Abstract
This study aims to investigate the adsorption characteristics of tetracycline from polluted waters using C-4-hydroxyphenylcalix[4]resorcinarene as an adsorbent. The adsorptive efficiency of C-4-hydroxyphenylcalix[4]resorcinarene is optimized by adjusting various operational parameters such as the adsorbent dosage, the pH, the contact time, the temperature, and the initial adsorbate concentration. The most efficient remediation is 96% at 10.0 mg/L tetracycline as the initial concentration, 0.05 mg/L C-4-hydroxyphenylcalix[4]resorcinarene, a contact time of 30 min, pH 5.6, and ambient temperature. The adsorption ability of C-4-hydroxyphenylcalix[4]resorcinarene toward tetracycline is also investigated in water with different characteristics, including solutions with and without the addition of background salts. The results show that C-4-hydroxyphenylcalix[4]resorcinarene can effectively remove tetracycline from aqueous solutions with an adsorption capacity of ca. 36.9 mg/g. The study also finds that the removal process followed pseudo-second order kinetics and Freundlich isotherm models. Moreover, the adsorption is spontaneous and exothermic, suggesting a thermodynamically favorable chemisorption process. In addition, the optimized method is successfully applied to remove tetracycline from various real natural water systems.

Introduction
Water is crucial in maintaining life and supporting various human activities; therefore, effectively managing and conserving water resources is vital. Antibiotic contamination in water resources is a growing concern worldwide. Their residues can enter water resources through wastewater discharge from hospitals, pharmaceutical industries, and livestock operations. Indeed, the safety and sustainability of our water resources are affected by contamination by antibiotics, potentially impacting the ecosystem and the health of human beings.
Among them, a highly used antibiotic is tetracycline (TC). The low cost and broad-spectrum activity of TC make it a popular choice for the treatment of various infections, especially in developing countries where access to more expensive antibiotics may be limited. However, the widespread use of TC has led to the presence of residual TC in water environments.1,2 The presence of even low levels of TC in the ecosystem may cause a significant risk to human and animal health. 3 Therefore, proper disposal management and effective treatment methods should be employed to minimize TC residues in water sources.
The purification of water contaminated with TC is a difficult task. The high resistance of TC to degradation and its small size renders the task of removing TC from water challenging using traditional treatment methods. 3 In addition, the different characteristics of water samples add to the complexity of the purification process. The removal of TC from aqueous environments has been successfully reported using various methods, including photocatalysis, redox reactions, biodegradation, and membrane filtration.4–8 However, such techniques may not be convenient for practical purification strategies, and more research is needed to develop effective and economical methods for TC removal from water systems.
Adsorption is a widely used technique for the treatment of water systems because of its high removal rate, cost-effectiveness, simplicity, reusability, and mitigation generating of harmful products.9,10 A variety of adsorbents, such as activated carbon, clay minerals, zeolites, and various types of biomaterials have been tested for the adsorption of TC from aqueous media.1,11–15 The choice of an adsorbent depends on availability, reproducibility, cost, and the specific application. A recent study reported a ca. 90% removal of TC using a high biochar dosage (1.0 g) as the adsorbent. 13 Another study demonstrated a 100% removal of TC at a high pH value. 16 Furthermore, a zeolite-based adsorbent afforded a ca. 98% removal within 60 min. 2 In this regard, it is crucial to develop a rapid treatment method that can be operated under ambient conditions with a minimal amount of adsorbent.
Calix[n]arenes are basket-shaped macromolecules with a hydrophilic lower rim due to phenolic side groups and a hydrophobic upper rim due to aromatic and aliphatic side groups. Noticeably, a wide range of calixarene derivatives and their inclusion complexes are used in controlled drug delivery. 17 Along with their unique molecular conformation and receptor properties, the intrinsic pores of the calixarene structure appear as promising adsorbents and stimulate water treatment studies.18–23 Moreover, the ability to modify the upper or lower rim of the calixarene molecule with various functional groups enhances its ability to bind toxic contaminants. These supramolecular compounds can act as molecular hosts and form inclusion complexes with guests such as ions and small molecules.17,20,24,25 A recent study successfully applied p-piperdinocalix[4]arene to remove Reactive Blue (RB-19) dye. 18 Another recent study used cyclodextrin-calixarene as an effective adsorbent to remediate Pb2+ ions from wastewater. 26 The present study aims to develop a more efficient and cost-effective method for practical applications in water purification, which is an urgent issue due to the potential environmental and health hazards caused by TC residues in the ecosystem. Consequently, it is essential to note that more effort is required to develop methods that operate rapidly under ambient conditions with a minimal amount of adsorbent for TC removal from water systems.
This study proposes using easily prepared C-4-hydroxyphenylcalix[4]resorcinarene (HPCR) as an adsorbent for the adsorption of TC from simulated real water samples. Batch adsorption tests were conducted using different ratios of sorbent and TC in aqueous media, with the pH, contact time, and temperature being varied. The kinetics, isotherms, and thermodynamics of TC adsorption were also investigated.
Results and discussion
This work aims to provide a reliable and efficient solution for TC removal from water using a regenerable adsorbent material. This information can be used to develop a practical and efficient adsorption process to purify water contaminated by TC. HPCR (

