Abstract
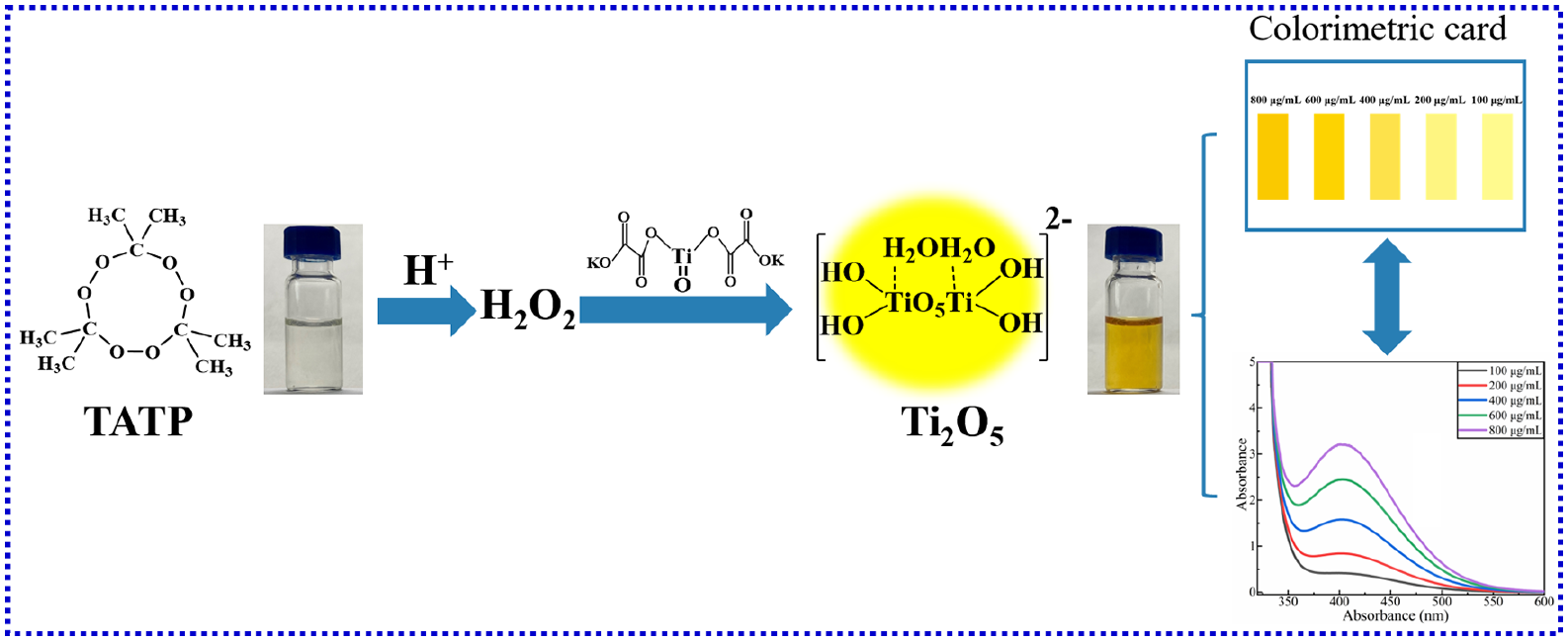
In this work, a visual colorimetric method for the rapid and selective detection of triacetone triperoxide is reported. This visual colorimetric method is based on the reaction between potassium titanyl oxalate and hydrogen peroxide (H2O2) released from triacetone triperoxide degradation. Potassium titanyl oxalate can selectively react with H2O2 to form peroxo-titanic acid (an orange complex), enabling the colorimetric detection of triacetone triperoxide. Based on the theory that triacetone triperoxide produces hydrogen peroxide under acidic conditions, acid types, acid concentration, response time, visual limit of detection, and reactants ratio are systematically studied simultaneously for this colorimetric method. Under sulfuric acid concentration is 60%, the proposed method can almost detect triacetone triperoxide instantly, and the color of the solution reaches the maximum within 1 min and remains stable with a visual limit of detection as low as 3.0 × 10−5 mol/L. Interference experiments were carried out on other kinds of explosives (hexamethylene triperoxide diamine, trinitrotoluene, etc.). The use of colorimetric card brings great convenience to the rapid, qualitative, and semi-quantitative on-site detection of triacetone triperoxide. Because of its rapidity, high sensitivity, simplicity, and selectivity, the proposed visual colorimetric method can serve as a valuable and promising reference for triacetone triperoxide’s rapid, qualitative on-site detection.
