Abstract
One organic compound [HMCA]2 (
One organic compound [HMCA]2 (
Introduction
In recent years, in the field of chemistry and materials, inorganic–organic hybrid materials have attracted increasing attention from chemists and material scientists, due to their potential applications in semiconductors, 1 optics, 2 magnetism, 3 solar cells, 4 medical materials, 5 and so on.
Quinoline compounds are widely used in many fields such as in the coordination of metal ions, 6 for selective recognition of anions, 7 for printing and dyeing auxiliaries, 8 as antimalarials, 9 and so on, because of their unique conjugated structure, good spectral properties, high thermal stability, and excellent biological activity. Quinoline is a type of nitrogen-containing heterocyclic naphthalene compound, which can react with many raw materials to form quinoline derivatives. Quinoline derivatives are heterocyclic compounds that are widely used as antibacterial, bactericidal, anti-allergic, antidepressant, antihypertensive, antitumor, and antimalarial agents.10–13 Quinoline carboxylic acid derivatives are multidentate organic ligands containing nitrogen atoms and carboxyl oxygen atoms which can coordinate with metal ions. The coordination modes of the carboxyl groups vary at different pH values, resulting in more coordination modes. The coordination mode can exhibit various structures and unique properties.
Based on the interest in quinoline carboxylic acids, a series of transition and lanthanide metal complexes with 3-hydroxy-2-methylquinoline-4-carboxylate acid (HMCA) as the main ligand were synthesized and reported by Yi’s group.14–19 In this work, all of the complexes are mononuclear complexes with monodentate or bidentate chelated coordination. In continuation of our work, we report here the solvothermal synthesis, X-ray crystal structure, solid-state photoluminescence, solid-state UV-Vis diffuse reflectance and magnetic properties, as well as the time-dependent density functional theory (TDDFT) calculations for three complexes, namely, [HMCA]2 (
Results and discussion
Synthesis and crystal structures
The synthesis of HMCA is based on relevant references.
20
Complexes
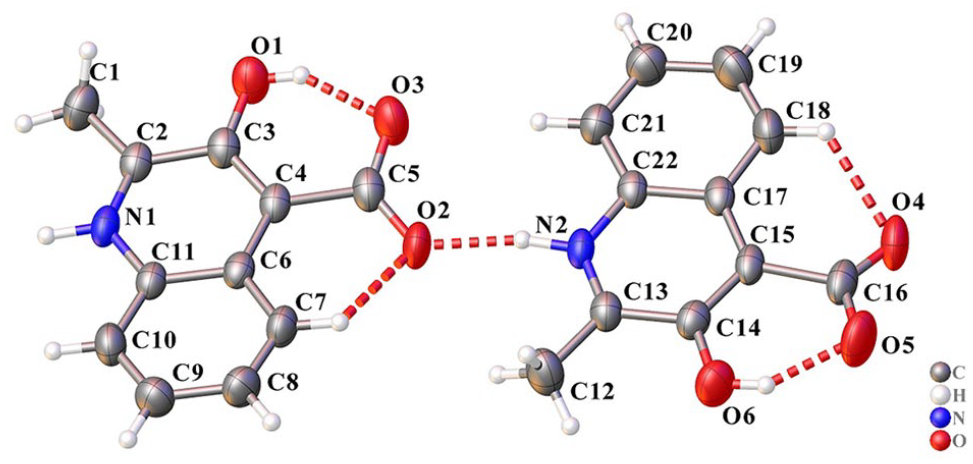
The molecular structure of the HMCA ligand.