The synthesis of HPCR (
Optimization of different parameters
Impact of adsorbent dosage (dosage effect)
The influence of the adsorbent dosage on the adsorptive removal and capacity was investigated in the range from 0.05 to 0.2 g/L under optimal conditions. Figure 1 shows that removal of TC improved on decreasing the adsorbent dosage, reaching a maximum removal efficiency of 96% at a dosage of 0.05 g. The adsorption percentage of TC was slightly lowered at higher adsorbent doses and almost remained constant. It was also observed that the adsorption capacity decreased dramatically as the adsorbent dosage increased. This trend could be attributed to a decrease in the availability of active sites resulting from the accumulation of adsorbent material. Thus, the optimum amount of the adsorbent was 0.05 g.

Impact of the adsorbent dosage on the adsorptive removal of tetracycline (TC) and the capacity. Conditions: pH, 5.6; contact time, 30 min; temperature, 25°C; TC initial concentration, 10.0 mg/L. Bars represent the standard deviation of three measurements.
Electrolyte effect
The adsorption ability of HPCR toward TC was also investigated in water solutions with different characteristics, including solutions with or without the addition of background salts. The impact of various common electrolytes (CH3COONa, KCl, NaCl, and AgNO3) on the percentage of TC adsorption was explored under optimal conditions, and the results are shown in Figure 2. It was found that the presence of NaCl did not significantly affect the efficiency of TC removal. In contrast, adding other electrolytes affects the adsorption ability of HPCR in the order KCl < CH3COONa < AgNO3, where KCl has the most negligible impact, and AgNO3 has the most. A similar result was reported in the literature.27,28 This is likely because such electrolytes may alter the pH of the solutions, negatively impacting adsorption. Such electrolytes (CH3COONa, KCl, NaCl, and AgNO3) can also change the ionic strength of the solution, altering the surface charge of the adsorbent, and competing with TC for adsorption sites on the adsorbent, leading to a decrease in the percentage of TC adsorption. In addition, AgNO3 is a potent oxidizing agent and can potentially oxidize TC, thereby significantly decreasing its adsorption.

Influence of different electrolytes (concentrations: 0.5 and 0.1 M) on the percentage adsorption of tetracycline (TC). Conditions: adorbent dosage, 0.05 g/L; pH, 5.6; contact time, 30 min; temperature, 25°C; TC initial concentration, 10.0 mg/L.
Impact of pH
The pH of aqueous systems plays a causative role, affecting the removal percentage of TC and its adsorption efficiency onto the adsorbent. Therefore, reference samples with different pH values (2–6) were tested under the optimized conditions. Figure 3(a). shows that the best adsorption of 96% was obtained at a pH of 5.6, indicating that almost neutral pH values are more favorable for adsorption. In contrast, the adsorption percentage of TC was slightly reduced at a pH of 6. This behavior may be attributed to deprotonation of both the TC molecule and the adsorbent, causing slightly negatively charged surfaces and electrostatic repulsion. 19 At low pH values, protonated forms of TC predominate and so do not favor adsorption onto the adsorbent surface. 29 In addition, the decrease in adsorption at lower pH is also attributed to the abundance of H+ ions that compete with TC for active sites on the adsorbent surface, leading to a decrease in the adsorption percentage. Therefore, a pH of 5.6 was maintained for subsequent experiments.

