Abstract
The chiral ionic liquid CILTPT-MS [CH3(OCH2CH2)16CD]+[CH3SO3]− (CD = cinchonidine) is found to exhibit cloud point character. Due to this cloud point character, a CILTPT-MS-stabilized chiral nano-Pt catalyst is prepared and utilized to realize transfer in a H2O–1-pentanol biphasic system by simply changing the temperature. Therefore, a fluorine-free, thermoregulated phase-transfer catalysis system is developed and applied to the asymmetric hydrogenation of α-ketoesters with enantiomeric excess of >99% and excellent conversion. In addition, the chiral nano-Pt catalyst can be easily separated and reused efficiently.
Keywords
A fluorine-free thermoregulated phase-transfer catalysis system is developed and applied to the asymmetric hydrogenation of α-ketoesters with high enantiomeric excess and good recyclability.

Introduction
The technology behind obtaining optically pure chiral molecules plays an increasingly important role in pharmaceutical research.1–3 Although many highly efficient homogeneous chiral catalysts have been reported to synthesize chiral molecules,4–6 the simple separation and recycling of chiral catalysts remains a big problem.7,8 Fortunately, heterogeneous chiral catalysis can overcome the abovementioned problems, but for many heterogeneous chiral catalysts, the enantioselectivity problem limits their practical applications. 9 Therefore, it is worthwhile to develop new catalytic systems that not only achieve the separation and recycling of chiral catalysts, but can also lead to high enantioselectivities.
In the past decade, the phase transfer of a catalyst in an aqueous–organic biphasic system has been extensively studied as a way to realize the separation and recycling of catalysts. 10 Among these studies, ionic liquids (ILs), as effective phase-transfer vehicles, have received significant attention.11–14 In our previous study, a thermoregulated phase-transfer catalysis system was developed based on the cloud point (Cp) of the chiral ionic liquid CILTPT [CH3(OCH2CH2)16CD]+[PF6]‒ (CD = cinchonidine). 15 The CILTPT-stabilized chiral Pt nanocatalyst could realize transfer in a H2O–1-pentanol biphasic system. Hence, the chiral Pt nanocatalyst can be easily separated and recycled. However, up to now, almost all the ILs involved in phase transfer of a catalyst are limited to using fluorine-containing anions,10–13 which are expensive and environmentally unfriendly. This of course pertains to our previously reported thermoregulated phase-transfer catalysis system. 15 Therefore, developing new thermoregulated phase-transfer catalysis systems without fluorine-containing anions is important.
In this paper, CILTPT-MS [CH3(OCH2CH2)16CD]+[CH3SO3]‒ (CD = cinchonidine) was investigated and found to exhibit Cp character. As a result of the Cp character, a chiral nano-Pt catalyst stabilized by CILTPT-MS was prepared, which can easily transfer between the H2O–alcohol biphasic system by changing temperature. The first fluorine-free, thermoregulated phase-transfer catalysis system containing a CILTPT-MS-stabilized chiral nano-Pt catalyst has been developed and applied to the asymmetric hydrogenation of α-ketoesters.
Results and discussion
The Cp detection of CILTPT-MS
Our investigations started with the Cp detection of CILTPT-MS. The experimental results indicated that the Cp was higher than 100 °C. Taking into consideration the literature reports that inorganic salts have obvious effects on decreasing the Cp,16–18 the effects of different inorganic salts on the Cp were investigated. As shown in Figure S1 (see the Online Supporting Information), a decrease in Cp was observed on increasing the inorganic salt concentration. This may be due to the decreasing number of hydrogen bonds between water and the polyether chains in CILTPT-MS when inorganic salts are present. 19 Among these inorganic salts, K3PO4·3H2O has the most dramatic effect on decreasing the Cp. The corresponding Cp was 51 °C when the K3PO4·3H2O concentration was 4.8 wt%. Besides, the influence of the CILTPT-MS concentration on Cp was also detected (Figure 1). The corresponding Cp was 74 °C when the CILTPT-MS concentration was 0.5 wt%. The results in Figure 1 indicate that the Cp evidently decreased on increasing the CILTPT-MS concentration. Furthermore, the Cp decreased to 61 °C when the CILTPT-MS concentration was increased to 3.0 wt%.

