Abstract
A series of 6-alkoxy-3-aryl-[1,2,4]triazolo[4,3-
A series of novel 6-alkoxy-3-aryl-[1,2,4]triazolo[4,3-
Introduction
Cancer is one of the most important problems in public health and is the second leading cause of death in the world. 1 Currently, cancer chemotherapy is one of the most effective methods to treat cancer, which represents a constant, global, and interdisciplinary research effort and can heavily promote and extend the quality of life. New antitumor therapies point to novel compounds with potently selective anticancer effects and must therefore exhibit a cytotoxic effect on malignant cells, without damaging normal cells.2–4
Most of the known synthetic anticancer drugs are heterocyclic compounds, and several of them correspond to nitrogen heterocycles.5–7 In the last decades, a large number of 1,2,4,5-tetrazine derivatives have been synthesized and their antitumor activity has been reported, which has led to them emerging as a promising and attractive scaffolds.8–11 Among them, a 1,4-dihydro-1,2,4,5-tetrazine derivative, N,

Structures of 1,2,4,5-tetrazine derivatives with anticancer activities.
Our research group has synthesized numerous [1,2,4]triazolo[4,3-
Results and discussion
Chemical synthesis
Scheme 1 details the synthesis and structures of the title compounds. The starting material
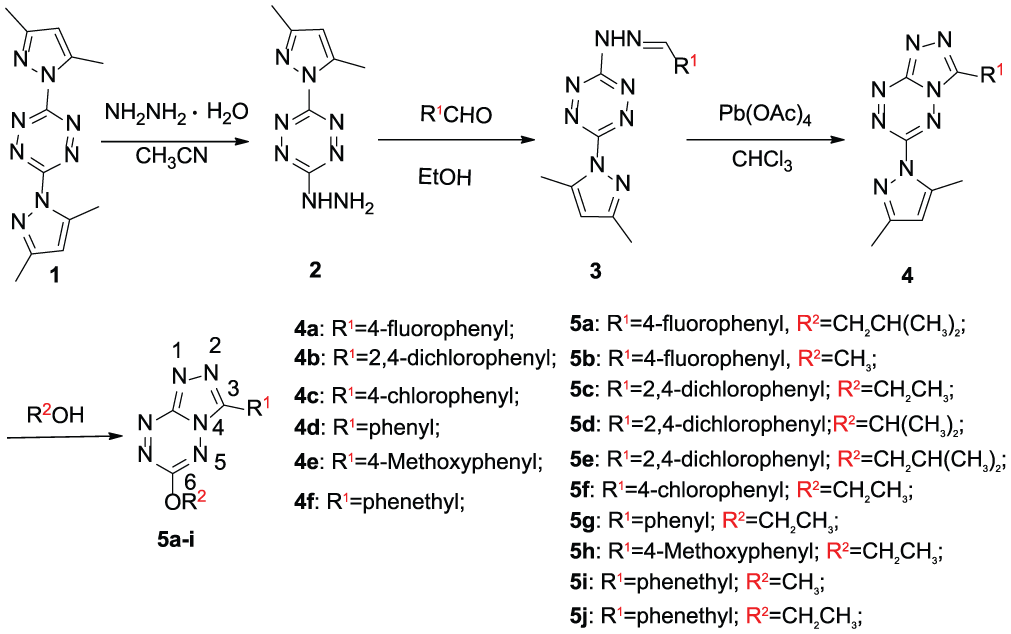
Synthesis of the target compounds
The structures of the synthesized compounds were confirmed from their 1H NMR, 13C NMR, infrared (IR), and elemental analysis data. All of these compounds provided satisfactory spectroscopic data, which were consistent with the assigned structures. It should be noted in this context that to the best of our knowledge, all of the title products are reported for the first time. The 1H NMR and 13C NMR spectra of the title compounds are given in the supplemental data.
Anticancer activity in vitro
To test the antitumor activities of the synthesized compounds, we evaluated the antiproliferative activities of compounds
Binding mode analysis
To gain a better understanding of the potency of the target compounds and to guide further structure–activity relationship (SAR) studies, we continued to investigate the interaction effect between compound
The binding modes of compound

(a) Compound
The two nitrogen atoms of the triazole ring formed two hydrogen bonds with the same amido hydrogen of LYS1110 (bond lengths: 1.94 and 2.79 Å, respectively), and the π–π interaction was predicted to form between the two phenyl rings in
Experimental
Materials and methods
Melting points (m.p.s) were obtained on an XRC-1 apparatus and are uncorrected (Beijing Technical Instrument Co.). IR spectra were recorded as KBr disks of solid materials on a Nicolex FI-IR-170 instrument. The 1H NMR spectra were run on a Bruker AC400 (400 MHz) spectrometer. Compounds were dissolved in DMSO-
General procedure for the synthesis of compounds 4a–f
Lead tetraacetate (5.0 g, 11.4 mmol) in chloroform (10 mL) was added to a refluxing mixture of compound
Compounds
3-(4-Chlorophenyl)-6-(3,5-dimethyl- 1H-pyrazol-1-yl)-[1,2,4]triazolo[4,3- b][1,2,4,5]tetrazine (
4c
)
Yellow solid, m.p. 198–200 °C; IR (KBr, cm−1): 2976, 1529, 1396, 1452, 1036, and 830. MS (ES):
6-(3,5-Dimethyl- 1H-pyrazol-1-yl)-3-phenethyl-[1,2,4]tri-azolo[4,3- b][1,2,4,5]tetrazine (
4f
)
Red solid, m.p. 143–145 °C; IR (KBr, cm−1): 3260, 3071, 1589, 1488, and 1418; MS (ES):
General procedure for the synthesis of compounds 5a–j
Compound
3-(4-Fluorophenyl)-6-isobutoxy-[1,2,4]triazolo[4,3- b][1,2,4, 5]tetrazine (
5a
)
Yellow solid, m.p. 128–130 °C; IR (KBr, cm−1): 3074, 2974, 1606, 1547, 1057, and 861. 1H NMR (400 MHz, DMSO-
3-(4-Fluorophenyl)-6-methoxy-[1,2,4]triazolo[4,3- b][1,2,4, 5]tetrazine (
5b
)
