Abstract
A fast and efficient Selectfluor-triggered fluorination/cyclization reaction of o-hydroxyarylenaminones has been successfully developed. The reaction successfully provides an expedient method for the synthesis of 3-fluoro-chromones promoted by potassium carbonate, which shows readily available starting materials and is easy to operate. In addition, a plausible mechanism of this tandem cyclization reaction was proposed where 4H-chromen-4-one, 2-(dimethylamino)-3,3-difluorochroman-4-one, and 3,3-difluoro-2-hydroxychroman-4-one were not found to be the reactive intermediates. Moreover, these novel compounds have been obtained in moderate to good yields, and their structures have been confirmed by 1H NMR, 13C NMR, and high-resolution mass spectrometry.
A direct fluorination/cyclization of o-hydroxyarylenaminones by Selectfluor-triggered has been successfully developed. The reaction successfully provides an expedient method for the synthesis of 3-fluoro-chromones promoted by potassium carbonate under mild reaction conditions, which shows readily available starting materials and operational simplicity. In addition, 4H-chromen-4-ones; 2-(dimethylamino)-3,3-difluorochroman-4-ones; and 3,3-difluoro-2-hydroxychroman-4-ones were not to be intermediates in this strategy. Accordingly, a plausible reaction mechanism was proposed. Moreover, these novel compounds have been obtained in moderate to good yields, and their structures have been confirmed by 1H NMR, 13C NMR, infrared spectroscopy, and high-resolution mass spectrometry.

Fluorinated organic compounds have attracted great attention in the field of medicinal chemistry owing to their unique physical and chemical properties, bioavailability, lipophilicity, and metabolic stability.1–4 This is especially true in the pharmaceuticals and agrochemicals, where fluorine is often considered a bioisostere of hydrogen. At present, about 30% of all agrochemicals (such as pyroxsulam and fluxapyroxad and so on) and 20% of all pharmaceuticals (such as 5-fluorouracil and norfloxacin and so on) incorporate at least one fluorine atom. 5 As such, many efforts have been put into the development of fluorinated molecules based on the C–F bond formation in the past decade.6–10
Chromones and their derivatives are found in numerous natural products and pharmaceuticals11–13 that show many biological activities, such as monoamine oxidase inhibitors and antitubercular activity.14–18 They also exist widely in pigments, dyes, and essential nutrients for the human body.19–22 Consequently, the development of effective methods to construct these heterocyclic scaffolds, especially C3-functionalized analogs, has been studied extensively.23–31
Furthermore, enaminones have been recognized as a class of available and powerful synthetic building blocks owing to their versatile reactivity in a variety of organic transformations,32–38 which were widely used in syntheses of many heterocyclic39–47 and fused heterocyclic compounds.48–56 Recently, many efforts have been devoted to develop new methods for the synthesis of 3-functionalized chromones from o-hydroxyarylenaminones via the electrophile-triggered cyclization reaction (Scheme 1(a)).29,30,57–66 As far as the reactions of enaminones with fluorination reagents are concerned, a difluorination of enaminones to access difluorinated carbonyl compounds by virtue of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate), cheap and easily available) as the fluorine source was reported by Shreeve’s group in 2005. 67 In 1996, Bolós’s group 68 synthesized 3-fluoro-chromones from o-hydroxyarylenaminones by using 1-fluoro-2,4,6-trimethylpyridinium triflate (NFTP) as the fluorine source, but it still suffers from more expensive fluorination reagent and substrate scope. Therefore, the development of an efficient method using Selectfluor as a fluorine source would be valuable. In 2017, the groups of Zhao 69 and Xu 70 developed a Selectfluor-triggered tandem cyclization of o-hydroxyarylenaminones to obtain 3,3-difluorinated 2-amino-substituted chromanones, respectively (Scheme 1(b)). In addition, the groups of Kandula 71 and Zhao 72 also reported a Selectfluor-triggered tandem cyclization strategy for constructing 3-fluoro-chromones via direct elimination of 3,3-difluorinated 2-amino chromanones at room temperature in a short time ago. As our ongoing interest in enaminone chemistry,73–82 herein, we report a fast and efficient Selectfluor-triggered fluorination/cyclization reaction of o-hydroxyarylenaminones for the construction of 3-fluoro-chromones promoted by potassium carbonate (Scheme 1(c)). Interestingly, this reaction is mechanistically different compared to the previously reported methods71,72 where 2-(dimethylamino)-3,3-difluorochroman-4-one.

Reaction of enaminones with Selectfluor for the synthesis of 3-fluoro-chromones.
