Abstract
The complex [Zn(CH3OH)4(MCA)2] (3-hydroxy-2-methylquinoline-4-carboxylic acid) has been synthesized through hydrothermal reactions and characterized by single-crystal X-ray diffraction. Its solid-state photoluminescence spectrum shows an emission in the blue region, which is assigned to ligand-to-metal charge transfer on the basis of time-dependent density functional theory calculations. Solid-state diffuse reflectance measurements show the existence of a narrow optical band gap of 1.73 eV.
Keywords
A novel zinc(II) complex shows blue violet photoluminescence emission, which can be attributed to ligand-to-ligand charge transfer (LMCT), as revealed by TDDFT calculations. Solid-state diffuse reflectance measurement shows the existence of a narrow optical band gap of 1.73 eV.
Introduction
In recent years, more and more coordination compounds possessing group-12 elements (Zn, Cd, and Hg) have been synthesized, because such compounds may possess attractive physicochemical properties and useful applications in many fields such as photoluminescent materials, photoelectric materials, nonlinear optics materials, and semiconductors, as well as their relevance to the important role played in biological systems by zinc.1–4
Quinoline carboxylic acids, as O-donor ligands, are very interesting units in the construction of inorganic–organic hybrid compounds with extended structures because of their carboxyl oxygen atoms and hydroxyl oxygen atoms, which bridge metal ions.5–8 Up to now, there are few reports on the complexes of quinoline carboxylic acids.
Based on this, we are interested in the crystal engineering of group-12 element-containing compounds with 3-hydroxy-2-methylquinoline-4-carboxylic acid (HMCA) as a ligand. We report the solvothermal synthesis, X-ray crystal structure, photoluminescent, and semiconductor properties, as well as time-dependent density functional theory (TDDFT) calculations for a novel zinc complex [Zn(CH3OH)4(MCA)2] (
Experimental
General procedure
The reagents and chemicals for the synthesis of the title compound were analytical reagent grade, commercially available and applied without further purification. Infrared spectra were obtained with a PE Spectrum-One Fourier-transform infrared (FT-IR) spectrometer using KBr disks. Elemental microanalyses of carbon, hydrogen, and nitrogen were performed on an Elementar Vario EL elemental analyzer. 1H NMR spectra were measured on Bruker Avance 400 MHz instrument with dimethyl sulfoxide (DMSO) as solvent. Photoluminescence measurements were performed on a F97XP photoluminescence spectrometer. Solid-state ultraviolet (UV)-vis reflectance spectroscopy was carried out with a TU1901 UV-Vis spectrometer equipped with an integrating sphere. A BaSO4 plate acted as a 100% reflectance reference, on which the finely ground powder was coated. TDDFT investigations were carried out by means of the Gaussian 03 suite of program packages.
Preparation of HMCA
HMCA was prepared according to the following reaction route (Scheme 1).

Reaction route.
Synthesis of isatin
Indigo (262 g) and K2Cr2O7 (147 g) were added into H2O (500 mL) and stirred. After cooling, K2Cr2O7 (147 g), H2O (300 mL) and dil. H2SO4 (500 mL) were added and kept stirring at 40°C and 45°C for 1.5 h. Then, the mixture was diluted with twice its volume of H2O, filtered off, dissolved with a KOH solution, filtered again, acidified by HCl and refiltered. Yield, 230 g (⩾90%); m.p. 210°C; high-resolution mass spectrometry (HRMS) m/z electrospray ionization (ESI) calcd for C8H5NO2 ([M+H]+)147.0320, found 147.0826.