Keywords
A visual colorimetric method combined with visual colorimetric card was reported for the rapid and selective detection of triacetone triperoxide, which could serve as a useful and promising reference for the rapid and qualitative on-site detection of triacetone triperoxide because of its rapidity, high sensitivity, simplicity, and selectivity.
Introduction
Triacetone triperoxide (TATP) is one kind of home-made explosive, which was first synthesized in the 19th century. 1 It is commonly used in terrorist attacks due to its simple preparation, 2 easily available precursors, 3 and quite explosive power. TATP can be easily obtained by acetone, hydrogen peroxide (H2O2), and acid. For example, TATP appeared in the 2005 London Underground bombing, the 2009 Northwest Airlines plane bombing and the 2016 Brussels terrorist attack.4–7 In addition, TATP does not contain any chromophores such as nitro or aromatic functional groups and does not show ultraviolet absorption and fluorescent, which makes it difficult to be detected by conventional methods.8–10 Trinitrotoluene (TNT) equivalency was calculated from air blast models as 0.92 for TATP and the one from incident overpressure is 0.70.11,12 Besides, TATP can easily volatilize at room temperature and decompose at high temperatures, and it may disappear completely during transportation. Consequently, it is urgent to develop an effective method for the rapid and selective on-site detection of TATP.
Owing to its structure and properties, TATP cannot be detected directly by conventional methods such as ultraviolet-visible (UV-Vis) spectroscopy. 13 The used detection methods mainly include chromatography, mass spectrometry, surface-enhanced Raman scattering (SERS), colorimetric assay, ion mobility spectroscopy, fluorescence spectroscopy, electrochemical methods, and so on.14–19 Although mass spectrometry is a sensitive technique, its high cost and inflexibility make it not suitable for practical applications.20,21 SERS technology has the advantages of fast analysis speed, simple operation, and abundant spectral information. However, the cost is relatively high, and the preparation process of SERS substrates is complex, which also limits its practical application.22,23
Based on the fact that TATP can easily decompose in acidic solution, their decomposition products such as H2O2 or acetone can be used as alternative targets for forensic scientist to indirectly detect TATP. 24 Colorimetric methods, fluorescence spectroscopy, and electrochemical methods are commonly used indirect methods for TATP detection. The fluorescence sensor, which is used for indirect detection through H2O2 and acetone, has the advantages of high sensitivity, portability, and simple operation.25,26 However, the synthesis process of fluorescent sensors is relatively complex, which also limits their practical application.
Colorimetry is another simple technique for the rapid, qualitative, and selective on-site detection of peroxide explosives.27–37 Colorimetric methods mainly include traditional colorimetric methods, colorimetric sensors, and colorimetric sensor arrays. Traditional colorimetry mainly uses various chemical reagents to detect the TATP. The colorimetric reagents for detecting H2O2 are potassium iodide, potassium permanganate, potassium/ammonia titanyl oxalate, and so on. Previous studies have shown that potassium iodide is prone to false positive results. 38 Potassium permanganate is unstable, and its solution is difficult to store for a long time.39,40 Several studies have used titanyl-based materials to detect H2O2 and TATP,41–43 but they did not systematically study the impact of various factors. Furthermore, the reported colorimetric sensors were sensitive and portable. However, they either require a complex synthesis process or long response times. For the above reasons, they are not suitable for on-site use and promotion at the grassroots level.
In recent years, reports on the detection of TATP by colorimetry have mainly focused on colorimetric sensors and colorimetric sensor arrays.27,44 These methods have the advantages of high sensitivity, high selectivity, fast analysis speed, and provide reliable qualitative results. However, it requires the operator to have professional knowledge, which leads to its limited application.
