Abstract
Fibres from a purple 1860s Victorian-era silk dress were extracted and analysed by liquid chromatography–mass spectrometry. The analysis was predominantly mauveine A and mauveine B with lesser amounts of other mauveine chromophores. The mauveine provenance of the dress was confirmed and the authenticity of the mauveine established as from WH Perkins Greenford factory by comparison with museum standards. Fibres from a 25-year-old silk bow tie were also analysed by liquid chromatography–mass spectrometry, which analysed as predominantly mauveine A with a much smaller amount of mauveine B. Strips of silk were dyed with museum-stored mauveine, and after leaving for 6 months in the light or the dark, the same liquid chromatography–mass spectrometry mauveine analysis shows that mauveine B was not degrading to mauveine A on silk.
Keywords

Introduction
WH Perkin made his accidental discovery of mauve 1 in 1856 and this was followed by H Caro’s method 2 in 1860. WH Perkin had financial assistance from his father to build a factory and exploit the discovery and develop the British coal-tar dye industry.3,4 Up to this point colours came from natural sources and were less stable and not readily available. 5 Mauve was the first synthetic aniline dye to be commercialised. 6 The development relied upon quantities of coal-tar which was produced as a by-product of the illuminating gas industry. The pyrolysis of coal or oil gave illuminating gas for lighting but created by-products from which benzene and toluene could be distilled. It was Michael Faraday in 1825 who was given a clear liquid by the Portable Gas Company which enabled him to discover benzene. 