Abstract
A series of ((4-([1,2,4]triazolo[4,3-b][1,2,4,5] methyl) benzo-hydrazide derivatives was designed, synthesized, and evaluated for their inhibition activities against five tumor cells and c-Met kinase in vitro. These compounds were fully characterized by 1H NMR, 13C NMR, MS, and elemental analysis. Antitumor experiments indicated that some compounds exhibited significant inhibition activities against A549 and Bewo. Especially, the IC50 values of
A series of ((4-([1,2,4]triazolo[4,3-b][1,2,4,5] methyl) benzohydrazide derivatives were designed, synthesized, and evaluated for their inhibition activities against five tumor cells and c-Met kinase in vitro.
Introduction
Cancer is a class of diseases in which a group of cells display uncontrolled growth, invasion, and sometimes metastasis.1,2 According to the World Health Organization (WHO), cancer is the second leading cause of death after cardiovascular disease all around the globe and accounted for 8.8 million deaths in 2015 worldwide.3,4
c-Met is a unique member of the receptor tyrosine kinases (RTKs) expressed in both normal and malignant cells. It is a cell-surface receptor for hepatocyte growth factor (HGF), a pleiotropic cytokine that conveys a unique combination of pro-migratory, anti-apoptotic, and mitogenic signals.5,6 Under normal physiological conditions, the pleiotropic effects of the HGF/c-Met axis are essential for embryogenesis and tissue homeostasis.7,8 Aberrant HGF/c-Met signaling has been identified in a wide range of human malignancies, including brain, colorectal, gastric, lung, stomach, breast, and head and neck cancers.9,10 Moreover, both c-Met overexpression and MET amplification have been associated with poor clinical outcomes for cancer patients. Of particular note, amplification of the MET oncogene is observed in epidermal growth factor receptor (EGFR)-mutant non-small-cell lung carcinomas after tyrosine kinase inhibitor failure.11–14 Therefore, c-Met has become one of the most promising therapeutic targets in anticancer drug discovery. 15
There have been a number of studies on developing small molecule c-Met inhibitors, and various c-Met inhibitors have been reported.
16
Among these, benzoyl hydrazide derivatives

Examples of c-Met inhibitors: benzoyl hydrazide and triazolotetrazine derivatives.
Recently, a series of triazolotetrazine derivatives has been synthesized and found to exhibit significant anticancer activities. 6-Alkylamino-[1,2,4]triazolo[4,3-b][1,2,4,5]tetrazine derivatives (
Based on the above information, we designed a series of new triazolotetrazine derivatives with benzohydrazide groups at the 6-position to investigate further how these substituents located at the 6-position of the [1,2,4]triazolo[4,3-b][1,2,4,5]tetrazine ring influence antitumor activity.
Results and discussion
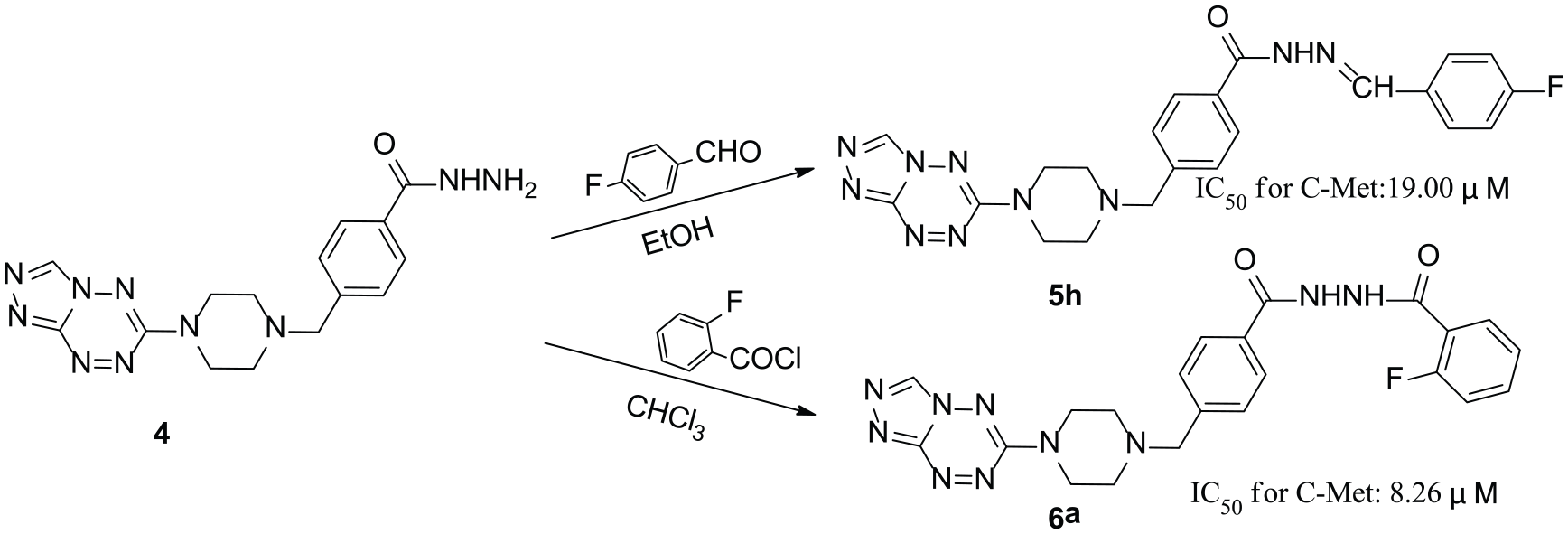
