Abstract
This paper covers the synthesis, in vitro urease inhibition, enzyme kinetic parameters, and anti-oxidant studies of a novel series of quinazolinone derivatives containing an acyl hydrazone skeleton. Compounds

Introduction
The urease enzyme, EC 3.5.1.5, which belongs to the super family of amidohydrolases and phosphotriesterases, catalyzes the hydrolysis of urea into ammonia and a carbamate in micro-organisms, animals and various plant species.1,2 This reaction promoted by the urease enzyme, results in a sudden increase in the overall pH, which causes negative effects on the health of animals and humans. The enzyme activity has been shown to have a link with several clinical conditions such as pyelonephritis, kidney stones, and gastrointestinal ulcers. Urease inhibitors are impressive against a number of serious infections caused by the secretion of urease by Helicobacter pylori. These bacteria may be responsible for numerous gastroduodenal disorders, together with gastric cancer, duodenal ulcers, peptic ulcers, urolithiasis, and even urinary catheter encrustation.3,4 Difficulties associated with these kinds of infectious illnesses and other diverse sickness conditions are the synthesis and development of novel pharmacologically active compounds with minimum side effects. Urease inhibitors are known to be potent anti-ulcer drugs. Such potent urease inhibitors are generally divided into two classes as follows: (1) substrate structural analogs like hydroxamic acid and (2) those which affect the mechanism of action, like phosphoramidate, lansoprazole, omeprazole, thiol compounds, quinines, and the Schiff base derivatives. 4
Quinazolinone derivatives containing an acyl hydrazone skeleton are a class of fused heterocycles that is of considerable interest because of their diverse pharmacological properties and activities. Such activity includes anti-fungal, anti-hypertensive, anti-microbial, anti-viral, anti-tumor, anti-convulsant, anti-inflammatory, anti-cancer, and so on.5–10 Enzyme inhibition studies, which are important in pharmaceutical research, have led to the discovery of a wide variety of drugs that are useful in treating a number of diseases. According to recent studies, several quinazolinone derivatives with diverse side groups have anti-urease activity.11–16 In our recent studies, we investigated quinazolinone derivatives, that were very active inhibitors of jack bean urease. However, there is still a considerable need for alternative or novel treatments of H. pylori associated with diseases.13–15 This work describes the synthesis some new quinazolinone derivatives containing an acyl hydrazone skeleton with different side groups (methyl and methoxy) to determine the inhibition properties of these compounds, the enzyme kinetic parameters, and to investigate their anti-oxidant activities.
Results and discussion
Chemistry
In this study, a novel series of quinazolinone derivatives containing acyl hydrazone skeletons was synthesized according to the pathways outlined in Scheme 1. A total of 12 compounds, 10 of which are novel, were synthesized and the structures were confirmed by 1H NMR, 13C NMR, and elemental analysis. A literature method was used for the synthesis;
16
however, ethylimido(3-methoxy)benzyl acetate hydrochloride and ethylimido(3-methyl)benzyl acetate hydrochloride were used as the starting compounds instead of ethylimidobenzyl acetate hydrochloride. Ethylimido(3-methoxy)benzyl acetate hydrochloride and ethylimido(3-methyl)benzyl acetate hydrochloride, as starting compounds, were prepared according to the literature.16,17 In the second step of our experimental studies, compounds

Synthetic pathway toward the target compounds.
Next, the acetohydrazide compounds
Cupric reducing anti-oxidant activity and ferric reducing anti-oxidant capacity anti-oxidant activity results
The anti-oxidant capacities of the newly synthesized compounds were determined using ferric reducing anti-oxidant capacity (FRAP) and cupric reducing anti-oxidant activity (CUPRAC) assays. The CUPRAC and FRAP of mainly electron transfer-based methods were applied for the synthesized and natural compounds. The CUPRAC method is based on the absorbance measurement of the CUPRAC chromophore, Cu(I)-neocuproine (Nc) chelate, formed as a result of the redox reaction of anti-oxidants with the CUPRAC reagent, a bis(neocuproine)copper(II) cation [Cu(II)-Nc], where the absorbance is recorded at the maximum light absorption wavelength of 450 nm. The orange-yellow color is due to the Cu(I)-Nc charge-transfer complex formed. Increased absorbance of the reaction mixture indicates a higher reducing ability.