7 The benzene from coal-tar distillation contained about 10% toluene which could not be separated; so when it was nitrated and reduced to aniline, it always contained a small amount of ortho and para-toluidine. 8 WH Perkin’s original source of small quantities of aniline 9 was probably based on AW Hofmanns 10 and FF Runges work. 11 Aniline dyes have a signature from this mixture of amines which can indicate provenance and purity of the aniline. These building blocks can be located in the mauveine chromophore.12,13 For example, the bottle of mauveine from H Caro in the Deutsche Museum 14 and Schnucks mauveine in the Manchester Museum of Science and Industry is largely pseudomauveine indicating that purer aniline was used for its synthesis. 15 However, WH Perkin used toluidine-rich mixtures of aniline to make mauveine as the yields are improved. 8 When repeated today and analysed by liquid chromatography–mass spectrometry (LC-MS), this experiment gives four mauveine chromophores assigned as A, B, B2 and C (Figure 1 and Table 1).16,17
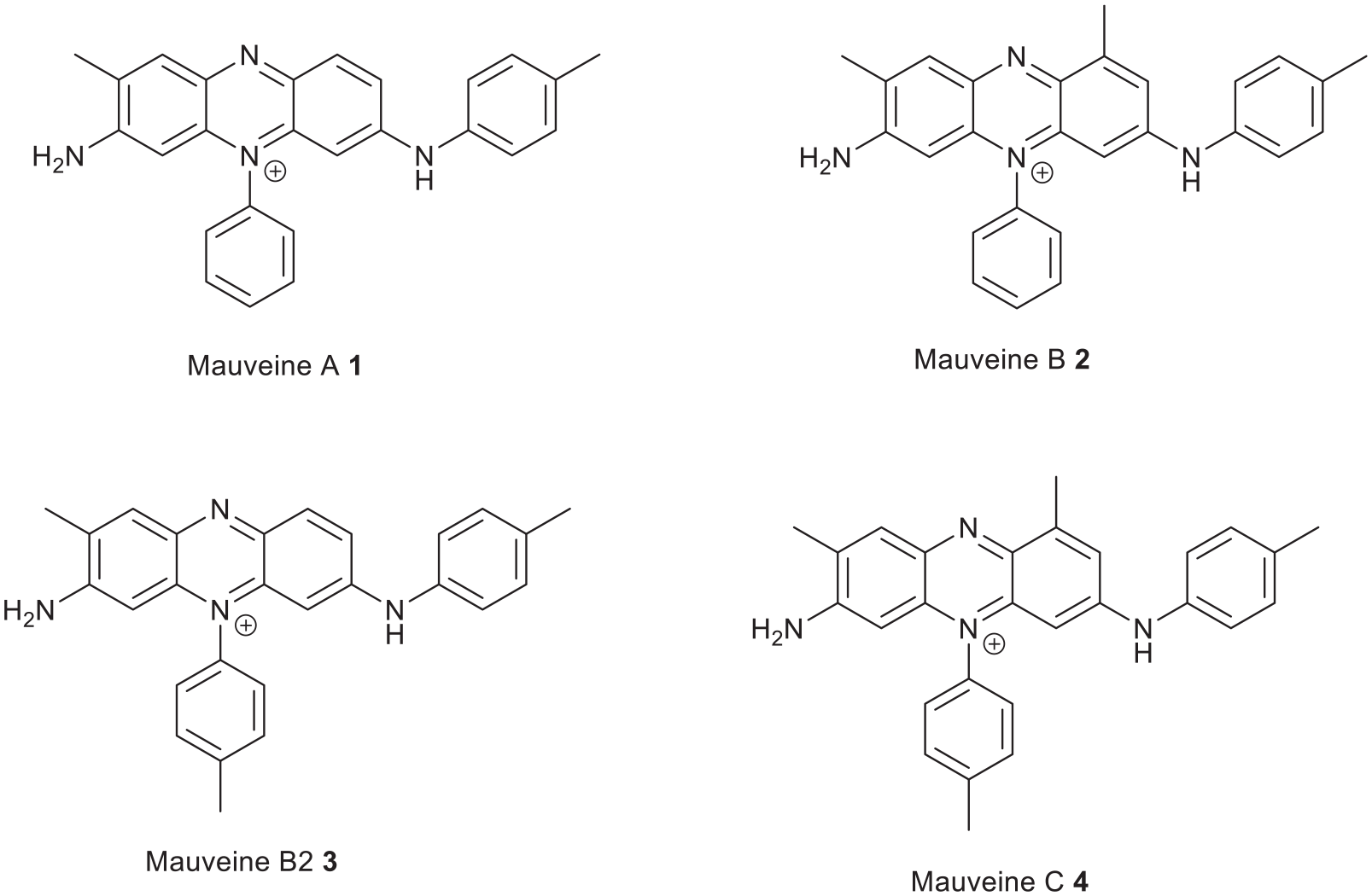
Structure of mauveine A, B, B2 and C.
Mauveine chromophores: name, formulae and molecular weights.
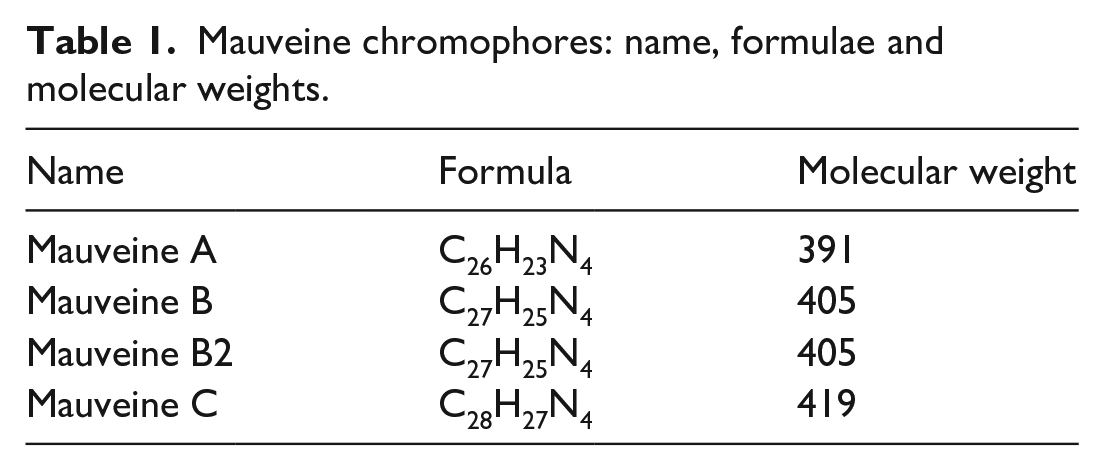
These have been characterised by spectroscopic methods.12,18–22 A crystal structure determination proved the structure of pseudomauveine (mauveine A1 lacking methyl groups) and a mauveine A isomer.
