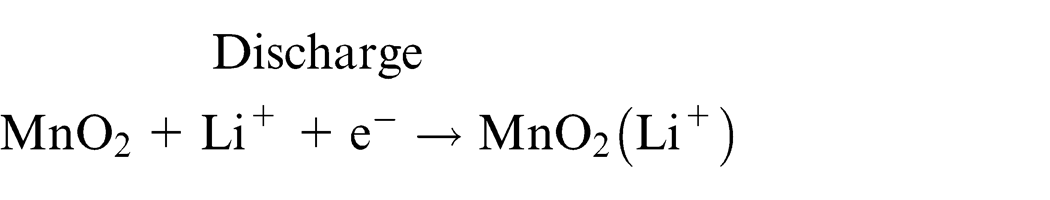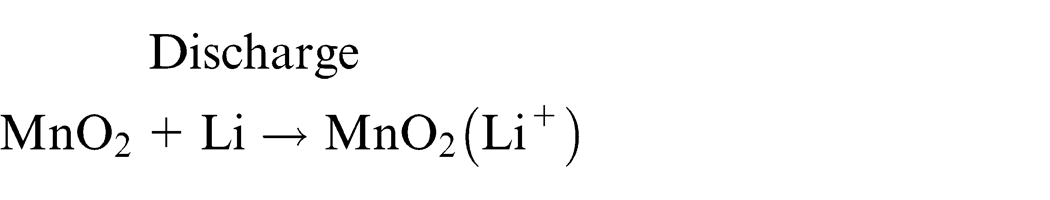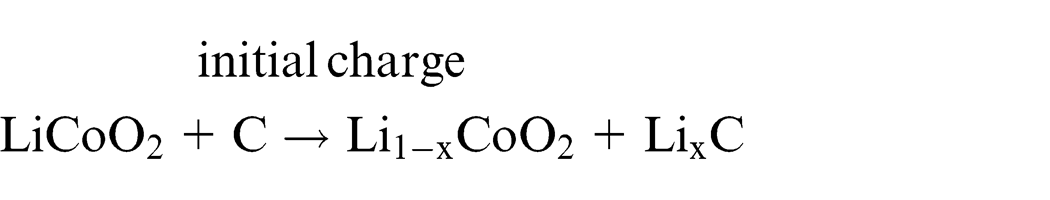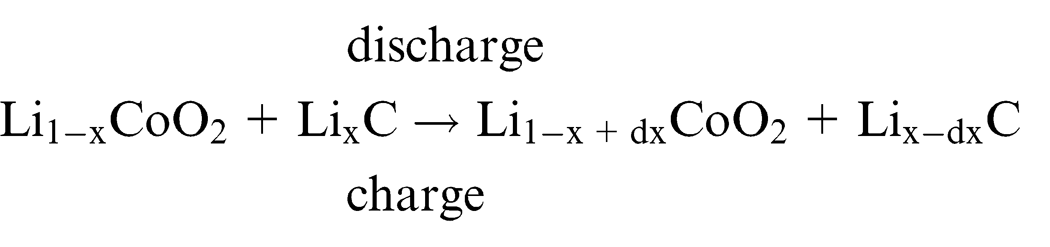Abstract
This study focused on a newly proposed recycling process to recover electrodic powder enriched in cobalt (Co) and lithium (Li) from spent lithium primary and ion batteries. In addition, this new process was designed to prevent the explosion of batteries during thermal treatment under an inert atmosphere. Spent lithium mixed batteries were heated over the range of 300 °C to 600 °C for 2 h, and each component was completely separated inside the reactor after the experiment. Electrodic powder was successfully recovered from the bulk components through sieving; the powder contained several pieces of metals. The electrodic powder obtained was examined using X-ray diffraction, energy dispersive X-ray spectroscopy, and atomic absorption spectroscopy. Additionally, an image of the powder was taken using scanning electron microscopy. The cobalt and lithium were mainly recovered at approximately 32–35 wt% and 3–5 wt% in the electrodic powder, respectively.
