Abstract
A one-pot method for the synthesis of aromatic ethers using hypervalent iodine(III) reagents obtained from the corresponding iodoaryl compounds is developed. In this concise method, six diaryl ethers and three heterocyclic aromatic ethers are synthesized in good yields. Furthermore, possible mechanisms for the syntheses of the hypervalent iodine reagents and construction of the aromatic ethers are proposed.

Introduction
Hypervalent iodine(III) reagents have been known for over a century, but these compounds have only recently been applied extensively in many important organic transformations.1–3 Diaryl ethers are common structural features in numerous biologically active compounds and natural products, some of which are potential drugs. Numerous methods have been developed to synthesize diaryl ethers, and new routes for a wide range of biologically active compounds have been devised.4–8 Heterocyclic aromatic ethers are likely to exhibit higher potent biological activity than the corresponding diphenyl ethers. However, many of the reported methods for the synthesis of heterocyclic aromatic ethers have limited efficiency.
9
In this study, we report a concise and efficient method for the preparation of trivalent iodine compounds
Results and discussion
In our laboratory, we developed an efficient and inexpensive method for preparing diacetoxyiodoarenes10–12 in ideal yields from corresponding iodoarenes with sodium perborate tetrahydrate. 13 The optimized method for the preparation of diacetoxyiodoarenes from iodoarenes is described in section “Experimental” (Scheme 1).

Preparation of diacetoxyiodoarenes.
The ortho-iodo aromatic ethers can be synthesized smoothly from the corresponding phenols with the diacetoxyiodoarenes. In particular, the 4-pyridone also could be oxidized to ortho-iodo aromatic ethers via diacetoxyiodoarenes. The detail experiment of the preparation of aromatic ethers is described in section “Experimental” (Scheme 2).

Construction of aromatic ethers.
A possible mechanism for the construction of diacetoxyiodoarenes has been proposed based on the experimental results (Scheme 3). The starting material is oxidized to an unstable intermediate iodosyl benzene
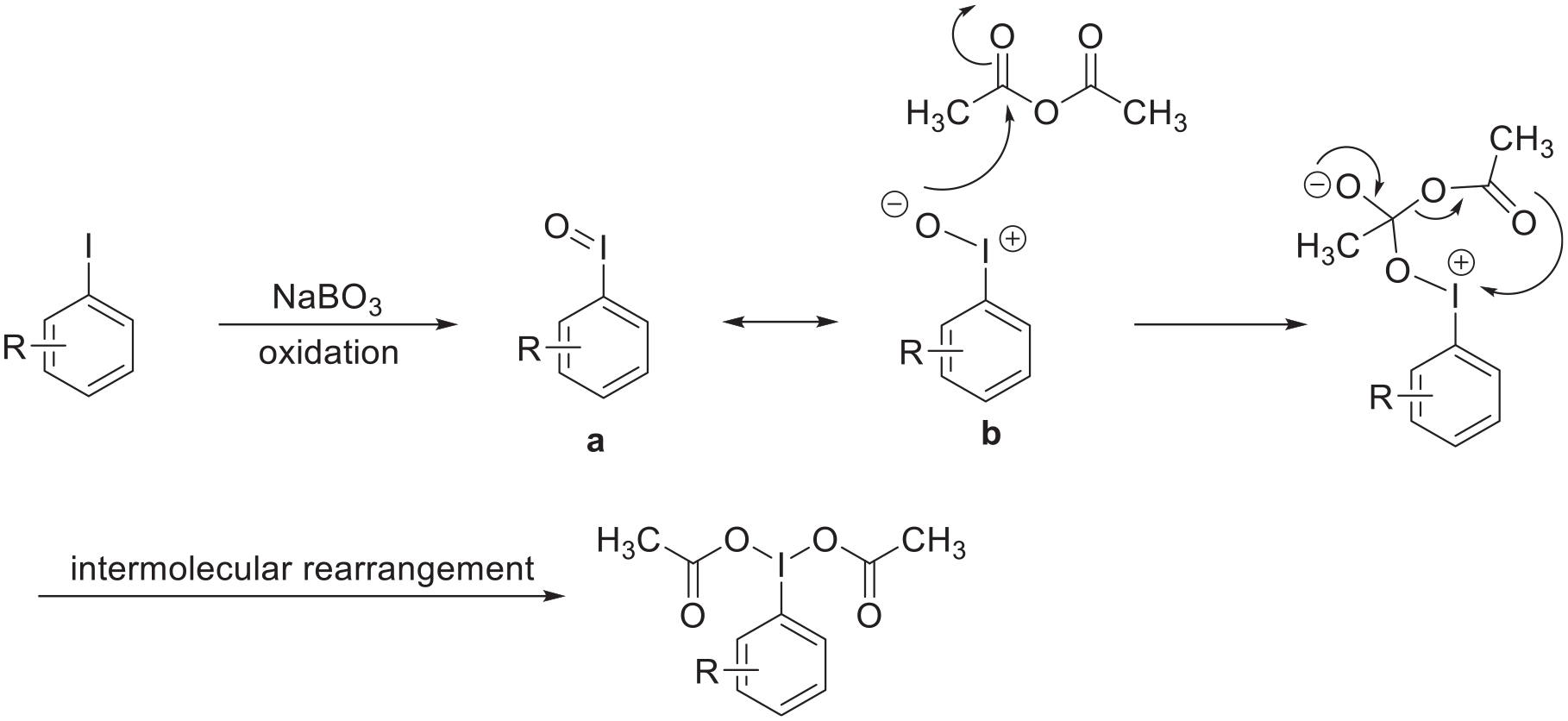
Possible mechanism for the construction of the diacetoxyiodoarenes.
