Abstract
A practical method is developed for the synthesis of oxazolidinone derivatives, an important class of heterocyclic compounds. The effect of bases and solvents on this cyclization reaction is discussed and a simple new base–solvent system (triethylamine in toluene) is found to be the most effective. The reaction conditions developed here are mild and no by-products are observed. Moreover, using optimized conditions, a number of differently substituted propargylic alcohols are cyclized to the corresponding N-substituted-4-methylene-oxazolidinones in yields of up to 99%.

Introduction
Oxazolidinones, an important class of heterocyclic compounds, are ubiquitous in natural products and materials. As important intermediates in organic synthesis, they are widely used to construct the skeletons of diverse pharmaceuticals.1–4 Among the numerous oxazolidinones, N-substituted-4-methylene-oxazolidinones usually show excellent antibacterial and anticancer properties. Consequently, there is increasing interest being paid toward the synthesis of N-substituted-4-methylene-oxazolidinones.5–11 Most of these approaches are focused on nucleophilic cyclization via transition-metal-catalyzed strategies. However, the high catalyst loading in some of these processes results in a high economic cost.12–16 In addition, the residual heavy transition metals in the final products also present a major problem. Therefore, the development of efficient strategies and transition-metal-free processes for the assembly of oxazolidinones is an important target in organic synthesis.
The transformation of alkynes via nucleophilic addition, especially for oxygen or nitrogen heteroatoms, is endowed with great synthetic value. Although this strategy has been used to construct oxazolidinone derivatives through intermolecular or intramolecular nucleophilic addition of acetylenic triple bonds with nitrogen components, examples are still scarce. Zhang and co-workers 17 reported a new route for the synthesis of N-substituted-4-methylene-oxazolidinones via the carbonylation of propargylic alcohols with amines using CO2 as the carbonyl source under high pressure, high temperature, and in the presence of ionic liquids. The reaction occurs via intramolecular nucleophilic addition of the acetylenic triple bond with the amino component (Scheme 1(a)). Schmalz et al. 18 have provided some simple routes to furnish N-substituted-4-methylene-oxazolidinones via cyclization of o-propargyl carbamates under palladium, gold, and base-catalyzed conditions (Scheme 1(a)). Liu et al. 19 have developed transition-metal-catalyzed protocols for producing oxazolidinones via domino intermolecular nucleophilic addition between propargylic alcohols and p-toluenesulfonyl isocyanate. Herein, we describe a facile, one-pot preparation of 4-alkylideneoxazolidinones having an N-tosyl group. The reactions proceed under mild conditions in good yields starting from propargylic alcohols and p-toluenesulfonyl isocyanate in the presence of a catalytic amount of triethylamine (Scheme 1(b)). This proved to be an efficient method for preparing oxazolidinones by exo-cycloisomerization of propargylic carbamates catalyzed by a base.

Synthesis of N-substituted-4-methylene-oxazolidinones: (a) Previous work and (b) this work.
Results and discussion
Initially, the cyclization of 1-phenylprop-2-yn-1-ol (see the Supporting Information for details) (
Optimization of the reaction conditions. a
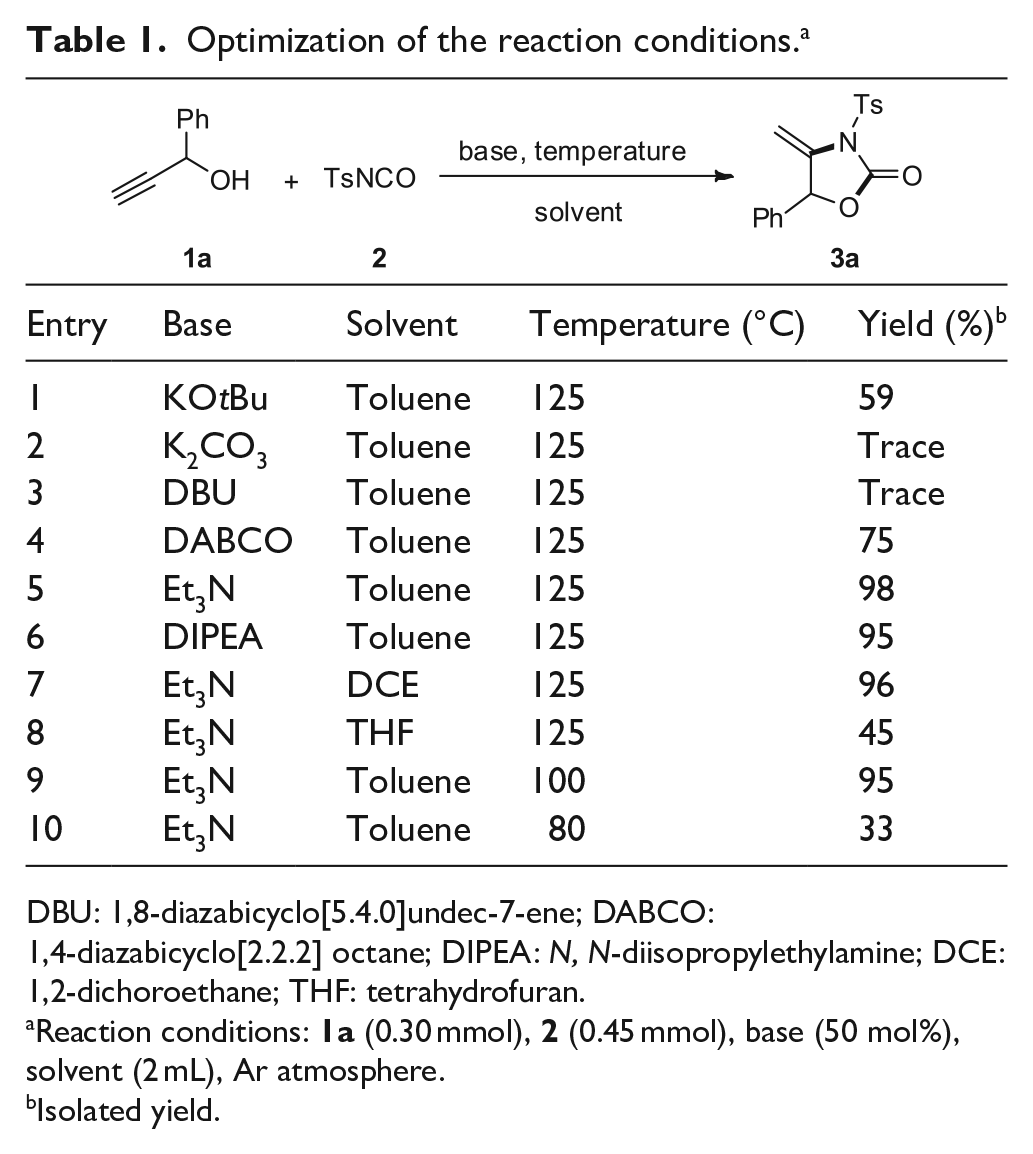
DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DABCO: 1,4-diazabicyclo[2.2.2] octane; DIPEA: N, N-diisopropylethylamine; DCE: 1,2-dichoroethane; THF: tetrahydrofuran.
Reaction conditions:
Isolated yield.
With optimized conditions in hand, the scope of the cyclization of p-toluenesulfonyl isocyanate with various propargylic alcohols was investigated, and the results are summarized in Table 2. First, a number of 1-arylprop-2-yn-1-ols were evaluated, and they were all found to be suitable substrates for the cyclization with p-toluenesulfonyl isocyanate (
Screening the scope of propargylic alcohols