Effects of the CILTPT-MS concentration on Cp (2.5 wt% K3PO4·3H2O).
The thermoregulated phase-transfer of a chiral nano-Pt catalyst
Based on the Cp of CILTPT-MS, the thermoregulated phase-transfer of CILTPT-MS in a H2O–organic biphasic system was attempted in the presence of K3PO4·3H2O. The results clearly showed that the CILTPT-MS possessed thermoregulated phase-transfer properties in a H2O–alcohol (such as 1-butanol, 1-pentanol, 1-hexanol, and cyclohexanol) biphasic system. Furthermore, the UV-Vis spectra also proved the phase-transfer phenomenon of the CILTPT-MS in the H2O–1-pentanol biphasic system (Figure 2). However, the phase transfer did not occur when the organic phase was toluene or n-heptane.

UV-Vis spectra of CILTPT-MS before and after phase transfer in a H2O–1-pentanol biphasic system upon heating and cooling (the original concentration of CILTPT-MS in H2O was 0.1 wt%).
After investigating the phase-transfer properties, the CILTPT-MS was used as a stabilizer of a nano-Pt catalyst. The results indicated that the chiral nano-Pt catalyst stabilized by CILTPT-MS dispersed very well and exhibited a mean diameter of 1.9 ± 0.4 nm (see Figure S2, A1-2 in the Online Supporting Information). More excitingly, the chiral nano-Pt catalyst also demonstrated the thermoregulated phase-transfer properties in the H2O–alcohol biphasic system. Also, the phase-transfer temperatures of the chiral nano-Pt catalyst from H2O to different alcohols were established. The experimental results showed that the chiral nano-Pt catalyst exhibited distinct phase-transfer temperatures in different alcohols (Table 1).
The phase-transfer temperatures of the CILTPT-MS-stabilized chiral nano-Pt catalyst in H2O–organic biphasic systems.

Organic solvent: 2.000 g, H2O: 1.000 g, CILTPT-MS: 20.0 mg, CILTPT-MS/Pt: 10:1 (mol/mol), K3PO4·3H2O: 50.0 mg.
Taking the H2O–1-pentanol biphasic system as an example, the thermoregulated phase-transfer properties of the chiral nano-Pt catalyst were determined and are shown in Figure 3. The effects of different factors, such as the amount of H2O and 1-pentanol, and the concentration of K3PO4·3H2O, on the phase-transfer temperature were investigated. The results indicated that the phase-transfer temperatures can be controlled within a broad range from 31 °C to >100 °C by altering the K3PO4·3H2O concentration and the amounts of 1-pentanol and H2O (see Table S1 in the Online Supporting Information).

The thermoregulated phase-transfer properties of the chiral nano-Pt catalyst in a H2O–1-pentanol biphasic system.
In addition, with the purpose to test the stability of the chiral nano-Pt catalyst, continuous phase-transfer experiments in the H2O–1-pentanol biphasic system were attempted through heating and cooling. No sign of aggregation was visually noticed after five transfer cycles. Furthermore, the transmission electron microscope (TEM) images of the freshly prepared chiral nano-Pt catalyst and that after five cycles showed that the particle size had only slightly increased from 1.9 to 2.2 nm (see Figure S2, B1-2 in the Online Supporting Information). These results confirmed that the CILTPT-MS-stabilized chiral nano-Pt catalyst displayed good stability.