(a) Dependence of the percentage adsorption of tetracycline (TC) on the pH, and (b) the pH point of zero charge (pHPZC). Other conditions are as described in Figure 2. Bars represent the standard deviation of three trials.
Figure 3(b) illustrates the impact of pH on the charge of the adsorbent surface. The pH point of zero charges (pHPZC) was 5.6, implying that the adsorbent surface has a net zero charge at this pH value. It represents the pH at which the surface of the adsorbent has a balanced charge, meaning that there are equal amounts of positively and negatively charged groups. It is known that the adsorbent has a positively charged surface with a solution pH lower than pHPZC, and its surface may be negatively charged as the solution pH increases above the pHPZC value. It was noticed that the solution pH can significantly affect the adsorption process; therefore, knowing the pHPZC value can provide insights into how the pH influences the adsorption. Briefly, a change in the solution pH can lead to a change in the charge on the adsorbent surface, which is consistent with the pH-dependent adsorption results described above. The results are in good agreement with those obtained previously. 30
Effect of contact time and the removal kinetics
Adsorption is a multistep process influenced by various factors, so kinetic studies were conducted to explore the TC removal rate by HPCR. Figure 4(a) shows the adsorptive capacity of HPCR as a function of time. It is remarkable that the adsorption process started quickly and then leveled off as equilibrium was reached. It is worth noting that TC adsorption onto the HPCR surface may become more difficult as the adsorption process progresses as the HPCR surface is rapidly filled.

(a) Impact of the contact time on the sorption capacity of tetracycline (TC), and the kinetic plots of (b) pseudo-first order and (c) pseudo-second order. Other conditions are as described in Figure 2.
The adsorption mechanism of TC was further characterized using the most common kinetic models. Herein, pseudo-first order (Figure 4(b)) and pseudo-second order (Figure 4(c)) models were applied. They are expressed by equations (1) and (2), respectively. The adsorption capacity of the adsorbent in mg of TC per gram of HPCR at equilibrium (qe) and at time t (qt) was computed from the experimental data. The extracted parameters of both models are given in Table 1
Kinetic parameters for the adsorption of TC.

TC: tetracycline.
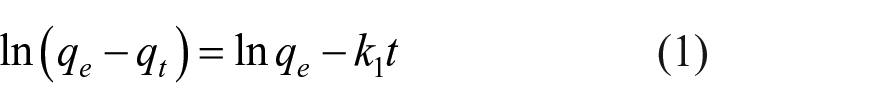

The reaction rate constants of adsorption are represented by k1 and k2, respectively, as shown in Table 1.
These models help to understand the TC adsorption mechanism on HPCR and how it progresses over time. The fitting level was evaluated using the correlation coefficient (R2) as well as the comparability when comparing the experimental (qe, exp) and calculated (qe, cal) adsorbed amount of TC at equilibrium. The kinetic results indicated that the pseudo-second order model better fitted the adsorption data. The obtained results suggest that the chemisorption mechanism may control the adsorption process between TC and the HPCR surface.
Influence of the initial TC concentration and the removal isotherms
The adsorption isotherms could be used to describe the relationship between the adsorbed amount of TC and its concentration in the surrounding solution at equilibrium. They can provide information about the adsorption capacity, the nature of the adsorbent-adsorbate interactions, and the type of adsorption (monolayer or multilayer). The Langmuir and Freundlich isotherms are utilized often in describing adsorption data based on different assumptions about the adsorption process. 31 The Langmuir model postulates monolayer adsorption on a homogeneous surface, where the adsorption sites have the same binding energy. The Freundlich model, on the other hand, supposes multilayer adsorption on a heterogeneous surface, where the adsorption sites have different binding energies.
The impact of the TC concentration on the adsorption was explored within a specific range of initial concentrations, from 10 to 50 mg/L under the optimized conditions. This range was chosen based on the expected concentration range of TC in the system being studied. Figure 5(a) depicts a graph showing the relationship between the initial concentration of TC and its removal efficiency. As the initial TC concentration increases from 10 to 50 mg/L, the removal efficiency reduced significantly from 96% to 74%. This means that the adsorption process is less effective in removing TC at higher initial concentrations. This is likely attributed to a number of factors, such as increased competition for adsorption sites and saturation of the adsorbent material. In contrast, the adsorptive capacity of HPCR was found to increase on increasing the TC concentration, indicating the good adsorptive ability of HPCR toward TC. The obtained results are in good agreement with prior literature data. 31