Yellow solid, m.p. 178–180 °C; IR (KBr, cm−1): 3099, 2977, 1605, 1559, 1052, and 852; 1H NMR (400 MHz, DMSO-
3-(2,4-Dichlorophenyl)-6-ethoxy-[1,2,4]triazolo[4,3- b][1, 2,4,5]tetrazine (
5c
)
Yellow solid, m.p. 146–148 °C; IR (KBr, cm−1): 3098, 2979, 1596, 1549, 1052, and 826; 1H NMR (400 MHz, DMSO-
3-(2,4-Dichlorophenyl)-6-isopropoxy-[1,2,4]triazolo[4,3- b][1, 2,4,5]tetrazine (
5d
)
Yellow solid, m.p. 128–130 °C; IR (KBr, cm−1): 3094, 2988, 1547, 1399, 1053, and 824; 1H NMR (400 MHz, DMSO-
3-(2,4-Dichlorophenyl)-6-isobutoxy-[1,2,4]triazolo[4,3- b][1,2, 4,5]tetrazine (
5e
)
Yellow solid, m.p. 168–170 °C; IR (KBr, cm−1): 3058, 2977, 1583, 1538, 1038, and 821; 1H NMR (400 MHz, DMSO-
3-(4-Chlorophenyl)-6-ethoxy-[1,2,4]triazolo[4,3- b][1,2,4,5] tetrazine (
5f
)
Yellow solid, m.p. 220–222 °C; IR (KBr, cm−1): 3090, 2994, 1539, 1054, and 851; 1H NMR (400 MHz, DMSO-
6-Ethoxy-3-phenyl-[1,2,4]triazolo[4,3- b][1,2,4,5]tetrazine (
5g
)
Yellow solid, m.p. 120–122 °C; IR (KBr, cm−1): 3068, 2986, 1573, 1541, 1054, 747, and 705; 1H NMR (400 MHz, DMSO-
3-(4-Methoxyphenyl)-6-ethoxy-[1,2,4]triazolo[4,3- b][1,2, 4,5]tetrazine (
5h
)
Yellow solid, m.p. 143–145 °C; IR (KBr, cm−1): 3086, 2991, 1529, 1044, and 850; 1H NMR (400 MHz, DMSO-
3-Phenylethyl-6-methoxy-[1,2,4]triazolo[4,3- b][1,2,4,5] tetrazine (
5i
)
Yellow solid, m.p. 98–100 °C; IR (KBr, cm−1): 3098, 2945, 1579, 1551, 1041, 745, and 707; 1H NMR (400 MHz, DMSO-
3-Phenylethyl-6-ethoxyl-[1,2,4]triazolo[4,3- b][1,2,4,5] tetrazine (
5j
)
Yellow solid, m.p. 83–85 °C; IR (KBr, cm−1): 3024, 2999, 1601, 1541, 1037, 748, and 698; 1H NMR (400 MHz, DMSO-
Biological evaluation
In vitro cancer cell growth inhibition assay
The antiproliferative activities of the compounds
Molecular docking
Molecular docking was performed with the Surflex-Dock program interfaced with Sybyl-X 2.0. The programs adapted an empirical scoring function and a patented searching engine. 22 The ligand was docked into the corresponding protein binding site guided by protomol, which is an idealized representation of a ligand that makes every potential interaction with a binding site. In this work, the crystal structure of c-Met complexed with 2-benzyl-5-{4-[(6,7-dimethoxyquinolin-4-yl)oxy]-3-fluorophenyl}-3-methylpyrimidin-4(3H)-one (PDB entry: 3EFJ) was extracted from the Brookhaven Protein Database (PDB; http://www.rcsb.org/pdb). At the beginning of docking, all the water and ligands were removed and random hydrogen atoms were added. Next, the receptor structure was minimized over 10,000 cycles using the Powell method in Sybyl-X 2.0. All the compounds were constructed using a sketch molecular module. Hydrogen and Gasteiger-Hückel charges were added to every molecule. Then, their geometries were optimized by the conjugate gradient method in the TRIPOS force field. The energy convergence criterion is 0.001 kcal mol−1. Finally, ligand-based mode was adopted to generate the “protomol,” leaving the threshold at their default value of 1.
Conclusion
In summary, several of 6-alkoxy-3-aryl-[1,2,4]triazolo[4,3-
Supplemental Material
Supplementary_information-1 – Supplemental material for Synthesis and biological evaluation of novel 6-alkoxy-3-aryl-[1,2,4]triazolo[4,3-b][1,2,4,5] tetrazines
Supplemental material, Supplementary_information-1 for Synthesis and biological evaluation of novel 6-alkoxy-3-aryl-[1,2,4]triazolo[4,3-b][1,2,4,5] tetrazines by Run-Jie Shi, Zhen-Zhen Yang, Ye-Tao Gao, Wen-Jin Cai, Can Ye, Feng Xu and John Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Science and Technology Innovation project of college students in Zhejiang (2018R477002), the Zhejiang Public Welfare Technology Research Project (LGF19B020001), and the Research Project of Taizhou Vocational and Technical College (2016ZJ08).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