The synthetic routes to the 6-substituted-[1,2,4]triazolo[4,3-b][1,2,4,5] tetrazines are summarized in Scheme 1. Methyl 4-(piperazin-1-ylmethyl) benzoate

Synthetic routes to the target compounds
The chemical structures of the compounds synthesized were elucidated on the basis of 1H NMR, 13C NMR, and elemental analysis. In the 1H NMR spectra of compounds
To test the antitumor activities of the compounds synthesized, we evaluated antiproliferative activities of compounds
Antitumor activities against A549, Bewo, Hela, HepG2, and HT29 cell lines in vitro (IC50 in μM) a .

Average of three independent experiments.
Cisplatin was used as positive control.
To examine whether the compounds inhibit c-Met kinase, we screened compounds
To gain a better understanding of the potency of the compounds studied and to guide further SAR studies, we proceeded to examine the interaction of compound
The binding modes of compound

(a) Compound
The two nitrogen atoms of the triazolotetrazine ring formed two hydrogen bonds with the same amido hydrogen of ARG1086 (bond length: 2.23 and 2.60 Å, respectively), and the oxygen atom of benzoyl group formed one hydrogen bond with the amido hydrogen of ASP1222 (bond length: 2.12 Å). A C–H—π interaction was predicted to form between the methyl hydrogen of LEU1157 and the o-chlorophenyl of
Conclusion
In summary, a series of ((4-([1,2,4]triazolo[4,3-b][1,2,4,5] tetrazin-6-yl)piperazin-1-yl) methyl) benzohydrazide derivatives has been prepared and evaluated for anticancer activity against five cancer cell lines (A549, Bewo, Hela, HepG2, and HT29). Compounds
Experiment
Materials and methods
Melting points were carried out on a XRC-1 apparatus and are uncorrected (Beijing Technical Instrument Co., Beijing, China). Infrared spectra were recorded using KBr disks for solid materials on a Nicolex FI-IR-170 instrument. The 1H NMR and 13C NMR spectra were run on a Bruker AC400 (400 MHz). Compounds were dissolved in DMSO-d6, and chemical shifts were referenced to TMS (tetramethylsilane). Mass spectra were obtained on an Agilent 1260 Ion Trap LC/MS 500 analysis system. Elemental analyses were performed on a Thermo-Finnigan Flash EA 1112 instrument. TLC was carried out on silica gel UV-254 plates.
General procedure for the synthesis of Methyl 4-((4-([1,2,4] triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl) benzoate (3)
Step 1. Methyl 4-(bromomethyl)benzoate (7.3 g, 30 mmol) in dichloromethane (50 mL) was added dropwise to a stirred solution of anhydrous piperazine (12.9 g, 0.15 mol) and dichloromethane (150 mL) at 0 °C. The mixture was stirred at room temperature overnight. After the reaction was over (the reaction course was monitored by TLC (AcOEt)), the mixture was washed by a saturated aqueous NaHCO3 solution (2 × 100 mL) and dried over Na2SO4. Finally, the solvent was evaporated under reduced pressure and the resulting product was used directly in the next step (yield: 5.6 g, 75.2%).
