrode (graphite anode), separator, and positive electrode (metallic oxide cathode), as shown in Figure 1 with a single spherical particle representing respective electrode of Li-ion batteries in the discharge/charge process according to the literature. 24 –26 During charge process, Li-ions diffuse to the surface of metallic oxide particles in the cathode, where they extract from the lithiated particles, travel through the electrolyte separator, and insert and diffuse in solid phase of graphite particles in the anode. Simultaneously, electrons emitted from the cathode transmit through the external circuit toward the anode. During discharge, positive Li-ions and electrons flow in the reverse direction (see Figure 1).

Schematic illustration of a lithium-ion battery during discharge/charge.
Evolution of stresses within an SPM
For a spherical particle, several groups provided evidence for Li-ion diffusion and phase transformations in mechanical properties. Qi et al. 27 showed that material properties change substantially upon Li insertion. Deshpande et al. 9 –11,15 considered the effects of elastic properties dependent on solute concentration on DISs in single-phase systems. Furthermore, analytic solutions for stresses 3,4,8 –11,14 –17,19 –23 can help elucidate the effects of Li-ions diffusion on DISs. We study DISs within an SPM as Li-ions insert and remove in Li-ion battery electrodes under galvanostatic charging strategies.
Solid mechanics of DIS
The stresses induced by Li-ion diffusion are considered in an SPM of radius
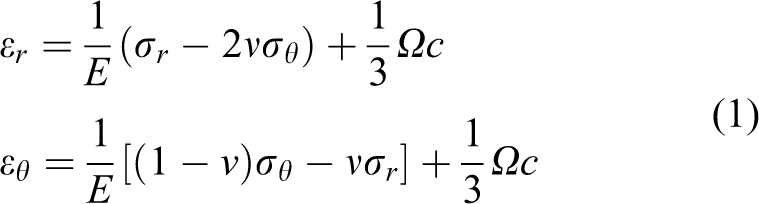
where
For infinitely small formulation of deformation, the radial and tangential strains of the spherically symmetric particle are given by

where

The boundary condition

where
One principal shear stress equals zero and the other two are both
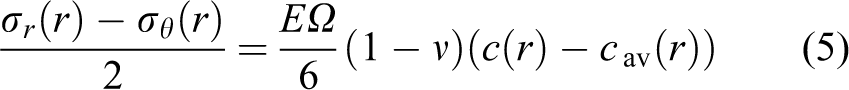
Thus, the stresses distributed at any given time and location can be obtained under the conditions that the concentration distribution is known. 4
DIS under galvanostatic operation conditions
The stress evolution within the spherical particles under galvanostatic condition is studied in this article since Li-ion batteries applied in EVs are mostly charged with a typical CC–constant voltage (CV) charging strategy. A simple relevant equation of Li-ion diffusion within a spherical SP of radius

where symbol
Considering that the particle parameters of different batteries will be different, equation (6) would be transformed into dimensionless form as follows

Then, the governing equation (6) in dimensionless form is shown by the following equation
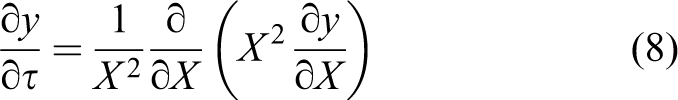
This condition denotes that the current is a constant and the ionic flux is invariant at electrode surface. The initial and boundary conditions subjected to galvanostatic control in dimensionless form are given by
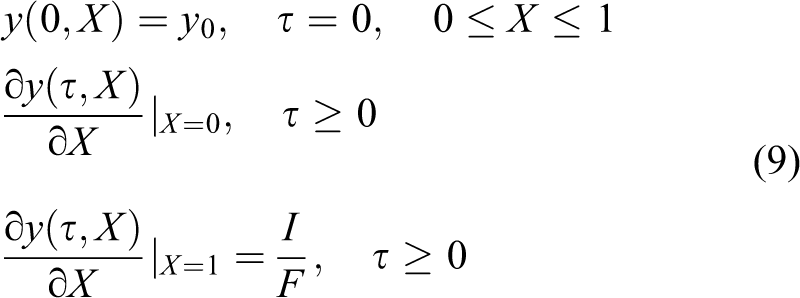
The analytic solution to address the diffusion equation (equation (8)) with initial and boundary conditions (equation (10)) subjected to galvanostatic control is as follows 29,30

where
Utilizing equation (10), the average concentrations of Li-ions can be expressed as

Equation (11) can be used to solve for stresses after equation (3) is substituted into it.

