Abstract
A ruthenium nanocatalyst is utilized for the first time for the highly efficient and selective hydrogenation of 1,5-cyclooctadiene under atmospheric hydrogen pressure. Under the optimized reaction conditions, the conversion of 1,5-cyclooctadiene and the selectivity for cyclooctene are >99% and 95%, respectively. The turnover frequency is 451 h−1, which is higher than that ever reported for Ru complex catalysts.
Keywords

Introduction
The selective hydrogenation of 1,5-cyclooctadiene (1,5-COD) is an important method for the synthesis of cyclooctene (COE), which is of vital significance for the synthesis of polymers, epoxides, cyclic alcohols, and other intermediates.1–5 In recent years, due to their high catalytic efficiency, a wide variety of noble transition-metal nanoparticles have been extensively reported for the selective hydrogenation of 1,5-COD, such as Pd,6–10 Rh, 11 Pt, 12 and Ru13,14 nanoparticles. Until now, most reactions were carried out under high hydrogen pressure to obtain good catalytic activity. As for the atmospheric hydrogenation of 1,5-COD, only a limited number of attempts have been carried out with expensive Pd,15–21 Rh, 22 and Pt 23 nanoparticles as catalysts. The use of cheaper Ru nanocatalysts for this atmospheric hydrogenation reaction has not been reported.
In our previous work, a type of Ru nanocatalyst stabilized by the thermoregulated ligand Ph2P(CH2CH2O)22CH3 (LP1000) was synthesized and was utilized for thermoregulated phase-transfer in an aqueous/1-pentanol biphasic system. 24 During heating, this Ru nanocatalyst can transfer from the aqueous phase to the substrate-containing 1-pentanol phase and enable the catalyst and the substrate to react in the same phase. Following the reaction, and cooling of the Ru nanocatalyst, it can return to the aqueous phase and simplify separation of the products. This thermoregulated phase-transfer Ru nanocatalyst (TPT-Runano) combines the merits of a monophasic reaction and biphasic separation. Therefore, in this paper, TPT-Runano has been explored as a catalyst for the selective hydrogenation of 1,5-COD under atmospheric hydrogen pressure.
Results and discussion
The TPT-Runano catalyst was prepared by reduction of Ruthenium(III) chloride hydrate (RuCl3·xH2O) in the presence of hydrogen using LP1000 as a stabilizer. Figure 1 shows the ultraviolet–visible (UV–vis) absorption spectra of the mixture of RuCl3·xH2O and LP1000 before and after reduction. Before reduction, the characteristic absorption peak was observed at 382 nm. After reduction, this characteristic peak disappeared completely, which indicated the formation of Ru0. 25

UV–vis absorption spectra of the mixture of LP1000 and RuCl3·xH2O. UV–vis: ultraviolet–visible.
To further explore the chemical valence of the Ru element, X-ray photoelectron spectra (XPS) characterization was performed (Figure 2). We note that two peaks appear at binding energies of 484.2 eV and 462.0 eV, corresponding to Ru

XPS spectra of TPT-Runano catalyst. XPS: X-ray photoelectron spectra; TPT: thermoregulated phase-transfer; a.u.: arbitrary unit.
Furthermore, the TEM image and particle size histogram (Figure 3) show that the average particle size of the newly prepared Ru nanoparticles was 1.4 ± 0.3 nm.