Portable devices are suitable for TATP detection at crime scenes, but they are expensive, limiting their wide application. 22 Compared with portable devices, the colorimetric sensing method is simple, sensitive, specific, low-cost, and efficient, so it is incredibly suitable for detecting TATP at crime scenes. 45
Based on the above analysis, we chose the colorimetric method to detect TATP using potassium titanyl oxalate (PTO) as the colorimetric reagent. On the basis of previous studies, we systematically studied the influence of various influencing factors and the visual detection limit of the method on titanium oxalate salts. The response times of this method are much shorter than the previous reports. The proposed method can almost detect TATP instantly, and the color of the solution reaches the maximum within 1 min and remains stable with a visual limit of detection as low as 3.0 × 10−5 mol/L. What is more, the color card is made based on the visual detection results of the method for the rapid, qualitative, and semi-quantitative on-site detection of TATP. In brief, this method of detecting TATP has the advantages of simple operation, low-cost, high sensitivity and selectivity, and short response time. It is incredibly suitable for being widely applied at the grassroots level and lays a theoretical foundation for developing detection kits. These findings are very beneficial for the prevention and rapid qualitative analysis of real explosion cases.
Results and discussion
Degradation of TATP
It has been reported that TATP can be decomposed into H2O2 in acid.46,47 The decomposition mechanism is shown in Figure 1. According to this mechanism, we first tested the degradation efficiency of sulfuric acid (H2SO4), hydrochloric acid (HCl), formic acid (HCOOH), and acetic acid (CH3COOH) for TATP by comparing the color depth of the solution (Figure 2). PTO was added to the mixed solution of H2SO4 and acetone solution of TATP, and the solution turned yellow immediately. Under the conditions of sulfuric acid aqueous solution, TATP is decomposed into diacetone diperoxide (DADP) and H2O2. Partial DADP can be further decomposed into acetone and H2O2. PTO selectively reacts with H2O2 and forms peroxo-titanic acid (PTA, an orange complex), which makes the solution yellow.

Decomposition mechanism of TATP in acid.

Visualization results of TATP decomposition in different acids. The concentration of H2SO4 is 3 mol/L, and the concentration of HCl, HCOOH, CH3COOH is 6 mol/L.
An interesting phenomenon was observed in the presence of HCl. No noticeable color change was detected in the mixed solution when PTO was added to the mixed solution of HCl and acetone solution of TATP. By contrast, when acetone solution of TATP and PTO were first mixed, the color changed immediately with the addition of HCl. This phenomenon can be explained based on the decomposition mechanism in Figure 1. When HCl and TATP are mixed, TATP decomposes into acetone and H2O2. Then, HCl and H2O2 produce Cl2, which is rapidly dissolved in water to generate hypochlorous acid (HClO). Under acidic conditions, acetone transforms into an enol form. Finally, hypochlorous acid and enol form can continue to react to produce various forms of chloroacetone (C3H6-xClxO, 1⩽x⩽6).46,47 In the above process, acetone and H2O2 were consumed, and there is no remaining H2O2 and potassium titanate to form orange PTA. When TATP was added to the mixed solution of HCl and PTO, there was no time for the reaction described in the mechanism. H2O2 produced by TATP decomposition reacted with PTO and then produced a yellow complex instantaneously.
However, the solution has no obvious color change after the introduction of HCOOH or CH3COOH. Moreover, in the presence of HCOOH or CH3COOH, a large number of white acicular crystals were formed. The crystal precipitates were determined to be PTO by Fourier transform-infrared (FTIR) analysis. Based on the above results, H2SO4 and HCl were chosen for the subsequent experiment.
Influence of acid and TATP concentration
In the wavelength range of 190–600 nm, the UV-Vis absorption spectra of the reagents were recorded to determine the peak position of the target compound, orange PTA. As shown in Figure 3, all the reagents have an absorbance peak of approximately 200 nm. The absorption peak of PTA is observed at nearly 381 nm. Due to the n→π* electronic transitions of sulfur in H2SO4 and carbonyl in HCOOH, CH3COOH, acetone, and PTO, absorbance occurs at nearly 200 nm. However, the UV-Vis absorbance of H2O2 is attributed to n→σ* transition of lone pair electrons. The charge transfer transition may be attributed to the interaction between chloride ion and water molecule for HCl. TATP has no UV absorption. At 381 nm, PTA shows UV absorbance due to the electron transfer of the peroxy bond in the ligand to Ti4+. The absorption peak at 381 nm is unique to the reaction solution and does not exist in other reagents. Therefore, the intensity of the peak at 381 nm is used to evaluate the influence of various factors on the experiment.

