Abstract
Chemical investigation of the EtOAc-soluble extract of the leaves of Apocynum venetum allowed the isolation of seven flavonoids including a new compound named 4′-hydroxy-7-O-(4-hydroxybenzyl)-3-methoxy-6-prenylflavone (
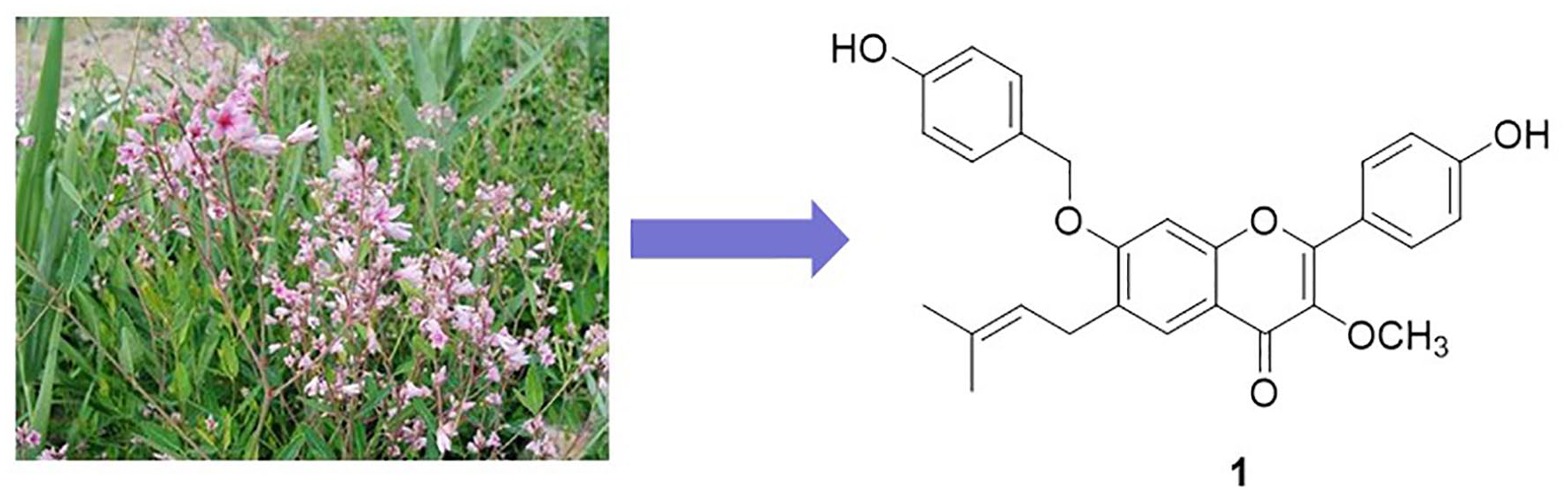
Introduction
Apocynum venetum L. (Apocynaceae) is a small perennial shrub that is widely distributed in the temperate regions of Europe, North America, and Asia.
1
In China, A. venetum commonly grows in the salt marshes of the Yellow River delta, and its flowers and leaves are used as a medicine and as tea.
2
As a traditional Chinese medicine, the leaves of A. venetum are used to treat neurasthenia, hypertension, nephritis, and heart disease.
3
Pharmacological studies showed that the extract of A. venetum possesses significant antioxidant, anti-hyperlipidemic, anti-hyperglycemic effect and reverses the effects of depressive-like behaviors.4–7 Phytochemical investigations on A. venetum revealed that flavonoids were the major active constituents which exhibited various pharmacological activities including anti-hypertensive, antioxidant, anti-depressant, anti-anxiety, hepatoprotective, and cardiotonic effects.1,2,8–10 During our studies on finding novel natural products with potent anti-inflammatory activity, we found that the EtOAc-soluble extract of the leaves of A. venetum showed moderate inhibitory activity on nitric oxide (NO) production in lipopolysaccharide (LPS)-induced mouse peritoneal macrophages (RAW264.7). Thus, a series of studies focused on the leaves of A. venetum were carried out. As a result, a new flavonoid named 4′-hydroxy-7-O-(4-hydroxybenzyl)-3-methoxy-6-prenylflavone (
Results and discussion
Structural elucidation
Compound

Key HMBC correlations of compound
Moreover, six known compounds

Structures of compounds
Analysis of the biological activity results
Compounds
Anti-inflammatory activities of compounds