Introduction
A primary lithium battery is used when recharging or reuse is impossible; the secondary lithium ion battery (LIB) can be recharged and reused. A lithium primary battery refers to a battery using lithium or a lithium mixture as the cathode. Depending on the chemical materials or design used for the battery, the battery produces a voltage of 1.5–3.7 V, which amounts to approximately twice the output voltage of manganese or alkaline batteries. Lithium primary batteries have been widely used for over 30 years and are known for their useful properties such as long shelf life, constant voltage output, wide operating temperature range, and low self-discharge. Therefore, the batteries are applied in various fields such as information technology (IT), medical, and military.1,2 However, lithium primary batteries contain hazardous materials such as lithium (the metal anode material), flammable solvents, and potentially explosive components (the lithium perchlorate electrolyte), which can lead to exothermic activity and a runaway reaction above a certain temperature. 3
LIBs are a type of secondary batteries in which the lithium ions move from the cathode to anode in the discharge process. Upon recharging, lithium ions move from the anode to cathode to recover the original positions. LIBs are different from lithium primary batteries (primary batteries cannot be recharged or reused) and from lithium ion polymer batteries (polymer batteries use a solid polymer as the electrolyte). LIBs are divided into three parts: anode, cathode, and electrolyte. A variety of materials may be used for each of these components. Commercially, the most widely used anode material is graphite (C). For cathode materials, layer-structured lithium cobalt oxide (LiCoO2), lithium iron phosphate (LiFePO4), and lithium manganese oxide (LiMnO2) are in use; titanium disulphide (TiS2) was also used initially. Depending on the materials used for the cathode, anode, and electrolyte, the voltage, life, capacity, and stability of LIBs may vary greatly. Recently, nanotechnology is enhancing battery production and performance. LIBs are widely used as electrochemical sources in portable electronic applications, including mobile phones, personal computers, and video cameras, because of their favourable performance and electrochemical properties; these batteries are also likely to play an important role in providing power for future electric automobiles. 4 With the increasing use of IT, electronic equipment, military supplies, and vehicles, the generation of spent lithium primary and ion batteries has increased significantly. The recycling of lithium mixed batteries has increasingly become important because their safe disposal may become serious problems because of the presence of flammable and toxic materials. However, spent lithium mixed batteries are not generally classified as a dangerous waste.5–7 Additionally, an economic benefit could be achieved by recovering the major components of lithium mixed batteries.8–9
Over the past decade, several recycling technologies have been investigated. These technologies are mainly classified as physical and chemical processes. Generally, chemical processes have been widely adopted and basically consist of acid leaching, chemical precipitation, solvent extraction, filtration, or other processes. Unfortunately, these processes could generate another pollutant (acid solutions) after the recovery process. Therefore, an objective of this present study is to develop a new recycling process for recovering valuable metals from spent lithium primary batteries and LIBs. This process should be eco-friendly and cost-saving. Additionally, the process employs thermal treatment to form electrodic powders, unlike previously developed leaching operations that dissolve all materials. This newly proposed thermal treatment technique allows the safe handling of spent LIBs and efficient concentration of valuable metals in the electrodic powders.
Materials and methods
Spent lithium primary and ion batteries were used in this study. Notably, the identical brand of batteries has been used to maintain the reproducibility of experiments. Depending on the chemical materials used for the cathode or electrolyte, lithium primary batteries are mainly divided into two structures (Figure 1). The lithium-thionyl chloride cell uses thionyl chloride (SOCl2) as the cathode, lithium metal as the anode, and thionyl chloride as the electrolyte. This cell is known to have a high energy density. In this battery, porous carbon material is used as the anode collector to receive electrons from the outside circuit. However, the lithium-thionyl chloride cell is not generally commercially sold; these batteries are primarily being used in industry, particularly for alarm devices requiring operation at a low current for a long time. The most common lithium battery, lithium manganese dioxide, is used widely among consumers; this battery employs lithium salt dissolved in an organic solvent as the electrolyte, metal lithium as the cathode, and manganese dioxide as the anode. 10 The electrochemical system consists of a cathode (which is a mixture of manganese dioxide), a carbon black conducting agent, and a binder. The electrolyte is a solvent solution of lithium perchlorate (LiClO2) in propylene carbonate. The water content is below 50 ppm. The anode is made of a lithium foil pressed into a stainless steel anode can. Lithium primary batteries are assembled in a high-purity argon atmosphere. The electrode half reactions and cell reactions for lithium primary batteries are as follows.