TEM image and particle size histogram of the newly prepared TPT-Runano catalyst. TEM: transmission electron microscopy; TPT: thermoregulated phase-transfer.
With these results in hand, we investigated the catalytic performance of TPT-Runanocatalyst for the atmospheric hydrogenation of 1,5-COD. Different reaction conditions were explored and the results are displayed in Table 1. First, the influence of the reaction temperature was investigated in the range of 50 °C-90 °C. When the temperature was increased from 50 °C to 80 °C, the conversion increased from 34% to >99% and the selectivity increased from 87% to 95% (Table 1, entries 1–3). On further increasing the temperature to 90 °C, the conversion remained >99%, while the selectivity dropped from 95% to 89% (Table 1, entry 4). Subsequently, the influence of the reaction time was studied. When the reaction time was extended from 15 to 25 min, the conversion and selectivity increased to >99% and 95%, respectively (Table 1, entries 3, 5, and 6). Prolonging the reaction time to 30 min decreased the selectivity and the by-product was mainly cyclooctane (COA) (Table 1, entry 7). Finally, we investigated the effect of the 1,5-COD/Ru molar ratio on the reaction. The results indicated that the conversion was always >99% on increasing the molar ratio from 50 to 200, but the selectivity increased from 79% to 95% (Table 1, entries 3, 8, and 9). When the molar ratio was further increased to 250, both the conversion and selectivity decreased (Table 1, entry 10). Thus, optimized reaction conditions for the atmospheric hydrogenation of 1,5-COD catalyzed by TPT-Runano catalyst were established. Under these conditions, the conversion of 1,5-COD was >99% and the selectivity for COE was 95% (Table 1, entry 3). Also, the TOF was 451 h–1, which is highest ever reported for a Ru complex catalyst. 28
TPT-Runano-catalyzed selective hydrogenation of 1,5-COD under atmospheric hydrogen pressure a .
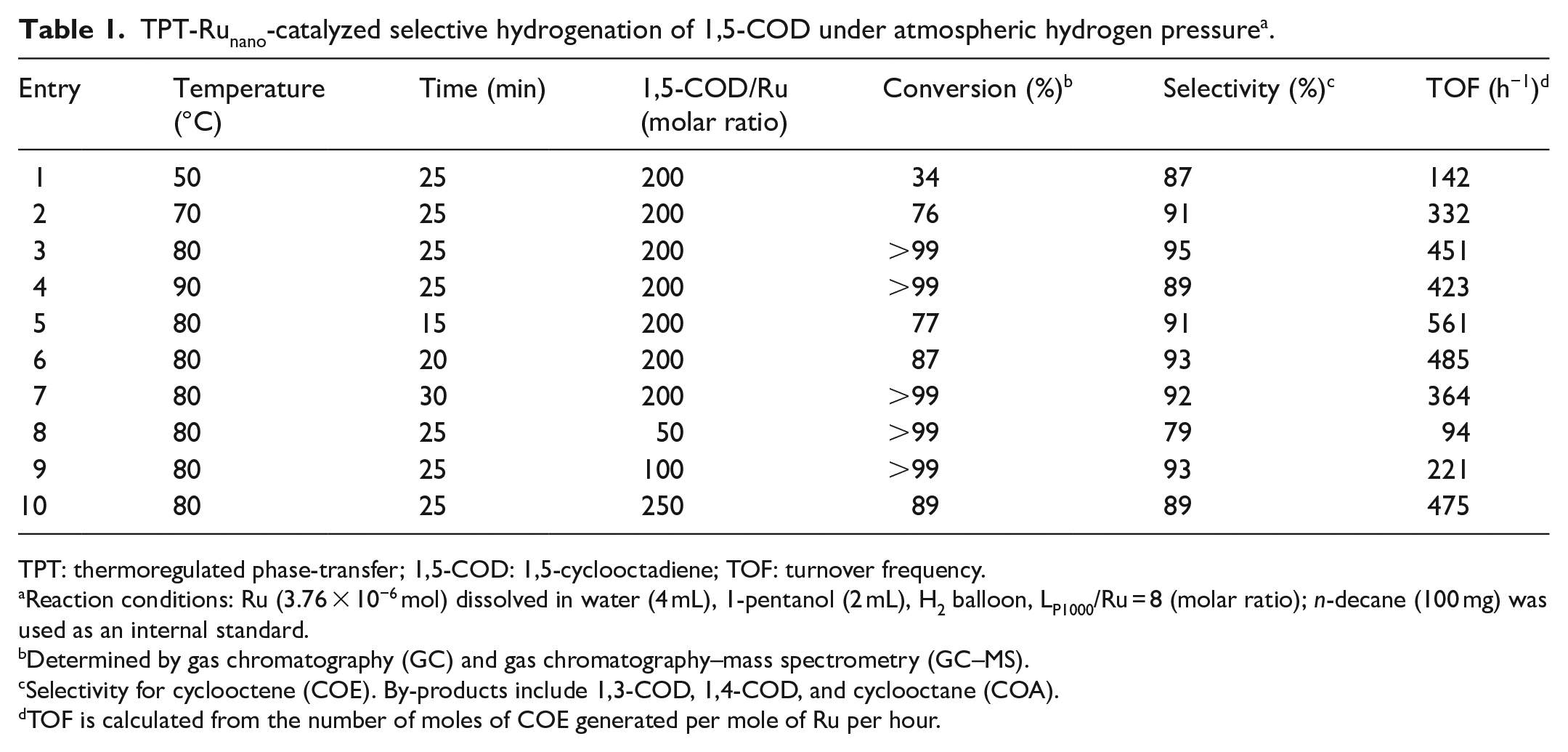
TPT: thermoregulated phase-transfer; 1,5-COD: 1,5-cyclooctadiene; TOF: turnover frequency.
Reaction conditions: Ru (3.76 × 10−6 mol) dissolved in water (4 mL), 1-pentanol (2 mL), H2 balloon, LP1000/Ru = 8 (molar ratio); n-decane (100 mg) was used as an internal standard.
Determined by gas chromatography (GC) and gas chromatography–mass spectrometry (GC–MS).
Selectivity for cyclooctene (COE). By-products include 1,3-COD, 1,4-COD, and cyclooctane (COA).
TOF is calculated from the number of moles of COE generated per mole of Ru per hour.
After optimizing the reaction conditions, the reusability of the TPT-Runano catalystwas examined. First, by separation from the product, the TPT-Runano catalyst was directly reused in the second cycle. The results indicated that the conversion decreased from >99% to 86% and the selectivity dropped from 95% to 62%. To investigate the possible reasons, a TEM image was obtained after the first cycle. As shown in Figure 4, the size of the TPT-Runano particles had changed to 1.8 ± 0.2 nm, now being larger than the 1.4 ± 0.3 nm size of the freshly prepared TPT-Runano particles. Also, leaching of Ru after the first cycle was detected by inductively coupled plasma atomic emission spectroscopy (ICP-AES). The data showed that 4.9 wt% (weight percentage) of Ru was lost in the 1-pentanol phase. Based on the above results, the reusability experiments on the TPT-Runano catalyst were performed by adding LP1000 (13.2 mg) from the second cycle onwards to avoid the size of the nanoparticles increasing and by prolonging the reaction time (35 min for the second cycle, 45 min for the third cycle, 55 min for the fourth cycle, and 65 min for the fifth cycle) due to Ru leaching. As shown in Figure 5, the TPT-Runano catalyst could be used five times without any evident loss of conversion or selectivity.