19
The anti-oxidant effects were classified into two groups; compounds
CUPRAC and FRAP test results of all the newly synthesized compounds.

CUPRAC: cupric reducing anti-oxidant activity; FRAP: ferric reducing anti-oxidant capacity.
CUPRAC values are given as mM TEAC (Trolox equivalent anti-oxidant capacity) obtained from the [Trolox]-absorbance calibration graph (r2 = 0.999). FRAP values are expressed as mM FeSO4.7H2O equivalent of the per gram of compound, calculated from the reference Fe2SO4.7H2O linear graph (r2 = 0.999). The CUPRAC and FRAP values of the compounds are expressed as the mean ± SD in triplicate.
Mean value ± SD.
DPPH and ABTS radical-scavenging assays
The DPPH (2,2-diphenyl-1-picrylhydrazyl) method is based on the fact that the free radical is purple in color and that the purple color of DPPH decays in the presence of an anti-oxidant. The color changes from purple to yellow after reduction, which can be quantified by a decrease in the absorbance at a wavelength of 517 nm. The results are expressed as % radical-scavenging activity at three different final concentrations of 100, 25, and 6.25 µg mL−1 (Table 2). Compounds
Radical-scavenging properties of the synthesized compounds and BHT (butylated hydroxytoluene) in the ABTS and DPPH assays.
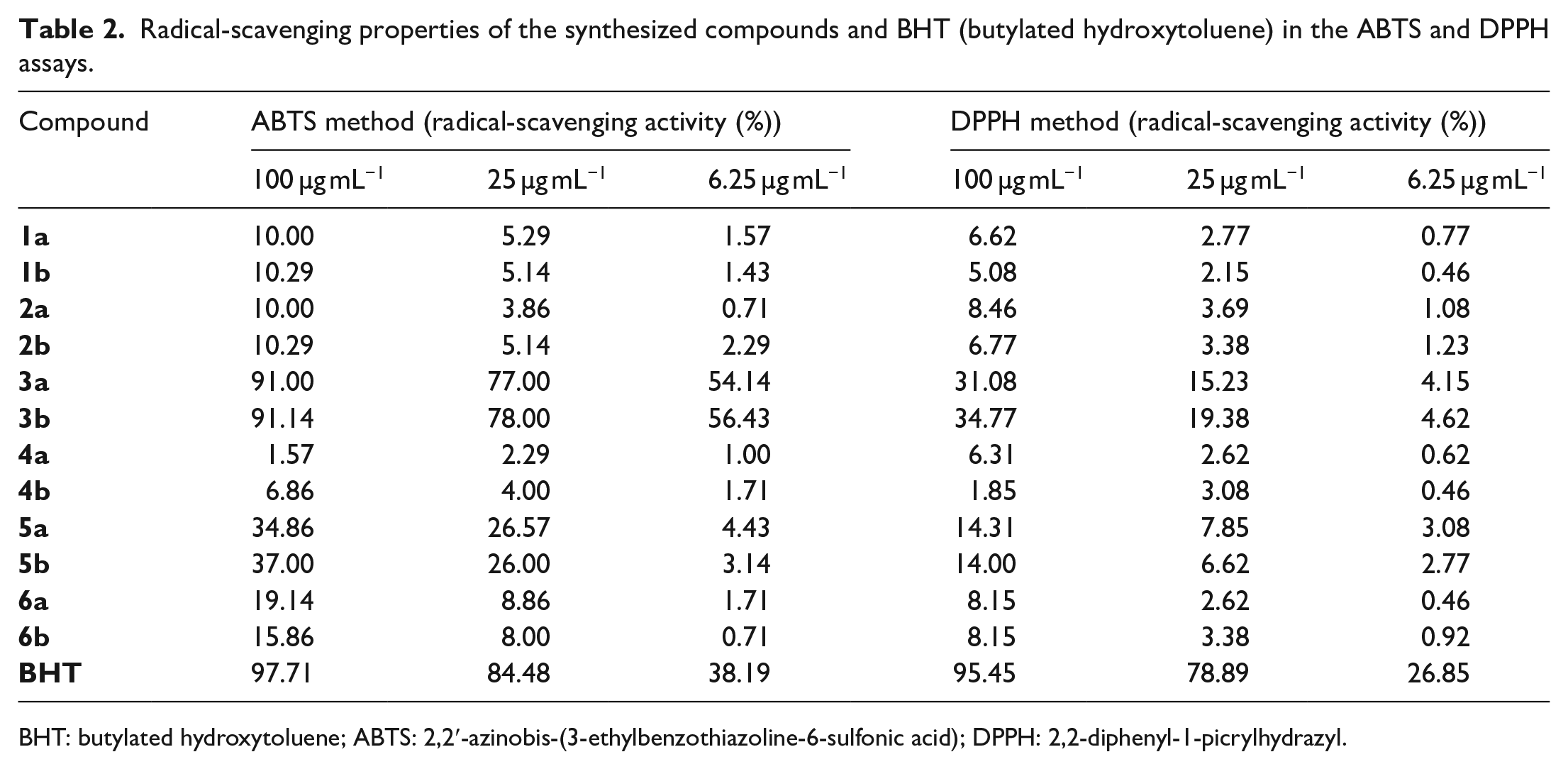
BHT: butylated hydroxytoluene; ABTS: 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid); DPPH: 2,2-diphenyl-1-picrylhydrazyl.