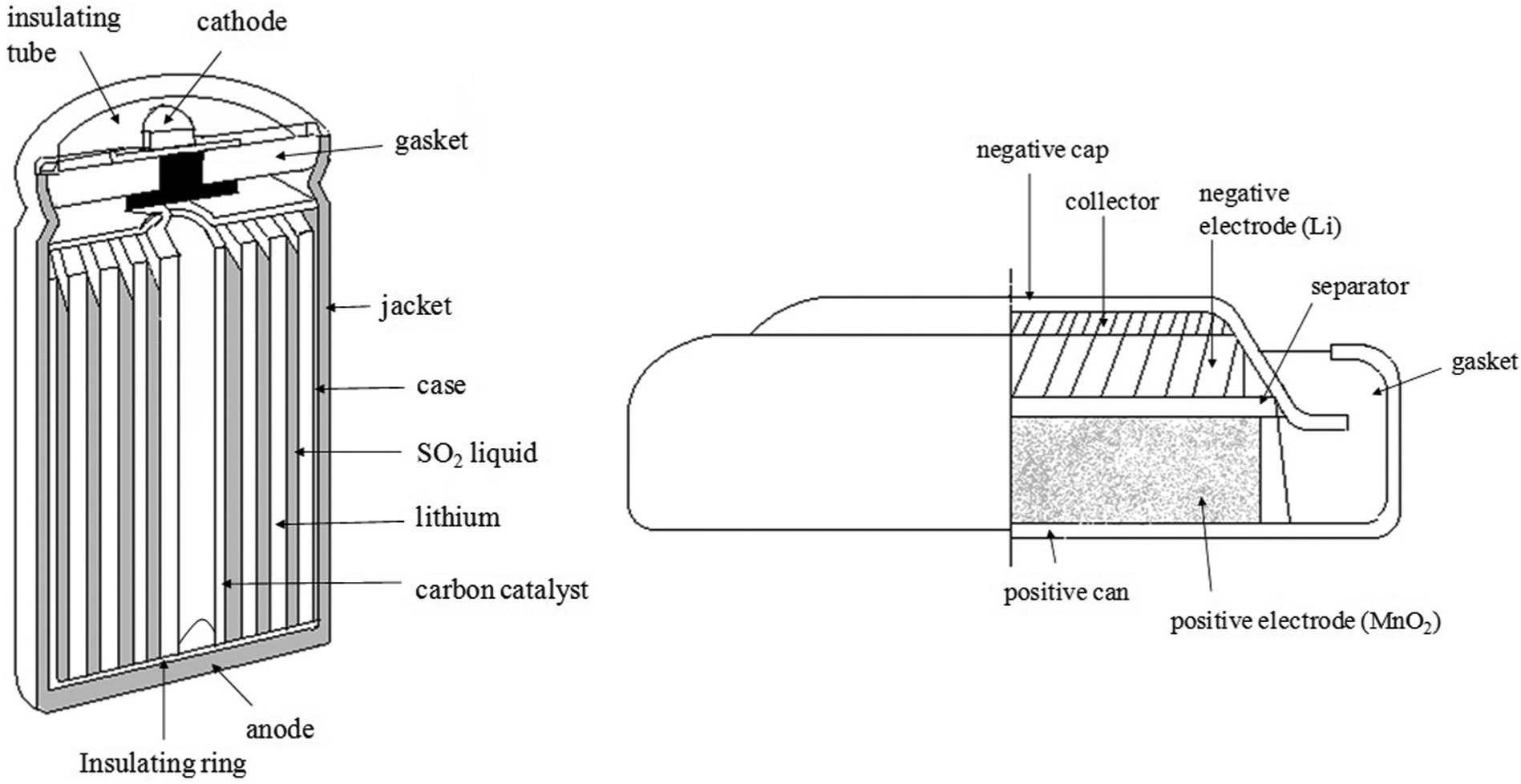
Structure of the different types of lithium primary batteries.
Positive electrode reaction
Negative electrode reaction
Total reaction
The conventional structure of LIBs mainly consists of a cathode, electrolyte, separator, anode, gasket, gas release valve, and sealing plate (Figure 2). 11 The cathode is the positive electrode of the battery, that is, the source of positive ions (Li+) and sink of negative ions (e−). In layered cathodes, the composition is denoted by LiMO2, in which M corresponds to Co, Ni, Mn, or V. 11 LiCoO2 is currently the most popular option (despite the high cost of cobalt) because it is relatively easy to prepare a high-quality electrode with a layered structure. The anode is the negative electrode of the LIB, consisting of layered graphitic carbon. The separator provides a barrier preventing the short circuiting of the cathode and anode. This barrier should be sufficiently porous to allow the electrolyte to freely connect the two electrodes. Separators are commonly made of polytetrafluoroethylene (PTFE) and other inert polymer membranes with defined porosities and ionic conductivities. Commonly used electrolytes for LIBs include propylene carbonate and mixtures of these solvents.12–14
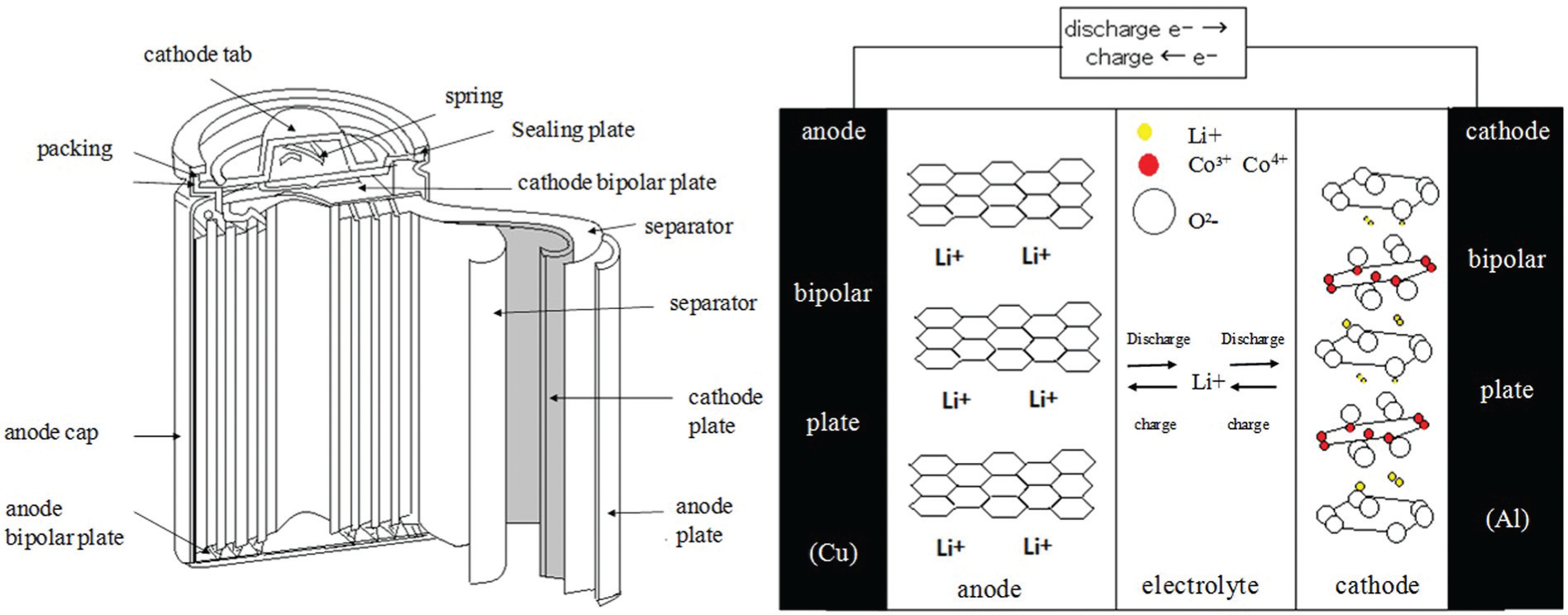
Structure and principle of operation of a lithium ion battery. 11
Lithium ions migrate between the cathodes and anodes, leading to battery charging and discharging. During charging, lithium ions are dedoped from the cathodes, whereas the interlayers of carbon in the anodes are doped with lithium. During discharge, however, lithium is dedoped from between the carbon layers in the anodes; the compound layers in cathodes are doped with lithium. Therefore, when charging initiates, lithium ions migrate from the lithium compound of the cathode to the carbon material of the anode. 15 The structure and principle of operation of a LIB is illustrated in Figure 2, and the chemical reactions in the two electrodes can be simply expressed as follows16–18
Subsequent discharge reactions occur through the migration of lithium ions from the anode to the cathode