TEM image and particle size histogram of the TPT-Runano catalyst after the first cycle. TEM: transmission electron microscopy; TPT: thermoregulated phase-transfer.

The reusability of the TPT-Runano catalyst for the atmospheric hydrogenation of 1,5-COD. TPT: thermoregulated phase-transfer; 1,5-COD: 1,5-cyclooctadiene; COE: cyclooctene.
To gain more information on the catalytic reaction, mercury-poisoning experiments were carried out, which are commonly used to determine whether a catalytic reaction is homogeneous or heterogeneous. 29 As previously established, the conversion of 1,5-COD was >99% under the optimized conditions (Table 2, entry 1). However, when Hg (0.75 g, Hg/Ru = 1000, molar ratio) was added before the reaction, the conversion of 1,5-COD decreased sharply to 15% (Table 2, entry 2) indicating the evident poisoning effect of Hg. Moreover, another experiment was designed to further test the poisoning effect. When the reaction was carried out for 15 min, the conversion of 1,5-COD was 77% (Table 2, entry 3). Hg was then added and the reaction was continued for another 10 min (total time = 25 min). The conversion was 78% (Table 2, entry 4), which implies that the reaction nearly stopped completely when Hg was added to the catalytic system. These results in Table 2 show that the catalytic process might be heterogeneous.
Mercury-poisoning experiments on the selective hydrogenation of 1,5-COD catalyzed by the TPT-Runano catalyst a .