Figure 2 shows the Li-ion concentration, radial, tangential, and shear stresses of SPM in the insertion process under galvanostatic condition. The algebraic expression, normalized by
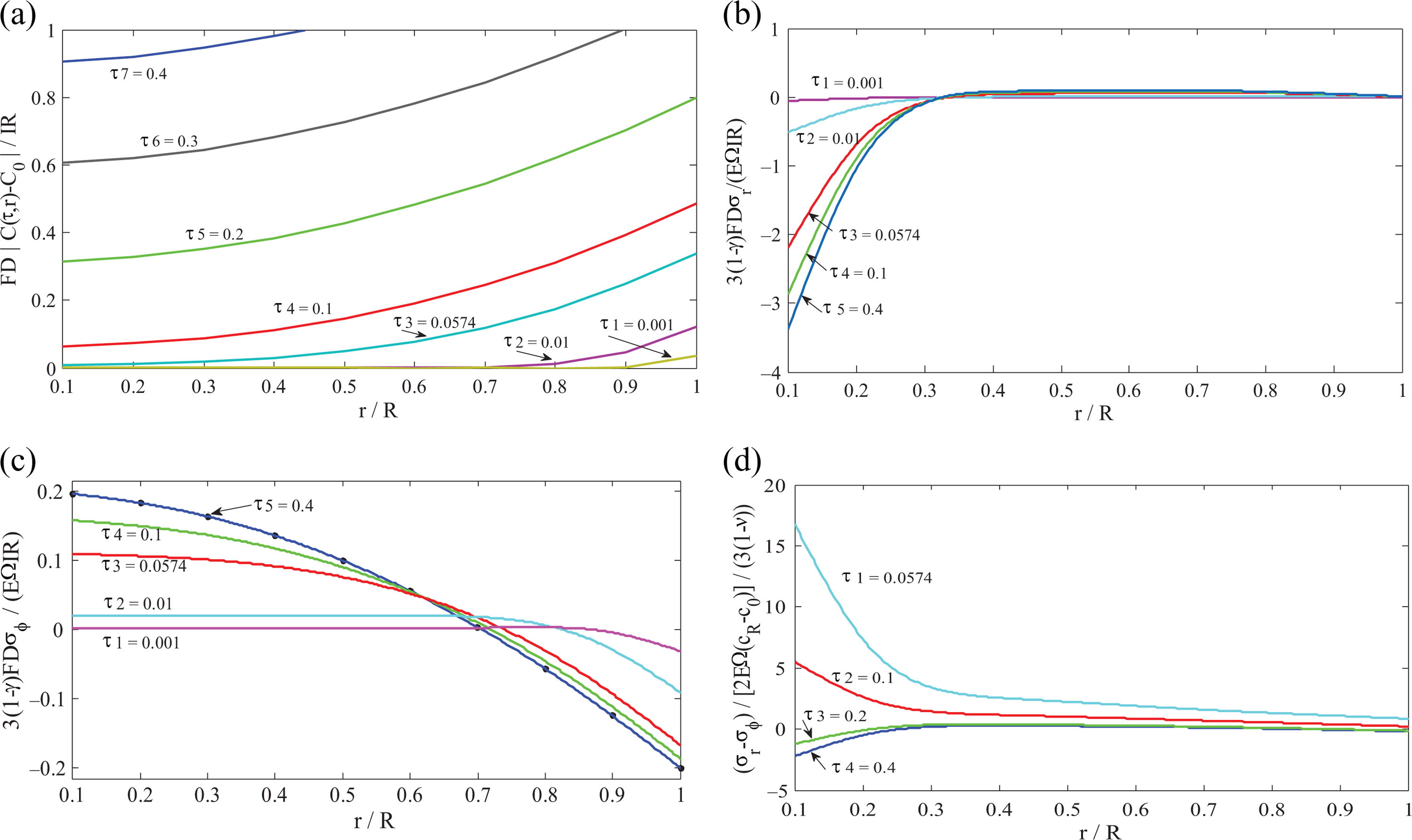
Characteristic profile of SPM in the insertion process under galvanostatic control: (a) Li-ion concentration; (b) radial stress; (c) tangential stress; and (d) shear stress. SPM: single particle model.
The current sign in equation (10) is defined as follows: positive current expresses for insertion process and negative corresponds to extraction. The Li+ concentration increases with position and time as depicted in Figure 2(a). During Li-ion insertion process, the radial stress appears in the form of compressive stress at the center of the sphere and approximates to zero close to the sphere surface. It increases with time near the sphere center and reaches maximum at the center (see Figure 2(b)).
The tangential stress appears as tensile stress near the sphere center and compressive close to the surface. The maximum of tensile tangential stress appears at the center before the Li-ions reach there and decreases monotonically toward the surface; then, it reverses and expresses as compression stress. Afterwards, it increases along the direction of sphere surface. Moreover, the tangential stress increases in magnitude with time at any location and finally tends to a steady state (see Figure 2(c)).The shear stress reaches the maximum at the location of sphere center and decreases toward the surface for all times and reaches a steady state afterwards. In addition, the shear stress decreases before dimensionless time
Validation
To demonstrate the complex causality between electrochemistry and mechanical degradation of anode in Li-ion batteries, we opted to study mesocarbon microbeads (MCMB) anode as a representative material that undergoes interactions and subsequent DISs during electrochemical cycling. The investigated commercial batteries used in this work have a normal capacity rating of 87 Ah and contain carbon material in anodes. The fresh battery was first charged at 1C rate to 4.2 V and discharged to 3.0 V subsequently. 1C rate means the current is 87 A. During each cycle, the battery was operated in a CC charge mode and then underwent a CC discharge until its voltage reaches 3.0 V. The rest time between charge and discharge is 2 h. The stop criterion for the cycling tests is state of health (SOH) = 80% at 25°C. The surface and structure morphology of anode obtained from dismantled cells before and after cycled were investigated by XRD, SEM, and TEM.
The graphitic MCMB is spherical and contains somewhat randomly oriented graphitic domains. It consists of graphene sheets staggered in either an AB (hexagonal, the most common form) or ABC (rhombohedral) stacking arrangement. Upon insertion of Li-ions during charge, the graphite was lithiated, and every fourth layer was filled before the next layers begin to take up Li. However, lithiation may be initiated at several different sites on the surface of a graphite grain, and the lithiated layers did not correspond to one another at different nucleation sites. Hence, the expansion of each grain may represent a large fraction of the expansion of a fully lithiated grain. 