In the present study, spent mixed batteries consisted of lithium primary batteries and LIBs with weight ratio of 4:1. The end-of-life mixed batteries used in this experiment are shown in Figure 3, and 10 pieces of total batteries were fed continuously into the reactor until the thermal treatment was completed. For the thermal treatment experiment, products collected from the identical manufacturer per battery type have been used to maintain the reproducibility of the composition of metal values in the mixed batteries.

End-of-life mixed batteries used in this study.
The newly proposed recycling process to recover electrodic powders enriched in various valuable metals is shown in Figure 4. This process mainly focused on preventing the explosion of batteries during thermal treatment under an inert atmosphere because the oxidation of metallic compositions in the presence of air during thermal treatment might result in a dramatic reaction able to produce a dangerous explosion. End-of-life spent batteries were collected and charged in a steel vessel. The thermal treatment in this work was performed at different temperatures, ranging from 300 °C to 600 °C, for 2 h in electric furnace with a SiC heater. Spent batteries were placed inside the reactor; the reactor was then completely sealed, and ultra-high-purity argon was injected at the rate of 3 L/min to maintain an inert atmosphere inside the furnace during the experiments. The temperature was increased at 10 °C/min and was maintained for 2 h after obtaining the target temperature. After thermal treatment, the temperature decreased at a rate of 10 °C/min to room temperature. A sieving operation was performed to separate fine powders (electrodic powder) less than 105 µm (75 mesh) from bulk components that contained pieces of metals. The collected powders were further extracted by chemical leaching, whereas the larger factions were further separated into ferrous metals, non-ferrous metals, and non-metals by an Eddy Current Separator. To remove organic solvents from the powders, another heat treatment was conducted by maintaining the temperature at 200 °C for 2 h using a muffle furnace.