AH: aminoguanidine hydrochloride.
IC50 values represent the means ± SEM of three parallel measurements.
Positive control.
Conclusion
In this study, a new flavonoid named 4-hydroxy-7-O-(4-hydroxybenzyl)-3-methoxy-6-prenylflavone (
Experimental
General
UV spectra were obtained on a Hitachi U-3310 UV/vis spectrometer (Hitachi, Tokyo, Japan). IR spectra were recorded with a Nicolet Avatar 370 FTIR spectrophotometer (Nicolet, Wisconsin, USA). Nuclear magnetic resonance (NMR) spectra were acquired on a Bruker AV-500 MHz spectrometer with tetramethylsilane (TMS) as an internal standard (Bruker, Karlsruhe, Germany). Mass spectra were obtained on a QTOF2 high-resolution mass spectrometer (Micromass, Wythenshawe, UK). Column chromatography was conducted using silica gel 60 (100 and 200 μm particle size, Yantai Xinde Chemical Co., Ltd, Yantai, China) and RP-18 (150-63 μm particle size, Merck, Darmstadt, Germany). Thin-layer chromatography (TLC) was performed with precoated silica gel GF254 glass plates (Qingdao Marine Chemical Co., Ltd, Qingdao, China). High-performance liquid chromatography (HPLC) was carried out using a Shimadzu System LC-20AT pump equipped with a SPD-10Avp UV detector (Shimadzu, Tokyo, Japan), and a YMC ODS-A column (250 mm × 4.6 mm, 5 μm).
Plant material
The leaves of A. venetum were collected in Tengchong, Yunnan Province, P.R. China, and authenticated by Professor Qiaofeng Wu (College of Pharmacy, Zhejiang Chinese Medical University). A voucher specimen of the plant (no. 20200724) was deposited at the College of Pharmacy, Zhejiang Chinese Medical University, Zhejiang, P.R. China.
Extraction and isolation
The dried leaves of A. venetum (10.0 kg) were extracted three times with 75% MeOH under reflux, and the solution was concentrated in vacuo to yield the extract (1.8 kg). This extract was suspended in H2O, partitioned successively with petroleum ether (PE), CH2Cl2, EtOAc, and n-BuOH. The EtOAc extract of A. venetum showed moderate inhibitory activity toward NO production with an IC50 value of 64.3 ± 0.2 μg/mL. Thus, the EtOAc fraction (161.5 g) was subjected to silica gel column chromatography eluting with a gradient of CH2Cl2-MeOH (from 100:0 to 0:1) to give 12 fractions (Fr.1-Fr.12). Fr.6 (13.9 g) was applied to silica gel column using a gradient of PE-EtOAc (from 100:0 to 0:1) and was separated into 12 fractions (Fr.6.1-Fr.6.12). Furthermore, Fr.6.7 (1.8 g) was subjected to the RP-18 column and eluted using MeOH-H2O (2:8 to 1:0) to afford 15 fractions (Fr.6.7.1-Fr.6.7.15). Fr.6.7.5 (121.2 mg) was further purified by HPLC and eluted with a gradient of 55%–65% MeOH in H2O at a flow rate of 3.0 mL/min over 70 min. This resulted in the isolation of compounds
4′-Hydroxy-7-O-(4-hydroxybenzyl)-3-methoxy-6-prenylflavone (
Anti-inflammatory assay
The anti-inflammatory activities were tested by measuring the inhibitory activity toward NO and TNF-α production in mouse macrophage RAW264.7 cells. 14 Briefly, RAW264.7 cells were cultured to the log phase, and seeded into 24-well plates at 1 × 105 cells/well and the MTT method was used to evaluate the effect of different concentrations (100, 50, 25, 12.5, 6.25, 3.125 μM) of tested samples on cell proliferation of cells in response to LPS. A Griess kit was used to measure the effect of these sample concentrations on NO secretion of RAW 264.7 cells in response to LPS and aminoguanidine hydrochloride was used as the positive control. The levels of TNF-α were determined using an ELISA (enzyme-linked immunosorbent assay) kit (Solarbio, Beijing, P.R. China) according to the manufacturer’s instructions. TNF-α was determined from a standard curve and silybin was used as the positive control. Experiments were performed at least three times.
Supplemental Material
sj-doc-1-chl-10.1177_17475198211073871 – Supplemental material for Flavonoids from the leaves of Apocynum venetum and their anti-inflammatory activity
Supplemental material, sj-doc-1-chl-10.1177_17475198211073871 for Flavonoids from the leaves of Apocynum venetum and their anti-inflammatory activity by Hong-Min Fu, Chun-Ling Yin, Zhi-Yong Shen and Ming-Hua Yang in Journal of Chemical Research
Footnotes
Acknowledgements
We are grateful for a grant from The Third Affiliated Hospital of South China University, and the Department of Instrumental Analysis of Zhejiang Chinese Medical University for the measurement of UV, IR, HRESIMS, and NMR spectra.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