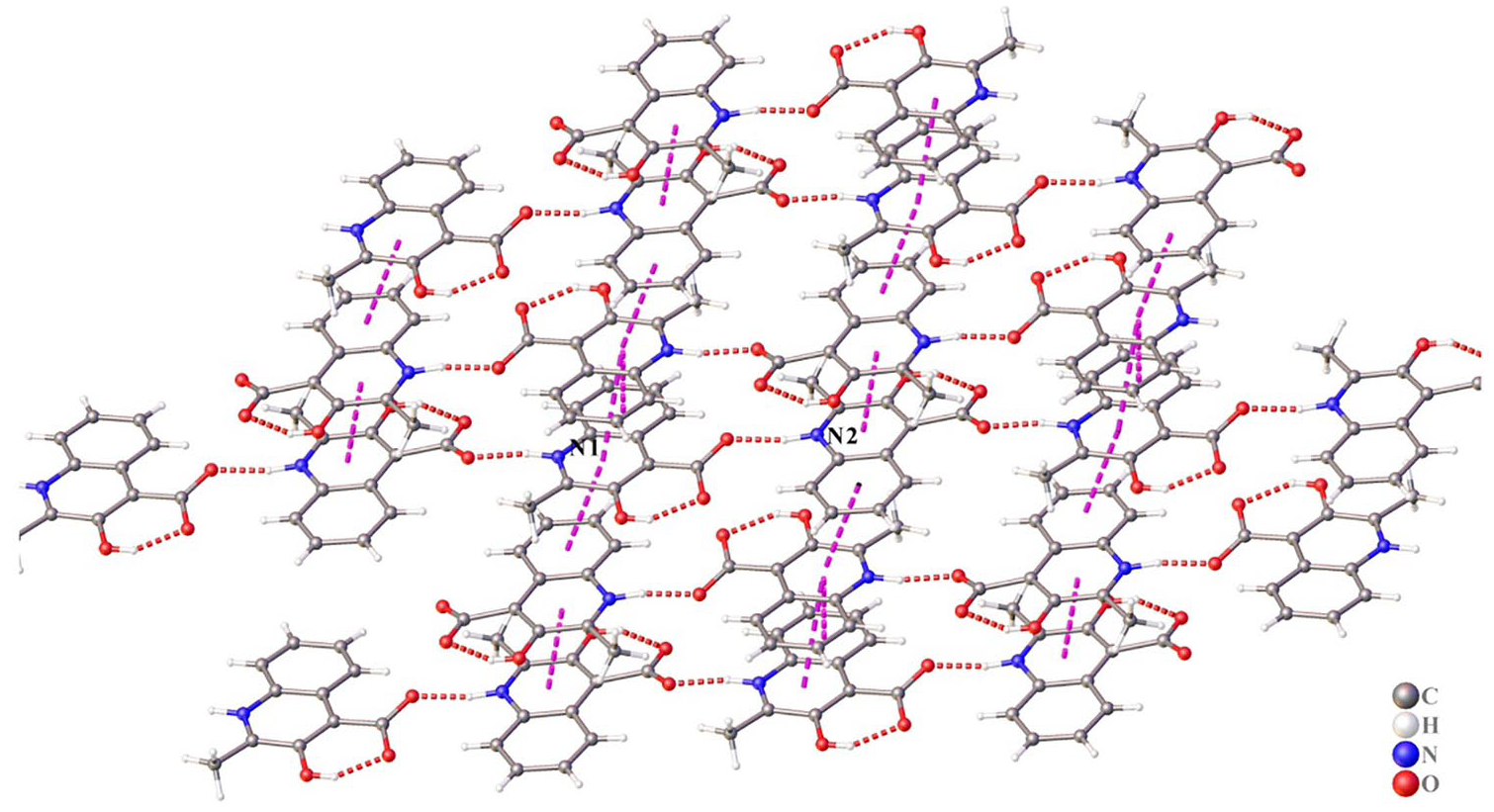
One-dimensional structure of the hydrogen bonds in the HMCA ligand.