Step 2. 6-(3,5-dimethyl-1H-pyrazol-1-yl)-[1,2,4]triazolo[4,3-b][1,2,4,5]tetrazine
General procedure for the synthesis of ((4-([1,2,4]triazolo [4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl) methyl) benzohy-drazide (4)
Methyl 4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5] piperazin-1-yl)methyl)benzoate
General procedure for the synthesis of compounds 5a-5i
Intermediate
General procedure for the synthesis of compounds 6a-6h
Intermediate
Methyl 4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)benzoate (3)
Orange solid, m.p. 141–143 °C; 1H NMR (400 MHz, DMSO-d6): δ 9.48 (s, 1H, CH), 7.95 (d, 2H, J = 8.0 Hz, Ar), 7.52 (d, 2H, J = 8.0 Hz, Ar), 3.85 (brs, 7H, 2CH2+CH3), 3.65 (s, 2H, CH2), 2.58 (t, 4H, J = 6.0 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 166.6, 155.6, 150.1, 144.2, 136.9, 129.7(2C), 129.5(2C), 128.9, 61.8, 52.6(2C), 52.3(2C), 44.7. ESI-MS m/z (%): 377 [(M+Na)+, 100]. Anal. calcd (%) for C16H18N8O2: C, 54.23; H, 5.12; N, 31.62; found: C, 54.10; H, 5.11; N, 31.70.
((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)benzohydrazide (4)
Orange solid, m.p. 176–178 °C; 1H NMR (400 MHz, DMSO-d6): δ 9.77 (s, 1H, NH), 9.48 (s, 1H, CH), 7.82 (d, 2H, J = 7.8 Hz, Ar), 7.43 (d, 2H, J = 7.8 Hz, Ar), 3.84 (t, 4H, J = 6.2 Hz, 2CH2), 3.61(s, 2H, CH2), 2.57(t, 4H, J = 6.2 Hz, 2CH2), 2.38(brs, 2H, NH2). 13C NMR (100 MHz, DMSO-d6): δ 166.2, 156.6, 136.9, 132.6, 129.6, 129.4, 129.2 (2C), 127.5 (2C), 61.9, 53.1 (2C), 52.3 (2C). ESI-MS: m/z (%): 377 [(M+Na)+, 100]. Anal. calcd (%) for C15H18N10O: C, 50.84; H, 5.12; N, 39.53; found: C, 50.95; H, 5.10; N, 39.44.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(4-(dimethylamino)benzylidene) benzohydrazide (5a)
Yield: 78%. Orange solid, m.p. 149–151 °C. 1H NMR (400 MHz, DMSO-d6): δ 11.57 (s, 1H, NH), 9.46 (s, 1H, CH), 8.30 (s, 1H, N=CH), 7.88 (d, 2H, J = 8.0 Hz, Ar), 7.51 (m, 4H, Ar), 6.75 (d, 2H, J = 7.8 Hz, Ar), 3.86 (t, 4H, J = 5.8 Hz, 2CH2), 3.63 (s, 2H, CH2), 2.97 (s, 6H, 2CH3), 2.58 (t, 4H, J = 5.8 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.0, 155.6, 151.9, 150.1, 149.0, 142.0, 136.9, 133.1, 129.3(2C), 128.8(2C), 128.0(2C), 122.1, 112.3(2C), 61.9, 52.3(2C), 44.7(2C), 40.2(2C). ESI-MS: m/z (%): 486 [(M+H)+, 40], 508 [(M+Na)+, 100]. Anal. calcd (%) for C24H27N11O: C, 59.37; H, 5.60; N, 31.73; found: C, 59.51; H, 5.57; N, 31.63.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(3,4-dihydroxybenzylidene)benzohydrazide (5b)
