Abstract
In order to investigate the changes in the spectroscopic properties of dimeric boron-dipyrromethenes (BODIPYs), four BODIPY derivatives are synthesized, including a monomer BODIPY in which a furyl group is substituted at the meso position and a dimer BODIPY with a furan group as a bridge. The four synthesized BODIPY derivatives are characterized through nuclear magnetic resonance and mass spectrometry. Photophysical properties such as ultraviolet–visible absorbance and the fluorescence emission of monomers (

Introduction
Boron-dipyrromethene (BODIPY) derivatives were first reported by Kreuzer in the late 1960s as tricyclic complexes in which two dipyrrole units are connected by a methine bridge and centered on a boron atom. 1 BODIPY has attracted great interest from the scientific community for decades because of its high fluorescence quantum efficiency, narrow emission bandwidth, and excellent thermal and photochemical stability.2–5 Several types of BODIPYs have been used as fluorescent probes for cell imaging and for the investigation of biological processes in vivo.6–8 BODIPYs are widely used to label biologically essential molecules such as proteins, nucleotides and fatty acids, or to label intracellular organelles such as mitochondria and receptors.9–14 Recently, BODIPYs have emerged as promising candidates in various fields such as electrochemical luminescence (ECL), organic solar cells, photodynamic therapy (PDT), logic gates, fluorescence thermometers, gas probes, and organic transistors.15–22
Dimeric chromophores have often been found in Nature, such as in photosynthetic bacterial proteins and in natural light harvesting systems. 23 Such covalent pairs of chromophores in Nature have a long-standing interest in the scientific community. Since the BODIPY dimer was first reported by Akkaya, various kinds of derivatives have been synthesized and studied.24–26 In particular, the photophysical properties of BODIPY can be fine-tuned for various applications through structural modification of the boron-dipyrine core and oligomerization including dimers. β–β-linked dimers, meso–meso-linked dimers and fused aromatic-linked dimers have been recently synthesized.27–34 These BODIPY dimers show increased Stokes shifts and red-shifted luminescence. They can be used in organic solar cells or in biological applications, and have attracted significant attention as singlet oxygen photosensitizers, near infrared (NIR) fluorescence probes and electrochemiluminescence (ECL) sensors.35–37 Although reports on covalently bonded BODIPY dimers are increasing, there is still a need to study the photophysical properties of heterocyclic bridged BODIPY dimers.38–41
There have been previous reports of BODIPY dimers with furan bridges, such as

Synthetic routes toward (a)

Molecular structures of the furan-bridged BODIPY derivatives studied in this work.
Results and discussion
The synthesis of the furan-bridged BODIPY derivatives is depicted in Scheme 1. Furfural was condensed with pyrrole in the presence of a catalytic amount of trifluoroacetic acid to form dipyrranes
The BODIPY derivatives
The ultraviolet (UV)–visible (UV-Vis) absorption and fluorescence spectra of BODIPY derivatives