21
Their occurrence was explained by suggesting that one common intermediate leads to mauveine A and B2 and that another common intermediate leads to mauveine B and C
23
(Figure 2). We showed how the ratio of aniline to p-toluidine influences the chromophore ratio in both pairs. It was shown by LC-MS that more p-toluidine over aniline can suppress the formation of mauveine A and B just leaving mauveine B2 and C.
24
In another run, an excess of aniline, with less o-toluidine, favoured mauveine A and B2, suggesting common intermediate
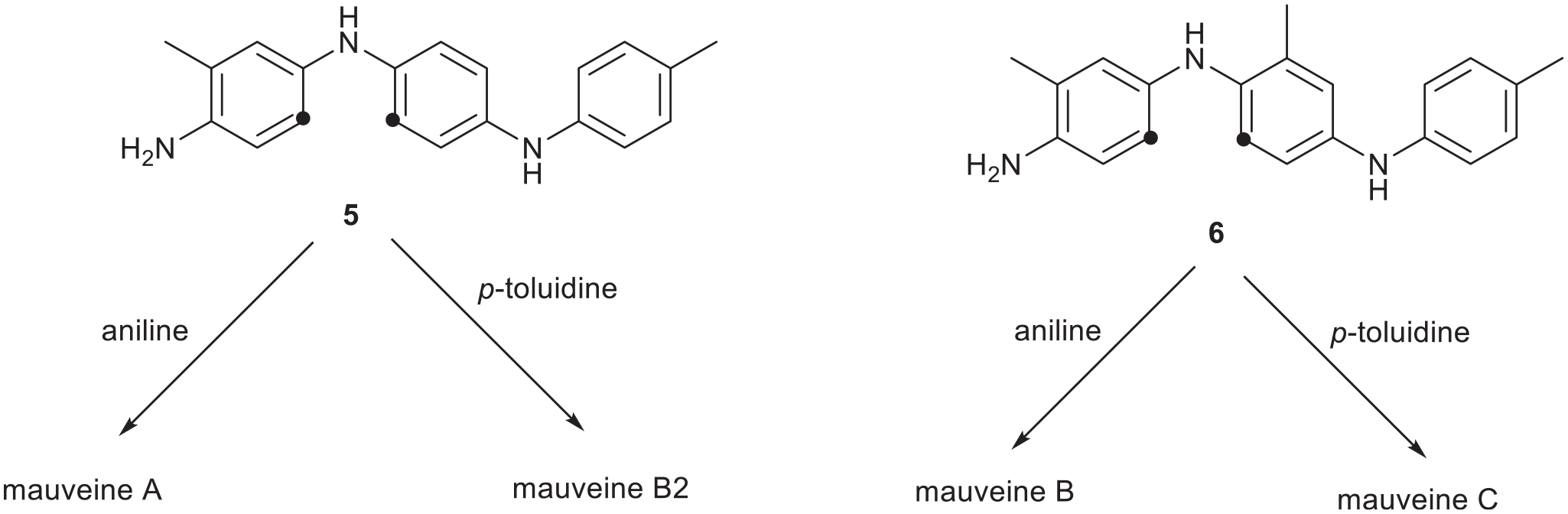
A summary of some possible common intermediates leading to different mauveines. The dots indicate where an amine would cyclise.
However, WH Perkin’s mauveine, now stored in a number of locations, has a very different hallmark.26–31 It is only mauveine A and B, so WH Perkin appears to have improved his method to select two chromophores over four without publishing this. Traces of modified mauveine intermediates have been detected in black precipitate. 32 With this knowledge of mauveine provenance, 33 we have analysed the mauveine extracted from a silk dress 34 and from a more recent silk bow tie. 35 This may be the first fully dyed dress to be analysed and it comes from the right era. 30 One previous mauveine containing dress has been identified. 36 The mauveine signature can help us decide if a dress item was made from WH Perkin’s mauveine and hence not only confirm if it is dyed with mauveine but also approximate its date of manufacture.