(a) Impact of the initial tetracycline (TC) concentration on the adsorption removal and capacity, and isotherm plots of (b) Langmuir and (c) Freundlich for the TC adsorption. Other conditions are as described in Figure 2. Bars represent the standard deviation of three trials.
The Langmuir model is described by equation (3). The Langmuir equation relates the adsorption capacity of HPCR (qe, mg/g) to the concentration of TC in the solution (Ce, mg/L) at equilibrium. The maximum monolayer adsorption capacity (qm) was calculated and is presented in mg of TC per g of adsorbent, and kL (L/mg) is the Langmuir constant. The Freundlich model, described by equation (4), has parameters qe, Ce, n, and KF, where kF (mg1−1/n·g−1·L−1/n) is the Freundlich constant and n represents the surface heterogeneity of the adsorbent


The results in Figures 5(b) and (c) and Table 2 show that the Langmuir and Freundlich models fit well with the adsorption data and exhibit high R2 values. Meanwhile, it is observed that the R2 value is higher for the Freundlich isotherm as compared with the Langmuir isotherm, indicating the best fit to the observed adsorption data. Therefore, the multilayer adsorption mechanism onto a heterogeneous surface defined by the Freundlich isotherm dominates the TC adsorption process, which agrees with the previous literature data. 32 According to this isotherm model, the maximum adsorption capacity was ca. 36.9 mg/g. The 1/n (0.469) and RL (0.178, separation factor) values were less than unity, confirming the favorability of TC adsorption onto HPCR.
Isotherm parameters for TC removal by HPCR.

TC: tetracycline; HPCR: hydroxyphenylcalix[4]resorcinarene.
A comparison between our prepared adsorbent and others reported in the literature is summarized in Table 3. It can be seen that the removal efficiency of the adsorbent used in this study is higher than those of others reported in previous studies. In addition, the superiority of our adsorbent can also be depicted in terms of saving chemicals and time.
Comparison of the TC removal by HPCR with other sorbents.

TC: tetracycline; HPCR: hydroxyphenylcalix[4]resorcinarene.
Impact of temperature and the adsorption thermodynamics
Different thermodynamic parameters providing insight into the nature of the adsorption process and the driving forces behind it were investigated. In the current study, ΔS0 (J/mol/K), ΔH0 (kJ/mol), and ΔG0 (kJ/mol) were computed using the following equations



According to these equations, the parameters of the adsorption thermodynamics can be calculated by measuring the distribution coefficient (Kd), the change in entropy (ΔS0), the change in enthalpy (ΔH0), and the change in Gibbs free energy (ΔG0) at a specific temperature. The distribution coefficient (Kd) measures the partitioning of the adsorbate between the adsorbent and the aqueous phases. It can be used to estimate the adsorptive capacity of HPCR. A positive value of ΔS0 suggests an entropy-favored adsorption process and a negative value of ΔH0 implies it is exothermic in nature. A negative value for ΔG0 reveals the spontaneity and favorability of the adsorption process, indicating the higher stability of the adsorbate on the adsorbent surface than in solution. Understanding these thermodynamic parameters can help to optimize the adsorption process for a specific application.
The results of the temperature effect found that TC removal was reduced significantly on raising the temperature, as shown in Figure 6(a). The ΔS0 and ΔH0 values are presented in Table 4 and computed from the plot of ln Kd versus 1/T (Figure 6(b)), which in turn can be used to calculate ΔG0.