Yield: 85%. Orange solid, m.p. 268–270 °C. 1H NMR (400 MHz, DMSO-d6):δ 11.61 (s, 1H, NH), 9.47 (brs, 3H, CH+2OH), 8.27 (s, 1H, N = CH), 7.88 (d, 2H, J = 7.1 Hz, Ar), 7.49 (d, 2H, J = 7.1 Hz, Ar), 7.25 (s, 1H, Ar), 6.92(d, 1H, J = 7.3 Hz, Ar), 6.79 (d, 1H, J = 7.3 Hz, Ar), 3.87 (t, 4H, J = 5.9 Hz, 2CH2), 3.64 (s, 2H, CH2), 2.59 (t, 4H, J = 5.9 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.1, 155.5, 150.1, 148.7, 148.4, 146.2, 142.1, 136.8, 132.9, 129.3 (2C), 128.0 (2C), 126.2, 121.1, 116.0. 113.1, 61.8, 52.2 (2C), 44.7 (2C). ESI-MS: m/z (%): 497 [(M+Na)+, 100]. Anal. calcd (%) for C22H22N10O3: C, 55.69; H, 4.67; N, 29.52; found: C, 55.81; H, 4.63; N, 29.44.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(3-hydroxy-4-methoxybenzylidene) benzohydrazide (5c)
Yield: 82%. Orange solid, m.p. 210–212 °C. 1H NMR (400 MHz, DMSO-d6): δ 11.68 (s, 1H, NH), 9.48 (s, 1H, CH), 9.37 (s, 1H, OH), 8.30 (s, 1H, N = CH), 7.89 (d, 2H, J = 8.1 Hz, Ar), 7.51 (d, 2H, J = 8.1 Hz, Ar), 7.28 (s, 1H, Ar), 7.06 (d, 1H, J = 8.4 Hz, Ar), 6.98 (d, 1H, J = 8.4 Hz, Ar), 3.87 (t, 4H, J = 6.1 Hz, 2CH2), 3.81 (s, 3H, OCH3), 3.65 (s, 2H, CH2), 2.59 (t, 4H, J = 6.1 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.2, 155.6, 150.2, 150.1, 148.3, 147.3, 142.2, 136.9, 132.9, 129.3 (2C), 128.1 (2C), 127.6, 120.8, 112.7. 112.3, 61.9, 56.0, 52.3 (2C), 44.8 (2C). ESI-MS: m/z (%): 511 [(M+Na) +, 100]. Anal. calcd (%) for C23H24N10O3: C, 56.55; H, 4.95; N, 28.67; found: C, 56.40; H, 4.91; N, 28.78.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(3,4-dimethoxybenzylidene)benzohydrazide (5d)
Yield: 88%. Orange solid, m.p. 185–187 °C. 1H NMR (400 MHz, DMSO-d6): δ 11.75 (s, 1H, NH), 9.48 (s, 1H, CH), 8.39 (s, 1H, N=CH), 7.90 (d, 2H, J = 8.0 Hz, Ar), 7.51 (d, 2H, J = 8.0 Hz, Ar), 7.35 (s, 1H, Ar), 7.20 (d, 1H, J = 8.3 Hz, Ar), 7.03 (d, 1H, J = 8.3 Hz, Ar), 3.87 (t, 4H, J = 6.0 Hz, 2CH2), 3.83 (s, 3H, OCH3), 3.82 (s, 3H, OCH3), 3.66 (s, 2H, CH2), 2.60 (t, 4H, J = 6.0 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.3, 155.6, 151.2, 150.2, 149.5, 148.4, 142.2, 136.9, 133.4, 129.4 (2C), 128.1 (2C), 127.5, 122.4, 111.9. 108.5, 61.8, 56.0, 55.9, 52.3 (2C), 44.7 (2C). ESI-MS: m/z (%): 503 [(M+H)+, 100], 525 [(M+Na)+, 80]. Anal. calcd (%) for C24H26N10O3: C, 57.36; H, 5.21; N, 27.87; found: C, 57.47; H, 5.24; N, 27.80.
5-4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(4-hydroxybenzylidene)benzohydrazide (5e)
Yield: 75%. Orange solid, m.p. 148 °C (dec.). 1H NMR (400 MHz, DMSO-d6):δ 11.66 (s, 1H, NH), 9.99 (brs, 1H, OH), 9.48 (s, 1H, CH), 8.35 (s, 1H, N = CH), 7.89 (d, 2H, J = 8.1 Hz, Ar), 7.56 (d, 2H, J = 8.5 Hz, Ar), 7.50 (d, 2H, J = 8.1 Hz, Ar), 6.84 (d, 2H, J = 8.5 Hz, Ar), 3.87 (t, 4H, J = 5.9 Hz, 2CH2), 3.65 (s, 2H, CH2), 2.59 (t, 4H, J = 5.9 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.1, 159.9, 155.6, 150.2, 148.5, 142.2, 136.9, 133.0, 129.3 (2C), 128.0 (2C), 125.8 (2C), 119.6, 116.2 (2C), 62.1, 52.2 (2C), 44.8 (2C). ESI-MS: m/z (%): 481 [(M+Na)+, 100]. Anal. calcd (%) for C22H22N10O2: C, 57.63; H, 4.84; N, 30.55; found: C, 57.75; H, 4.80; N, 30.48.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(2,4-dichlorobenzylidene)benzohydrazide (5f)