Discussion
Mauveine analysis by LC-MS
The LC-MS charts show the data from the mauveine analysis of the dress fibres, silk bow tie and a control piece of silk. The senior curator of a museum kindly provided dress fibres from a Victorian dress and retired scientist turned professional artist O Meth-Cohn kindly provided silk fibres from a 25-year-old bow tie (Figure 3). The supplementary letters explain how the dress was uncovered. Dilute solutions of mauveine in MeOH were obtained by extracting fibres in a vial with MeOH/TFA then evaporating to dryness. Trifluoroacetic is used because it is a strong acid, non-nucleophilic and volatile. A dilute solution of the mauveine extract in methanol is injected into the instrument which elutes the mauveine down a reverse phase silica column. This separates the chromophores by their size. The smallest elute first followed by the larger ones which spend longer in the column pores. Hence pseudomauveine elutes first, then monomethyl mauveines, then mauveine A, then B and then C. They are detected by a 550 nm detector which matches to their ultraviolet-visible (UV-Vis) absorption spectrum. The horizontal scale is the plot of the time taken for the mauveine chromophores to elute. There are two vertical scales. One is for the UV-Vis absorption (mAU or milli Atomic Units) and one is for the mass spectrometer (ESMS or Electro Spray Mass Spectral counts) which records the molecular weight and accurate mass. This is helpful to characterise the compound and is colour coded. Four key mauveine chromophores are shown in Figure 1 although many more occur in small amounts.

Left: A mauve shade silk dress from the Victorian era in Gunnersbury Park and Museum. Right: A Charles Rees mauveine dyed bow tie worn by Charles Rees.
Analysis of mauveine extracted from a Victorian silk dress
Figure 4 shows an LC-MS chart for the analysis of mauveine extracted from silk fibres taken from a dress of the Victorian-era house in Gunnersbury Park and Museum. Table 2 shows the mauveine relative peak areas. The sample is very dilute as only a few silk fibres were extracted. Microscopic examination of the extracted fibres revealed that all the dark coloured dye had been extracted. The sample showed a pale colouration to the naked eye, but no UV-Vis absorption was detected. This is the black line in Figure 4. For this reason, the UV-Vis curve is removed in Figure 5, which makes the chart easier to see. The mass spectrometer clearly detected both mauveine A (orange peak at m/z 391) and mauveine B (green peak at m/z 405). These chromophores are drawn in the introduction. Other minor chromophores were detected such as C group chromophores (blue line at m/z 419) and a monomethyl mauveine (red line at m/z 377). The dress is dyed with mauveine and outside of this range no other chromophores were detected. These data agree with the provenance of the museum for the origins of the dress. The predominance of both mauveine A and B chromophores is typical of mauveine stored in a number of museums that was made in WH Perkins factory. The only other UK mauveine manufacturer was H Caro, but his bottled mauveine and possibly that found in some unusual stamps was of a very different composition.14,17

Liquid chromatography–mass spectrometry (LC-MS) chart for the analysis of mauveine extracted from fibres from a Victorian dress in Gunnersbury Park and Museum. The black line is the UV-Vis absorption which was not detected here because of the dilute sample.
Relative peak areas for mauveine A and mauveine B in Figure 4.


The same as in Figure 4, but without the UV-Vis absorption.
The orange and green lines are owing to mauveine detected by the mass spectrometer (orange: mauveine A and green: mauveine B). Other minor mauveine chromophores are present.
