Abstract
A Co(NO3)2 method to promote cyclization of benzoyl hydrazone for the formation of 2,5-diphenyl-1,3,4-oxadiazoles has been developed. The reaction proceeded smoothly and was promoted by Co(NO3)2 under air at 110 °C in DCE; 16 examples of products were obtained.
Graphical abstract

Introduction
2,5-Disubstituted-1,3,4-oxadiazoles are important scaffolds which have been found in many natural and synthetic molecules with unique physical characteristics1–3 and remarkable biological activities (Figure 1).4,5

Representative examples of 2,5-disubstituted 1,3,4-oxadiazoles.
In view of their great importance, the synthesis of 2,5-diaryl-1,3,4-oxadiazoles has gained much attention in recent decades and many useful synthetic procedures for their preparation have been developed. For example, arylation of 1,3,4-oxadiazoles (Scheme 1(a)),6–8 aminocarbonylation reaction of aryl halides with chloroform and tetrazoles (Scheme 1(b)),9–12 oxidative dehydrogenation of

Synthetic approaches toward 2,5-Disubstituted 1,3,4-oxadiazoles.
As shown in Scheme 2, many different methods have been developed for the synthesis of 2,5-substituted-1,3,4-oxadiazoles from aldehydes and acyl hydrazides (Scheme 1(h)). For example, halogen,20–22 organic peroxide,23,24 inorganic oxides, 25 sulfur and oxygen, 26 and palladium (0) 27 can all promote oxidative cyclization of acylhydrazones to 2,5-substituted 1,3,4-oxadiazoles. Similarly, photooxidation 28 and electrooxidation27–31 also have been applied to this reaction. In addition, we have shown that cobalt nitrate plays a special role in organic synthesis and is different from other cobalt salts and nitrates of other metals.32,33 Inspired by this work and our continuing efforts on C−H bond activation reactions,34–37 we here report the development of an efficient method for the Co(NO3)2-mediated, one-pot synthesis of unsymmetrical 2,5-diaryl-1,3,4-oxadiazoles. This method requires mild conditions with high substrate tolerance and good selectivity; high yields of products were obtained.

Synthetic of 2,5-Disubstituted 1,3,4-oxadiazoles from aldehydes and acyl hydrazides.
Results and discussion
Scheme 3. Scale-up experiment
First, we investigated the influence of the nitrate (Table 1, entries 1–5). We observed that the best reproducible yield was obtained when Co(NO3)2•6H2O in DCE was used (Table 1, entry 1). Several other solvents, such as 1,4-dioxane, THF, CH3CN, toluene, and acetone were examined, but the yield was not as good (Table 1, entries 6–10). Finally, the yield of
Optimization of reaction conditions.
a