Yield: 94%. Orange solid, m.p. 147–149 °C. 1H NMR (400 MHz, DMSO-d6): δ 11.75 (s, 1H, NH), 9.48 (s, 1H, CH), 8.39 (s, 1H, N=CH), 7.90 (d, 2H, J = 8.0 Hz, Ar), 7.51 (d, 2H, J = 8.0 Hz, Ar), 7.35 (s, 1H, Ar), 7.20 (d, 1H, J = 8.3 Hz, Ar), 7.03 (d, 1H, J = 8.3 Hz, Ar), 3.87 (t, 4H, J = 5.7 Hz, 2CH2), 3.66 (s, 2H, CH2), 2.60 (t, 4H, J = 5.7 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.6, 155.5, 150.3, 149.9, 143.1, 136.9, 135.6, 134.4, 129.9 (2C), 128.6 (2C), 128.3, 126.0, 120.9, 111.7. 100.0, 62.5, 52.2 (2C), 44.8 (2C). ESI-MS: m/z (%): 511 [(M+H) +, 100]. Anal. calcd (%) for C22H20Cl2N10O: C, 51.67; H, 3.94; N, 27.39; found: C, 51.79; H, 3.93; N, 27.32.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(4-nitrobenzylidene)benzohydrazide (5g)
Yield: 90%. Orange solid, m.p. 153–155 °C. 1H NMR (400 MHz, DMSO-d6): δ 12.19 (s, 1H, NH), 9.48 (s, 1H, CH), 8.56 (s, 1H, N=CH), 8.32 (d, 2H, J = 8.5 Hz, Ar), 8.01 (d, 2H, J = 8.5 Hz, Ar), 7.93 (d, 2H, J = 8.0 Hz, Ar), 7.54 (d, 2H, J = 8.0 Hz, Ar), 3.88 (t, 4H, J = 6.1 Hz, 2CH2), 3.66 (s, 2H, CH2), 2.60 (t, 4H, J = 6.1 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.7, 155.6, 150.2, 148.3, 145.6, 141.1, 136.9, 132.3, 129.4 (2C), 128.5 (2C), 128.3, 124.6 (4C), 61.9, 52.3 (2C), 44.7 (2C). ESI-MS: m/z (%): 488 [(M+H)+, 100]. Anal. calcd (%) for C22H21N11O3: C, 54.21; H, 4.34; N, 31.61; found: C, 54.03; H, 4.36; N, 31.69.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(4-fluorobenzylidene)benzohydrazide (5h)
Yield: 91%. Orange solid, m.p. 182–184 °C. 1H NMR (400 MHz, DMSO-d6): δ 11.89 (s, 1H, NH), 9.48 (s, 1H, CH), 8.46 (s, 1H, N=CH), 7.90 (d, 2H, J = 8.0 Hz, Ar), 7.80 (dd, 2H, J1 = 8.4 Hz, J2 = 5.6 Hz, Ar), 7.52 (d, 2H, J = 8.0 Hz, Ar), 7.32 (t, 2H, J = 8.8 Hz, Ar), 3.88 (t, 4H, J = 5.8 Hz, 2CH2), 3.65 (s, 2H, CH2), 2.59 (t, 4H, J = 5.8 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.6 (J = 241 Hz), 163.4, 155.6, 150.2, 147.0, 142.4, 136.9, 132.7, 131.4, 129.7 (2C) (J = 15 Hz), 129.4(2C), 128.2 (2C), 116.4 (2C) (J = 22 Hz), 61.9, 52.3 (2C), 44.8 (2C). ESI-MS: m/z (%): 461 [(M+H)+, 5], 483 [(M+Na)+, 100]. Anal. calcd (%) for C22H21FN10O: C, 57.38; H, 4.60; N, 30.42; found: C, 57.52; H, 4.62; N, 30.32.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-benzylidenebenzohydrazide (5i)
Yield: 87%. Orange solid, m.p. 213–215 °C. 1H NMR (400 MHz, DMSO-d6): δ 11.86 (s, 1H, NH), 9.47 (s, 1H, CH), 8.47 (s, 1H, N=CH), 7.92 (d, 2H, J = 7.0 Hz, Ar), 7.73 (d, 2H, J = 7.0 Hz, Ar), 7.46–7.53 (m, 5H, Ar), 3.88 (t, 4H, J = 5.9 Hz, 2CH2), 3.66 (s, 2H, CH2), 2.60 (t, 4H, J = 5.9 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 163.4, 155.6, 150.2, 148.1, 142.4, 136.9, 134.8, 132.7, 130.6, 129.3(4C), 128.2(2C), 127.6(2C), 61.9, 52.3(2C), 44.8(2C). ESI-MS: m/z (%): 465 [(M+Na)+, 100]. Anal. calcd (%) for C22H22N10O: C, 59.72; H, 5.01; N, 31.66; found: C, 59.85; H, 4.98; N, 31.57.