The asymmetric hydrogenation of α-ketoesters catalyzed by the chiral nano-Pt catalyst
After the construction of this novel thermoregulated phase-transfer catalysis system, its catalytic efficiency was evaluated in the asymmetric hydrogenation of α-ketoesters. Such compounds have been studied extensively, serving as effective precursor for synthesizing optically active α-hydroxy esters.20–30 However, among the numerous reports, only a few studies are available that focus on recycling of the chiral catalyst and in which the enantiomeric excess (ee) values still need improvement.30–34 Therefore, with the purpose of achieving high ee values and recycling of the chiral catalyst, the asymmetric hydrogenation of methyl benzoylformate (MBF) was investigated as a model substrate and the reaction conditions were systematically explored (Table 2). First, the CILTPT-MS/Pt molar ratio was investigated (Table 2, entries 1–4). The experimental results indicated that the ee increased from 80% to >99% with the molar ratio increasing from 10 to 12.5, while the ee began to decrease when the molar ratio was increased further. Next, the reaction temperature was investigated in the range of 30–50 °C (Table 2, entries 2 and 5–7). At 40 °C, the conversion of MBF increased to >99% from 78%, and the ee increased simultaneously (Table 2, entry 2 vs entry 5). On further increasing the temperature to 50 °C, the conversion of MBF remained unchanged; however, the ee began to decrease (Table 2, entries 2, 6, and 7). In addition, the H2 pressure has a significant effect on the ee. The results indicated that when the H2 pressure was higher than 2 MPa, the ee decreased significantly (Table 2, entries 2, 8, and 9). Therefore, the optimized reaction conditions for the asymmetric hydrogenation of MBF were established with >99% conversion and >99% ee (see Table 2, entry 2).
Asymmetric hydrogenation of MBF catalyzed by the CILTPT-MS-stabilized chiral nano-Pt catalyst.
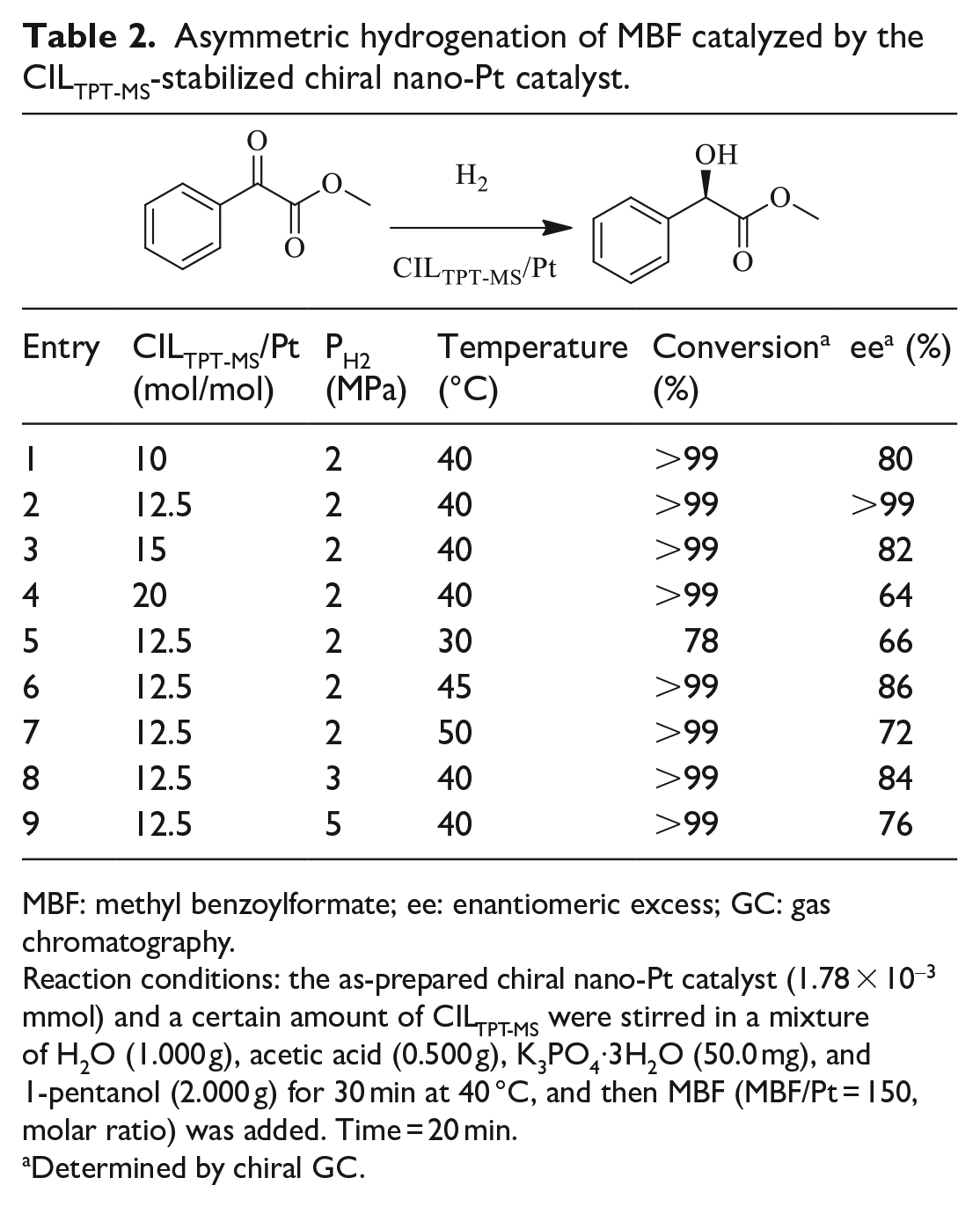
MBF: methyl benzoylformate; ee: enantiomeric excess; GC: gas chromatography.