Analysis of mauveine extracted from a 25-year-old silk bow tie
Figure 6 shows an LC-MS chart for the analysis of mauveine extracted from silk fibres taken from a 25-year-old bow tie. The bow tie was dyed with museum-stored mauveine with a typical composition of mauveine A and B. Table 3 shows the relative peak areas. In 1994, four of these bow ties were made from silk and museum-housed mauveine. Figure 3 shows a well-known recipient wearing one of them. The LC-MS chart was unusual because instead of the roughly equal ratio of mauveine A to mauveine B expected, or the slight predominance of mauveine A, mauveine A predominates over mauveine B by about 4:1. The dye may not have all extracted from the silk fibres which could explain this result or the fibres are not evenly dyed. Mauveine B is still the second-most-intense chromophore present. Some control experiments were set up to verify the result.

Liquid chromatography–mass spectrometry (LC-MS) chart for the analysis of mauveine extracted from silk fibres from a 25-year-old bow tie.
Relative peak areas for mauveine chromophores in Figure 6.

Analysis of mauveine extracted from silk stored in the daylight or dark
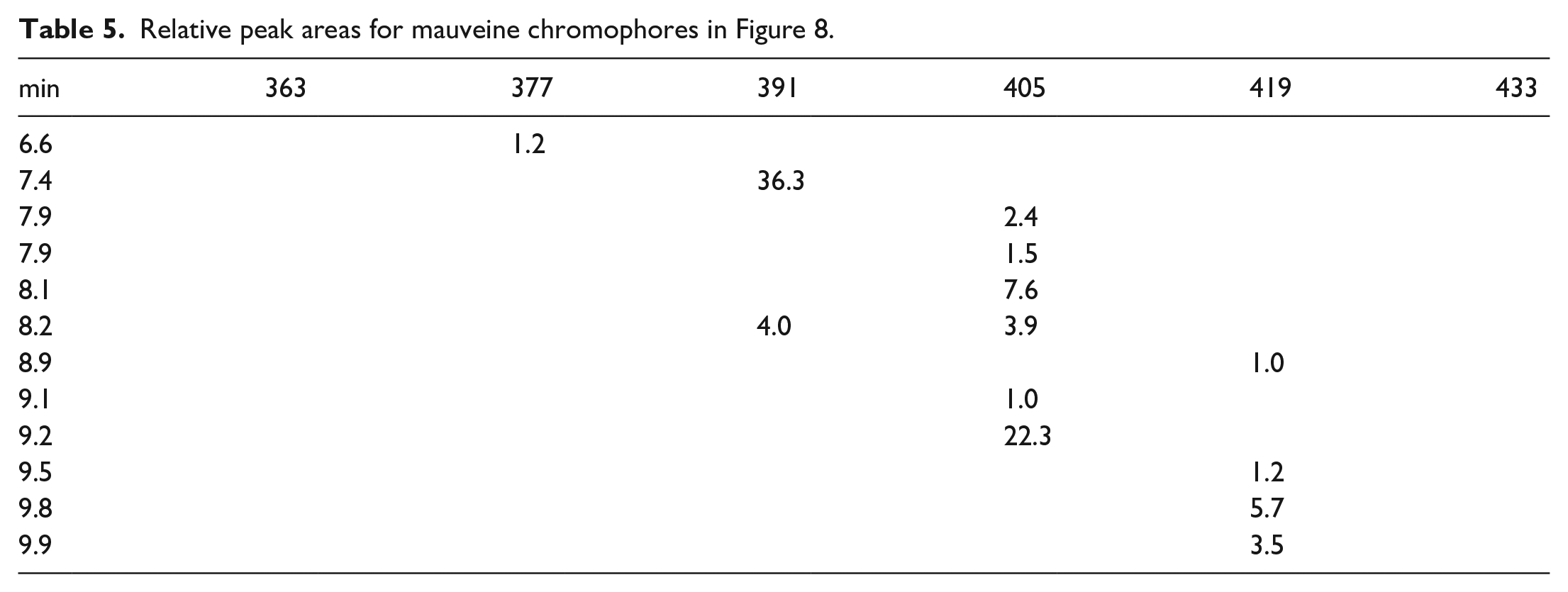
Owing to the predominance of mauveine A over mauveine B in the extracted fibres from the bow tie a silk strip was dyed with museum-stored mauveine. This was cut in half and one half was left in daylight for 6 months and one half was left in the dark for 6 months. Figure 7 and Table 4 show data for the silk strip left in daylight for 6 months and Figure 8 and Table 5 show data for the silk strip left in the dark for 6 months. Both of the LC-MS charts look the same; so this shows that light-catalysed decomposition of either mauveine A or B is not occurring. Oxidative demethylation of mauveine B into mauveine A does not occur on silk. In Supplemental Material, we showed that silk drying for 2 days after dyeing gives the same analysis (Figure 1 SI and Table 1 SI), that TFA-treated mauveine is not decomposed, (Figure 2 SI and Table 2 SI) and a check on the museum-stored mauveine (Figure 3 SI and Table 3).