N′-(4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6yl)piperazin-1-yl)methyl)benzoyl)-2-fluorobenzohydrazide (6a)
Yield: 75%. Yellow solid, m.p. 184–186 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.67 (s, 1H, NH), 10.42 (s, 1H, NH), 9.54(s, 1H, CH), 7.36–8.31 (m, 8H, Ar), 4.11(s, 2H, CH2), 3.86 (t, 4H, J = 6.2 Hz, 2CH2), 2.98 (t, 4H, J = 6.2 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 165.6, 164.0, 162.1 (J = 253 Hz), 155.4, 150.2, 148.3, 137.0, 133.6 (J = 6.5 Hz), 130.6 (J = 2.4 Hz), 128.5 (2C), 128.2 (2C), 125.1 (J = 2.8 Hz), 124.6, 122.7 (J = 15.3 Hz), 116.8 (J = 32.3 Hz), 58.7, 50.1(2C), 41.9(2C). ESI-MS: m/z (%): 477 [(M+H)+, 100]. Anal. calcd (%) for C22H21FN10O2: C, 55.46; H, 4.44; N, 29.40; found: C, 55.35; H, 4.46; N, 29.45.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(4-chlorobenzoyl)benzohydrazide (6b)
Yield: 70%. Yellow solid, m.p. 178–180 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.69 (s, 1H, NH), 10.66 (s, 1H, NH), 9.60 (s, 1H, CH), 8.02 (d, 2H, J = 7.0 Hz, Ar), 7.95 (d, 2H, J = 7.9 Hz, Ar), 7.79 (d, 2H, J = 7.0 Hz, Ar), 7.62 (d, 2H, J = 7.9 Hz, Ar), 4.66 (s, 2H, CH2), 3.86 (t, 4H, J = 6.1 Hz, 2CH2), 3.25 (t, 4H, J = 6.1 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 165.7, 165.3, 155.3, 150.2, 137.3, 137.1, 132.0, 131.7, 129.9 (3C), 129.2 (4C), 128.3 (2C), 58.9, 50.3 (2C), 42.0 (2C). ESI-MS: m/z (%): 515 [(M+Na)+, 100]. Anal. calcd (%) for C22H21ClN10O2: C, 53.61; H, 4.29; N, 28.42; found: C, 53.70; H, 4.26; N, 28.35.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(4-Nitrobenzoyl)benzohydrazide (6c)
Yield: 70%. Yellow solid, m.p. 178–180 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.98 (s, 1H, NH), 10.76 (s, 1H, NH), 9.57 (s, 1H, CH), 8.33 (d, 2H, J = 8.6 Hz, Ar), 8.17 (d, 2H, J = 8.4 Hz, Ar), 8.00 (d, 2H, J = 7.4 Hz, Ar), 7.74 (d, 2H, J = 7.4 Hz, Ar), 4.11(s, 2H, CH2), 3.86 (t, 4H, J = 6.3 Hz, 2CH2), 3.14 (t, 4H, J = 6.3 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 166.3, 165.7, 155.4, 150.5, 150.2, 137.0, 136.8, 131.2 (2C), 129.8, 129.5 (2C), 128.2 (2C), 128.0, 124.2 (2C), 62.2, 50.7 (2C), 42.8 (2C). ESI-MS: m/z (%): 504 [(M+H)+, 100]. Anal. calcd (%) for C22H21N11O4: C, 52.48; H, 4.20; N, 30.60; found: C, 52.61; H, 4.18; N, 30.52.
N′-(4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl) piperazin-1-yl)methyl)benzoyl)-3-chlorobenzohydrazide (6d)
Yield: 83%. Yellow solid, m.p. 181–183 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.74 (s, 1H, NH), 10.69 (s, 1H, NH), 9.58(s, 1H, CH), 8.01 (s, 1H, Ar), 7.98 (d, 2H, J = 7.5 Hz, Ar), 7.90 (d, 1H, J = 7.8 Hz, Ar), 7.75 (d, 2H, J = 7.5 Hz, Ar), 7.70 (d, 1H, J = 7.8 Hz, Ar), 7.59 (t, 1H, J = 7.8 Hz, Ar), 4.27 (s, 2H, CH2), 3.86 (t, 4H, J = 6.2 Hz, 2CH2), 3.19 (t, 4H, J = 6.2 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 165.7, 165.0, 155.4, 150.2, 137.0, 134.9, 133.9, 133.3, 132.3, 131.5, 131.2, 129.8 (2C), 128.2 (2C), 127.7, 126.7, 59.4, 50.6(2C), 42.5(2C). ESI-MS: m/z (%): 493 [(M+H) +, 20], 515 [(M+Na)+, 30]. Anal. calcd (%) for C22H21ClN10O2: C, 53.61; H, 4.29; N, 28.42; found: C, 53.72; H, 4.25; N, 28.36.