Based on the experiment results and previous reports,
14
a possible mechanism for the formation of heterocyclic aromatic ethers is shown in Scheme 4. Initially, the lone pair of the hydroxy group attacks the iodine of the diacetoxyiodoarene, which loses an acetic acid to form intermediate

A possible mechanism for the formation of heterocyclic aromatic ethers.
Conclusion
In this work, we report an efficient method for synthesis of diacetoxyiodoarenes via intramolecular rearrangement, and three diacetoxyiodoarenes were prepared smoothly via this method. Then, the front freshly prepared compounds are used to oxidize some phenols, and the corresponding six diaryl ethers and three heterocyclic aromatic ethers are synthesized in good yields. Both of the possible mechanisms of preparing diacetoxyiodoarenes and constructing diaryl ethers are proposed.
Experimental
All chemical reagents were obtained from commercial suppliers (Aldrich, Tokyo Chemical Industry (TCI), Aladdin, Macklin, and Bidepharm Pure Chemical Industries) and were used without further purification. Anhydrous solvents were obtained via standard protocols. All non-aqueous reactions were carried out under an Ar atmosphere. Thin-layer chromatography (TLC) was performed on silica gel 60 F254 glass plates precoated with a 0.25-mm thickness of silica gel (Yantai Jiangyou). Column chromatography was carried out on Cica Silica Gel 60N (spherical, neutral, 40–50 μm or 63–210 μm). 1H and 13C NMR spectra were obtained on a Varian UNITY plus 300 (300 MHz for 1H and 75 MHz for 13C) instrument, with CDCl3 as the solvent an d internal reference. IR spectra were measured on a JNM FTIR-460 Plus spectrometer. Mass spectra were recorded on JEOL D-200, JEOL JMS-GCmate II, SHIMAZU GC-MS-QP 500, or JEOL AX 505 spectrometers. Melting points were recorded with a Yanagimoto micro melting point apparatus and are uncorrected.
Optimized method for the preparation of diacetoxyiodoarenes from iodoarenes
NaBO3·4H2O (10 mmol) was slowly added portionwise over 30 min to a stirred solution of iodoarene
General procedure for the preparation of aromatic ethers
A mixture of 4-pyridone or substrate
Diacetoxyiodobenzene (4 )
Yield: 91%; white solid; m.p. 161–163 °C (lit. 15 161.2–162.2 °C). 1H NMR (300 MHz, CDCl3): δ 1.98 (6 H, s), 7.46 (2 H, d, J = 7.6 Hz), 7.58 (1 H, td, J = 7.6 Hz, J = 1.1 Hz), 8.08 (2 H, dd, J = 7.6 Hz, J = 1.1 Hz); 13C NMR (75 MHz, CDCl3): δ 20.18 (q), 20.18 (q), 121.38 (s), 130.77 (d), 131.59 (d), 134.76 (d), 176.19 (s).
p-(Diacetoxyiodo)toluene (5 )
Yield: 89%; white solid; m.p. 109–110 °C. 1H NMR (300 MHz, CDCl3): δ 1.97 (6 H, s), 2.40 (s, 3 H), 7.27 (2 H, d, J = 8.1 Hz), 7.95 (2 H, d, J = 8.1 Hz); 13C NMR (75 MHz, CDCl3): δ 20.7 (q), 21.6 (q), 119.2 (s), 131.8 (d), 134.5 (d), 142.8 (s), 176.3 (s). IR (KBr, cm−1): 3385, 3321, 2261, 1631, 1441, 1385, 1193; MS-ES: m/z = 336 (M+); HRMS-ES: m/z [M]+ calcd for C11H13IO4: 335.9859; found: 335.9849.
o-(Diacetoxyiodo)toluene (6 )
Yield: 92%; white solid; m.p. 100–102 °C. 1H NMR (300 MHz, CDCl3): δ 1.98 (6 H, s), 2.72 (3 H, s), 7.22-7.28 (1 H, m), 7.50-7.52 (2 H, m), 8.16 (1 H, d, J = 7.42 Hz); 13C NMR (75 MHz, CDCl3): δ 20.13 (q), 25.39 (q), 126.83 (s), 128.05 (d), 130.49 (d), 132.37 (d), 136.82 (d), 140.20 (s), 175.94 (s). IR (neat, cm−1) 3435, 1649, 1614, 1367, 1293, 1011, 764, 669; MS-ES: m/z = 336 (M+); HRMS-ES: m/z [M]+ calcd for C11H13IO4: 335.9859; found: 335.9851.