Reaction conditions: the as-prepared chiral nano-Pt catalyst (1.78 × 10-3 mmol) and a certain amount of CILTPT-MS were stirred in a mixture of H2O (1.000 g), acetic acid (0.500 g), K3PO4·3H2O (50.0 mg), and 1-pentanol (2.000 g) for 30 min at 40 °C, and then MBF (MBF/Pt = 150, molar ratio) was added. Time = 20 min.
Determined by chiral GC.
With optimized conditions in hand, the recyclability of the chiral nano-Pt catalyst was evaluated. After the reaction, the upper alcohol phase containing the products can be simply separated and the lower catalyst-containing aqueous phase was reused in the next cycle. As shown in Table 3, the chiral nano-Pt catalyst showed good recyclability without loss of ee after the catalyst had been used 3 times. Although the conversion decreased to 86% in the fourth cycle, prolonging the reaction time from 20 min to 30 min also achieve a conversion of >99% (Table 3, entry 4).
Recyclability of the chiral nano-Pt catalyst.

ee: enantiomeric excess; MBF: methyl benzoylformate; GC: gas chromatography.
Reaction conditions: the as-prepared chiral nano-Pt catalyst (1.78 × 10-3 mmol) and CILTPT-MS 5.0 mg were stirred in a mixture of H2O (1.000 g), acetic acid (0.500 g), K3PO4·3H2O (50.0 mg), and 1-pentanol (2.000 g) for 30 min at 40 °C, and then MBF (MBF/Pt = 150, molar ratio) was added.
Determined by chiral GC.
In the recycling experiments, the factors that may lead to a decrease in the conversion were further investigated. First, the size of the chiral nano-Pt catalyst was determined by TEM (Figure 4), and the results indicated that the diameter of the chiral nano-Pt catalyst had slightly increased from 2.6 ± 0.4 nm (freshly prepared) to 3.0 ± 0.4 nm after three cycles. In addition, the Pt leaching into the 1-pentanol phase was detected by inductively coupled plasma atomic emission spectroscopy (ICP-AES) at the end of the reaction. These results are presented in Table S2 (see the Online Supporting Information) and indicate that the Pt leaching gradually decreased in each cycle experiment. The average Pt leaching was 1.2 wt%. Therefore, we speculate that the increasing Pt nanoparticle size and the Pt leaching maybe the reasons for the decreased catalytic performance during the recycling experiments.

TEM micrographs and size histograms of the chiral nano-Pt catalysts: (a) freshly prepared; (b) after three cycles.