N′-(4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin -1-yl)methyl)benzoyl)-3-fluorobenzohydrazide (6e)
Yield: 85%. Yellow solid, m.p. 207–209 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.72 (s, 1H, NH), 10.70 (s, 1H, NH), 9.60(s, 1H, CH), 8.02 (d, 2H, J = 8.2 Hz, Ar), 7.81 (d, 3H, J = 8.2 Hz, Ar), 7.72 (d, 1H, J = 8.3 Hz, Ar), 7.46–7.67 (m, 2H, Ar), 4.47 (s, 2H, CH2), 3.87 (t, 4H, J = 6.0 Hz, 2CH2), 3.51 (t, 4H, J = 6.0 Hz, 2CH2). 13C NMR (100 MHz, DMSO-d6): δ 165.6, 165.0, 162.4 (J = 242 Hz), 155.3, 150.2, 137.1, 135.1 (J = 6.9 Hz), 133.8, 132.2, 131.4 (J = 7.0 Hz), 129.9(2C), 128.3(2C), 124.1 (J = 2.7 Hz), 116.2 (J = 21.8 Hz), 114.7 (J = 22.7 Hz), 58.7, 50.2(2C), 41.9(2C). ESI-MS: m/z (%): 477 [(M+H)+, 20]. Anal. calcd (%) for C22H21FN10O2: C, 55.46; H, 4.44; N, 29.40; found: C, 55.59; H, 4.40; N, 29.29.
N′-(4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin -1-yl)methyl)benzoyl)-3-methylbenzohydrazide (6f)
Yield: 65%. Yellow solid, m.p. 174–176 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.63 (s, 1H, NH), 10.54 (s, 1H, NH), 9.61 (s, 1H, CH), 8.02 (d, 2H, J = 8.2 Hz, Ar), 7.81 (d, 1H, J = 8.2 Hz, Ar), 7.73–7.75 (m, 3H, Ar), 7.38–7.42 (m, 2H, Ar), 4.47 (s, 2H, CH2), 3.87 (t, 4H, J = 5.9 Hz, 2CH2), 3.52 (t, 4H, J = 5.9 Hz, 2CH2), 2.37 (s, 3H, CH3). 13C NMR (100 MHz, DMSO-d6): δ 166.4, 165.6, 155.3, 150.2, 138.3, 137.1, 134.0, 132.9, 132.2, 130.2, 128.9 (2C), 128.5, 128.3 (2C), 126.9, 125.0, 58.7, 50.1 (2C), 41.9 (2C), 21.4. ESI-MS: m/z (%): 473 [(M+H)+, 7], 495 [(M+Na)+, 6]. Anal. calcd (%) for C23H24N10O2: C, 58.46; H, 5.12; N, 29.64; found: C, 58.61; H, 5.10; N, 29.43.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-(cyclohexanecarbonyl)benzohydrazide (6g)
Yield: 54%. Yellow solid, m.p. 185–187 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.36 (s, 1H, NH), 9.85 (s, 1H, NH), 9.56 (s, 1H, CH), 7.92 (d, 2H, J = 7.6 Hz, Ar), 7.69 (d, 2H, J = 7.6 Hz, Ar), 4.20 (s, 2H, CH2), 3.85 (t, 4H, J = 5.8 Hz, 2CH2), 3.12 (t, 4H, J = 5.8 Hz, 2CH2), 2.24–2.29 (m, 1H, CH), 1.63–1.74 (m, 4H, 2CH2), 1.15–1.40 (m, 6H, 3CH2). 13C NMR (100 MHz, DMSO-d6): δ 175.1, 165.4, 155.4, 150.1, 137.0, 133.3, 131.2, 129.7 (2C), 128.1 (2C), 59.6, 52.7, 50.8 (2C), 42.5 (2C), 29.5 (2C), 25.6 (3C). ESI-MS: m/z (%): 465 [(M+H)+, 60]. Anal. calcd (%) for C22H28N10O2: C, 56.88; H, 6.08; N, 30.15; found: C, 57.98; H, 6.06; N, 30.11.
