Abstract
A novel up-conversion luminescence tetranuclear sodium complex [Na4(5-(carboxymethyl 2-oxo-propyl) amino isophthalic acid)(H2O)9] is synthesized by a solvothermal reaction, and its crystal structure is determined by single-crystal X-ray diffraction. The title complex crystallizes as a triclinic system with the P
Keywords

Introduction
Due to their fascinating properties, numerous complexes have been widely used in biology, materials, and chemistry. Therefore, in recent decades, complexes have attracted more and more attention by materials and chemical scientists.1–5 In complexes, especially due to the superior properties of rare earth metals in magnetism, semiconductors and photoluminescence, organic–inorganic hybrid complexes have received significance.6–9 In recent years, many researchers have devoted their attention to the design, preparation, and characterization of new rare earth metal and transition-metal materials. So far, scientists have undertaken a large number of rare earth or transition element studies to reveal the practical applications of materials. Such materials in the field of light-emitting diodes (LEDs), magnets, electrochemical displays, luminescent probes, and so on, are related to the magnetic and photoluminescence properties of rare earth elements.10–12
As is well-known, the chemical and physical properties of rare earth materials are mainly related to the 4f electronic configuration of lanthanide ions. For example, if the 4f electrons of a lanthanide material can be transferred effectively, then photoluminescence is possible. However, in many cases, due to the low absorption coefficients of lanthanide ions, the conversion between 4f electron orbitals is impossible; hence, the photoluminescence quantum yield of lanthanide materials is fairly low. In order to increase the absorption coefficient of lanthanide ions and to promote the conversion between 4f electron orbits, scientists usually employ a strategy called the antenna effect.13–16 In order to achieve this strategy, scientists typically use organic molecules with conjugated structures as antenna ligands to bind to lanthanide ions. Antenna ligands, such as heterocyclic molecules or aromatic carboxylic acids, can absorb the excited light energy and transfer it to lanthanide ions, after which, the lanthanide ions can emit photoluminescence.17,18 However, there have been few reports regarding the coordination of ligands with the metal atoms of the first main group. 19
In the design and preparation of complexes, it is very important to choose a good ligand. Carboxylic acids not only have many coordination modes, such as single-tooth coordination, symmetrical chelation coordination, asymmetric chelation coordination, single-oxygen-bridge coordination, oxygen-bridge coordination, and so on, but also have strong coordination ability.20,21 Therefore, carboxylic acid compounds are the preferred flexible ligands for researchers.
Our group has recently been studying the crystal structures and physicochemical properties of tetracarboxylic acids. In this work, we report the preparation, crystal structure, and semiconductor and photoluminescence properties of a novel tetranuclear sodium complex: [Na4(5-(carboxymethyl 2-oxo-propyl) amino isophthalic acid)(H2O)9] (1), which forms a three-dimensional (3D) supramolecular network.
Results and discussion
Synthesis
The synthesis of the ligand was prepared by adding chloroacetic acid to alkaline solution for substitution, and then acidification, as shown in Scheme 1.

Synthesis route of the ligand HL.
Crystal structure
Single-crystal X-ray diffraction measurements revealed that the title complex is a neutral molecule that crystallizes in the space group P

An ORTEP drawing of the title complex with 50% thermal ellipsoids.
The crystallographically independent Na5 ion is hexa-coordinated and displays a distorted octahedal structure. The Na5 ion is bound by two oxygen atoms of a 5-(dicarboxymethylamino)(isophthalate) anion ligand, three oxygen atoms of three-coordinated water molecules, and one Na2 ion. The Na2 ion is also hexa-coordinated and displays a distorted octahedral structure. The Na2 ion is bound by four oxygen atoms of four-coordinated water molecules and two Na+ ions (Na3+ and Na5+). The Na3 ion is octa-coordinated and displays a dodecahedral structure. It is bound by six oxygen atoms of six-coordinated water molecules and two Na+ ions (Na2+ and Na2#+). The Na4 ion is hepta-coordinated and displays a slightly distorted pyramidal structure. It is bound by five oxygen atoms of three 5-(dicarboxymethylamino)(isophthalate) anion ligand, one-coordinated water molecule and two Na+ ions (Na#+ and Na4#+). The Na1 ion is four-coordinated and located in the center of the plane composed of four-coordinated water molecules. In the crystal structure, There is no π···π stacking interaction, but there are a large number of hydrogen bonds (16 groups of intermolecular hydrogen bonds and 2 groups of intramolecular hydrogen bonds in the crystal structure of the form O-H···O (see Table 3 and Figure 2 for details) and van der Waals attractions yielding a 3D supramolecular structure. The crystal packing is presented in Figure 3.

The hydrogen bonds present in the title complex (magenta dashes).