Normalized (a) absorption and (b) emission spectra of the BODIPYs
Photophysical properties of
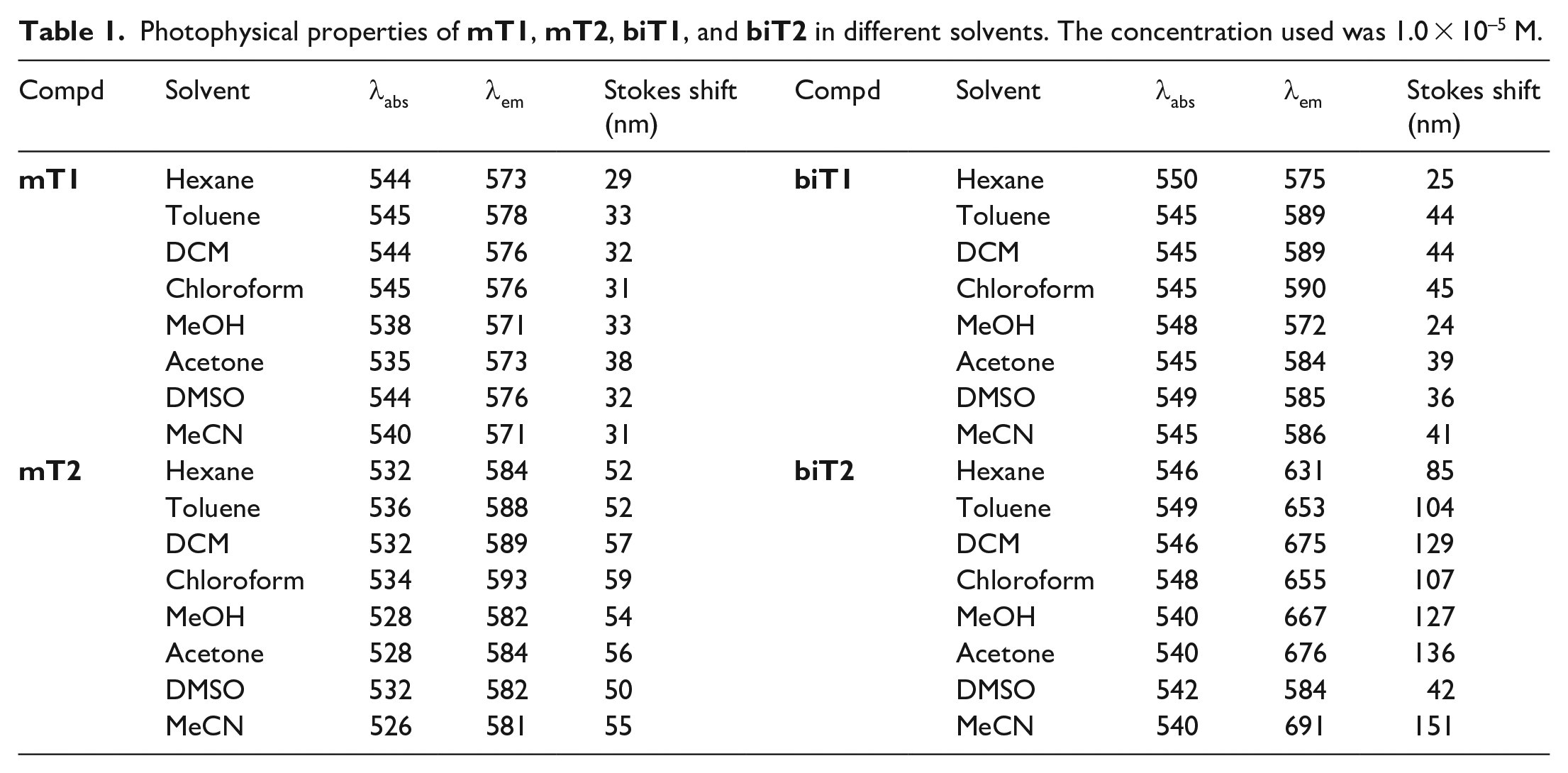
The wavelength of the main absorption band of
A comparison of the emission spectra of the BODIPY derivatives is shown in Figure 2(b), and the solvatochromism data is summarized in Table 1. The emission maxima of
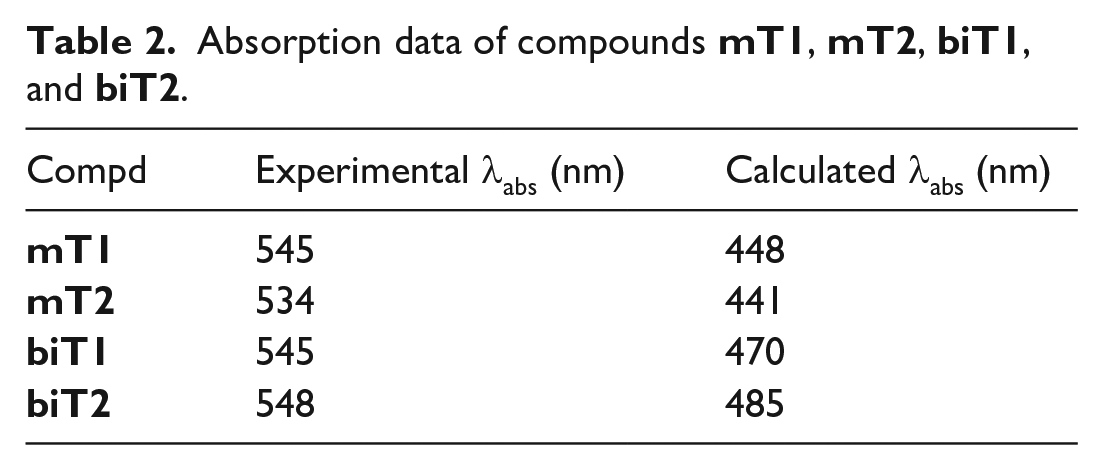
In order to gain detailed insights of the absorption bands, density functional theory (DFT) calculations were carried out using Gaussian 16 to optimize the geometries of the four derivatives and obtain molecular orbitals (MO).
47
All calculations were performed at the B3LYP/6-31+G* level without any solvation options. The resulting optimized geometries were double-checked with frequency calculation. Delocalization of the frontier molecular orbitals (FMO) of the four compounds represents important electronic bonds between the BODIPY unit and the furan group. Interestingly, the HOMO–LUMO band gap for
In the frontier molecular orbitals of the four molecules, the distribution of the HOMO and LUMO levels reflects the strong donor–acceptor interactions (Figure 3). In