Iodo-4-phenoxypyridine (7 )
Yield: 94%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 6.56 (1H, d, J = 5.77 Hz), 7.09-7.12 (2 H, m), 7.25-7.31 (1 H, m), 7.41-7.48 (2 H, m), 8.29 (1 H, d, J = 5.77 Hz), 8.86 (1 H, s); 13C NMR (75 MHz, CDCl3): δ 85.38 (s), 111.01 (d), 120.65 (d), 125.73 (d), 130.18 (d), 150.36 (d), 153.66 (s), 158.43 (d), 163.74 (s); IR (neat, cm−1): 3038, 2360, 1562, 1464, 1397, 1270, 1199, 882; MS-ES: m/z = 297 (M+); HRMS-ES: m/z [M]+ calcd for C11H8INO: 296.9651; found: 296.9652.
3-Iodo-4-(p-tolyloxy)pyridine (8 )
Yield: 91%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.37 (3 H, s), 6.53 (1 H, d, J = 5.77 Hz), 6.97 (2 H, d, J = 9.52 Hz), 7.20 (2 H, d, J = 9.52 Hz), 8.25 (1 H, d, J = 5.77 Hz), 8.83 (1 H, s); 13C NMR (75 MHz, CDCl3): δ 20.92 (q), 85.15 (s), 110.69 (d), 120.46 (d), 130.44 (d), 135.44 (s), 150.25 (d), 151.28 (s), 158.25 (d), 163.93 (s); IR (neat, cm−1): 3034, 1567, 1504, 1462, 1271, 1201, 1016, 885; MS-ES: m/z = 311 (M+); HRMS-ES: m/z [M]+ calcd for C12H10INO: 310.9807; found: 310.9799.
3-Iodo-4-(o-tolyloxy)pyridine (9 )
Yield: 95%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.16 (3 H, s), 6.41 (1 H, d, J = 5.49 Hz), 7.03 (1 H, d, J = 7.69 Hz), 7.19-7.32 (4 H, m), 8.28 (1 H, d, J = 5.77 Hz), 8.86 (1 H, s); 13C NMR (75 MHz, CDCl3): δ 16.07 (q), 84.64 (s), 109.89 (d), 121.14 (d), 126.19 (d), 127.55 (d), 130.26 (s), 131.87 (d), 150.12 (d), 151.54 (s), 157.96 (d), 163.42 (s); IR (neat, cm−1): 3035, 2360, 1563, 1464, 1271, 1181, 888; MS-ES: m/z = 311 (M+); HRMS-ES: m/z [M]+ calcd for C12H10INO: 310.9807; found: 310.9791.
Methyl 3-iodo-4-(p-tolyloxy)benzoate (13 )
Yield: 85%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.36 (3 H, s), 3.90 (3 H, s), 6.72 (1 H, d, J = 8.52 Hz), 6.95 (2 H, d, J = 7.97 Hz), 7.19 (2 H, d, J = 7.97 Hz), 7.89 (1 H, dd, J = 8.52, 1.92 Hz), 8.52 (1 H, d, J = 1.92 Hz); 13C NMR (75 MHz, CDCl3): δ 20.91 (q), 52.26 (q), 86.48 (s), 115.82 (d), 119.80 (d), 125.85 (s), 130.47 (d), 131.06 (d), 134.49 (s), 141.25 (d), 152.91 (s), 161.12 (s), 165.13 (s); IR (neat, cm−1) 2863, 1720, 1588, 1505, 1477, 1433, 1253; MS-ES: m/z = 368 (M+); HRMS-ES: m/z [M]+ calcd for C15H13IO3: 367.9909; found: 367.9909.
Methyl 5-iodo-2-methoxy-4-(p-tolyloxy)benzoate (14 )
Yield: 91%; white solid; m.p. 77–79 °C. 1H NMR (300 MHz, CDCl3): δ 2.36 (3 H, s), 3.69 (3 H, s), 3.87 (3 H, s), 6.35 (1 H, s), 6.94 (2 H, d, J = 8.52 Hz), 7.19 (2 H, d, J = 8.52 Hz), 8.30 (1 H, s); 13C NMR (75 MHz, CDCl3): δ 20.91 (q), 52.08 (q), 56.18 (q), 75.09 (s), 101.54 (d), 116.12 (s), 119.36 (d), 130.46 (d), 134.28 (s), 142.43 (d), 152.98 (s), 161.10 (s), 161.31 (s), 164.50 (s); IR (neat, cm−1): 2948, 1687, 1588, 1505, 1438, 1276, 1103, 834; MS-ES: m/z = 398 (M+); HRMS-ES: m/z [M]+ calcd for C16H15IO4: 398.0015; found: 398.0010.