UV-Vis absorbance of various reagents.
Due to the existence of acetone in the reaction solution and different water content in each concentration of acid, the solutions have different polarities, which leads to a redshift in the absorption peak of PTA, as shown in Figure 4. Therefore, the highest absorbance was selected for further analysis. According to Figure 5, the absorbance is positively correlated with the concentration of TATP and acid. As for H2SO4, the absorbance increases with the increase in the concentration of TATP and acid, but it decreases when the concentration exceeds 60%. This may be because the high concentration of H2SO4 leads to a decrease in the content of water and free H+, which weakens their effect. By contrast, as for HCl, the absorbance increases with the increase in the concentration of TATP and HCl. Furthermore, the absorbance never decreases because the acid concentration is too high. Overall, it can be concluded that when H2SO4 is used, its concentration should not exceed 60%, but HCl has no such concentration limit.

Red shift of absorption peak for 800 μg/mL of TATP. (a) UV-Vis curves at different concentrations of H2SO4. (b) UV-Vis curves at different concentrations of HCl.

Influence of acid and TATP concentration on this colorimetric method. (a) UV-Vis curves at different concentrations of TATP and H2SO4. (b) UV-Vis curves at different concentrations of TATP and HCl.
Effect of the molar ratio of PTO to TATP
According to the possible synthesis mechanism of PTA, a series of experiments were designed to determine the optimal proportion of PTO and TATP. The formation process of PTA is shown in Figure 6(a). After the hydrolysis of PTO, Ti4+ ① was formed. Ti4+ ① continuously reacted with H2O2 to form PTA ③. Two PTA ③ was polymerized into product ④.48,49 In the entire process, one Ti4+ consumes five hydrogen peroxides (Figure 6(a)). As shown in Figure 1, one TATP generates three hydrogen peroxides. Based on the above two mechanisms, the molar concentration of PTO was selected as 0.75, 1.5, 3, 6, 12, 18, 24, 30 times that of TATP (PTO: H2O2=0.25, 0.5, 1, 2, 4, 6, 8, 10), and the proportion of reagents was examined to obtain the optimal visual results. It can