Synthesis of HMCA
Isatin (1.0 mol) and KOH (1.0 mol) were dissolved into a sufficient amount of water and filtered. The filtrate and 1.0 mol KOH were added into chloroacetone (2.0 mol), and hydrochloric acid was added dropwise to adjust to pH = 7, and the solution was then filtered. Yield ⩾95% and m.p. 225°C. IR peaks (KBr, cm−1): 3433(vs), 3125(w), 3043(w), 2869(w), 2499 (m), 2040(m), 1621(m), 1553 (s), 1500 (m), 1462(m), 1410(m), 1242(vs), 1160(m), 1014(w), 906(m), and 686(s); HRMS m/z (ESI) calcd for C11H9NO3 ([M+H]+) 203.0582, found 203.0548; 1H NMR (400 MHz, DMSO) δ 9.15 (s, 1H), 7.93 (d, J = 8.2 Hz 1H), 7.64 (t, J = 7.7 Hz, 1H), 7.60–7.52 (m, 3H), 2.70 (s, 3H).
The melting points of isatin and HMCA are in reasonable agreement with those reported in the literature. 9
Preparation of complex 1
HMCA (1 mmol, 203 mg), (CH3COO)2Zn·6H2O (0.5 mmol, 109 mg), methanol (9.5 mL) and distilled water (0.5 mL) were mixed in a 25-mL Teflon-lined stainless steel autoclave. The autoclave was heated to 200°C in an oven and kept there for 3 days and then allowed to cool. When the autoclave reached room temperature, yellow block crystals were obtained and used to collect the single-crystal X-ray data. Yield 60% based on (CH3COO)2Zn·6H2O. IR peaks (KBr, cm−1): 3434(s), 3338(vs), 3122(w), 2962(w), 2740(w), 2500 (s), 2018(w), 1656(m), 1608(s), 1580 (s), 1524(s), 1434(m), 1408(m), 1242(vs), 1158(m), 1019(s), 912(s), 870(s), 812(w), and 764(s); Anal. Calcd for C26H32N2O10Zn: C, 52.23; H, 5.39; N, 4.69; found: C, 52.32; H, 5.31; N, 4.68%.