(a) The variation of TC percentage adsorption with solution temperature and (b) thermodynamics of the removal of TC by HPCR. Other conditions are as described in Figure 2. Bars represent the standard deviation of three trials. TC: tetracycline; HPCR: hydroxyphenylcalix[4]resorcinarene.
Thermodynamic parameters for TC removal by HPCR.

TC: tetracycline; HPCR: hydroxyphenylcalix[4]resorcinarene.
The negative ΔG0 value implies the thermodynamic favorability of the TC adsorption process. Similarly, the negative ΔH0 value suggests the exothermic nature of the removal process. The results from the thermodynamic analysis further support the earlier observation described above in which the adsorption capacity decreased as the temperature increased. The spontaneity of the adsorption process was found to be reduced as the temperature was increased, and thus heat diminished the adsorption of TC onto HPCR.
Recyclability of the adsorbent
A successful reusability test would indicate that the adsorbent has good stability and can be reused multiple times without significantly decreasing its adsorption efficiency. This would result in cost savings in the treatment process and reduced waste generated from disposal of the adsorbent. The results of the reusability test can provide valuable information for the practical application of HPCR as an adsorbent for the removal of TC from water. Herein, the used HPCR was first rinsed with dH2O and dried in an electric oven. After that, HNO3 solution was used to regenerate the adsorbent, which was then separated and rinsed at least five times with dH2O.
This study also evaluated the potential for recycling and reusing the adsorbent material; the results are presented in Figure 7. The results of the recycling experiments showed that HPCR had a relatively poor reuse capacity, with a significant drop in the adsorption capacity after the first desorption cycle (from 96% to 78%). After the second cycle, the prepared adsorbent maintained its adsorption capacity (ca. 62%). This poor performance may be due to structural alterations of HPCR after interacting with TC. In addition, the adsorption–desorption process may result from a combination of chemical and physical processes, and the specific conditions of the aqueous solution containing the TC may affect the results. 19 This study suggests HPCR may be a promising adsorbent material for removing TC from aqueous solutions.