be seen in Figure 6(b) that when the molar ratio is less than 24 (PTO: H2O2 = 6:1), the absorbance increases gradually. When the molar ratio exceeded 24, the absorbance was stable and reached its maximum. The results were independent of the type of acid. These experimental results indicated that when the molar ratio of PTO and TATP was 24 or higher, the absorbance reached the maximum.
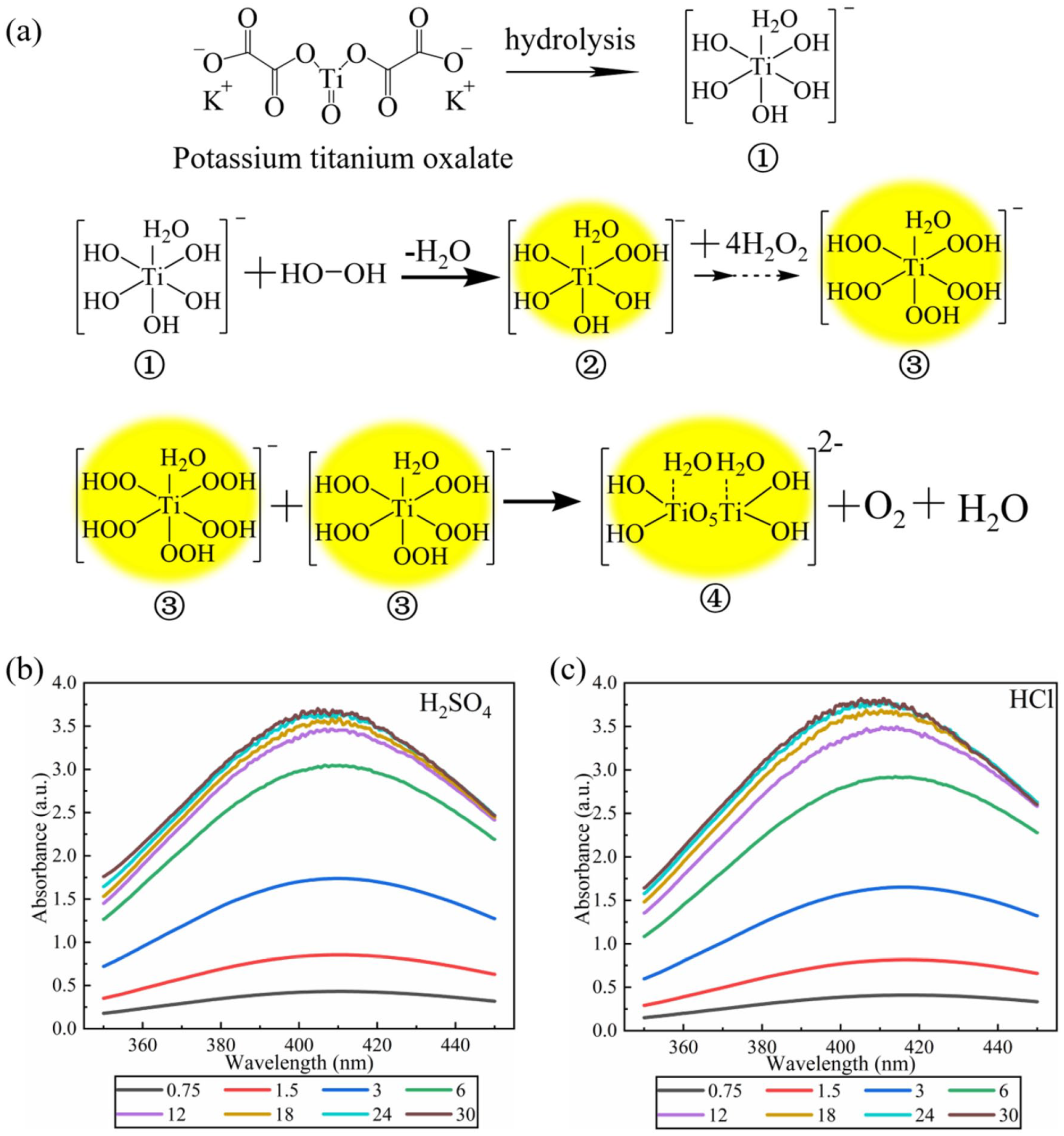
(a) Reaction mechanism of PTO with H2O2. (b) UV-Vis curves at different PTO/TATP mole ratio with H2SO4 as the initiator. (c) UV-Vis curves at different PTO/TATP mole ratio with HCl as the initiator.
Effect of reaction time
Figure 7(a) shows that in the experimental group with H2SO4, the reaction rate was low when the acid concentration was less than 20%. The reaction rate was significantly accelerated when the acid concentration exceeded 30%. When the acid concentration reached 70%, the absorbance became stable within 1 min, indicating that TATP was completely decomposed. When the absorbance of the reaction solution was stable, it was confirmed that there was no TATP in the reaction solution based on SPME-GC-MS analysis results according to the method Anikó Kende reported, 14 indicating TATP had been almost completely decomposed. In the experimental group with HCl (Figure 7(b)), the reaction rate uniformly increased with the increasing of acid concentration. When the acid concentration was 20%, TATP was almost completely decomposed within 10 min. When the acid concentration exceeded 30%, the reaction rate remained almost unchanged. When the acid concentration exceeded 35%, the absorbance became stable within 1 min, which suggested that TATP was completely decomposed. To achieve the same reaction rate, the concentration of H2SO4 must be much higher than that of HCl. Moreover, the concentrated H2SO4 was extremely dehydrating and dangerous. Therefore, HCl is a better choice. However, when HCl is used, the addition order of reagents should be highly considered.

Relationship between the time and UV-Vis absorbance. (a) UV-Vis curves of solution with time under different concentrations of H2SO4. (b) UV-Vis curves of solution with time under different concentrations of HCl.