Moreover, with the purpose of obtaining further information on the nature of the chiral nano-Pt catalyst, mercury poisoning experiments were conducted, which are widely used to determine whether the catalyst was heterogeneous or not.35,36 The experimental results are shown in Table 4. When Hg (Hg/Pt = 1000, molar ratio) was added before the start of the reaction, the conversion of MBF was only 4% (Table 4, entry 2). Compared with the reaction without adding Hg, the addition of Hg almost made the chiral nano-Pt catalyst completely lose its catalytic activity and the MBF conversion was reduced from >99% to 4% (Table 4, entry 1 vs entry 2). Furthermore, another experiment was designed to examine the effect of Hg on the reaction. Thus, Hg was added to the system when the reaction had proceeded for 10 min. The conversion of MBF remained unchanged at 46%, even if the reaction time was further extended to 20 min (Table 4, entry 3 vs entry 4). It is evident from the above data that the addition of Hg poisoned the chiral nano-Pt catalyst. Therefore, in this catalytic process, the chiral nano-Pt catalyst might be heterogeneous.
Mercury poisoning experiments on the asymmetric hydrogenation of MBF catalyzed by the chiral nano-Pt catalyst.

MBF: methyl benzoylformate; GC: gas chromatography.
Reaction conditions: 1.78 × 10-3 mmol of Pt, CILTPT-MS/Pt = 12.5 (molar ratio), H2O (1.000 g), K3PO4·3H2O (50.0 mg), 1-pentanol (2.000 g), and acetic acid (0.500 g), Temp = 40 °C, PH2 = 2 MPa, MBF (MBF/Pt = 150:1, mol/mol).
Determined by chiral GC using cyclohexane as an internal standard.
Based on the above experimental results, the mechanism may be that a chiral microenvironment is formed by the adsorption of the quinoline ring in the chiral ionic liquid CILTPT-MS on the Pt nanocatalyst surface, which is consistent with the reported literature. 37 In addition, the polyether chain in the chiral ionic liquid CILTPT-MS can interact with the Pt nanocatalyst and contribute to the stable adsorption of CILTPT-MS on Pt nanocatalyst surface and therefore leads to the better stability of the chiral microenvironment on Pt nanocatalyst surface.
Finally, to explore the generality of the chiral nano-Pt catalyst, different α-ketoesters were employed as substrates and the results are summarized in Table 5. Under the optimized reaction conditions, four chiral α-hydroxy esters were obtained with >99% ee and >99% conversion (Table 5, entries 1–4).
Asymmetric hydrogenation of different α-ketoesters.

ee: enantiomeric excess; GC: gas chromatography.
Reaction conditions: H2O (1.000 g), 1-pentanol (2.000 g), K3PO4·3H2O (50.0 mg), and acetic acid (0.500 g), Temp = 40 °C, PH2 = 2 MPa.
The conversion and ee values were determined by chiral GC.
Chiral nano-Pt catalyst (1.78 × 10-3 mmol), substrate/CILTPT-MS/Pt = 100:10:1, molar ratio, time = 30 min.
Chiral nano-Pt catalyst (1.78 × 10-3 mmol), substrate/CILTPT-MS/Pt = 100:12:1, molar ratio, time = 40 min.
Chiral nano-Pt catalyst (3.56 × 10-3 mmol), substrate/CILTPT-MS/Pt = 100:8:1, molar ratio, time = 120 min.
Chiral nano-Pt catalyst (1.78 × 10-3 mmol), substrate/CILTPT-MS/Pt = 100:12:1, molar ratio, time = 60 min.
Conclusion
The CILTPT-MS was found to exhibit Cp character. On the basis of the Cp, the first fluorine-free, thermoregulated phase-transfer catalysis system has been developed and explored for the asymmetric hydrogenation of α-ketoesters. High ee values and conversions were obtained for the asymmetric hydrogenation of α-ketoesters, and the chiral nano-Pt catalyst showed good recyclability. More importantly, it is expected that this fluorine-free, thermoregulated phase-transfer catalysis system will provide an approach for designing more environmentally friendly phase-transfer catalytic systems.