Reusability of HPCR. Conditions are as described in Figure 2. Bars represent the standard deviation of three trials. HPCR: hydroxyphenylcalix[4]resorcinarene.
Proposed adsorption mechanism(s)
The adsorption process depends on the chemical nature of adsorbates, the surface properties of adsorbents, and the conditions under which the adsorption occurs. The properties of the adsorbent surface, including its active area, pore size, and chemical functional groups, may also be involved in determining its adsorption capacity and efficiency. 22 Similarly, organic macromolecular steric hindrance refers to the interference of larger organic molecules with the adsorption process. 22 If the contaminants in the wastewater are large organic molecules, they may not fit into the pores of the adsorbent, hindering their removal through adsorption. On the contrary, heterogeneous adsorption sites refer to the presence of different active sites on the adsorbent surface with varying adsorption properties. This may affect the adsorption efficiency, as specific sites may more easily adsorb particular contaminants. The adsorbent–adsorbate interaction is also critical, as it determines the strength of the adsorption and, thus, the amount of adsorbate that can be removed. The charge of the adsorbent surface affects the interaction, and the presence of specific chemical functional groups may also enhance the selectivity of the adsorption process.
Overall, combining the results from the kinetic, isotherm, and thermodynamic models, along with the effect of pH provides a detailed understanding of the TC adsorption mechanism. Thus, the adsorption of TC onto HPCR is likely to be a combination of various mechanisms: electrostatic interactions, pore adsorption, hydrogen bonding, and π−π interactions. The results show how well the pseudo-second order model describes the TC adsorption process, suggesting that it proceeds via electrostatic and chemisorption interactions. A chemical interaction is expected between the abundant hydroxy group moieties of the adsorbent surface and nucleophilic atoms (nitrogen and oxygen) in the TC molecules. Other processes that may also impact the adsorption affinity include aromaticity and hydrophobicity. 22
In addition, the highest adsorption capacity was found at the point of zero charges, where fewer repulsive forces are present between the adsorbent and adsorbate, leading to a higher degree of adsorption. The Freundlich isotherm model demonstrated the multilayer adsorption of TC onto the heterogeneous surface of the HPCR.
Application to real water samples
A total of 12 real water samples representing various sources, including Al-Shoubak Pool, King Talal Dam, and the wastewater treatment facility located at Mutah University, Jordan, were collected. Herein, the water samples were used to assess the effectiveness of the proposed method for removing TC under various conditions. All of the water samples were filtered, analyzed, and found to have no detectable amount of TC. As a result, a specific amount of TC was spiked into the samples to give a final concentration of 10 mg/L. Adsorption experiments were conducted, and the removal efficiency was found to be 96%, 83%, 80%, and 76% from the simulated tap water, Al-Shoubak Pool, King Talal Dam, and wastewater samples, respectively. The removal efficacy of our sorbent was higher than that reported in the literature utilizing CuCoFe2O4@Ch when applied to wastewater samples. 32 The results showed that the proposed method was highly effective and worked well in the presence of other ionic species and contaminants commonly found in actual water samples.
Conclusion
This study highlights the potential use of HPCR as an efficient adsorbent for the removal of TC from aqueous solutions. The highest removal efficiency (96%) and adsorption capacity (36.9 mg/g) were determined at 10.0 mg/L TC as the initial concentration, 1.0 mg/L HPCR, a contact time 30 min, pH 5.6, and room temperature. The adsorption ability of HPCR toward TC was investigated in samples of water with different characteristics. The study found that adsorption followed pseudo-second order kinetics and the Freundlich isotherm model. Accordingly, the multilayer adsorption of TC onto a heterogeneous surface of HPCR proceeded mainly via chemisorption. The thermodynamic results also revealed that the adsorption process is spontaneous and exothermic. Our study also examined the reusability of the HPCR adsorbent by testing its ability to be regenerated and reused. Unfortunately, a slightly poor reuse capacity was observed throughout the recycling experiments.
Overall, this study provides valuable insights into the use of HPCR as a promising adsorbent for TC removal from water systems. However, further research is required to understand the underlying adsorption–desorption mechanisms and how to improve the recycling potential of HPCR. Also, it is important to note that additional work is needed to investigate the scalability and long-term stability of this method for practical use.
Materials and methods
Chemicals and instrumentation
All chemicals and reagents were used as-received, and deionized water (18.2 µΩ cm-1) was used to prepare all solutions. p-Hydroxybenzaldehyde (C7H6O2, 98%) and resorcinol (C6H6O2, 98%) were obtained from Sigma Aldrich. Tetracycline (C22H24N2O8) was purchased from Amresco (OH, USA). Hydrochloric acid (HCl, 37%), potassium chloride (KCl, 99.5%), and nitric acid (HNO3, 70%) were of analytical grade and were purchased from Sigma Aldrich. Sodium chloride (NaCl), anhydrous sodium acetate (CH3COONa), silver nitrate (AgNO3, 99%), and ethanol (C2H6O, 99.5%) were obtained from Merck. A pH meter (HANNA instruments, HI5521-02) was utilized to determine the pH of solutions. A Perkin–Elmer Model lambda 25 UV–Vis spectrophotometer was employed to analyze the residual TC content at λmax = 280 nm.
Preparation of the calixarene
HPCR was synthesized by a single-step organic reaction, as reported previously.
35
The full characterization of HPCR, which are comparable with our results (data not shown), have already been reported in previous work.35 Scheme 1 shows the single-step synthesis of HPCR. An ice-cold solution of resorcinol (
Batch adsorption experiments
Standard solutions of TC were prepared by weighing a specific quantity of TC and dissolving it in deionized water. The stock solution was appropriately diluted with deionized water to the desired concentrations. Aluminum foil was used to wrap 250 mL Erlenmeyer flasks to eliminate the effect of light throughout the batch adsorption experiments. To evaluate the impact of various operational parameters on TC adsorption, point of zero charges (PZC), adsorbent dosage (0.05, 0.1, 0.15, and 0.2 g/L), pH (2, 3, 4, 5, 5.6, and 6), contact time (5, 15, 25, 30, 35, 60, and 120 min), temperature (25, 30, 35, 40, and 45°C), and initial adsorbate concentration (10, 30, and 50 mg/L) were investigated.
The study used batch adsorption experiments. The adsorbent and TC were mixed in different ratios (%w/w) in aqueous media (50 mL) and equilibrated with an orbital shaker over different time periods. Next, a 0.22-μm filter membrane was used to separate the adsorbent. The residual TC content was analyzed in the filtrate by using a UV–Vis spectrophotometer before and after adsorption. The adsorbent and TC were mixed in different ratios (%w/w) by varying the HPCR dosage from 0.05 to 0.2 g/L and the initial TC concentration from 10 to 50 mg/L under optimal conditions. The effect of pH on the TC adsorption was examined in the range from 2 to 6 using 1.0 g/L of HPCR and 10 mg/L of TC as the initial concentration. The adsorptive capacity of HPCR was also studied over different contact times and temperatures under the optimized conditions. All experiments were conducted under controlled conditions by shaking at 250 r/min for 30 min at room temperature. Solutions of 1 M HCl and 1 M NaOH were used to adjust and maintain the initial pH of the solutions at 5.6. The adsorption experiments were repeated three times, and the averaged results are presented.
The adsorption amount (equation (8)) and removal efficiency (equation (9)) were computed based on the initial (Ci) and final (Ct) amounts of TC as well as the mass of adsorbent (W), and the volume of the aqueous media (V) used. 2