The packing diagram of the title complex with dashed lines representing hydrogen bonding interactions.
Solid-state diffuse reflectance spectrum
In order to help understand the conductivity of insoluble complexes, the technique of solid-state UV-Vis diffuse reflection was developed. Under excitation with ultraviolet light, the electrons in a complex absorb energy, transition from the ground state to the excited state and absorb in the ultraviolet region. In this way, information on the conductivity of a material is obtained.
We measured the UV-Vis diffuse reflectance spectrum (DRS) on a solid-state sample of the title complex on the basis of barium sulfate as a reference giving 100% reflectivity at room temperature. Data processing is mainly based on the formula proposed by Tauc, Davis, and Mott, commonly known as the Tauc plot. (ahv)1/n = C(hv – Eg),22,23 where a is the absorbance index, h is the Planck constant, v is the frequency, C is the constant, and Eg is the semiconductor band gap width. The result showed that the title complex is a wide-band-gap semiconductor (Figure 4). The energy band gap of 2.91 eV is greater than those of reported photovoltaic materials with high energy bands.24,25 Therefore, we can believe that the title complex may be a candidate material for wide-band-gap semiconductors.
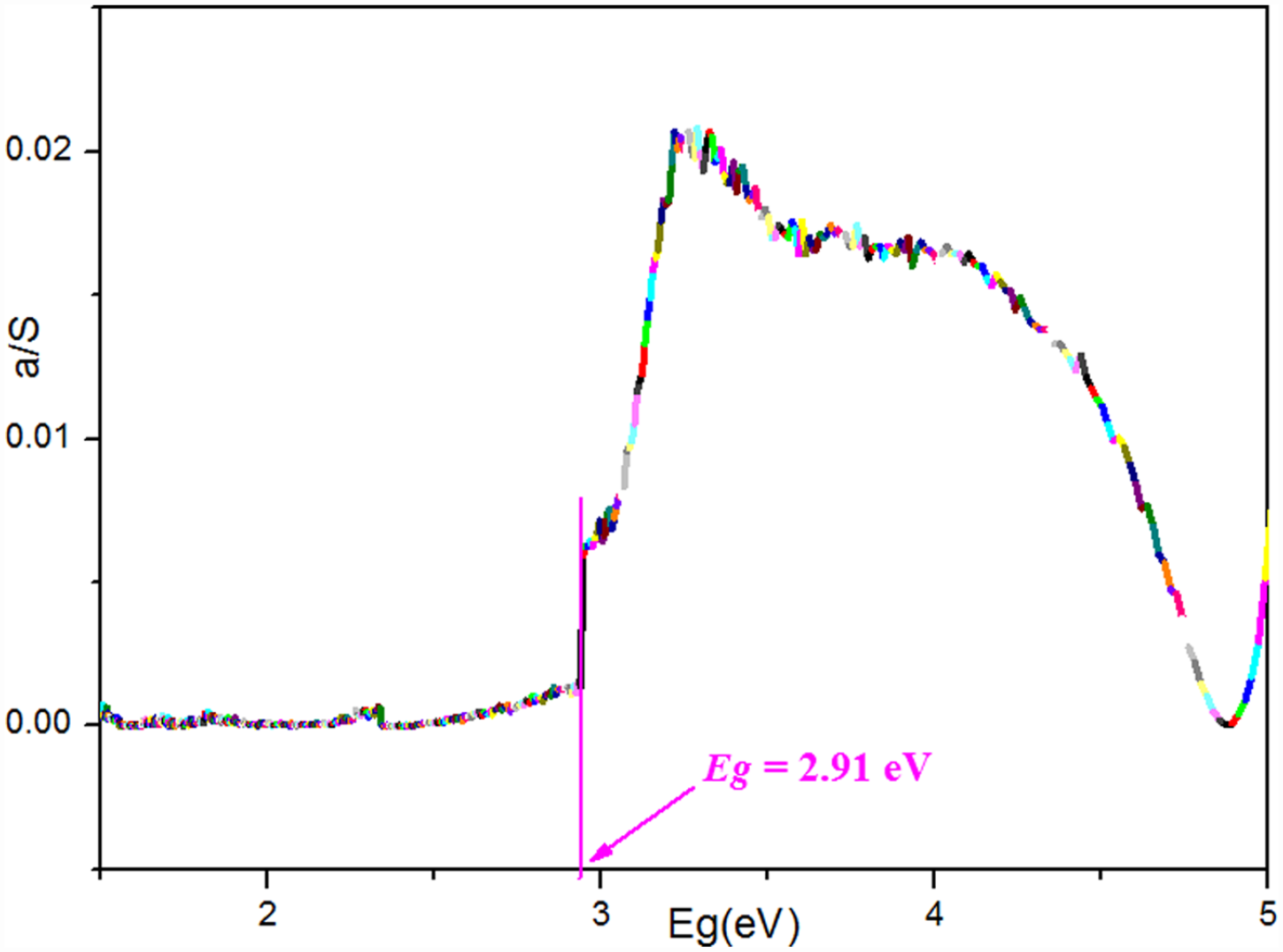
The solid-state UV-Vis DRS of the title complex.
Solid-state photoluminescence
The photoluminescence of coordination complexes has received more and more attention. Based on this, we have measured the photoluminescence of solid powder samples of the ligand and the title complex at room temperature and the results are shown in Figure 5, Figure 5(a) shows the solid-state fluorescence spectrum of the ligand, and Figure 5(b) shows the solid-state fluorescence spectrum of the title complex. It is evident that the photoluminescent spectrum in Figure 5(b) displays an effective energy absorption in a wavelength range of 700–900 nm. Upon excitation at 791 nm, the emission spectrum was characterized by a sharp band at 527 nm in the blue region. As a result, the title complex is a typical up-conversion blue-light-emitting material. The emission band located in the blue region with a CIE1931 chromaticity coordinate of (0.2068, 0.3072), as shown in Figure 6(b). The quantum yield of the title complex was 0.3%. In order to reveal the nature of the photoluminescence properties of the title complex, the solid-state photoluminescence spectrum of the ligand was recorded Figure 5(a) and Figure 6(a). Upon excitation at 564 nm, the emission spectrum of the ligand was characterized by a sharp band at 853 nm. The photoluminescence spectra for the title complex and the ligand are very similar. Hence, the photoluminescence of the title complex obviously originates from the ligands.