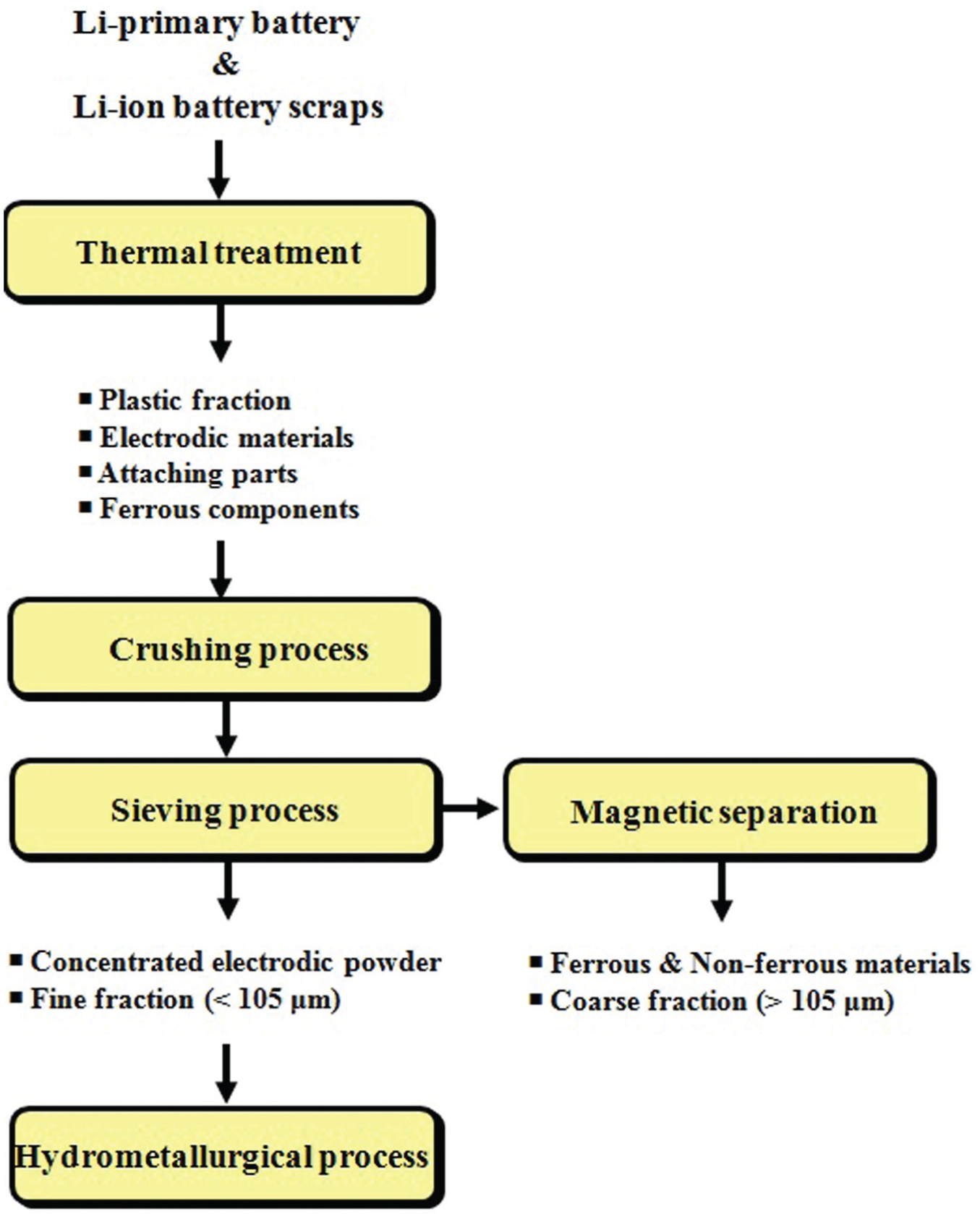
Proposed flow diagram for recycling spent LIBs using a thermal treatment process.