2-iodo-4-nitro-1-(p-tolyloxy)benzene (15 )
Yield: 79%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.39 (3 H, s), 6.71 (1 H, d, J = 9.07 Hz), 6.98 (2 H, d, J = 8.52 Hz), 7.24 (2 H, d, J = 8.52 Hz), 8.10 (1 H, dd, J = 9.07, 2.75 Hz), 8.73 (1 H, d, J = 2.75 Hz); 13C NMR (75 MHz, CDCl3): δ 20.97 (q), 85.66 (s), 114.63 (d), 120.22 (d), 125.13 (d), 130.77 (d), 135.33 (d), 135.51 (s), 142.66 (s), 152.17 (s), 162.78 (s); IR (neat, cm−1): 3093, 2923, 1576, 1518, 1465, 1342, 1260893; MS-ES: m/z = 355 (M+); HRMS-ES: m/z [M]+ calcd for C13H10INO3: 354.9706; found: 354.9713.
Methyl 3-iodo-4-(o-tolyloxy)benzoate (16 )
Yield: 89%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.19 (3 H, s), 3.90 (3 H, s), 6.54 (1H, d, J = 8.79 Hz), 6.97 (1 H, d, J = 7.69 Hz), 7.14-7.31 (3 H, m), 7.87 (1 H, dd, J = 8.79, 1.92 Hz), 8.54 (1 H, d, J = 1.92 Hz); 13C NMR (75 MHz, CDCl3): δ 16.25 (q), 52.25 (q), 85.67 (s), 114.38 (d), 120.56 (d), 125.38 (d), 125.54 (s), 127.38 (d), 130.10 (s), 131.15 (d), 131.74 (d), 141.30 (d), 152.90 (s), 160.57 (s), 165.15 (s); IR (neat, cm−1): 2950, 2360, 1720, 1581, 1478, 1433, 1283, 1252, 1111; MS-ES: m/z = 368 (M+); HRMS-ES: m/z [M]+ calcd for C15H13IO3: 367.9909; found: 367.9910.
Methyl 5-iodo-2-methoxy-4-(o-tolyloxy)benzoate (17 )
Yield: 86%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.21 (3 H, s), 3.65 (3 H, s), 3.87 (3 H, s), 6.18 (1 H, s), 6.94 (1 H, d, J = 7.97 Hz), 7.12-7.31 (3 H, m), 8.32 (1 H, s); 13C NMR (75 MHz, CDCl3): δ 16.28 (q), 52.05 (q), 56.11 (q), 74.27 (s), 100.07 (d), 115.65 (s), 119.99 (d), 125.23 (d), 127.32 (d), 129.86 (s), 131.72 (d), 142.50 (d), 152.87 (s), 160.91 (s), 161.25 (s), 164.48 (s); IR (neat, cm−1): 2949, 2360, 1731, 1592, 1488, 1244, 1093, 781; MS-ES: m/z = 398 (M+); HRMS-ES: m/z [M]+ calcd for C16H15IO4: 398.0015; found: 398.0010.
2-iodo-4-nitro-1-(o-tolyloxy)benzene (18 )
Yield: 85%; colorless oil. 1H NMR (300 MHz, CDCl3): δ 2.17 (3 H, s), 6.54 (1 H, d, J = 9.07 Hz), 7.01 (1 H, d, J = 7.69 Hz), 7.19-7.34 (3 H, m), 8.09 (1 H, dd, J = 9.07, 2.75 Hz), 8.75 (1 H, d, J = 2.75 Hz); 13C NMR (75 MHz, CDCl3): δ 16.13 (q), 85.03 (s), 113.45 (d), 120.90 (d), 125.25 (d), 126.15 (d), 127.67 (d), 130.13 (s), 131.98 (d), 135.39 (d), 142.56 (s), 152.32 (s), 162.06 (s); IR (neat, cm−1): 3093, 2359, 1574, 1518, 1462, 1341, 1258, 745; MS-ES: m/z = 355 (M+); HRMS-ES: m/z [M]+ calcd for C13H10INO3: 354.9705; found: 354.9713.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the National Natural Science Foundation of China (no. 81803399), the Science and Technology Research Project of Henan Province (nos 182102310300 and 192102310144), and the Colleges and Universities in Henan Province Key Scientific Research Project (nos 18A150028 and 19B350003) for the financial support.