The pre-formed radical monocation of 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS•+) is generated by the oxidation of ABTS with potassium persulfate and is reduced in the presence of hydrogen-donating anti-oxidants. The influence of both the concentration of the anti-oxidant and the duration of the reaction on the inhibition of the radical cation absorption is taken into account when determining the anti-oxidant activity.
20
Compounds
Urease inhibition assay and kinetic parameters
The newly synthesized compounds were examined in terms of their urease inhibition potential. The percentage relative activities versus inhibitor concentrations were plotted separately for each organic molecule. The IC50 values were determined for the urease inhibitory activity of the synthesized compounds and for that of thiourea (Table 3). Compound
IC50 values of the synthesized compounds against to jack bean urease.

Thiourea was used the standard inhibitor.
Mean value ± SD.

Types of inhibition and the Lineweaver–Burk plots of thiourea (a),
Type of jack bean urease inhibition in the presence of compounds

Mean value ± SD.
Conclusion
A novel series of 2-(3-methyl/methoxybenzyl)quinazolin-4(3H)-one derivative containing ester, hydrazide, and acyl hydrazones functional groups on the second nitrogen atom has been synthesized and then their anti-urease, enzyme kinetic studies, and anti-oxidant activities have been screened. The anti-oxidant activities and free radical-scavenging properties of the synthesized compounds were clarified using various assays. Products
Materials and methods
Experimental
All the chemicals were supplied from Merck (Darmstadt, Germany), Sigma-Aldrich (St. Louis, MO, USA), and Fluka (Morris Plains, NJ 07950-2546 USA). The progress of all reactions was determined by TLC (aluminum sheets, silica gel 60F 2.54–0.2 mm thickness). Melting points were determined using a Stuart SMP30 melting point apparatus and are uncorrected. 1H NMR and 13C NMR (attached proton test (APT)) spectra were recorded on a Varian-Mercury 400 MHz spectrometer in dimethyl sulfoxide-d6 (DMSO-d6) using tetramethylsilane as an internal standard chemical shifts (δ values) are given in ppm. The elemental compositions were determined on a Carlo Erba 1106 CHN analyzer. The composition values were in agreement (±0.4%) with calculated values. Compounds
2-(3-Methylbenzyl)quinazolin-4(3H)-one (1a):
White solid (2.37 g); 95%; m.p. 203–204 °C. (CAS no. 1401675-28-2; m.p. 202–203 °C); 1H NMR (400 MHz, DMSO-d6): δ (ppm) 2.25 (s, 3H, CH3), 3.87 (s, 2H, CH2), 7.02–7.05 (m, 1H, ArH), 7.13–7.20 (m, 3H, ArH), 7.42–7.46 (m, 1H, ArH), 7.58–7.60 (m, 1H, ArH), 7.73–7.77 (m, 1H, ArH), 8.05 (m, 1H, ArH), 12.38 (s, 1H, NH). 24