Reaction conditions:
Isolated yields.
Nitro source (2.0 equiv.).
Nitro source (20 mol%)
Note: DCE = 1,2-dichloroethane.
With the optimized conditions in hand, we investigated the substrate scope of this reaction, beginning by varying the substituents on the benzene ring of the aromatic aldehyde (Table 2). Substrates derived from aromatic aldehydes with
Substrate scope. a
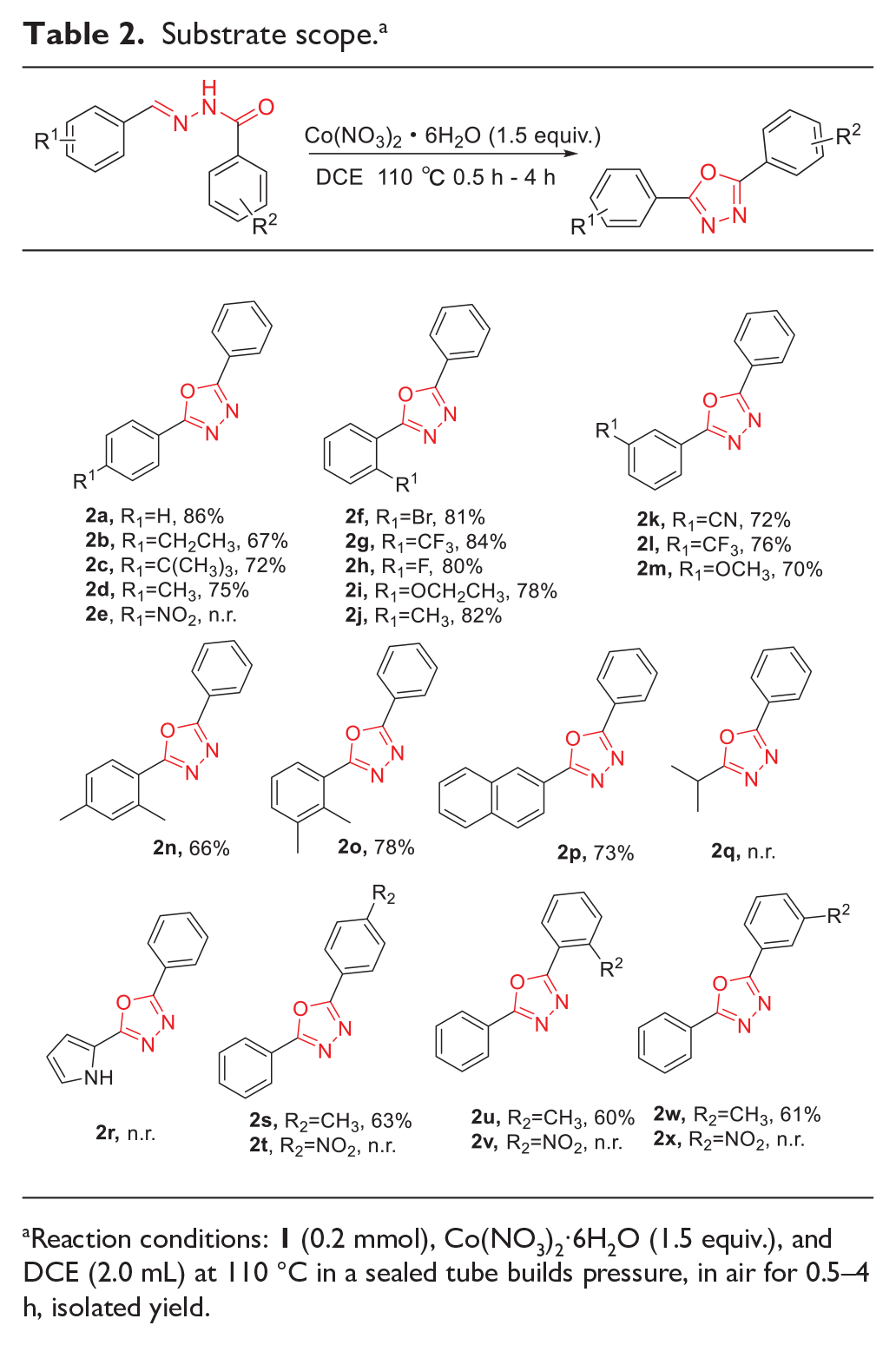
Reaction conditions:
A gram-scale preparation of

Scale-up experiment.
We then wanted to learn more about the mechanism of this reaction and conducted several control experiments to elucidate some of the key steps. First, we found that the reaction was inhibited when a radical scavenger such TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy) was added to the reaction mixture (Scheme 4(a)). Furthermore, employing 1-diphenylethylene as starting material and reacting under the standard conditions (Scheme 4(b)), we observed a clean transformation to the nitroolefin

Control Experiments.
On the basis of previous related studies14,38 and the results we obtained, a plausible reaction mechanism for this direct synthesis of 2,5-diaryl-1,3,4-oxadiazoles is proposed in Scheme 5. Initially, the compound

Plausible mechanism.
Conclusion
In conclusion, we have developed a facile synthesis of 2,5-diaryl-1,3,4-oxadiazoles via a cyclization protocol employing cobalt nitrate and a benzoyl hydrazone. This methodology works under mild conditions and provides a direct approach for the synthesis of various 2,5-diphenyl-1,3,4-oxadiazoles, which are useful in organic and pharmaceutical industries, from commercially available starting materials under mild reaction conditions.
Experimental section
General information
All the chemicals were obtained commercially and used without any prior purification. 1 H NMR spectra were recorded on a Bruker Advance II 400 spectrometer. All products were isolated by short chromatography on a silica gel (200–300 mesh) column using petroleum ether (60°C–90 °C) and ethyl acetate. Unless otherwise noted. All compounds were characterized by 1H NMR and 13 C NMR, which are consistent with those reported in the literature.
General procedure for synthesis of various 2,5-substituted 1,3,4-oxadiazoles
A mixture of the 2,5-substituted 1,3,4-oxadiazole (0.2 mmol) and Co(NO3)2•6H2O (1.5 equiv.) in CH2ClCH2Cl (2.0 mL) was stirred in sealed tube at 110 °C for 0.5–4 h. After cooling down to room temperature and concentrating in vacuum, the residue was purified by flash chromatography on a short silica gel column to afford the product.
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211045932 – Supplemental material for Cobalt(II) nitrate promoted cyclization of benzoyl hydrazone for the synthesis of 2,5-diphenyl-1,3,4-oxadiazole derivatives
Supplemental material, sj-pdf-1-chl-10.1177_17475198211045932 for Cobalt(II) nitrate promoted cyclization of benzoyl hydrazone for the synthesis of 2,5-diphenyl-1,3,4-oxadiazole derivatives by Xiaojun Li, Shangtie Liao, Yu Chen, Chengcai Xia and Guodong Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Academic promotion program of Shandong First Medical University (nos.2019LJ003) for financial support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.