Results and discussion
Initially, o-hydroxyarylenaminone (
Screening of the reaction conditions. a
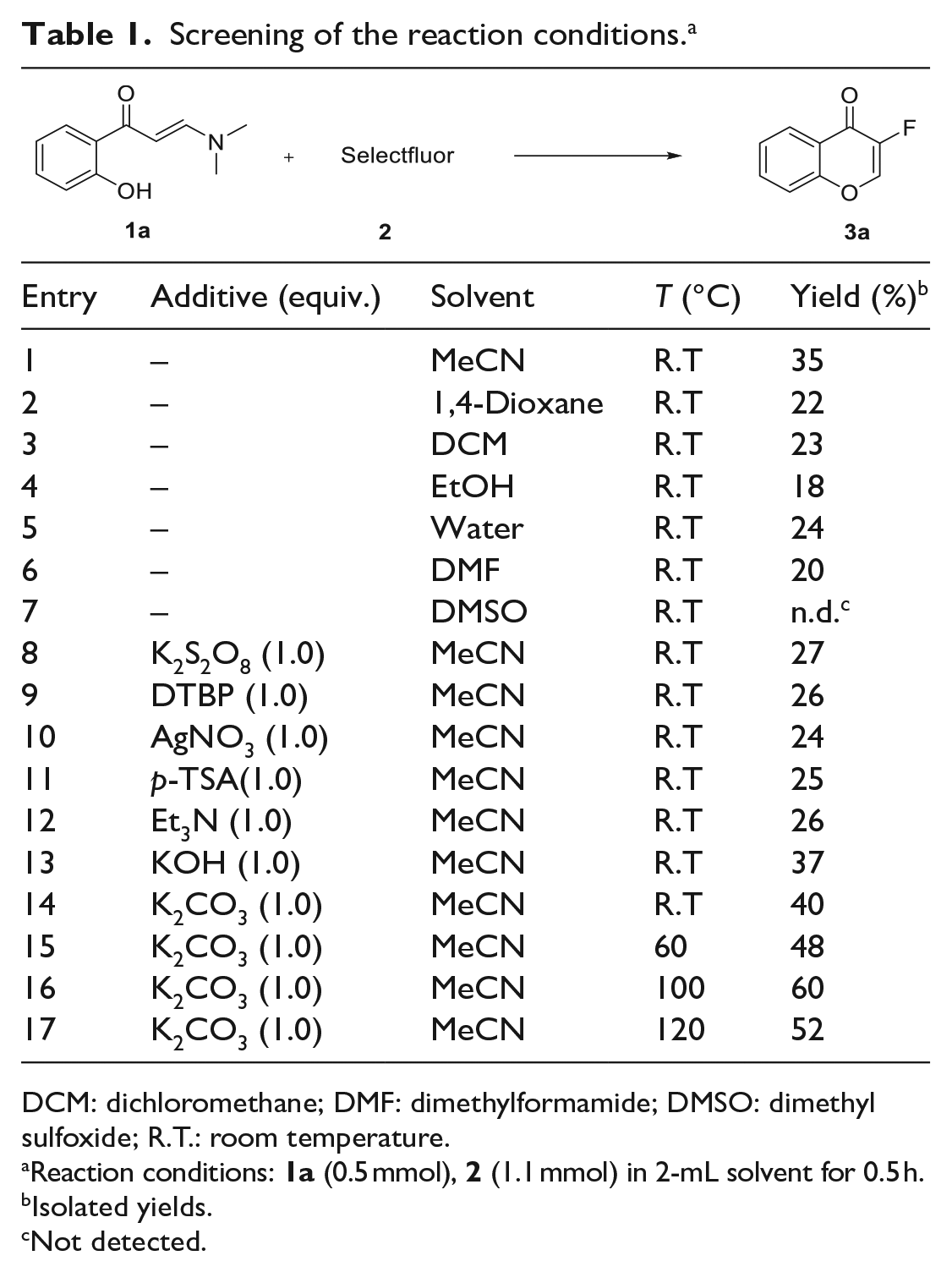
DCM: dichloromethane; DMF: dimethylformamide; DMSO: dimethyl sulfoxide; R.T.: room temperature.
Reaction conditions:
Isolated yields.
Not detected.