The solid-state photoluminescence spectra of the ligand and the title complex: (a) red curve: excitation; black curve: emission and (b) red curve: emission; blue curve: excitation.

The CIE chromaticity diagrams and chromaticity coordinates of the emission spectrum of the ligand (a) and title complex (b).
Theoretical calculations
In order to reveal the nature of the photoluminescence emission of the title complex, time-dependent density functional theory (TDDFT) calculations were performed at the B3LYP/6-31G* level using the Gaussian09 program. 26 The single-crystal X-ray diffraction data set of the title complex was used to truncate the ground-state geometry for the calculations; the X-ray diffraction data of the title complex was not been optimized further. The electron-density distributions of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of the title complex are shown in Figure 7. Clearly, it can be seen that the electron-density distribution of the HOMO resides completing on the p-orbital of the ligand HL(5-(Bis-carboxymethyl-amino)-isophthalic acid) with an energy of −3.051 Hartrees. However, the electron-density population of the LUMO is calculated to be −2.760 Hartrees. In light of these observations, the energy difference (0.291 Hartrees) between the HOMO and LUMO is small enough to allow for charge transfer between the HOMO and the LUMO. Thus, it is proposed that the essence of the photoluminescence of the title complex could be attributed to ligand-to-ligand charge transfer (LLCT)(from the HOMO of the p-orbital of ligand HL to the LUMO of the oxygen atoms).