The frontier molecular orbitals of the BODIPYs
Absorption data of compounds
Conclusion
In conclusion, we have synthesized and characterized four BODIPY derivatives (
Experimental
Materials
All the reagents and solvents used herein were purchased from commercial suppliers (Aldrich, Acros, and Merck) and used without further purification. Silica gel (60–120 mesh size) was used for flash column chromatography, and was purchased from Merck.
Equipment
High-resolution matrix-assisted laser desorption/ionization (MALDI) mass analysis was performed using an Autoflex III MALDI-TOF mass spectrometer (Bruker Daltonics, Germany) equipped with a smart beam laser as an ionization source. All the spectra were acquired with –19 kV accelerating voltage, a 100-Hz repetition rate, and in positive mode with an average of 500 shots. 1 H and 13 C NMR spectra were acquired using a Bruker Avance 500-MHz spectrometer.
Synthesis
2,8-Diethyl-5,5-difluoro-10-(furan-2-yl)-1,3,7,9-tetramethyl-5H-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinin-4-ium-5-uide (
5,5-Difluoro-10-(furan-2-yl)-3,7-dimethyl-5H-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinin-4-ium-5-uide (
10,10'-(Furan-2,5-diyl)bis(2,8-diethyl-5,5-difluoro-1,3,7,9-tetramethyl-5H-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinin-4-ium-5-uide) (
10,10'-(Furan-2,5-diyl)bis(5,5-difluoro-3,7-dimethyl-5H-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinin-4-ium-5-uide) (
Supplemental Material
sj-docx-1-chl-10.1177_17475198221143738 – Supplemental material for Photophysical properties of furan-bridged dimeric boron-dipyrromethene derivatives (BODIPYs)
Supplemental material, sj-docx-1-chl-10.1177_17475198221143738 for Photophysical properties of furan-bridged dimeric boron-dipyrromethene derivatives (BODIPYs) by Galam Jung, Namdoo Kim and Se Won Bae in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Science Research Program to Research Institute for Basic Sciences (RIBS) of Jeju National University through the National Research Foundation of Korea (NRF) funded by the Ministry of Education to S.W.B. (No. 2019R1A6A1A10072987) and by a research grant of Kongju National University in 2020 to N.K.
Supplemental material
Supplemental material (BODIPYS SI.docx) for this article (CHL-22-0147) is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
