Abstract
A series of

A series of novel
Introduction
Cancers are a class of diseases in which a group of cells display uncontrolled growth, invasion, and sometimes metastasis.1,2 It is major cause of morbidity and mortality worldwide, in every region of the world, irrespective of the level of human development. 3 In preclinical and clinical trials, it has been demonstrated that c-Met inhibitors exhibit antitumor activity in the treatment of multiple types of cancers. 4 Specific small-molecule inhibitors targeting the c-Met tyrosine kinase domain are one of the treatment strategies in clinical trials. 5
About 75% of Food and Drug Administration (FDA)-approved drugs are derivatives of nitrogen-containing heterocyclic compounds. 6 These N-heterocyclic products exhibit anticancer effects in different types of cancers through inhibiting cell growth and induction of cell differentiation and apoptosis. 7 The successful synthesis and antitumor activity of such nitrogen-containing heterocyclic moieties over the past few years are certainly a case for optimism. 7
Recently, a series of triazole tetrazine derivatives was synthesized and exhibited significant anticancer activities. 6-Alkylamino[1,2,4]triazolo[4,3-
Based on the above information, we have synthesized a series of derivatives containing a bis-triazole tetrazine ring symmetrical structure by substitution reaction with alkyl diamines (Figure 1). Our goal was to improve the antitumor activity of the target compounds.

Design strategy toward N-linked triazole tetrazine compounds.
Results and discussion
Chemical synthesis
The synthetic route to the

Synthetic route toward the target compounds
The chemical structures of synthesized compounds were elucidated on the basis of 1H NMR, 13C NMR and elemental analysis. In the 1H NMR spectra of compounds
Anticancer activity in vitro
To test the antitumor activities of the synthesized compounds

Average of three independent experiments.
Cisplatin was used as the positive control.
Binding mode analysis
To examine whether the compounds inhibit c-Met kinase, we screened products
To gain a better understanding of the potency of the studied compounds and to guide further SAR studies, we proceeded to examine the interactions of compounds
The binding modes of compounds
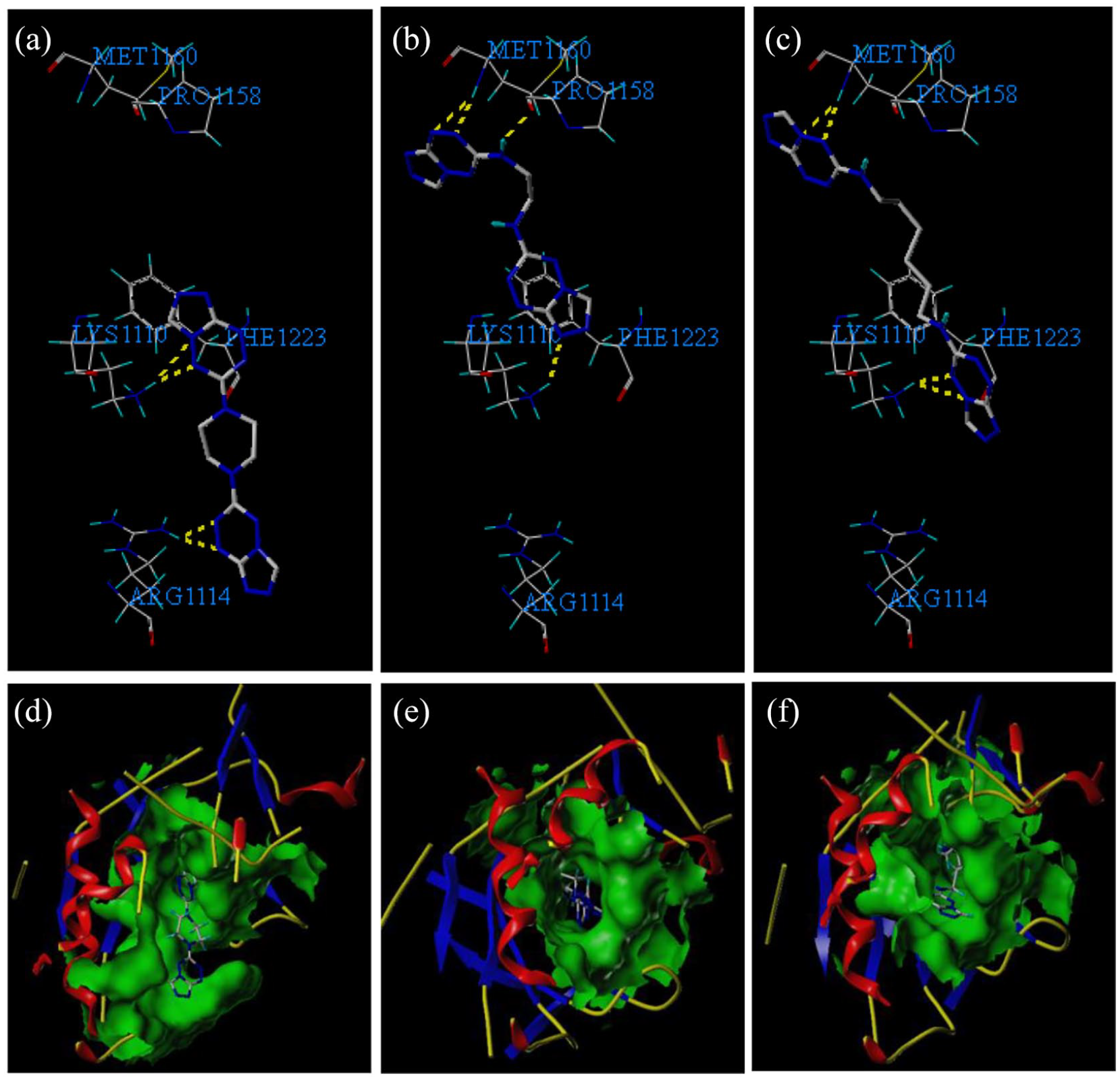
(a–c) Compounds
The geometries of the interactions are illustrated in Table 2. As can be seen, the order of the effect of molecular docking according to the Total-Score is
Geometries of interactions of compounds