Two-dimensional structure produced by hydrogen bonds and π. . .π stacking interactions in the HMCA ligand.
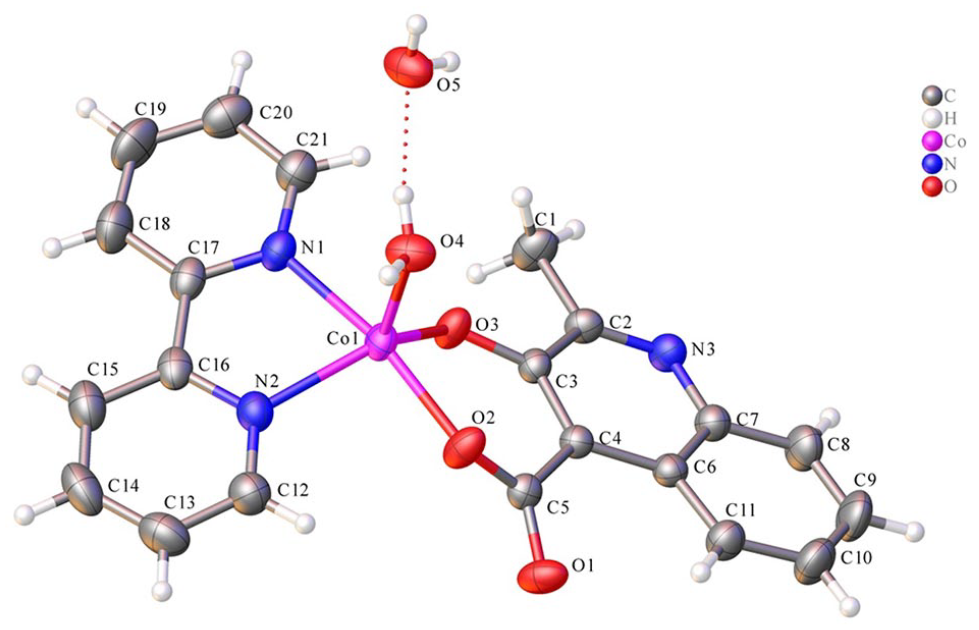
The molecular structure of the complex [Co(MCA)(bipy)(H2O)]·(H2O)
The hydrogen bonds present in [Co(MCA)(bipy)(H2O)]·(H2O)

The π. . .π stacking interactions in [Co(MCA)(bipy)(H2O)]·(H2O)
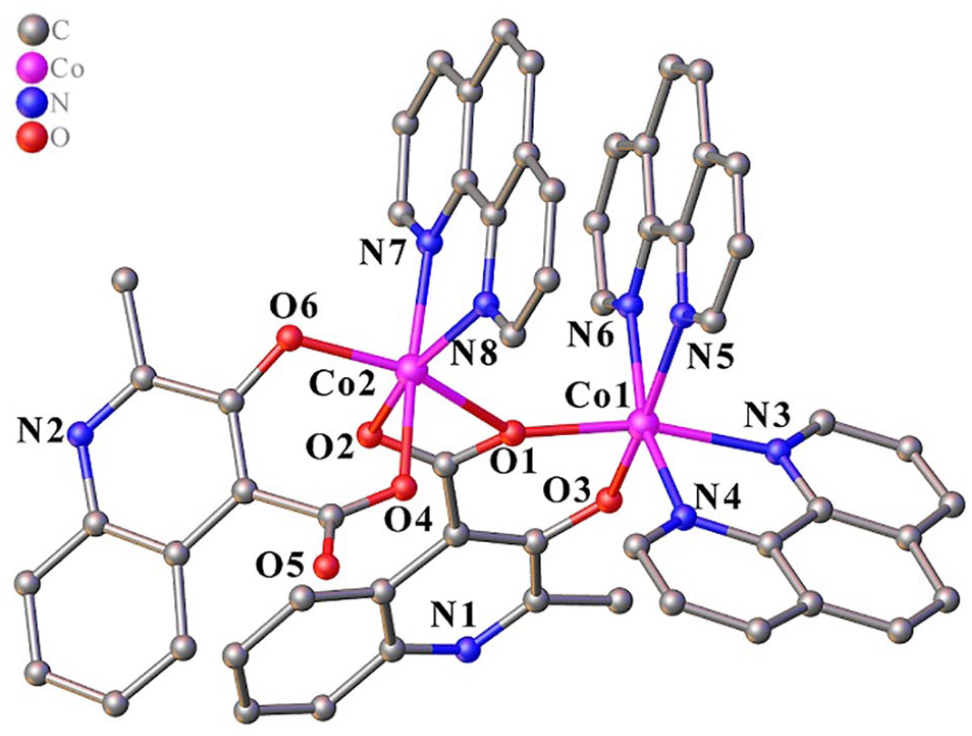
The molecular structure of the complex [Co2(MCA)2(Phen)3]·3(H2O)