Determination of visual detection limit
The previous experimental results suggested that 60% of H2SO4 and 30% of HCl provide the highest reaction rate, so these concentrations were selected for the subsequent experiment to determine the visual detection limit. As shown in Figure 8(a) and (b), the same phenomenon was found in the two experimental groups. The color became lighter with the decrease in the TATP concentration. According to the visual observation results of the experimental group with H2SO4, when the concentration of TATP was as low as 20 μg/mL, the noticeable yellow solution could still be found. However, when the concentration reached 2 and 5 μg/mL, the solution was nearly colorless. When the concentration was 10 μg/mL, the color of the solution showed a negligible change. As for HCl groups, they showed similar results under different concentrations of TATP solution. However, compared with the H2SO4 group, the color of the HCl group was lighter. It can be concluded that the minimum concentration of TATP that can be detected by visual observation under the condition of 60% of H2SO4 and 30% of HCl was 20 μg/mL.

Visual results of solutions with different TATP concentrations. (a) Visual results of solutions with different TATP concentrations under 60% H2SO4. (b) Visual results of solutions with different TATP concentrations under 30% HCl. (c) Colorimetric card made according to the visual results of 7(a) and (b).
In fact, the color change of the colorimetric experiment was not very convenient for on-site investigation. Therefore, we have standardized the above experimental results. Based on the visual results of the detection limit, the photos “before” and “after” the colorimetric experiment were obtained. According to the “three color” principle of Thomas Young, colorimetric change values of red, green, and blue (
Calibration curves for TATP determination
The last step of the experiment mainly focuses on the rapid and qualitative on-site detection of TATP at the crime scene. A correction curve should be established to realize the quantitative detection of TATP. Due to the red shift of absorption, the absorbance data at 404 nm are selected, and the results are shown in Figure 9. In the experimental group with H2SO4, the absorbance has a good linear relationship with TATP concentration as below,

Calibration curves for the measurement of TATP concentration. The red curve is the calibration curve of H2SO4 experimental group. The black curve is the calibration curve of HCl experimental group.
Specificity of the colorimetric method
The color of the interfering substance solution is shown in Figure 10(a), the solutions of 2,4-dinitrophenol (DNP) and 2,4,6-trinitrophenol (PA) were yellow, so they could be identified first and eliminated from interference. The other interfering substance has no obvious color. Then the saturated PTO and acid were added to the above solution, and the results are shown in Figure 10(b). As shown in Figure 10(b), the color of the solution in the experimental group of interferents did not change significantly except for the hexamethylene triperoxide diamine (HMTD) group. However, when 8% H2SO4 solution was used as initiator, the acetone solution of HMTD (200 μg/mL) quickly turned yellow within 1 min, and TATP solution (200 μg/mL) had no obvious color change (Figure 10(d)) within 20 min. In conclusion, the discrimination between TATP and HMTD can be achieved based on the visual colorimetric method.

Specificity of the colorimetric method toward to TATP. (a) Color of interfering substance solution. (b) Color of solutions after adding potassium titanium oxalate and acid. (c) UV-Vis absorbance data of Figure 9(b) at 400nm. (d) Visualization results of 8% sulfuric acid solution was used as initiator to distinguish TATP and HMTD.
Determination of TATP in actual samples
The applications of the reported method were appraised for the measurement of TATP in several real samples, including tap water, river water, soil, and the results of the experiments are summarized in Table 1. It suggests that the spiked recoveries for TATP through the current method were in the range of 96.6%–100.8%, and the relative standard deviation (RSD) was calculated to be 0.4%–4.0%. These results of the study demonstrate that the developed method has reliability and high accuracy. Therefore, it will exhibit bright prospects in the TATP assay of actual samples.
Results of the recovery of TATP from real samples.

TATP: triacetone triperoxide; RSD: relative standard deviation.