Results and discussion
A new electric furnace was newly designed to recover valuable materials after thermally treating spent mixed batteries. Using the current experimental apparatus, end-of-life batteries may be treated safely and efficiently by sorting the metal portions, obtaining an electrodic powder concentrated with valuable metals, and handling the toxic gases in electrolytes. The experimental apparatus used in the thermal treatment to recover valuable metals is shown in Figure 5. Figure 5 illustrates a side view of the main reactor and a top view of the cover for the thermal treatment furnace. The electric furnace (1) has been devised to treat mixed spent batteries in a stable manner and to efficiently treat the electrolyte and the gas produced in the closed system. The reactor should be able to withstand the explosiveness generated in the thermal treatment process and requires a material with high heat and corrosion resistance. The reactor was made of SUS 3103S; SUS 3103S has excellent heat, corrosion, and concentrated acetic acid resistance with good formability and weld-ability. In particular, this material has an excellent resistance to high-temperature oxidation and a good strength because of the high Ni-Cr content. The heating element (2) consists of SiC, which enables the temperature to be raised up to 1500 °C. The cover for the electric furnace is also made of SUS 310S material, consisting of 3ea of gas outlet (2), thermocouple (3), gas inlet (4), sample inlet (5), water cooler tube (6). To prevent inflow and outflow of inside and outside gas to and from the reaction furnace and the cover, O-ring (7) is installed, while water cooler tube (6) was placed above and below so that the O-ring may withstand high temperatures during the experiment.

A schematic diagram of the electric furnace newly developed for thermal treatment.
Accessory equipment of the electric furnace for thermal treatment was developed and installed to efficiently concentrate valuable metals and to handle toxic gases from mixed batteries (Figure 6). Thermal treatment was conducted at 500 °C for 2 h under an argon atmosphere, and mixed batteries were continuously added through charging tubes during the experiment. Off gases with toxic components were collected using a hood installed above the furnace. The off gases and dust passed through a dust collector to filter valuable materials and transport off gas to the scrubber. The scrubber reduced the toxicity of gases by neutralising the pH lever by washing the gases with a solution of sodium hydroxide (NaOH). Because of these accessory pieces of equipment installed with the electric furnace, hazardous materials from spent mixed batteries were efficiently collected and treated.

Recycling system for the recovery of valuable metals from spent mixed batteries.
Spent lithium primary and ion batteries from the identical brand manufacturer were heated at different temperatures (from 300 °C to 600 °C; Figure 7). As a result of the thermal treatment for 2 h at each temperature, the electrodic powder, copper electrode foil, separator, and metal case were completely separated inside the reactor after the experiment. The battery sheath and label were completely removed during thermal treatment, and electrodic powders were found to erupt out of the torn battery case. Regardless of an increase in temperature, spent batteries were easily disintegrated, and the components were found to be separated at above 300 °C, which might be a sufficient temperature for the thermal treatment stage.

Component and electrodic powder obtained after thermal treatment.
Overall, the recycling process separating the electrodic powder from components of spent batteries is shown in Figure 8. Spent lithium primary batteries and LIBs were fed into the electric furnace, and the thermal treatment was conducted at 500 °C for 2 h. Explosion occurred inside the reactor during the test, and then samples including the battery components were removed from the furnace. The samples that underwent a crushing process became small pieces of materials including the fine electrodic powder. A classification process was performed to screen concentrated powders from various components of spent batteries using a sieving machine. The electrodic powders less than 75 µm were recovered using a sieve with 105 mesh, whereas other components such as metal cases, caps, bottoms, separators, and electrode foils were captured on the sieve.

Overall recycling process to recover electrodic powder from spent lithium mixed batteries.
For the electrodic powders concentrated in the classification process, an analysis of the chemical composition was conducted with atomic absorption spectroscopy (AA) and energy dispersive X-ray spectroscopy (EDS). The results are displayed in Figure 9. According to the AA analysis, cobalt, lithium, copper, aluminium, and nickel were recovered and concentrated in the electrodic powder regardless of temperature. The major components of the powders were found to be cobalt and lithium; the largest amount of Co of approximately 32–35 wt% and Li of approximately 3–5 wt% may be attributed to the fact that cobalt and lithium are concentrated in the cathode in the battery. In addition, other materials (such as Cu, Al, and Ni) were also detected because a small amount of the case and electrode foil might contaminate the powder after the classification process. An EDS mapping was conducted for the concentrated powder, and Co, Cu, Al, and Fe and the gas phases of oxygen (O), chlorine (Cl), and fluorine (F) were observed as the major components. The fact that the cobalt composition displays the highest concentration is because lithium cobalt oxide (LiCoO2) is used as the cathode material that was concentrated in the powders. Aluminium and copper might be detected in the powders because of the pulverisation of the LIB and electrode foil, respectively. Substances such as O, Cl, and F are detected in the powder forms because of the organic solvents used for the electrolyte of the mixed batteries.