Reaction conditions:
Based on the above results and previous reports, a proposed reaction mechanism is shown in Scheme 2. First, 1-phenylprop-2-yn-1-ol (
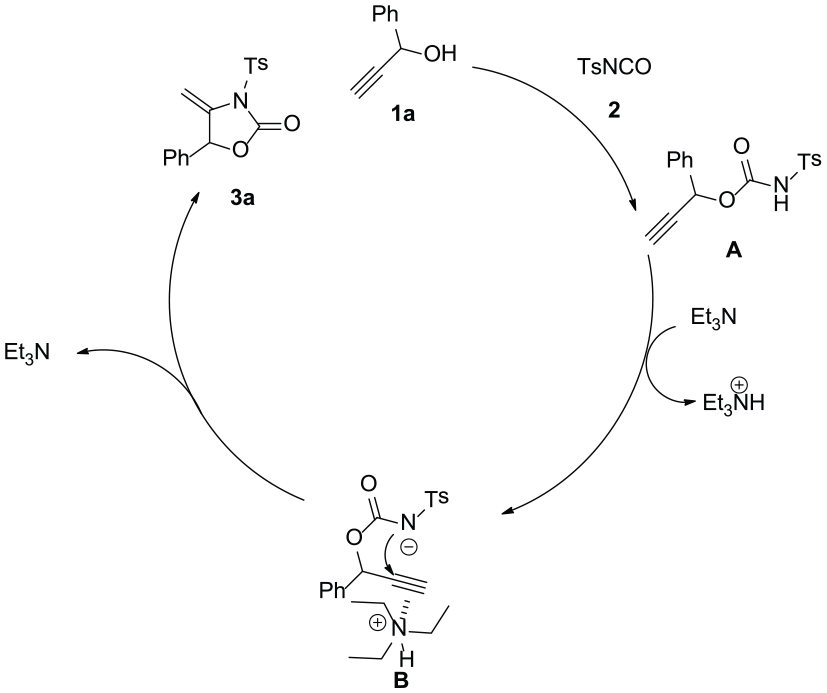
Proposed mechanism.
Conclusion
In conclusion, we have developed a mild and general cascade reaction of propargylic alcohols with p-toluenesulfonyl isocyanate for the synthesis of N-substituted-4-methylene-oxazolidinones. In the presence of a base–solvent system (triethylamine in toluene), a broad range of propargylic alcohols was tolerated in the present method. Notably, oxazolidinones are an important skeleton in natural products and exhibit a broad range of biological activities. Further work to probe the detailed mechanism and apply this reaction in organic synthesis is currently underway.
Experimental
Typical experimental procedure for the synthesis of 4-methylene-5-phenyl-3-tosyloxazolidin-2-one (
A dried 25 mL Schlenk tube equipped with a stir bar was loaded with
Supplemental Material
JCR_-Oxazolidinones-sup_st – Supplemental material for Synthesis of N-substituted-4-methylene-oxazolidinones via base-catalyzed cyclization of propargylic alcohols with p-toluenesulfonyl isocyanate
Supplemental material, JCR_-Oxazolidinones-sup_st for Synthesis of N-substituted-4-methylene-oxazolidinones via base-catalyzed cyclization of propargylic alcohols with p-toluenesulfonyl isocyanate by Shao-Feng Pi, Yue-Meng Guo, Zheng-Rui Zhou, Han-zhou Sun and Bing Yi in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the NSFC (Nos. 21202206 and 21772035), the Hunan Provincial Education Office General Project (18C0265), the Scientific Innovation Fund for Graduates of Hunan Province (CX2015B298), and the Scientific Innovation Fund for Graduates of the Central South University of Forestry and Technology (CX2015B20).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



