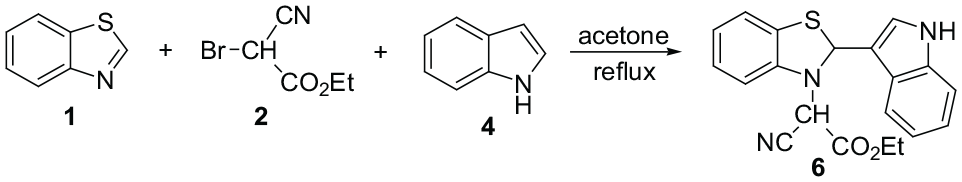
Abstract
Ethyl 2-(2-(1H-indol-3-yl)benzo[d]thiazol-3(2H)-yl)-2-cyanoacetate or acetate
Introduction
Multi-component reactions (MCRs) are economically and environmentally beneficial to the industry and have attracted much attention during the two recent decades. These reactions are perfectly applicable to organic synthesis and agrochemical chemistry.1,2 Due to their interest as natural and unnatural compounds, nitrogen heterocycles are of special interest in organic synthetic chemistry.
3
The sulfur- and nitrogen-containing aromatic heterocyclic compounds known as benzothiazoles and analogs play a crucial role in the synthesis of new drugs.
4
These compounds have different pharmacological and biological effects, and have different applications in the production of carbonic anhydrase enzyme inhibitory,5,6 anti-inflammatory,
7
anticancer,
7
antimicrobial,
8
antitumor,
8
Alzheimer,
9
antibacterial,
10
antiallergic,
11
and antioxidant
12
drugs. Benzothiazoles are also widely used in a variety of consumer and industrial products such as nonlinear optics (NLOs), textiles, α-glucosidase inhibitors, organic light-emitting diodes (OLEDs), fluorescent probes, and chemosensors.13–15 The indole moiety occurs in a number of natural products and alkaloids, and its derivatives are widely used in various medicine formulations. It has been reported that they exhibit numerous biological activities, including antirheumatoidal,
16
antioxidant,
16
antibacterial,
17
antiviral,
17
antiallergic,
18
and antileukemic
19
activities. In this article, we report the results of our studies involving the simple, novel, and catalyst-free reactions of benzothiazole, 2-methylbenzothiazole, or 2,5-dimethylbenzothiazole
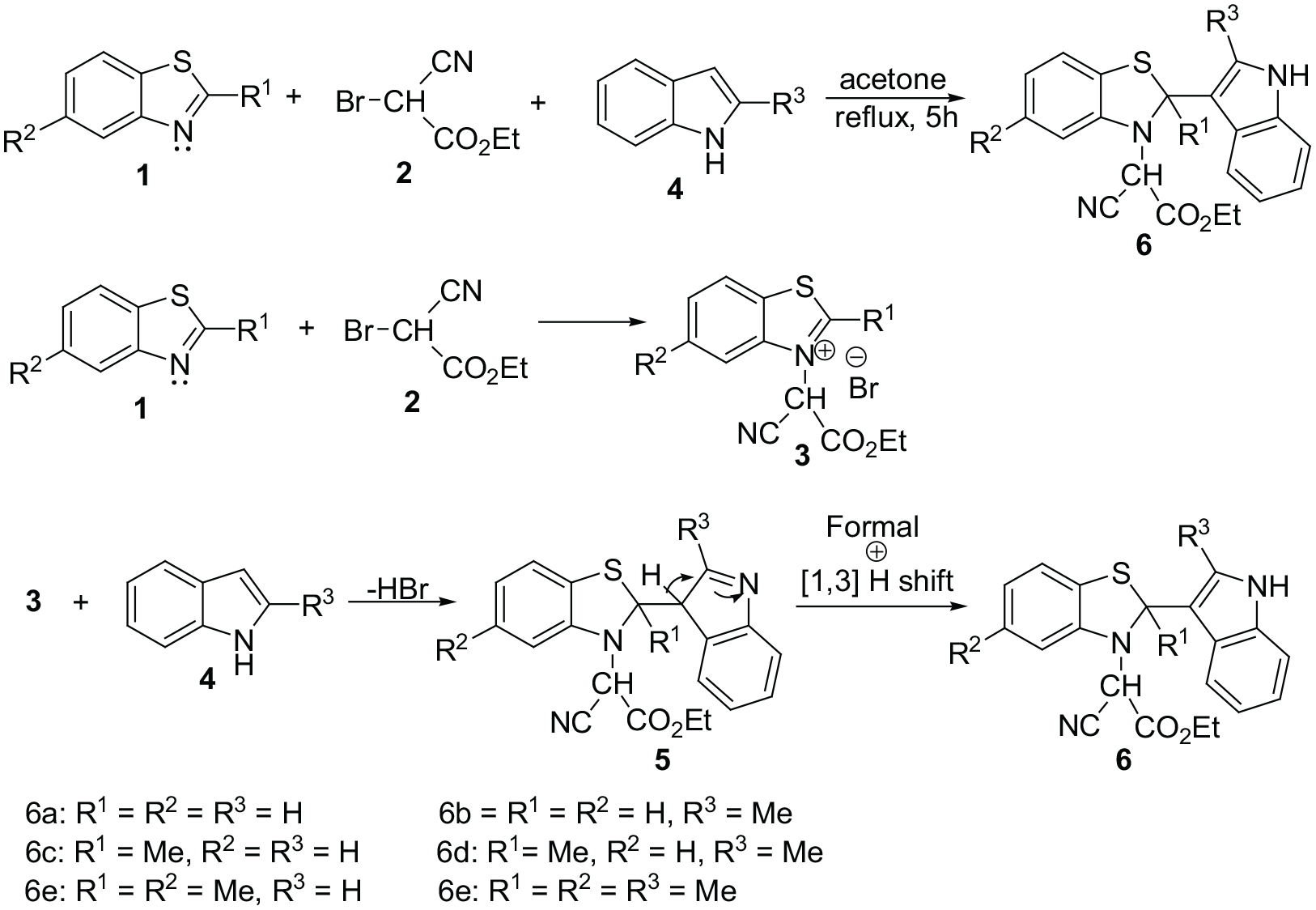
A plausible mechanism for the formation of compounds 6.