2-(3-Methoxybenzyl)quinazolin-4(3H)-one (1b):
White solid (2.55 g); 96%; m.p. 178–179 °C. (CAS no. 83988-49-2; m.p. 178–180 °C); 1H NMR (400 MHz, DMSO-d6): δ (ppm) 3.72 (s, 3H, OCH3), 3.89 (s, 2H, CH2), 6.80–6.98 (m, 3H, ArH), 7.20–7.22 (m, 1H, ArH), 7.46–7.74 (m, 3H, ArH), 8.06–8.08 (m, 1H, ArH), 12.37 (s, 1H, NH). 25
Ethyl [2-(3-methylbenzyl)-4-oxoquinazolin-3(4H)-yl]acetate (2a):
Yellowish solid (2.61 g); 88%; m.p. 102–103 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 1.08 (t, J = 8.0 Hz, 3H, CH3), 2.23 (s, 3H, CH3), 3.94 (q, OCH2, J = 8.0 Hz, 2H), 4.19 (s, 2H, CH2), 4.80 (s, 2H, –NCH2), 7.03–7.09 (m, 3H, ArH), 7.16–7.20 (m, 1H, ArH), 7.51–7.55 (m, 1H, ArH), 7.67–7.69 (m, 1H, ArH), 7.82–7.86 (m, 1H, ArH), 8.08–8.10 (m, 1H, ArH); 13C NMR (100 MHz, DMSO-d6): δ 14.29 (CH3), 21.41 (CH3), 41.45 (CH2), 45.94 (NCH2), 61.57 (OCH2), 114.01, 120.06, 123.41, 126.20, 126.62, 127.52, 128.05, 128.98, 129.62, 130.05, 135.27, 135.43, 138.20, 147.28, 155.90, 159.83, 161.74, 167.66. Anal. calcd for C20H20N2O3: C, 71.41; H, 5.99; N, 8.33; found: C, 71.47; H, 6.01; N, 8.38.
Ethyl [2-(3-methoxybenzyl)-4-oxoquinazolin-3(4H)-yl]acetate (2b):
Yellowish solid (3.20 g); 91%; m.p. 80–81 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 1.10 (t, CH3, J = 8.0 Hz, 3H), 3.70 (s, 3H, OCH3), 3.95 (q, J = 8.0 Hz, 2H, OCH2), 4.21 (s, 2H, CH2), 4.82 (s, 2H, –NCH2), 6.81–6.89 (m, 3H, ArH), 7.19–7.23 (m, 1H, ArH), 7.54–7.56 (m, 1H, ArH), 7.68–7.70 (m, 1H, ArH), 7.83–7.85 (m, 1H, ArH), 8.09–8.10 (m, 1H, ArH). 13C NMR (100 MHz, DMSO-d6): δ 14.25 (CH3), 41.44 (CH2), 45.95 (NCH2), 55.40 (OCH3), 61.58 (OCH2), 112.66, 115.09, 120.05, 121.25, 126.62, 127.44, 127.51, 130.15, 135.29, 137.00, 147.27, 155.81, 159.83, 161.73, 167.66. Anal. calcd for C20H20N2O4: C, 68.17; H, 5.72; N, 7.95; found: C, 68.23; H, 5.75; N, 7.99.
2-[2-(3-Methylbenzyl)-4-oxoquinazolin-3(4H)-yl]acetohydrazide (3a):
White solid (2.41 g); 75%; m.p. 220–222 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 2.25 (s, 3H, CH3), 4.09 (s, 2H, CH2), 4.25 (s, 2H, NH2), 4.58, 4.96 (s, 2H, NCH2, syn/anti conformers, 86:14), 7.02–7.07 (m, 3H, ArH), 7.18–7.20 (m, 2H, ArH), 7.49–7.53 (m, 1H, Ar), 7.62–7.64 (m, 1H, Ar), 7.79–7.83 (m, 1H, Ar), 9.36 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6): δ 21.44 (CH3), 41.49 (CH2), 44.82 (NCH2), 120.30, 126.03, 126.65, 127.18, 127.40, 128.08, 129.08, 129.49, 135.01, 135.81, 138.36, 147.33, 156.33, 161.80, 166.49. Anal. calcd for C18H18N4O2: C, 67.07; H, 5.63; N, 17.38; found: C, 6.11; H, 5.59; N, 17.43.