Experimental
Reagents and chemicals
Acetone (C3H6O, 99.7%), PTO (C4K2O9Ti·2H2O, ⩾98.5%), sulfuric acid (H2SO4, 95.0%–98.0%), hydrochloric acid (HCl, 36.0%–38.0%), hydrogen peroxide (H2O2, 30%), and acetic acid (C2H4O2, ⩾99.8%) were purchased from Sinopharm Chemical Reagent Co., Ltd. Formic acid (CH2O2, 88%) was purchased from Thermo Fisher Technology (China) Co., Ltd. Distilled water with a conductivity of 18.2 mΩ·cm was prepared in a Millipore purification system. All the chemical reagents were of analytical grade or guaranteed reagents. TATP was synthesized following the established procedures and washed to neutral without recrystallization, which included little DADP as a byproduct.50,51 1,3,5-trinitrobenzene (TNB), PA, pentaerythritol tetranitrate (PETN), 1,2,3-trinitroxypropane (NG), DNP, 1,3,5-trinitro-1,3,5-triazinane (RDX), 1,3,5,7-tetranitro-1,3,5,7-tetraazacyclooctane (HMX), 2,4,6-trinitrotoluene (TNT), 2,4-dinitrotoluene (DNT), HMTD were dissolved in acetone. Warning: TATP is an extremely sensitive organic peroxide with huge explosive power, so safety problems should be paid attention to in the process of synthesis, treatment, and storage.
Scheme of color reaction
Since H2O2 is the raw material of TATP synthesis and the detection target of the chemical color reaction, the PTO solution should be added to the sample solution first to confirm whether the sample contains unreacted H2O2. Based on the mechanism in Figure 1, the color of the mixed solution will quickly turn yellow when the sample contains residual H2O2. When it is determined that there is no H2O2 in the sample, acid is subsequently added to the mixed solution as an initiator. 1 mL of PTO solution was added to 1 mL of TATP acetone solution in a centrifuge tube. 1 mL of acid solution was then added for reaction. Unless otherwise specified, the PTO solution was saturated and the reaction time was 3 min. Then the mixed solution was transferred to a sample vial or a cuvette immediately. The visualization results of the mixed solution were recorded by the Canon EOS 6D camera.
Degradation of TATP
Aqueous solutions of sulfuric acid, hydrochloric acid, formic acid, and acetic acid with H+ concentration of 6.0 mol/L were used for the decomposition of TATP, where 1 mg/mL acetone solution of TATP was used. Based on the degree of color change, the decomposition effect of four kinds of acids on TATP was verified, and the acid with the highest decomposition rate was selected for the experiment because the presence of TATP needs to be rapidly confirmed at the crime scenes.
Influence of acid and TATP concentration
To study the influence of acid and TATP concentration on the chromotest experiment, it was necessary to determine the UV absorption peak of PTA. Appropriate amounts of acetone, PTO, sulfuric acid, hydrochloric acid, formic acid, acetic acid, and H2O2 were dissolved in deionized water to examine their UV-Vis absorption at 190–600 nm. The UV-Vis absorption of the reaction solution of H2O2 and PTO was also recorded at 190–600 nm to determine the absorption peak position of PTA.
A series of 70%, 60%, 50%, 40%, 30%, 20%, 10%, and 8% sulfuric acid solutions and 38%, 35%, 30%, 25%, 20%, 15%, 10%, 5% hydrochloric acid solutions were prepared. Under specific acid concentration, 800, 600, 400, 200, and 100 μg/mL TATP acetone solutions were tested. The solutions were mixed according to the scheme of color reaction. After 3 min, the mixed solutions were transferred to a cuvette and the UV-Vis absorbance spectra of these solutions were recorded at 400 nm.
Effect of the molar ratio of PTO to TATP
The concentrations of hydrochloric acid, sulfuric acid, and TATP-acetone solution were 30%, 60%, and 800 μg/mL (3.60 × 10−3 mmol/mL), respectively. The molar concentration of PTO was 0.75, 1.5, 3, 6, 12, 18, 24, and 30 times (PTO: H2O2=0.25, 0.5, 1, 2, 4, 6, 8, and 10) that of TATP. The solutions were mixed according to the scheme of color reaction. After 3 min, the mixed solutions were transferred to a cuvette, and the UV-Vis absorbance spectra of these solutions were recorded at 350–450 nm.
