Abstract
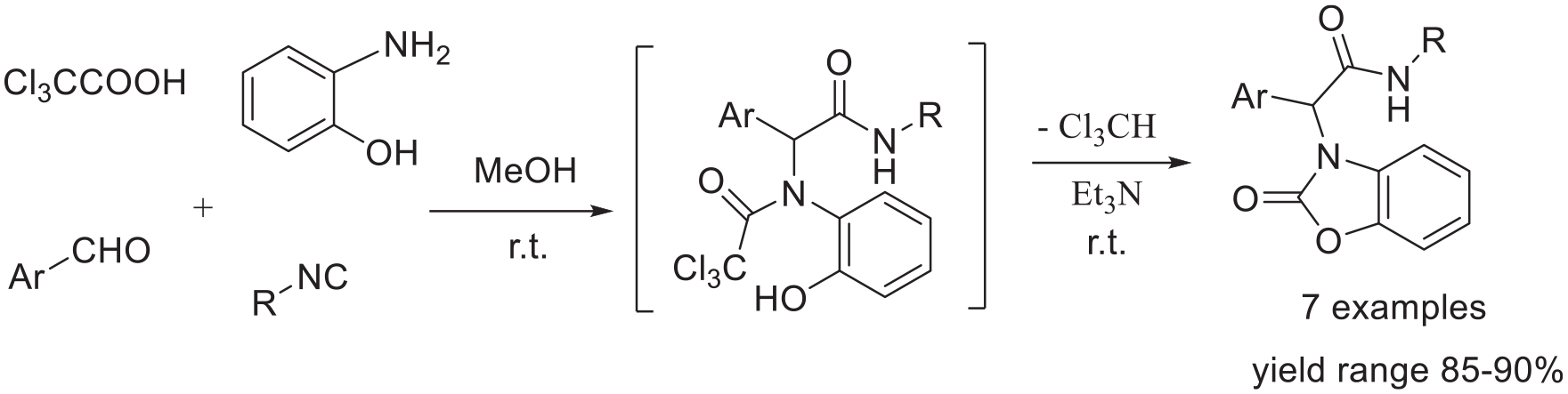
A one-pot, base-mediated approach to Acanthus ilicifolius Linn alkaloid 2-benzoxazolinone derivatives is developed. Starting from trichloroacetic acid, o-aminophenol, substituted benzaldehydes and alkyl isocyanides, the desired 2-benzoxazolinone derivatives are obtained in good yields via a tandem Ugi condensation and intramolecular haloform cyclization at room temperature in the presence of Et3N.
Introduction
Acanthus ilicifolius Linn is a mangrove plant growing in tropical coastal zones, which has antioxidant, hepatoprotective, anti-inflammatory and anti tumor activities.1,2 Researchers 3 discovered four new 2-benzoxazolinone-type alkaloids from the mangrove plant Acanthus ilicifolius, named acanthosides A-D (Figure 1). As an active substance of plant secondary metabolites, benzoxazolones play an important role in plant self-defense. The toxicological and pharmacological properties of benzoxazinoids have been reported as chemical resistance factors against insects, fungi, bacteria and viruses in many crop plants of the family Gramineae,4,5 as well as mutagenic activities, 6 can also be used as lead compounds for the development of new drugs.

Chemical structures of acanthosides A-D.
Urea fusion reactions with o-aminophenols or their hydrochloride salts 7 in harsh condition is one of the oldest and most widely employed syntheses of 2-benzoxazolinones. 8 Herein, Ugi reactions of o-aminophenol under mild condition with subsequent haloform cyclization are proposed for the synthesis of 2-benzoxazolinones.
The classical Ugi reaction is a four-component coupling reaction (U-4CR) between an amine, a carbonyl compound (aldehyde or ketone), a carboxylic acid, and an isocyanide. This 4-CR has been widely used in modern synthetic chemistry9-15 and has tentatively been used for direct bioconjugation.16-20 The construction of heterocyclic rings is a key stone of natural product and artificial drug synthesis, therefore, the Ugi reaction is now a cornerstone of isocyanide based multicomponent reactions (I-MCR) for the construction of natural products and diverse heterocyclic scaffolds.21-26 We synthesized 2-benzoxazolinones by Ugi reactions and introduced different substituents to synthesize a series of new compounds (Scheme 1).
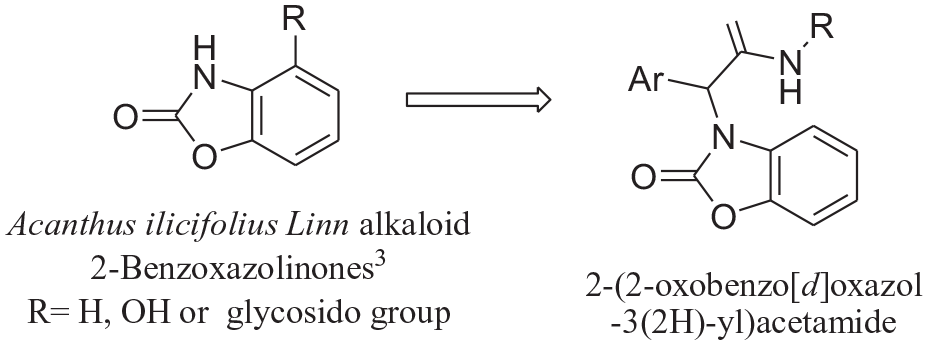
The strategy for the choice of 2-(2-oxobenzo[d]oxazol-3(2H)-yl) acetamides as targets.