X-ray structural determination
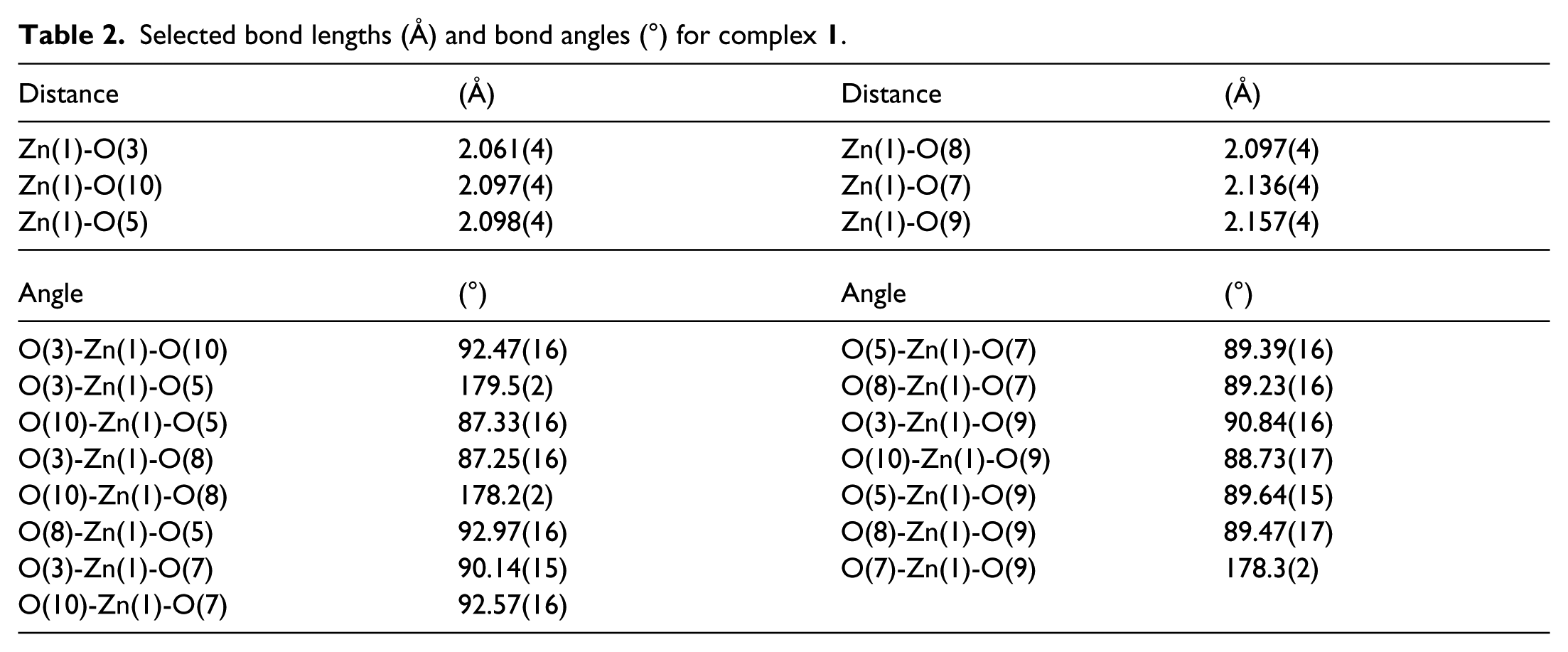
The X-ray diffraction data were collected on a SuperNova charge-coupled device (CCD) X-ray diffractometer with a carefully selected single crystal (dimensions of 0.17 × 0.08 × 0.05 mm). The X-ray source was graphite monochromated Mo-Kα radiation with λ = 0.71073 Å. The reduction and empirical absorption correction of the diffraction data were carried out with the CrystalClear software. The crystal structure is solved by direct methods and the Siemens SHELXTLTM V5 software 10 and refined with a full-matrix least-squares refinement on F2. All non-hydrogen atoms were located on the difference Fourier maps with applied anisotropic refinement. The hydrogen atoms were theoretically attached to their parent atoms and included in the structural factor calculations with assigned isotropic thermal parameters. 11 Reflections measured were 7434; the final R = 0.0354 for 353 parameters and 3972 observed reflections with I > 2σ(I) and wR = 0.0909 (w = 1/(σ2(Fo 2 ) + (0.0536 P) 2 + 0.0977 P), where P = (Fo2 + 2Fc2)/3); S = 1.115, (Δρ)max = 0.448, (Δρ)min = –0.542e Å−3 and (Δ/σ)max = 0. The selected bond distances and angles are shown in Table 1. Selected bond lengths and bond angles are presented in Table 2.