B3LPY predicted frontier molecular orbitals of the title complex.
Conclusions
In summary, a novel up-conversion tetranuclear sodium luminescent complex has been prepared and structurally determined by X-ray diffraction. Its crystal structure is characterized by a 3D supramolecular network including hydrogen bonding and van der Waals attractions. Solid-state photoluminescence measurements reveal that the title complex exhibits blue up-conversion emission with the emission band located at 527 nm, and displays a remarkable CIE chromaticity coordinates (0.2068, 0.3072). TDDFT calculations reveal that the emission can be attributed to ligand-to-ligand charge transfer (LLCT). Solid-state DRS measurements reveal that the title complex is a wide band gap semiconductor material with a band gap of 2.91 eV.
Experimental
Materials and instrumentation
All reagents and chemicals were of reagent grade, commercially available and were directly applied for the reaction. Infrared spectra were obtained with a PE Spectrum-One FT-IR spectrometer as KBr disks. NMR of the ligand was performed on a Bruker Avance 400 MHz spectrometer with DMSO-d6 as the solvent. Solid-state UV/Vis diffuse reflectance spectroscopy was performed on a computer-controlled TU1901 UV/Vis spectrometer. A finely ground powder sample was coated on barium sulfate for 100% reflectance. Photoluminescence characterization was performed on a F97XP photoluminescence spectrometer. TDDFT investigations were performed using the Gaussian09 suite of program package.
Synthesis of the ligand HL (5-(bis-carboxymethyl-amino)-isophthalic acid)
5-Amino-isophthalic acid (1 mmol, 181 mg), chloroacetic acid (3 mmol, 283.5 mg), NaOH (2 mmol, 80 mg), and distilled water were added to a 500 mL three-neck round-bottomed flask and the resulting mixture stirred. During the reaction, 10% NaOH solution was continuously added to keep the reaction at pH = 8 for 3 h. After the reaction was complete, the pH was adjusted to 4 with dilute hydrochloric acid to afford a light yellow solid. The obtained solid was filtered, washed, and dried. Yield: 282.15 mg (95%). IR (KBr, cm−1): 3445 (vs), 1699 (s), 1603 (vs), 1471 (m), 1241 (m), 1199 (m), 1091 (m), 995 (m), 758 (m), 676 (m). 1H NMR (400 MHz, DMSO-d6): δ: 7.82 (s, 1H), 7.24 (s, 2H), 4.05 (s, 4H). 13C NMR (101 MHz, DMSO-d6): δ: 174.30, 168.15, 147.48, 133.53, 118.60, 115.20, 60.68, and 57.48.
Synthesis of the title complex
5-(Bis-carboxymethyl-amino)-isophthalic acid (0.1 mmol), NaOH (0.5 mmol) and distilled water (20 mL) were mixed in a 25 mL Teflon-lined stainless-steel autoclave. The mixture was heated to 383 K and kept at this temperature for 1 week. After cooling the mixture slowly to room temperature, yellowish block crystals suitable for X-ray analysis were collected, washed and dried. Yield: 80% (based on sodium). IR (KBr, cm−1): 3418 (vs), 1606 (vs), 1565 (vs), 1451 (vs), 1424 (vs), 1399 (vs), 1368 (vs), 1312 (s), 1255 (s), 1184 (s), 998 (m), 801 (m), 707 (m), 644 (m), and 570 (m).
X-ray crystallographic determination
Single crystals of C12H25NNa4O17 were synthesized by the hydrothermal method. A suitable crystal was selected and collected on a SuperNova, (single source at offset/far) EosS2 diffractometer. The crystal was kept at 273 K during data collection. Using Olex2,
27
the structure was solved with the SHELXT
28
structure solution program using direct methods and refined with the SHELXL
29
refinement package using least squares minimization. All of the non-hydrogen atoms were generated based on the Fourier difference maps and were refined anisotropically. The hydrogen atoms, except for the lattice water (lattice water produces too many disordered states due to hydrogenation), were located theoretically and ride on their parent atoms. The refinement results displayed the title complex: triclinic, space group: P
Crystallographic data and structure analysis of the title complex.

Selected bond lengths (Å) and bond angles (°) for the title complex.

Symmetry operators: 1 = 2–x, 1–y, 2–z; 2 = 1–x, 2–y, –z; 3 = 1+x, +y, +z; 4 = –x, 2–y, –z; 5 = –1+x, +y, +z; 6 = –1–x, 1–y, –z; 7 = –1+x, +y, –1+z; 8 = –x, 1–y, 1–z.
Hydrogen bond lengths (Å) and bond angles (°) for the title complex.

Symmetry operators: 1 = 1–x, 1–y, 1–z; 2 = 1+x, +y, 1+z; 3 = 1–x, 1–y, 2–z; 4 = +x, 1+y, +z; 5 = 1–x, 2–y, 1–z; 6 = –x, 2–y, 1–z; 7 = –1+x, +y, +z; 8 = –x, 2–y, –z.
Supplemental Material
sj-doc-2-chl-10.1177_17475198231177955 – Supplemental material for Preparation, crystal structure, time-dependent density theory and properties of a novel organic-inorganic hybrid up-conversion luminescence tetranuclear complex
Supplemental material, sj-doc-2-chl-10.1177_17475198231177955 for Preparation, crystal structure, time-dependent density theory and properties of a novel organic-inorganic hybrid up-conversion luminescence tetranuclear complex by Wen-Quan Zeng, Wei Huang, Chuan-Hui He, Rong Zou, Wei-Kang Jin, Xiu-Guang Yi and Kun-Zhong Yang in Journal of Chemical Research
Supplemental Material
sj-pdf-1-chl-10.1177_17475198231177955 – Supplemental material for Preparation, crystal structure, time-dependent density theory and properties of a novel organic-inorganic hybrid up-conversion luminescence tetranuclear complex
Supplemental material, sj-pdf-1-chl-10.1177_17475198231177955 for Preparation, crystal structure, time-dependent density theory and properties of a novel organic-inorganic hybrid up-conversion luminescence tetranuclear complex by Wen-Quan Zeng, Wei Huang, Chuan-Hui He, Rong Zou, Wei-Kang Jin, Xiu-Guang Yi and Kun-Zhong Yang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the National Natural Science Foundation of China (grant no. 22168018), the Higher Education and Teaching Reform of Jiangxi Education Committee of China (grant no. JXJG-22-9-23), the Science and Technology Plan Project of Ji’an (grant no. 20211-025317), and the Doctoral Research Startup Foundation of Jinggangshan University (grant no. JZB1905) for supporting this work.
Data availability
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