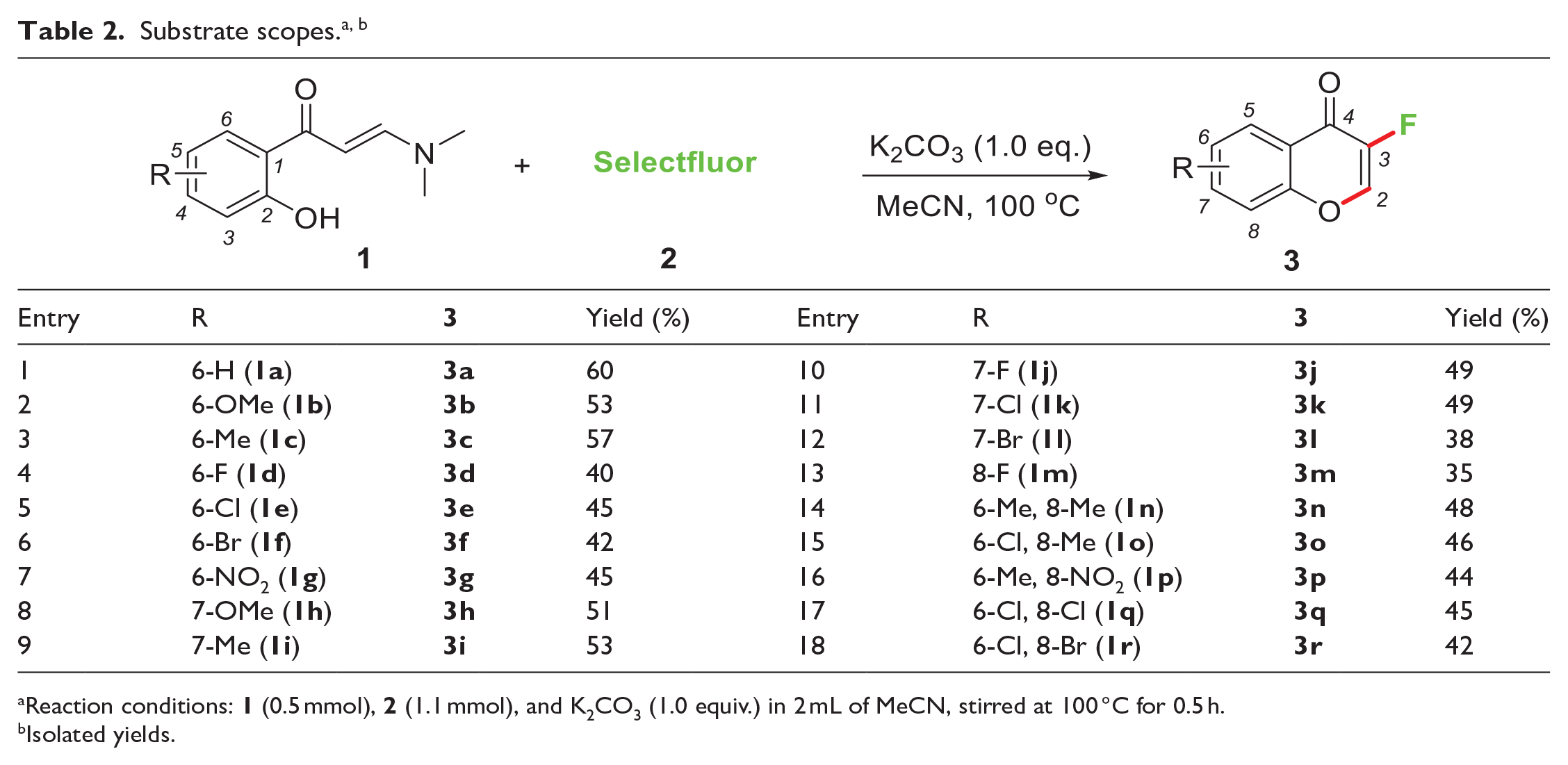
Under the optimized conditions, the substrate scope of this reaction was then investigated with a range of accessible o-hydroxyarylenaminones
Reaction conditions:
Isolated yields.
To gain insight into the mechanistic details of this fluorination/cyclization reaction, several control experiments were performed (Scheme 2). First, when the reaction of 4H-chromen-4-one

Control experiments.