The kinetic study examined the time when equilibrium is reached under the optimum conditions. The adsorption rate as a function of time was further analyzed using the pseudo-first order and pseudo-second order models. The isotherm mechanism was determined by examining the adsorption capacity as a function of the equilibrium TC amount and additionally explored using the Langmuir and Freundlich models. The adsorption thermodynamic was also performed by analyzing the relationship between the adsorptive capacity and the temperature, and then by determining the entropy, the enthalpy, and the Gibbs free energy changes. The results of this study provide insight into the potential of HPCR as a promising adsorbent and the impact of different parameters on the adsorption process.
Determining pHPZC
To further understand the mechanism of TC adsorption, various characterization techniques, such as point of zero charge pH (pHPZC), can be used to investigate the surface properties of adsorbents. Herein, a number of solutions in separate Erlenmeyer flasks with different initial pH values (2, 4, 6, 8, 10, and 12) were prepared containing HPCR (0.1 g) and 0.1 M KCl (50 mL, aqueous solution). The flasks were shaken and maintained at 250 r/min and 25°C for 48 h. The resulting mixtures were centrifuged, and the final pH was measured. The experimental results are plotted as a graph of ΔpH (change in pH) versus the initial pH. The pH at which ΔpH became zero was taken as the apparent PZC value of the HPCR. 32
Desorption and regeneration assays
In the reusability test, the TC adsorption efficiency was evaluated after the used HPCR had been treated with four different desorption agents: deionized water (dH2O), nitric acid (HNO3), hydrochloric acid (HCl), and ethanol (C2H5OH). Herein, HNO3 was found to be the best desorption agent. This test aimed to determine the stability and reusability of HPCR, as well as its ability to retain its adsorption properties after being washed with different desorption agents.
The used HPCR was first rinsed with dH2O and dried in an electric oven. After that, HNO3 solution (0.1 M, 50 mL) was used to regenerate the adsorbent over 30 min by shaking at 250 r/min, which was then separated and rinsed at least five times with dH2O. Once the adsorbent had been dried, it could be reused in further cycles to examine its adsorptive efficiency. Under the optimized conditions, the adsorption–desorption process was repeated four times to evaluate the reusability potential of the adsorbent.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the deanship of scientific research at Mutah University, Jordan, for supporting this work. This study was funded by a research grant from the Mutah University (decision no.: 525/2022). The funders had no role in the study design, data collection and analysis, decision to publish, or the preparation of the manuscript.