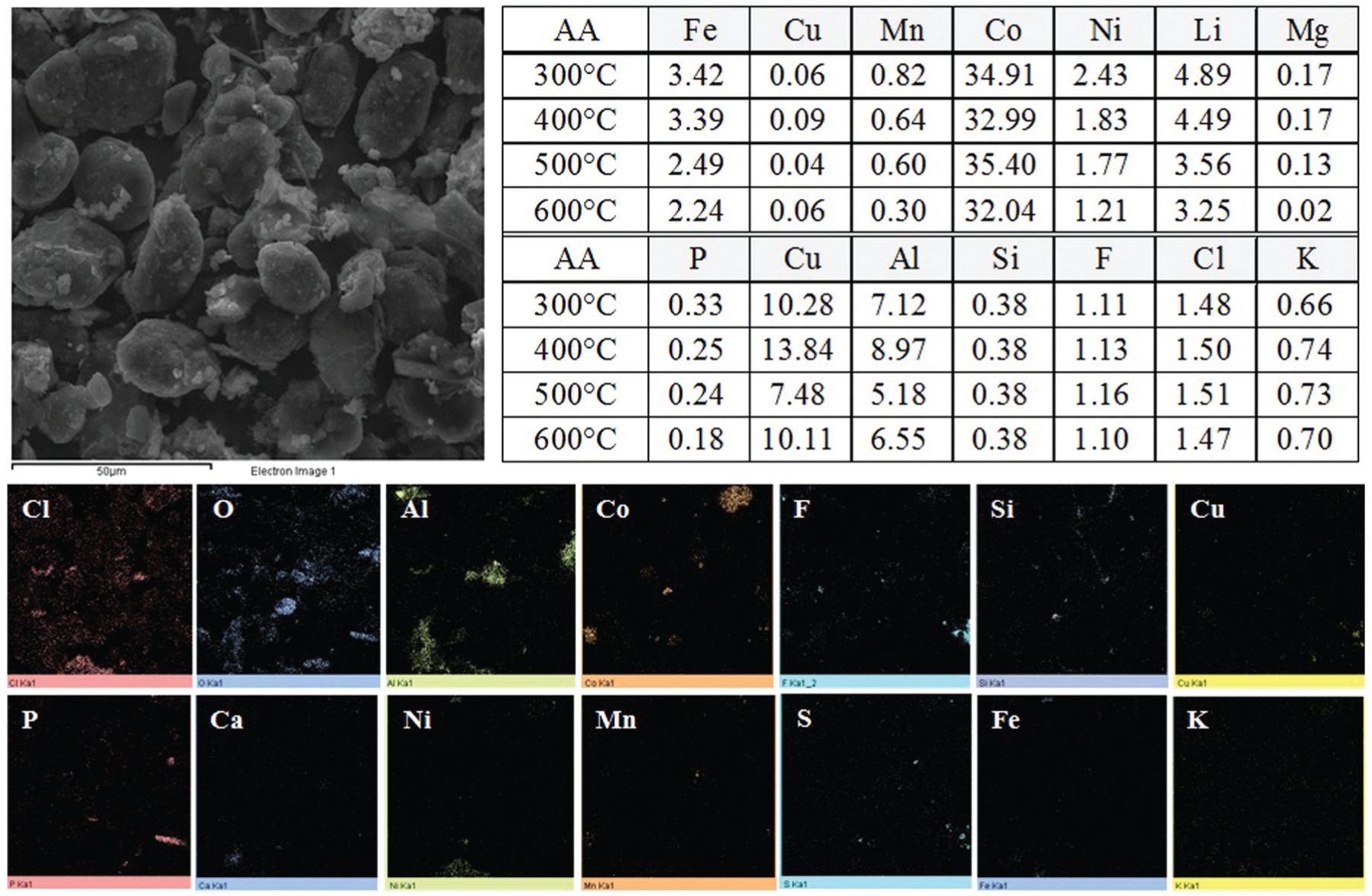
Chemical composition of the electrodic powder analysed using AA and EDS.
The electrodic powder obtained after thermal treatment at 500 °C for 2 h was examined using X-ray diffraction (XRD), and an image of the powder was taken using scanning electron microscopy (SEM; Figure 10). The observed major peak is lithium aluminium oxide (LiAlO2), which results from the metallic lithium used in the cathode materials and used as a salt dissolved in an organic solvent in the most common type of lithium ion cells. Other patterns observed in the XRD in Figure 10 could be considered as the anode material graphite (C) and the liquid electrolytes in lithium primary and ion batteries, which consists of lithium salts (such as SOCl2, FeS2, LiPF6, LiBF4, and LiClO4) in an organic solvent (such as thionyl chloride, iron sulphide, ethylene carbonate, dimethyl carbonate, and diethyl carbonate).