Structure determination summary for complex

Selected bond lengths (Å) and bond angles (°) for complex
Results and discussion
X-ray single-crystal diffraction analysis reveals that complex

An ORTEP drawing of compound

Packing diagram of compound
Photoluminescence measurements
To our knowledge, zinc and HMCA complexes can generally exhibit photoluminescence emission bands. Based on this consideration, and in order to reveal its potential photoluminescent properties, we carried out photoluminescence measurements on complex

Solid-state photoluminescence spectra of
Theoretical calculations
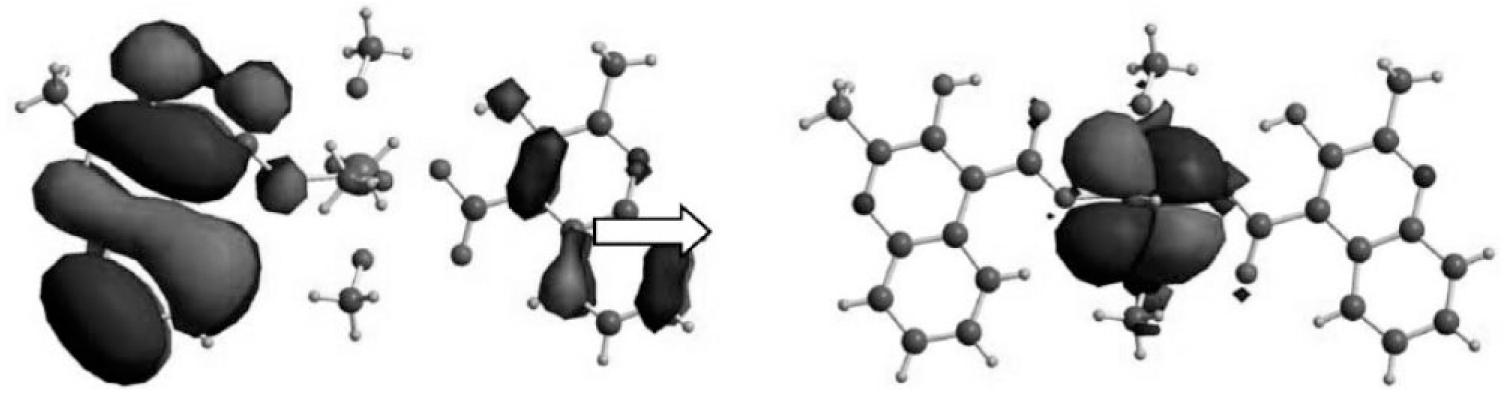
In order to reveal the nature of the photoluminescence emission of complex 1, TDDFT calculations were performed using the B3LYP function (Figure 4). The single-crystal X-ray diffraction data set of complex 1 was used to truncate the ground-state geometry for the calculation. Based on the geometry, a TDDFT study was carried out by means of the Gaussian 03 suite of programs, and the results reveal the characteristics of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of complex 1. Clearly, it can be seen that the electron density distribution of the singlet state of HOMO is located totally on the ligand moieties. However, the electron density population of the singlet state of the LUMO resides