2-[2-(3-Methoxybenzyl)-4-oxoquinazolin-3(4H)-yl]acetohydrazide (3b):
White solid (2.33 g); 69%; m.p. 200–202 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 3.71 (s, 3H, OCH3), 4.11 (s, 2H, CH2), 4.26 (s, 2H, NH2), 4.60 (s, 2H, NCH2), 6.79–6.85 (m, 3H, ArH), 7.24–7.26 (m, 2H, ArH), 7.52–7.82 (m, 3H, Ar), 9.37 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6): δ 41.47 (CH2), 44.85 (NCH2), 55.44 (OCH3), 112.67, 114.98, 120.29, 121.08, 126.65, 127.20, 127.39, 130.24, 135.02, 137.42, 147.31, 156.24, 159.90, 161.78, 166.50. Anal. calcd for C18H18N4O3: C, 63.89; H, 5.36; N, 16.56; found: C, 63.92; H, 5.32; N, 16.59.
General procedure compounds 4a, b, 5a, b and 6a, b
A solution of compound
2-[2-(3-Methylbenzyl)-4-oxoquinazolin-3(4H)-yl]-N’-[(1)-phenylmethylene]acetohydrazide (4a):
White solid (3.77 g); 92%; m.p. 230–231 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 2.17 (s, 3H, CH3), 4.12, 4.17 (s, 2H, CH2, syn/anti conformers, 78:22), 4.74, 5.19 [s, 2H, NCH2, anti/syn conformers, 19:81], 7.05–7.16 (m, 4H, ArH), 7.43–7.46 (m, 3H, ArH), 7.51–7.55 (m, 3H, ArH), 7.65–7.69 (m, 1H, ArH), 7.82–7.86 (m, 1H, ArH), 8.02 (s, 1H, N=CH), 8.09–8.17 (m, 1H, ArH), 11.76 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6): δ 21.39 (CH3), 41.74 (CH2), 45.17 (NCH2), 120.21, 126.11, 126.69, 127.32, 128.11, 129.28, 129.71, 130.52, 134.33, 135.11, 135.66, 138.28, 144.70, 147.70, 147.41, 156.35, 161.87, 168.14. Anal. calcd for C25H22N4O2: C, 73.15; H, 5.40; N, 13.65; found: C, 73.18; H, 5.43; N, 13.61.
2-[2-(3-Methoxybenzyl)-4-oxoquinazolin-3(4H)-yl]-N’-[(1)-phenylmethylene]acetohydrazide (4b):
White solid (3.74 g); 88%; m.p. 235–236 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 3.63, 3.71 [s, 3H, OCH3, syn/anti conformers, 60:30], 4.15, 4.17 [s, 2H, CH2, syn/anti conformers, 90:10], 4.75, 5.21 [s, 2H, NCH2, syn/anti conformers, 15:85], 6.86–7.85 (m, 12H, ArH), 8.03 (s, 1H, N=CH), 8.10–8.18 (m, 1H, ArH), 11.76 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6): δ 41.66 (CH2), 45.24 (NCH2), 55.33 (OCH3), 112.81, 115.01 (Ar-CH), 120.20 (Ar-C), 121.18, 126.69, 127.35, 127.44, 119.26, 130.16, 130.51, 134.33, 137.31, 144.71, 147.39, 156.28, 159.87, 161.86, 168.17. Anal. calcd for C25H22N4O3: C, 70.41; H, 5.20; N, 13.14; found: C, 70.38; H, 5.17; N, 13.17.
N’-[(1)-(4-hydroxyphenyl)methylene]-2-[2-(3-methylbenzyl)-4-oxoquinazolin-3(4H)-yl]acetohydrazide (5a):
White solid (3.62 g); 85%; m.p. 213–214 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 2.18, 2.24 [s, 3H, CH3, syn/anti conformers, 78:22], 4.10, 4.16 [s, 2H, CH2, syn/anti conformers, 80:20], 4.71, 5.15 [s, 2H, NCH2, anti/syn conformers, 24:76], 6.79–6.81 (m, 2H, ArH), 7.02–7.18 (m, 3H, ArH), 7.47–7.53 (m, 3H, ArH), 7.66–7.68 (m, 2H, ArH), 7.82–7.84 (m, 1H, ArH), 7.92 (s, 1H, N=CH), 8.09–8.11 (m, 1H, ArH), 9.91 (s, 1H, OH), 11.57 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6): δ 21.40 (CH3), 41.72 (CH2), 45.07 (NCH2), 116.12, 120.22, 125.35, 126.09, 126.70, 127.24, 127.44, 128.11, 129.07, 129.69, 135.08, 135.68, 138.28, 145.01, 147.41, 156.37, 161.87, 167.77. Anal. calcd for C25H22N4O3: C, 70.41; H, 5.20; N, 13.14; found: C, 70.39; H, 5.25; N, 13.10.