19 During discharge, the graphite was delithiated, and each grain may embody the contraction of a delithiated grain. The anode film mechanically failed due to the stress induced by the lattice parameter change during Li-ion intercalation/deintercalation. The average grain sizes of nanoparticles on the anode from a fresh and degraded cell are compared by XRD in Figure 3. Thus, the volume changes of the particle during Li-ion insertion and extraction resulted in a differential strain. It accumulated continuously and gave rise to stresses within the particle as the cycle increases. 9 Subsequently, cracks initiated, propagated preferentially at grain boundaries of the particle, and resulted in mechanical failure of the anode materials. They were also verified by the SEM and the high-resolution TEM micrographs (Figure 4), with an average particle size of 100 nm, which is in good agreement with the results obtained by XRD. 13 TEM images of lithiated anode display smooth edges of graphite (Figure 4(b)) with a very thin solid electrolyte interphase film. The crack and fracture could be observed on cycled anode from the TEM images (Figure 4(b)). The red arrows in Figure 4(b) point to the cracked nanoparticles visible on the graphitic layer surface. Besides, the edge of graphite sheet structure began to mellow and some circular particles with diameter of 100–150 nm appeared on the surface of the sheet, which can be associated with the Li plating and side reaction products produced at the reactions between anode surface and the electrolyte. They can also be observed in SEM and TEM images in Figure 4(b). By a unique combination analysis of SEM, TEM, and XRD, the results suggest that the mechanical failure of anode material can be caused by accumulated DISs within active particles. The stresses are initiated by repeated volume changes while the Li-ions insert/extract the host particles during charging/discharge.

XRD spectra of graphite-based anode from a fresh and degraded cell. XRD: X-ray diffraction.

SEM images of (a) fresh graphite-based anode before cycling tests and (b) degraded graphite-based anode after 500 cycles. SEM: scanning electron microscopy.
Conclusions
This article studied the evolution of stresses in a graphite-based anode of Li-ion batteries for EVs considering solid mechanics, diffusion theory, and electrochemical interfacial kinetics under galvanostatic condition. The profiles of Li-ion concentration, radial, tangential, and shear stresses in the SPM were analyzed and presented in the insertion and de-insertion process of the Li-ions in the particles. Furthermore, the evolution of phase structure and morphology for materials with anode demonstrates that a combination of XRD, SEM, and TEM can track the cause and effect of electrochemical and mechanical failure processes in a Li-ion battery for EVs. The experimental and analytical results show that large volume changes of anode materials occur during Li-ion insertion and extraction within the active particles when a battery discharges or charges. The accumulated changes lead to stresses within the active particles as the battery cycles, and they further lead to mechanical failure of the anode.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51575044).