The hydrogen bonds and C–H. . .Cg representation of complex

The π. . .π stacking interaction diagram of complex

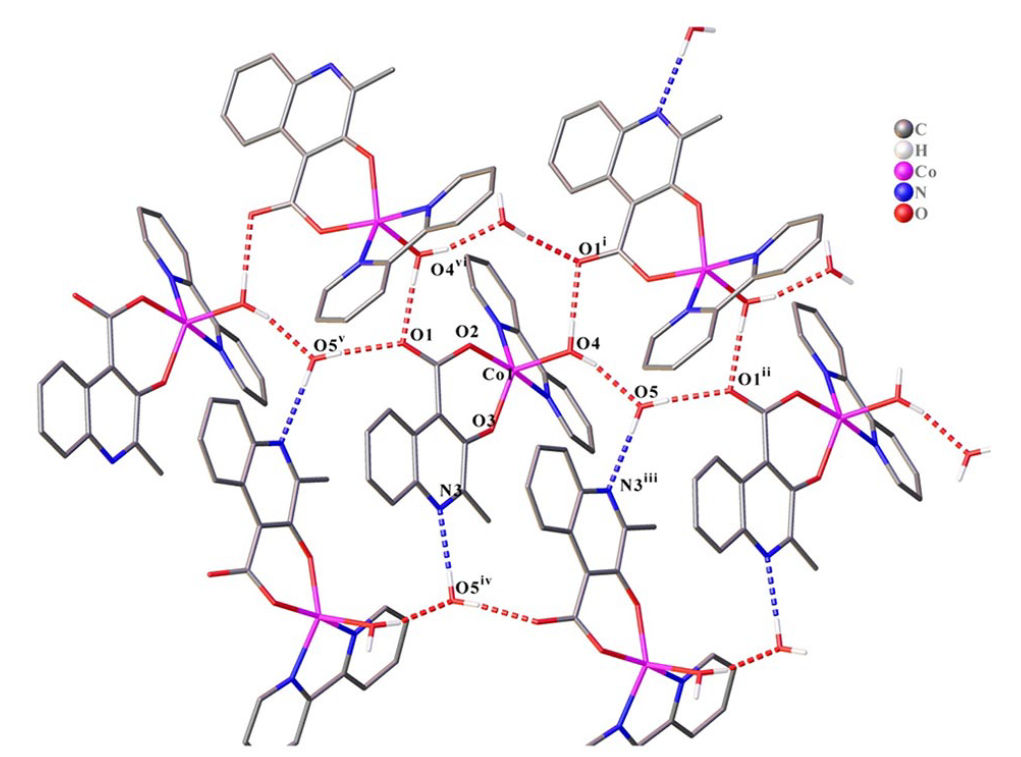
The packing diagram of complex
Photoluminescence
In recent years, increasing attention has been paid to the photoluminescence of coordination complexes. Coordination complexes containing transition elements and lanthanides have abundant 3/4d and 4f orbital electronic configurations because of their luminescent properties. The luminescent properties of the transition state complexes and lanthanide complexes have been studied. Some studies have shown that the complexes containing Co(II) ions have interesting photoluminescence properties. Based on the above considerations, we conducted photoluminescence spectral analysis on solid samples of

The solid-state excitation and emission spectra of the Co(II) complexes

CIE chromaticity diagrams and chromaticity coordinates of the emission spectra of the Co(II) complexes
TDDFT calculations
Based on the B3LYP function with basis set SDD for Co and 6-31-G* for C, H, O, and N elements to reveal the fluorescence innate character of the title Co (II) complexes, we calculated them with the Gaussian09 program
27
using the TDDFT. The ground state geometry was truncated from them single-crystal X-ray data without optimization. The characteristics of the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) of the complexes