N’-[(1)-(4-hydroxyphenyl)methylene]-2-[2-(3-methoxybenzyl)-4-oxoquinazolin-3(4H)-yl]acetohydrazide (5b):
White solid (4.02 g); 91%; m.p. 242–243 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 3.63, 3.70 [s, 3H, OCH3, syn/anti conformers, 90:10], 4.12, 4.17 [s, 2H, CH2, syn/anti conformers, 90:10], 4.71, 5.16 [s, 2H, NCH2, anti/syn conformers, 20:80], 6.79–6.86 (m, 3H, ArH), 7.22–7.25 (m, 2H, ArH), 7.47–7.53 (m, 3H, ArH), 7.66–7.68 (m, 2H, ArH), 7.81–7.84 (m, 1H, ArH), 7.91 (s, 1H, N=CH), 8.08–8.10 (m, 1H, ArH), 9.91 (s, 1H, OH), 11.57 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6): δ 41.64 (CH2), 45.15 (NCH2), 55.33 (OCH3), 112.79, 114.98, 116.12, 120.20, 121.16, 125.34, 126.68, 127.42, 129.09, 130.16, 137.33, 147.39, 156.31, 159.86, 161.84, 167.80. Anal. calcd for C25H22N4O4: C, 67.86; H, 5.01; N, 12.66; found: C, 67.82; H, 5.04; N, 12.62.
N’-[(1)-(2-hydroxyphenyl)methylene]-2-[2-(3-methylbenzyl)-4-oxoquinazolin-3(4H)-yl]acetohydrazide (6a):
White solid (3.79 g); 89%; m.p. 250–251 °C; 1H NMR (400 MHz, DMSO-d6): δ (ppm) 2.18, 2.24 [s, 3H, CH3, syn/anti conformers, 64:36], 4.11, 4.18 [s, 2H, CH2, syn/anti conformers, 63:37], 4.75, 5.17 [s, 2H, NCH2, anti/syn conformers, 43:57], 6.81–6.90 (m, 2H, ArH), 7.05–7.29 (m, 3H, ArH), 7.51–7.55 (m, 3H, ArH), 7.62–7.68 (m, 2H, ArH), 7.81–7.86 (m, 1H, ArH), 8.09 [s, 1H, N=CH), 8.33–8.38 (m, 1H, ArH], 10.04, 10.90 [s, 1H, OH, cis/trans conformers, 67:33], 11.69, 11.93 [s, 1H, NH, cis/trans conformers, 75:25]; 13C NMR (100 MHz, DMSO-d6): δ 21.38 (CH3), 41.71 (CH2), 45.13 (NCH2), 116.59, 119.83, 120.21, 126.10, 126.70, 127.45, 128.10, 129.01, 129.60, 135.08, 135.68, 138.29, 147.41, 156.37, 156.87, 161.86, 167.82. Anal. calcd for C25H22N4O3: C, 70.41; H, 5.20; N, 13.14; found: C, 70.44; H, 5.22; N, 13.10.