completely on the zinc moiety. In light of this observation, the essence of the photoluminescence emission of complex 1 can be attributed to ligand-to-metal charge transfer (LMCT) from the HOMO of the HMCA to the LUMO + 1 of the Zn.
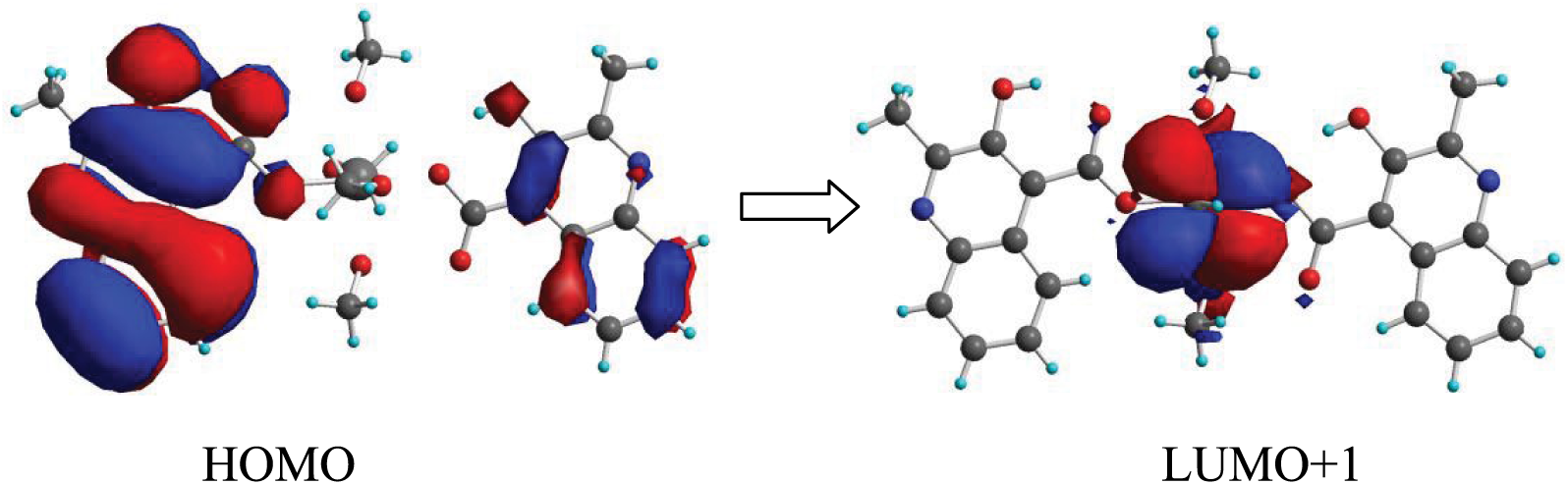
The electron density population of
Reflectance measurements
Solid-state UV-Vis diffuse reflectance spectra of complex
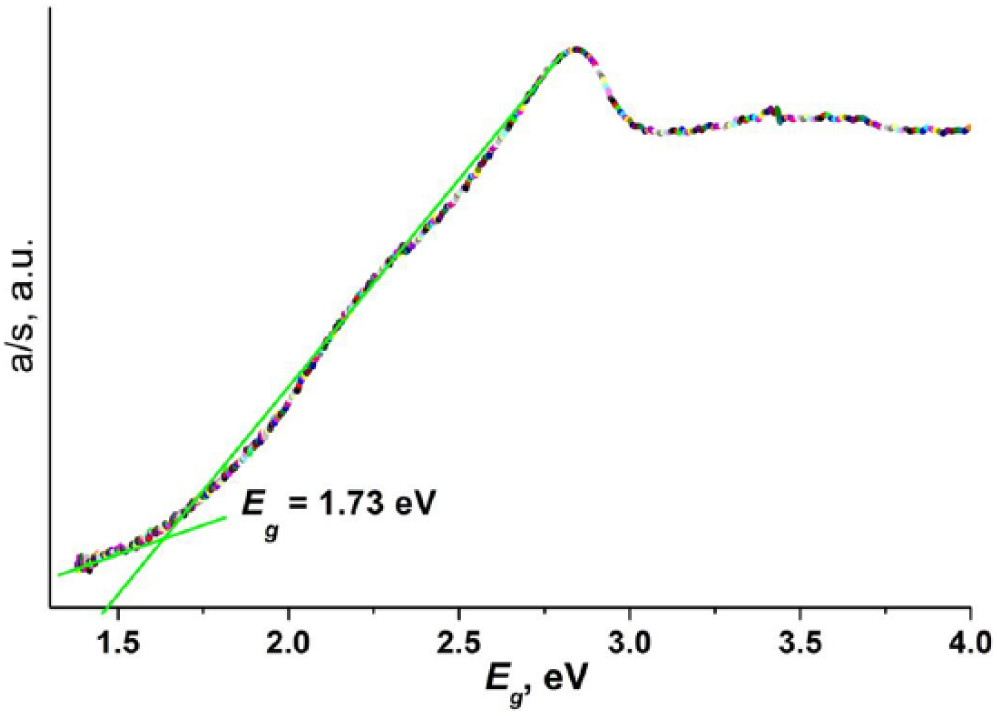
Solid-state diffuse reflectance spectrum of
Conclusion
In summary, a novel zinc complex [Zn(CH3OH)4(MCA)2] (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study received financial support from the National Natural Science Foundation of China (51363009, 21764007), Jiangxi Provincial Department of Education’s Item of Science and Technology (GJJ160745), the research topics of teaching reform in higher education institutions in Jiangxi (JXJG-17-9-14), the Science and Technology Plan project Fund of Jiangxi Provincial Health Planning Commission(20194083), and Natural Science Foundation Project of Jinggangshan University(JZ09029).
Supplemental material
Crystallographic data for the structural analysis have been deposited with the Cambridge Crystallographic Data Center, CCDC No. 1874445. Copies of this information may be obtained free of charge from the Director, CCDC, 12 Union Road, Cambridge, CBZ 1EZ, UK (fax: +44-1223-336033; e-mail: ![]() ).
).