1,5-COD: 1,5-cyclooctadiene; TPT: thermoregulated phase-transfer.
Reaction conditions: Ru (3.76 × 10−6 mol) dissolved in water (4 mL), 1-pentanol (2 mL), H2 balloon, LP1000/Ru = 8 (molar ratio), Temperature = 80 °C, 1,5-COD/Ru = 200 (molar ratio); n-decane (100 mg) was used as an internal standard.
Determined by gas chromatography (GC) and gas chromatography–mass spectrometry (GC–MS).
Conclusion
In summary, a Ru nanocatalyst has been explored for the first time for the selective hydrogenation of 1,5-COD at atmospheric hydrogen pressure, and was shown to be highly efficient. Under the optimized reaction conditions, >99% conversion and 95% selectivity for COE were achieved. The TOF was 451 h−1, being higher than that ever reported for a Ru complex catalyst. Inspired by the special case of 1,5-COD, the TPT-Runano catalyst will be then examined in the atmospheric hydrogenation of other alkenes to test selectivity in our laboratory.
Experimental section
Materials
Ruthenium(III) chloride hydrate (RuCl3·xH2O, 99.9%, 38% Ru) was purchased from Alfa Aesar. 1,5-COD and mercury were obtained from Aladdin. 1-Pentanol and n-decane were purchased from Kermel. All these chemicals were analytical grade and were used without further purification. LP1000 was synthesized as reported in the literature.30–32 1H NMR (400 MHz, CDCl3): δ = 7.49 − 7.27 (m, 10H), 3.72 − 3.50 (m, 95H), 3.37 (s, 3H), 2.40 (t, J = 7.7 Hz, 2H). 13C NMR (101 MHz, D2O): δ = 137.9 − 128.7, 71.1 − 69.4, 68.0, 58.1, 28.1. 31P NMR (162 MHz, D2O): δ = −23.3.
Characterization
The NMR data were obtained using Varian DLG400 or Bruker Avance II 400 spectrometers. The UV–Vis analyses were carried out on a Ruili UV–Vis 2100 spectrophotometer. The XPS were measured with an ESCALAB XI+ spectrometer. Transmission electron microscopy (TEM) data were recorded with a JEM-2000EX instrument. Gas chromatography (GC) was performed on a Tianmei 7900 GC with a 50 m OV-101 column and a flame ionization detection (FID) detector. Gas chromatography–mass spectrometry (GC–MS) was conducted on an Agilent 7000B instrument. ICP-AES analyses were recorded on an AVIO 500 instrument (PerkinElmer, USA).
Preparation of TPT-Runano catalyst
An aqueous solution of RuCl3·xH2O (4 mL, containing 3.76 × 10−6 mol of Ru), LP1000 (35.1 mg, 3.01 × 10−5 mol), and 1-pentanol (2 mL) were added to a 75 mL Teflon-lined stainless-steel autoclave, which was flushed three times with 1 MPa H2 and then inflated to 5 MPa with H2. After stirring the contents at 100 °C for 8 h, the autoclave was cooled to room temperature and depressurized. The aqueous phase displayed a brownish-black color, indicating formation of the TPT-Runano catalyst.
Atmospheric hydrogenation of 1,5-COD
In a typical experiment, the atmospheric hydrogenation of 1,5-COD was carried out in an autoclave, which was charged with the TPT-Runano catalyst, n-decane (100 mg; as an internal standard) and 1,5-COD (0.0822 g, 7.52 × 10−4 mol). The autoclave was flushed three times with 1 MPa H2 and the contents stirred at the scheduled temperature (80 °C) under atmospheric pressure (H2 balloon) for an appropriate time (25 min). Following the reaction, the autoclave was cooled to room temperature. The upper 1-pentanol phase containing the products was separated from the lower aqueous phase and analyzed by GC and GC–MS. Meanwhile, the lower aqueous phase containing the TPT-Runano catalyst could be reused in the next cycle.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the National Natural Science Foundation of China (No. 21173031).