Liquid chromatography–mass spectrometry (LC-MS) chart for the analysis of mauveine extracted from fibres from a dyed piece of silk left in the daylight for 6 months.
Relative peak areas for mauveine chromophores in Figure 7.
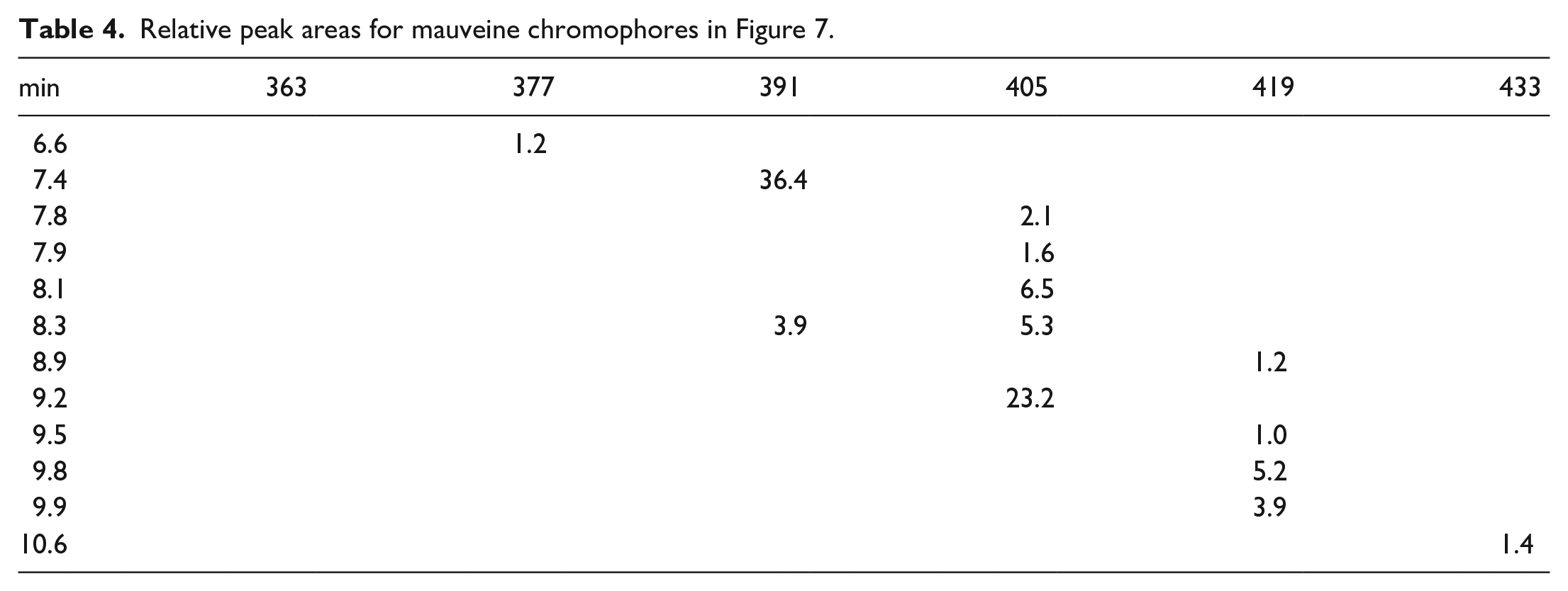

Liquid chromatography–mass spectrometry (LC-MS) chart for the analysis of mauveine extracted from fibres from a dyed piece of silk left in the dark for 6 months.