Based on the above observations and previous reports, a plausible mechanism for the synthesis of 3-fluoro-chromones
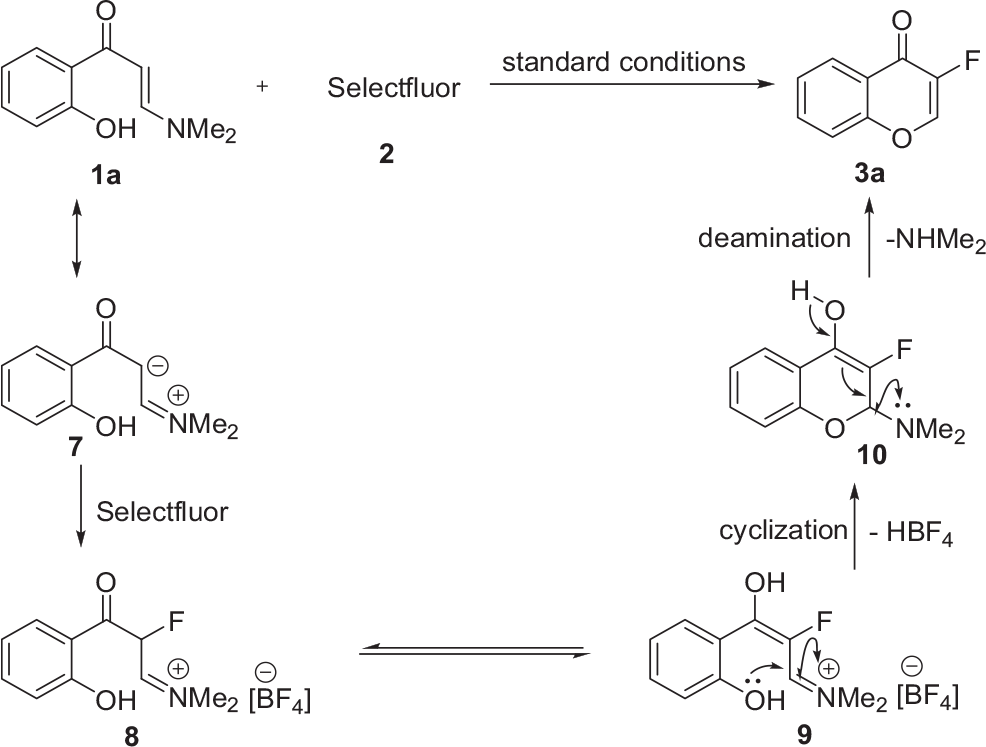
Proposed mechanism.
Conclusion
In conclusion, we successfully developed a facile and general approach accessing a range of 3-fluoro-chromones from o-hydroxyarylenaminones. Importantly, several species, such as 4H-chromen-4-ones; 2-(dimethylamino)-3,3-difluorochroman-4-ones; and 3,3-difluoro-2-hydroxychroman-4-ones, are proved not to be the reactive intermediates during the reaction process. This transformation involves the Selectfluor-triggered fluorination of the enaminone moiety, followed by subsequent intramolecular cyclization and deamination.
Experimental
All compounds were fully characterized by spectroscopic data. The nuclear magnetic resonance (NMR) spectra were recorded on a DRX600 spectrometer (1H: 600 MHz, 13C: 150 MHz), chemical shifts (δ) are expressed in ppm, and J values are given in Hz, and deuterated CDCl3 was used as solvent. The reactions were monitored by thin-layer chromatography (TLC) using silica gel GF254. The melting points were determined on XT-4A melting point apparatus and are uncorrected. High-resolution mass spectrometry (HRMS) was performed on an Agilent liquid chromatography–mass spectrometry (LC/MS) time-of-flight (TOF) instrument.
All chemicals and solvents were used as received without further purification unless otherwise stated. Column chromatography was performed on silica gel (200–300 mesh).
Compounds
General procedure for the synthesis of 3-fluoro-chromones 3a –3r
0.5-mmol-substituted o-hydroxyarylenaminones
3-Fluoro-4H-chromen-4-one
3-Fluoro-6-methoxy-4H-chromen-4-one
3-Fluoro-6-methyl-4H-chromen-4-one
3,6-Difluoro-4H-chromen-4-one
6-Chloro-3-fluoro-4H-chromen-4-one
6-Bromo-3-fluoro-4H-chromen-4-one
3-Fluoro-6-nitro-4H-chromen-4-one
3-Fluoro-7-methoxy-4H-chromen-4-one
3-Fluoro-7-methyl-4H-chromen-4-one
3,7-Difluoro-4H-chromen-4-one
7-Chloro-3-fluoro-4H-chromen-4-one
7-Bromo-3-fluoro-4H-chromen-4-one
3,8-Difluoro-4H-chromen-4-one
3-Fluoro-6,8-dimethyl-4H-chromen-4-one
6-Chloro-3-fluoro-7-methyl-4H-chromen-4-one
3-Fluoro-6-methyl-8-nitro-4H-chromen-4-one
6,8-Dichloro-3-fluoro-4H-chromen-4-one
8-Bromo-6-chloro-3-fluoro-4H-chromen-4-one
2-(Dimethylamino)-3,3-difluorochroman-4-one
3,3-Difluoro-2-hydroxychroman-4-one
Supplemental Material
Revised_Supporting_Information_Yu_Fuchao – Supplemental material for Selectfluor-triggered fluorination/cyclization of o-hydroxyarylenaminones: A facile access to 3-fluoro-chromones
Supplemental material, Revised_Supporting_Information_Yu_Fuchao for Selectfluor-triggered fluorination/cyclization of o-hydroxyarylenaminones: A facile access to 3-fluoro-chromones by Yanqin Wang, Biao Hu, Qiaohe Zhang, Siyun Zhao, Yuxuan Zhao, Biao Zhang and Fuchao Yu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received financial support from the National Natural Science Foundation of China (21402070 and 21961018) and the Analytical & Testing Foundation of Kunming University of Science and Technology (2017T20130137).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.