N’-[(1)-(2-hydroxyphenyl)methylene]-2-[2-(3-methoxybenzyl)-4-oxoquinazolin-3(4H)-yl]acetohydrazide (6b): White solid (3
84 g); 87%; m.p. 269–270 °C; 1H NMR (400 MHz, DMSO-d6):
δ (ppm) 3.65, 3.70 [s, 3H, OCH3, syn/anti conformers, 85:15], 4.10, 4.14 [s, 2H, CH2, syn/anti conformers, 90:10], 4.82, 5.08 [s, 2H, NCH2, anti/syn conformers, 85:15], 7.02–7.12 (m, 3H, ArH), 7.21–7.24 (m, 2H, ArH), 7.51–7.54 (m, 3H, ArH), 7.71–7.73 (m, 3H, ArH), 8.01 (s, 1H, N=CH), 8.09–8.11 (m, 1H, ArH), 10.03, 10.92 [s, 1H, OH, cis/trans conformers, 70:30], 11.68, 11.93 [s, 1H, NH, cis/trans conformers, 75:25]; 13C NMR (100 MHz, DMSO-d6): δ 41.65 (CH2), 45.16 (NCH2), 55.45 (OCH3), 112.82, 114.76, 116.12, 120.24, 121.23, 125.44, 126.58, 127.52, 129.03, 131.36, 137.35, 147.49, 156.56, 158.86, 162.44, 167.82. Anal. calcd for C25H22N4O4: C, 67.86; H, 5.01; N, 12.66; found: C, 67.83; H, 5.03; N, 12.69.
Anti-oxidant activity methods
CUPRAC and FRAP assays were applied to investigate the anti-oxidant activity of the newly synthesized compounds. In order to determine the cupric ion (Cu2+) reducing ability of the synthesized compounds, a previously published method was used.19,20,26 Trolox was used a standard, linear between 8.0 and 0.03125 mM (r2 = 0.999). CUPRAC values are expressed as mM Trolox per 1 mg of synthesized compound.
The reducing ability of the ferric tripyridyltriazine (Fe-III-TPTZ) complex was used for the total anti-oxidant capacity assay.20,26 FeSO4.7H2O was used as a positive control to construct a reference linear curve at six different concentrations (15.625–1000 µM). FRAP values are expressed as mM of FeSO4.7H2O equivalent of per gram of the compound, calculated from the reference FeSO4.7H2O linear graph (r2 = 0.999).
Free-radical-scavenging assays
The DPPH• (2,2-diphenyl-1-picrylhydrazyl) and ABTS•+ [2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)] methods were applied to analyze the free-radical-scavenging activity of the synthesized compounds using BHT as a standard.20,26 The DPPH radical has a purple color that decays in the presence of anti-oxidant agents; thus, the change in the absorbance was monitored at 517 nm using a UV-Vis spectrophotometer (1601UV-Shimadzu, Australia). The ability of the prepared compounds and BHT to scavenge the ABTS•+ radical was determined according to the literature.20,26 The decrease in the absorbance at 734 nm was measured using a UV-Vis spectrophotometer (1601UV-Shimadzu). All determinations for these assays were carried out three times, and the percentage scavenging was calculated from the formula

In vitro urease inhibition assay and kinetic studies
Jack bean urease inhibitory activity and kinetic studies on the synthesized compounds and thiourea were examined based on previously published studies.14,27–30 Thiourea (Sigma-Aldrich) was used as the standard inhibitor. The urease inhibition percentage was calculated as follows

where ODcontrol is the activity of the enzyme without the compound/standard and ODcompound is the activity of the enzyme with the compound/standard at different concentrations. The concentration of the inhibitor required for inhibiting 50% of the enzyme activity under the assay conditions is defined as the IC50 value. In order to calculate the IC50 values, different concentrations of the synthesized compounds and standard were assayed under the same reaction conditions. The enzyme activity was determined using urea as the substrate in the buffer, used in the urease inhibition studies (pH = 8.2). Increasing concentrations of the substrate (0.50–8.0 mg mL−1) and 200 µL of the urease enzyme solution were added to a test tube, and the mixture was incubated at 25 °C for 50 min. The absorbance was measured at 625 nm. The obtained data were plotted as 1/activity (1/V) against 1/substrate concentration (1/[S]) according to the Lineweaver–Burk graph. 28
Compounds
Supplemental Material
sj-docx-1-chl-10.1177_17475198221096568 – Supplemental material for Synthesis of quinazolinone derivatives containing an acyl hydrazone skeleton as potent anti-urease agents enzyme kinetic studies and anti-oxidant properties
Supplemental material, sj-docx-1-chl-10.1177_17475198221096568 for Synthesis of quinazolinone derivatives containing an acyl hydrazone skeleton as potent anti-urease agents enzyme kinetic studies and anti-oxidant properties by Nimet Baltaş in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