Total-Score represents the overall effect of molecular docking. The higher the score, the better the docking effect between compounds and receptors.
The nitrogen atoms all belong to the tetrazine rings,
The chiral carbon atom in PHE1223.
Cg1 is the centroid of the tetrazine ring of
Cg2 is the centroid of benzene ring of Phe1223.
Cg3 is the centroid of the tetrazine ring of
The enzyme surface models are shown in Figure 2(d)–(f), which reveal that molecules
Conclusion
In summary, a series of
Experimental
Materials and methods
Melting points were recorded on a XRC-1 apparatus and are uncorrected (Beijing Technical Instrument Co., Beijing, China). Infrared spectra were recorded as KBr disks for solid materials on a Nicolet FI-IR-170 spectrometer. The 1H NMR and 13C NMR spectra were run on a Bruker AC400 (400 MHz) spectrometer. Compounds were dissolved in DMSO-d6, and chemical shifts were referenced to TMS (tetramethylsilane). Mass spectra were obtained on an Agilent 1260 Ion Trap LC/MS 500 analysis system. Elemental analyses were performed on a Thermo-Finnigan Flash EA 1112 instrument. TLC was carried out on silica gel UV-254 plates.
Synthesis of compound 3
3-(3,5-dimethyl-1
Synthesis of compounds 4a-f; general procedure
Compound
N
1
, N
2
N
1
, N
3
N
1
, N
2
N
1
, N
4
N
1
, N
6
Biological evaluation
In vitro cancer cell growth inhibition assay
The antiproliferative activities of compounds
Molecular docking
Molecular docking was performed with the Surflex-Dock program interfaced with SybylX-2.0. The programs adapted an empirical scoring function and a patented searching engine.
19
The ligand was docked into the corresponding protein binding site guided by protomol, which is an idealized representation of a ligand that makes every potential interaction with a binding site. In this work, the crystal structure of c-Met complexed with 2-benzyl-5-(3-chloro- 4-((6,7-dimethoxyquinolin-3-yl)oxy)phenyl)pyrimidin-4(
c-Met kinase assay in vitro
HTRF(Homogeneous Time-Resolved Fluorescence) uses two fluorescence labels, europium cryptate (fluorescence donor, EuK) and crosslinked allophycocyanin (fluorescence acceptor, XL665). When both fluorescence molecules are in proximity (<10 nm), the energy of EuK excited by a nitrogen laser (λ = 340 nm) is transferred nonradiatively to XL665, resulting in long-lived emission at λ = 665 nm. The nonspecific fluorescence from unbound XL665 and from some other components in the media or plastic have short decay times, and their interference of the detection signal can be excluded by delaying the detection time. On the other hand, the free EuK excited by a nitrogen laser (λ = 340 nm), resulting in long-lived emission at λ = 620 nm, is used as a background signal. These two specific signals at 665 and 620 nm were measured with a multifunctional microplate reader, and the strength of the detection signal from the reaction system reflects the activity of the tested compounds against c-Met kinase. The HTRF experimental method is described as follows: (1) tested compounds (4 μL, diluted with buffer solution to nine different concentrations: 100000, 20000, 4000, 800, 160, 32, 6.4, 1.28 and 0.256 nM) and c-Met kinase (2 μL) were added to each well, to which was added a mixture (4 μL, v/v = 1:1) of TK Substrate-biotin (1 μM) and ATP (3 μM). Each well was kept at room temperature for 40 min. (2) The mixture (10 μL, v/v = 1:1) of SA-XL665 (0.125 μM) and TK antibody-cryptate (diluted to 100 times) was added to the above wells. After keeping the mixture at room temperature for 1 h, the fluorescence signal was measured with a multifunctional microplate reader.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the University-Enterprise Cooperation Project of Domestic Visiting Engineer of Universities in 2021 (FG2021232), the Science and Technology Innovation Activity Plan of College Students in Zhejiang Province in 2022 (2022R481A002), the Research and Cultivation Project of Taizhou Vocational and Technical College-Synthesis and Bioactivity Evaluation of New Tetrazine Antitumor Drugs (2021PY05) and the Taizhou Science and Technology Plan Project-Study on the Synthesis and Bioactivity of New Bistriazolo Tetrazine Drugs (1902gy27).