Herein, we report a direct synthesis of Acanthus ilicifolius Linn alkaloid 2-benzoxazolinone derivatives 6 from trichloroacetic acid (

Synthesis of 2-benzoxazolinone derivatives
Results and discussion
Based on tandem I-MCR/SN cyclization, we previously prepared (E)-2-aroyl-4-arylidene-5-oxopyrrolidine
27
and (E)-2-aroyl-4-arylidene-5-oxotetrahydro rhoplaladin analogs furan.
28
In the analogous way, seven Acanthus ilicifolius Linn alkaloid 2-benzoxazolinone derivatives
The structures of compounds
Conclusion
Based on a one-pot, base-mediated approach proceeding via a tandem Ugi condensation and intramolecular haloform cyclization at room temperature, seven novel 2-benzoxazolinone derivatives have been obtained. And in the intramolecular haloform cyclization step, the triethylamine is deprotonating the phenol to generate the phenoxide which then displaces the chloromethyl anion from the carboxyl carbon by an addition elimination process. The products were obtained in high yields under mild conditions using a simple procedure. During the experimental process, the Ugi reaction intermediates are not separated or purified, and can be directly used in the intramolecular haloform cyclization step.
Experimental section
General
Melting points were measured with an X-4 melting point instrument (uncorrected thermometer) produced by Beijing Ruili Analytical Instrument Co., Ltd. Mass spectrometry was performed with a Finnigan trace MS analyzer (direct injection method). Elemental analysis was performed using a Vario EL III analyzer. 1H NMR and 13C NMR spectra were measured at 400 MHz using model spectrometers. The solvent was CDCl3 with TMS as the internal standard.
General procedure for the preparation of 2-benzoxazolinone derivatives 6a-g
Trichloroacetic acid (
2-(2-Chlorophenyl)-N-cyclohexyl-2-(2-oxobenzo[d]oxazol-3(2H)-yl) acetamide (
2-(3-Chlorophenyl)-N-cyclohexyl-2-(2-oxobenzo[d]oxazol-3(2H)-yl) acetamide (
2-(3-Bromophenyl)-N-butyl-2-(2-oxobenzo[d]oxazol-3(2H)-yl) acetamide (
N-Cyclohexyl-2-(2-oxobenzo[d]oxazol-3(2H)-yl)-2-p-tolylacetamide (
N-Butyl-2-(4-methoxyphenyl)-2-(2-oxobenzo[d]oxazol-3(2H)-yl) acetamide (
N-Butyl-2-(4-fluorophenyl)-2-(2-oxobenzo[d]oxazol-3(2H)-yl) acetamide (
N-Cyclohexyl-2-(2-oxobenzo[d]oxazol-3(2H)-yl)-2-(o-tolyl) acetamide (
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211024609 – Supplemental material for One-pot synthesis of Acanthus ilicifolius Linn alkaloid 2-benzoxazolinone derivatives via a tandem Ugi 4-component coupling/haloform cyclization
Supplemental material, sj-pdf-1-chl-10.1177_17475198211024609 for One-pot synthesis of Acanthus ilicifolius Linn alkaloid 2-benzoxazolinone derivatives via a tandem Ugi 4-component coupling/haloform cyclization by Jun Zhu, Xiao-Qiong Tian, Ling-Qi Kong, Li-Na Ke, Feng-Ying Ran, Lun Wu, Hong-Mei Wang, Qin-Hua Chen and Xiao-Hua Zeng in Journal of Chemical Research
Footnotes
Author contributions
Zhu J, Tian XQ, Kong LQ and Wang HM contributed to the designing, conceptualization and synthesis. Ke LN, Ran FY and Wu L performed data analysis and statistical analysis. Chen QH and Zeng XH performed data acquisition and manuscript editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81872509), Bao’an TCM Development Foundation (2020KJCX-KTYJ-200), the Internal Research Project of Shenzhen Baoan Authentic TCM Therapy Hospital (BCZY2021003 and BCZY2021007), Baoan District Medical and Health Basic Research Project (2020JD491), the Chinese Medicine Research Fund of Health Commission of Hubei Province (ZY2021M051), the Hubei Province Health and Family Planning Scientific Research Project (WJ2021M063 and WJ2021M062), the Scientific Research Project of the Educational Commission of Hubei Province of China (B2020106), and the Postgraduate Innovation Project of Hubei University of Medicine (YC2020041).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