Relative peak areas for mauveine chromophores in Figure 8.
Conclusion
A silk dress from the Victorian-era dated approximately 1860 has been examined for its mauveine status. Fibres were collected by a senior curator, which were successfully posted in a small freezer bag. The fibres were extracted with a MeOH/TFA extractant at room temperature for 24 h followed by evaporation. TFA was used as an extractant because it is a strong acid, non-nucleophilic and volatile. The dry extract, with only faint purple colouration, was posted and then treated with a small quantity of MeOH. The extract solution was analysed by LC-MS as detailed in the “Experimental” section. The solution was very dilute and mauveine was not detected at 550 nm, but it was detected by the mass spectrometer. It consisted mainly of mauveine A with some mauveine B. The provenance of this dress as a mauveine-dyed silk dress is verified and the mauveine will have come from WH Perkins local Greenford factory as this type of composition rich in mauveine A and B is a trademark or hallmark of mauveine from his factory. Its composition resembles the many bottles of mauve stored in different museums. The fibres from a 25-year-old silk bow tie and related pieces of silk were treated in the same manner and extracted the same way. The mauveine analysis for the silk bow tie was not expected as the ratio of mauveine A/B should have been lower. It is known that the museum-stored mauveine which it was dyed with has a 1:1 composition of mauveine A and B. The investigator at that time was working on the separation of mauveine A from B, but is clear that a separated mixture was not used for any silk bow tie. Perhaps mauveine B was not extracted if it binds more strongly to the silk. The experiments on mauveine dyed silk pieces verify that mauveine B is not noticeably decomposed by light so this is unlikely to be a pathway here.
Experimental
LC-MS
For analytical separation, a Dionex 3000 system consisting of a binary HPLC-pump, cooled autosampler compartment, column oven and diode-array UV-Vis detector was used. A Kintex F5 column (150 × 2.1 mm, Phenomenex, UK) was used for separation with a water/acetonitrile (ACN) gradient (both 0.1% v/v formic acid) from 20% to 50% ACN in 10 min with a flow rate of 0.5 mL/min, column temperature 40 °C. The sample volume was 5 µL. The mass spectrometer (ESMS) used was an Exactive Orbitrap from Thermo Scientific with HESI-source connected to the UV-Vis detector (set to 550 nm) by a short length of PEEK-tubing. The ESMS was operated in positive-ion mode with a capillary voltage of 4.5 kV, S-lens RF level of 80, capillary temperature of 300 °C and a Probe heater temperature of 350 °C. Fragmentation for MS² was done in automatic mode. The ESMS was mass-calibrated and optimised as required. Exact mass extracted ion chromatograms were extracted manually with 5 ppm error.
Silk extraction
A few silk fibres from the dress or bow tie were treated with MeOH (2 mL) and trifluoroacetic acid (TFA) (3–4 drops) and sealed for 24 h. The solution was then allowed to evaporate and the sample posted for analysis dry. On receipt, it was diluted with a small amount of MeOH and analysed. TFA is used for the extraction because it is a strong, volatile and non-nucleophilic acid.
Supplemental Material
sj-pdf-1-chl-10.1177_17475198221103999 – Supplemental material for Liquid chromatography–mass spectrometry analysis of mauveine extracted from silk fabrics: A Victorian dress in Gunnersbury Park and Museum and a modern Charles Rees bow tie
Supplemental material, sj-pdf-1-chl-10.1177_17475198221103999 for Liquid chromatography–mass spectrometry analysis of mauveine extracted from silk fabrics: A Victorian dress in Gunnersbury Park and Museum and a modern Charles Rees bow tie by M John Plater and Andrea Raab in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are grateful to the senior curator of Gunnersbury Park and Museum for fibres of a Victorian-era silk dress and to Professor Otto Meth-Cohn for fibres of a silk bow tie.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