4-((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl)piperazin-1-yl)methyl)-N′-hexanoylbenzohydrazide (6h)
Yield: 61%. Yellow solid, m.p. 185–187 °C. 1H NMR (400 MHz, DMSO-d6): δ 10.42 (s, 1H, NH), 9.94 (s, 1H, NH), 9.58 (s, 1H, CH), 7.96 (d, 2H, J = 7.7 Hz, Ar), 7.78 (d, 2H, J = 7.7 Hz, Ar), 4.41 (s, 2H, CH2), 3.86 (t, 4H, J = 5.6 Hz, 2CH2),3.56 (t, 4H, J = 5.6 Hz, 2CH2), 2.19 (t, 2H, J = 7.3 Hz, CH2), 1.49–1.56 (m, 2H, CH2), 1.25–1.32 (m, 4H, 2CH2), 0.84–0.90 (m, 3H, CH3). 13C NMR (DMSO-d6, ppm): δ 175.0, 172.1, 165.3, 155.3, 150.1, 137.0, 133.7, 131.9 (2C), 128.3 (2C), 58.9, 50.2 (2C), 42.0 (2C), 34.2, 31.2, 25.2, 22.4, 14.4. ESI-MS: m/z (%): 453 [(M+H)+, 85], 475 [(M+Na)+, 27]. Anal. calcd (%) for C21H28N10O2: C, 55.74; H, 6.24; N, 30.95; found: C, 55.57; H, 6.26; N, 31.07.
In vitro cancer cell growth inhibition assay
The antiproliferative activities of the compounds
Molecular docking
Molecular docking was performed with the Surflex-Dock program interfaced with SybylX-2.0. The programs adapted an empirical scoring function and a patented searching engine. 22 The ligand was docked into the corresponding protein-binding site guided by protomol, which is an idealized representation of a ligand that makes every potential interaction with a binding site. In this work, The crystal structure of c-Met complexed with 5-phenyl-3-(quinolin-6-ylmethyl)-3,5,6,7-tetrahydro-4H-[1,2,3]triazolo[4,5-c]pyridin-4-one (PDB entry: 4DEH) was extracted from the Brookhaven Protein Database (PDB; http://www.rcsb.org/pdb). At the beginning of docking, all the water and ligands were removed and random hydrogen atoms were added. Next, the receptor structure was minimized over 10,000 cycles using the Powell method in SybylX-2.0. All the compounds were constructed using a sketch molecular module. Hydrogen and Gasteiger-Hückel charges were added to every molecule. Then their geometries were optimized by the conjugate gradient method in the TRIPOS force field. The energy convergence criterion is 0.001 kcal/mol. Finally, ligand-based mode was adopted to generate the “protomol,” leaving the threshold at their default value of 1.
c-Met kinase assay in vitro
HTRF uses two fluorescence labels: europium cryptate (fluorescence donor, EuK) and crosslinked allophycocyanin (fluorescence acceptor, XL665). When both fluorescence molecules are in proximity (<10 nm), the energy of EuK excited by nitrogen laser (λ = 340 nm) is transferred nonradiatively to XL665, resulting in long-lived emission at λ = 665 nm. The nonspecific fluorescence from unbound XL665 and from some other components in media or plastic have short decay times and can exclude their interference of the detection signal by delaying detected time. On the other hand, the free EuK excited by nitrogen laser (λ = 340 nm), resulting in long-lived emission at λ = 620 nm, which is used as a background signal. These two specific signals at 665 and 620 nm were measured by multifunctional microplate reader, and the strength of detection signal from the reaction system can reflect the activity of tested compounds against c-Met kinase. The experimental method of HTRF was described as follows: (1) tested compounds (4 μL, diluted with buffer solution to nine different concentrations: 100,000, 20,000, 4000, 800, 160, 32, 6.4, 1.28, and 0.256 nM) and c-Met kinase (2 μL) were added to each well, to which the mixture (4 μL, V/V = 1/1) of TK Substrate-biotin (1 µΜ) and ATP (3 μM) was added. Then each well was kept at room temperature for 40 min. (2) The mixture (10 µL, V/V = 1/1) of SA-XL665 (0.125 µΜ) and TK antibody-cryptate (diluted to 100 times) was added to the above wells, after keeping the mixture at room temperature for 1 h, the fluorescence signal was measured by multifunctional microplate reader. Original data can be obtained from Supplemental material.
Supplemental Material
Supplementary_information-1 – Supplemental material for Synthesis, biological evaluation, and molecular docking of ((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl) piperazin-1-yl)methyl) benzohydrazide derivatives
Supplemental material, Supplementary_information-1 for Synthesis, biological evaluation, and molecular docking of ((4-([1,2,4]triazolo[4,3-b][1,2,4,5]tetrazin-6-yl) piperazin-1-yl)methyl) benzohydrazide derivatives by Xiaoyu Wu, Feng Xu, Zhenzhen Yang, Zhonglu Ke, Lei Shi, Can Ye, Qidong Yan and Shijie Zhang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Zhejiang Public Welfare Technology Research Project (LGF19B020001), a scientific research project of the Education Department of Zhejiang Province (Y201840472), Taizhou Science and Technology Planning Project (1902gy27), and a research project of Taizhou Vocational and Technical College (2019YB04).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