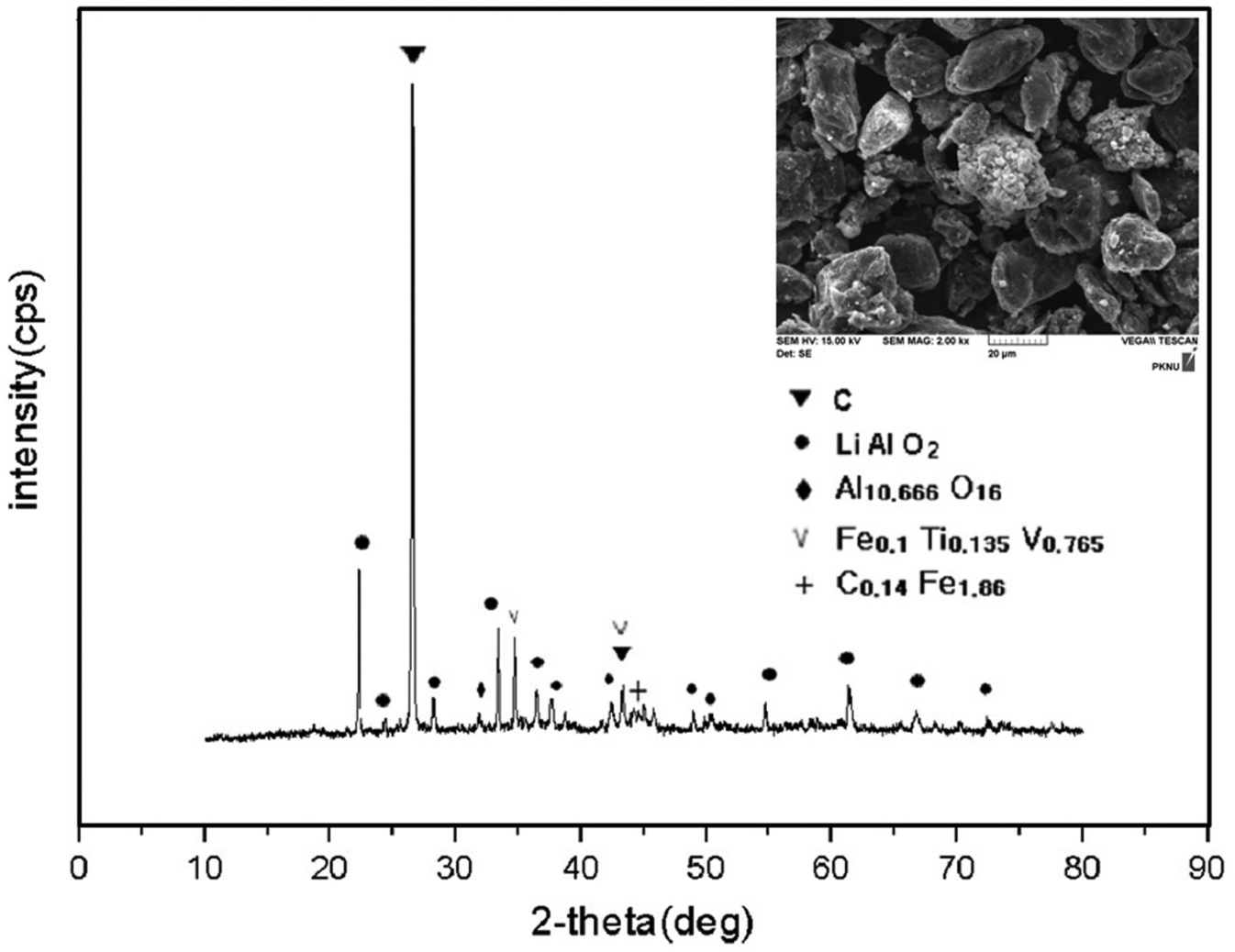
XRD patterns and a SEM image of the electrodic powder obtained at 500 °C.
Conclusion
A newly proposed recycling process to recover valuable metals in the electrodic powder resulting from spent lithium primary batteries and LIBs has been conducted using a thermal treatment method at different temperatures under an inert atmosphere. Valuable metals such as cobalt (32–35 wt%) and lithium (3–5 wt%) were concentrated in the electrodic powder. With the thermal treatment process, the end-of-life lithium mixed batteries could be treated in a stable manner; additionally, the explosion risks and environmental problems could be resolved effectively.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by the R&D Centre for Valuable Recycling (Global-Top Environmental Technology Development Program) funded by the Ministry of Environment (Project No.:GT-11-C-01-060-0).