Effect of reaction time
The above concentrations of sulfuric acid and hydrochloric acid solutions were examined, and the concentration of TATP acetone solution was 800 μg/mL. The mixed solutions were immediately transferred into a cuvette to examine the variation in the UV-Vis absorbance with time at 400 nm. The absorbance spectrum was recorded every 5 s for 10 min.
Determination of visual detection limit
In order to determine the visual detection limit, 1 mL of 60% sulfuric acid and 1 mL of 30% hydrochloric acid reacted with 100, 80, 60, 40, 20, 10, 5, and 2 μg/mL TATP acetone solutions. 1 mL of saturated PTO solution was added. After 3 min, the pictures were recorded and the color depth was analyzed visually.
Calibration curves for TATP determination
One milliliter of 60% sulfuric acid and 1 mL of 30% hydrochloric acid reacted with 800, 600, 400, 200, and 100 μg/mL TATP acetone solutions. One milliliter of saturated PTO solution was added. After 3 min, the mixed solutions were transferred to a cuvette, and the UV-Vis absorbance of the solution was recorded at 400 nm.
Specificity of the colorimetric method
Selectivity of the colorimetric method is also critical in achieving accurate detection of TATP. Consequently, the specificity of the method was evaluated by the execution of different control experiments using other explosives as the interfering substances (TNB, PA, PETN, NG, DNP, RDX, HMX, TNT, DNT, and HMTD). The concentration of the acetone solution of interferents was 200 μg/mL. In both groups, 60% H2SO4 was used as the reaction initiator. In order to effectively distinguish TATP and HMTD, an 8% sulfuric acid solution was also selected as the initiator for the HMTD group. The solutions were mixed according to the scheme of color reaction. The color change of mixed solutions was observed and recorded.
Determination of TATP in actual samples
Considering the actual application of the method, two water samples were obtained from a local river and tap water, while soil samples were taken from local parks. TATP powder was added to the above actual samples, and acetone was added after thorough mixing, so that the concentrations of TATP in the solution were 50, 100, and 200 μg/mL, respectively. After ultrasonication for about 3 min at room temperature, the solution was filtered by a 0.22 μm membrane. The solutions were mixed according to the scheme of color reaction. After 3 min, the mixed solutions were transferred to a cuvette, and the UV-Vis absorbance spectra of these solutions were recorded at 400 nm.
Characterizations
The UV-Vis absorbance spectra were recorded by the Shimadzu UV-2550 spectrophotometer. The absorbance spectra in the wavelength range of 190–600 nm were recorded by the spectrum module, while the absorbance versus time curves were recorded by the kinetic module. The UV absorption curves and other data curves were processed and obtained by Origin 2018. All of the pictures recorded by the camera were processed and cropped without other processing by Photoshop CS2.
Conclusion
In this study, a rapid, sensitive, simple, low-cost, and visual method was established for the quantitative and qualitative on-site detection of TATP. This method is based on the fact that TATP can decompose to produce H2O2 in the presence of acid, and H2O2 can selectively react with PTO to form PTA (an orange complex). Notably, the visual limit of detection of the proposed method was as low as 20 μg/mL (3.0 × 10−5 mol/L). The results verified that the proposed method could be effectively used for rapid, qualitative, semi-quantitative, quantitative, and accurate quantitative detection of TATP in the presence of H2SO4 and HCl. The most ideal acid is sulfuric acid, the optimum sulfuric acid concentration is 60%. When using sulfuric acid, there is no requirement for the use sequence of reagents. However, when using hydrochloric acid, it is necessary to pay attention to the use sequence of reagents, which increases the difficulty of on-site use. The proposed method for detecting TATP possesses several unique merits. First, it is extremely simple, low-cost, and convenient. The testing equipment and pre-synthesis of some materials are unnecessary, and the test can be simply completed by mixing several commercially available reagents without complex operations. In addition, the response time of this method is short. The proposed method can almost detect TATP instantly, and the color of the solution reaches its maximum within 1 min. Furthermore, a colorimetric card was developed based on the experimental results for the qualitative and semi-quantitative on-site detection of TATP. Therefore, the proposed method has great application potential in preventing terrorist attacks and bombing cases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the China National Key R&D Program (No. 25901) and the Public Security Theory and Soft Science Research Program (No. 2019LLYJWZZX073).