Experimental
Chemicals
The CILTPT-MS was prepared as reported. 38 MBF (99%), isoamyl pyruvate (97%), and ethyl 3-chlorobenzoylformate (96%) were obtained from J&K Chemical (Shanghai, China). PtCl4 (99.9%) was obtained from Alfa Aesar (Ward Hill, MA, USA). Ethyl 4-methyl-2-oxopentanoate was purchased from Ark Pharm (Chicago, USA). Pyruvic acid (Z)-3-hexenyl ester (95%) was purchased from TCI (Tokyo, Japan). Chemicals such as 1-pentanol, 1-hexanol, cyclohexane, cyclohexanol, 1-butanol, and acetic acid were purchased from Kermel (Tianjin, China). NaCl, KCl, Na2SO4, KBr, NaNO3, and K3PO4·3H2O were supplied from Sinopharm Chemical Reagent Co. Ltd. (Shanghai, China). All these chemicals were analytical grade and were used without further purification.
Characterization
1 H NMR and 13C NMR spectra were recorded on Varian (400 MHz) spectrometer (Varian, Inc., USA) and Bruker Avance (125 MHz) spectrometer (Bruker, Switzerland), respectively. Chiral gas chromatographic analyses were obtained using a Fuli 9790 GC instrument (FULI Instruments, China) with an Agilent CP-Chirasil-Dex column (25 m × 0.25 mm × 0.25 μm), flame ionization detector (FID), and N2 as the carrier gas. The particle size of the chiral nano-Pt catalyst was characterized by TEM, which was carried out by using a Tecnai G2 20 S-TWIN (200 kV) instrument (Thermo Fisher Scientific, Inc., USA).
Preparation of the chiral nano-Pt catalyst stabilized by CILTPT-MS
In a typical experiment, PtCl4 (0.60 mg, 1.78 × 10–3 mmol), the chiral ionic liquid CILTPT-MS (20.00 mg, 1.78 × 10‒–2 mmol), and H2O (0.400 g) were added to a Teflon-lined standard stainless-steel autoclave (75 mL). The mixture was stirred at 70 °C for 7 h under hydrogen (4 MPa). The autoclave was allowed to cool and depressurized. The change of the color of the solution to black indicated the formation of the chiral nano-Pt catalyst.
Catalytic applications
The hydrogenation of MBF served as a standard model reaction. The reaction was performed in a Teflon-lined standard stainless-steel autoclave (75 mL). The chiral nano-Pt catalyst, CILTPT-MS, H2O, K3PO4·3H2O, 1-pentanol, cyclohexane (internal standard), and acetic acid were injected into the autoclave and the mixture stirred for 30 min. Next, MBF was added and the autoclave was flushed with H2 three times and pressurized with H2 up to the desired pressure. The reactor was held at the given temperature for the appropriate amount of time. After completion of the reaction, the reactor was depressurized and allowed to cool. The 1-pentanol phase was separated and analyzed by chiral gas chromatography (GC). The enantiomeric excess is expressed as ee% = 100 × |R − S| / (R + S).
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211039133 – Supplemental material for A novel thermoregulated phase-transfer catalysis system for chiral nano-Pt-catalyzed asymmetric hydrogenation
Supplemental material, sj-pdf-1-chl-10.1177_17475198211039133 for A novel thermoregulated phase-transfer catalysis system for chiral nano-Pt-catalyzed asymmetric hydrogenation by Pu Chen and Yanhua Wang in Journal of Chemical Research
Supplemental Material
sj-pdf-2-chl-10.1177_17475198211039133 – Supplemental material for A novel thermoregulated phase-transfer catalysis system for chiral nano-Pt-catalyzed asymmetric hydrogenation
Supplemental material, sj-pdf-2-chl-10.1177_17475198211039133 for A novel thermoregulated phase-transfer catalysis system for chiral nano-Pt-catalyzed asymmetric hydrogenation by Pu Chen and Yanhua Wang in Journal of Chemical Research
Supplemental Material
sj-pdf-3-chl-10.1177_17475198211039133 – Supplemental material for A novel thermoregulated phase-transfer catalysis system for chiral nano-Pt-catalyzed asymmetric hydrogenation
Supplemental material, sj-pdf-3-chl-10.1177_17475198211039133 for A novel thermoregulated phase-transfer catalysis system for chiral nano-Pt-catalyzed asymmetric hydrogenation by Pu Chen and Yanhua Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the National Natural Science Foundation of China (No. 21373039).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