HOMO and LUMO of the title complexes
Solid-state UV-Vis diffuse reflectance spectroscopy
Diffuse reflectance spectroscopy (DRS) can be helpful to understand the conductance properties of materials; therefore, a study was carried out with the title Co(II) complexes. The solid-state UV-Vis diffuse reflectance spectra of power samples of the title Co(II) complexes

(a) The solid-state UV-Vis diffuse reflectance spectrum of the complex
Conclusion
In conclusion, we have reported the preparation of two novel cobalt complexes with HMCA, bipy, and Phen as mixed ligands under the solvothermal conditions and which have been structurally characterized by single-crystal X-ray diffraction. The complexes exhibit 2D, 3D and 1D chain-like structures, respectively. Solid-state photoluminescence reveals that the complexes
Experimental section
Measurements
The infrared spectra were recorded using a Nicolet iS10 spectrometer as KBr pellets. 1 H NMR spectra were measured on a Bruker Avance 400 MHz instrument with DMSO-d6 as the solvent. The photoluminescence of solid-state samples was investigated using an FX-97XP fluorescence spectrometer. The solid-state UV-Vis DRS was recorded using a TU-1901 UV-Vis spectrometer with an integrating sphere in the wavelength range of 200–900 nm. TDDFT investigations were carried out using the Gaussian 09 suite of program packages.
Syntheses
Commercially available reagents and chemicals that were used to synthesize the title compound are analytical reagent grade.
HMCA (1) Ligand
The synthesis of HMCA is based on relevant references Boudet et al. 20 as shown in Scheme 1.

Synthetic route of ligand HMCA (1).
Isatin: Yield: 116 g (90%); m.p.: 210 °C; HRMS m/z (ESI): calcd. for C8H5NO2 ([M + H]+):147.0320, found: 147.0826. IR (KBr, cm1): 3192 (vs), 1729 (vs), 1617 (vs), 1461 (s), 1332 (s), 946 (m), 770 (m), 661 (m).
HMCA (1): Yield: 96 g (95%); m.p.: 225 °C; HRMS m/z (ESI): calcd. for C11H9NO3 ([M + H]+): 203.0582, found: 203.0548. 1H NMR (400 MHz, DMSO): δ 9.15 (s, 1H), 7.93 (d, J = 8.0 Hz, 1H), 7.64 (t, J = 8.0 Hz, 1H), 7.60–7.52 (m, 2H), 2.70 (s, 3H). IR (KBr, cm1): 3391 (s), 2521 (s), 2045 (m), 1552 (m), 1462 (s), 1409 (vs), 1339 (s), 1286 (s), 1241 (vs), 1161 (m), 906 (m), 861 (vs), 766 (m), 687 (m).
X-ray crystallographic studies
The diffraction data were collected on a SuperNova CCD X-ray diffractometer using carefully selected single crystals of complexes
Crystal parameters of complexes

Selected bond lengths (Å) and bond angles (°) for the complexes

Hydrogen bond lengths (Å) and bond angles (°) for the complexes

Supplemental Material
SEI – Supplemental material for Preparation, crystal structures, properties, and time-dependent density functional theory of two cobalt complexes with 3-hydroxy-2-methyl-quinoline-4-carboxylate
Supplemental material, SEI for Preparation, crystal structures, properties, and time-dependent density functional theory of two cobalt complexes with 3-hydroxy-2-methyl-quinoline-4-carboxylate by Xiu-Guang Yi, Fei-Ping Lai, Yun-Yi Yan, Cong Zhang and Wen-Ping Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NSF of China (51363009), Jiangxi Provincial Department of Educations’s Item of Science and Technology (GJJ190550), and Doctoral Research Startup Foundation of Jinggangshan University (JZB1905).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