Benzothiazole, indole, and ethyl bromocyanoacetate.

Results and discussion
The reactions of benzothiazole, 2-methylbenzothiazole, or 2,5-dimethylbenzothiazole 1 and ethyl bromocyanoacetate 2 with indole or 2-methylindole 4 were conducted under reflux conditions in acetone and were complete within 5 h. The different stages of the reactions included mixing the benzothiazole with ethyl bromocyanoacetate for 2 h, followed by the addition of the indole and heating under reflux for 3 h. The 1H and 13C nuclear magnetic resonance (NMR) spectra of the crude products clearly indicated the formation of compounds
Conclusion
A new method to access a novel class of heterocyclic derivatives has been described. The new compounds
Experimental
Compounds
General Procedure (preparation of compound 6a )
A mixture of benzothiazole
Ethyl 2-(2-(1H-indol-3-yl)benzo[d]thiazol-3(2H)-yl)-2-cyanoacetate (6a)
Pale yellow powder, yield 92%, 0.33 g; m.p. 116–118 °C, IR (KBr) (vmax/cm−1): 1730. MS, m/z (%) = 318 (M-OEt, 87), 290 (M-CO2Et, 80), 251 (M-C5H6NO2, 79), 202 (M-C8H6N and OEt, 47), 116 (C8H6N, 74), 112 (C5H6NO2, 62), 73 (CO2Et, 56), 45 (OEt, 34). Anal. calcd for C20H17N3O2S (363.35): C, 66.11; H, 4.71; N, 11.56%, Found: C, 66.22; H, 4.79; N, 11.51%. 1H NMR (250.1, CDCl3), δ: 1.12 (3H, t, 3JHH = 7.3 Hz, CO2CH2CH3), 4.10 (2H, q, 3JHH = 7.3 Hz, CO2CH2CH3), 6.70–8.30 (11H, m, 9 ArH, 1 from S-CH and NC-CH), 8.86 (1Hbr, s, HN). 13C NMR (62.9 MHz, CDCl3), δ: 14.5 (CO2CH2CH3), 59.5 (CO2CH2CH3), 69.7 (NC-CH), 92.3, (S-CH), 103.3, 106.1, 108.7, and 109.8 (4 aromatic carbons), 117.6 (CN), 118.6, 120.5, 121.7, 122.1, 124.3, 124.9, 127.2, 132.7, 146.5, and 147.6 (10 aromatic carbons), 169.2 (C=O).
Ethyl 2-cyano-2-(2-(2-methyl-1H-indol-3-yl)benzo[d]thiazol-3(2H)-yl)acetate (6b)
Pale yellow powder, yield 92%, 0.35 g; m.p. 109–111 °C, IR (KBr) (vmax/cm−1): 1728. MS, m/z (%) = 333 (M-Et and Me, 88), 332 (M-OEt, 83), 362 (M-Me, 92), 304 (M-CO2Et, 76), 247 (M-C9H8N, 58), 130 (C9H8N, 69). Anal. calcd for C21H19N3O2S (377.38): C, 66.84; H, 5.07; N, 11.13%, Found: C, 66.92; H, 5.12; N, 11.19%. 1H NMR (250.1 MHz, CDCl3), δ: 1.15 (3H, t, 3JHH = 7.2 Hz, CO2CH2CH3), 2.46 (3H, s, Me), 4.11 (2H, q, 3JHH = 7.2 Hz, CO2CH2CH3), 6.67–8.32 (10H, m, 8 ArH, 1 from S-CH and NC-CH), 8.83 (1Hbr, s, HN).13C NMR (62.9 MHz, CDCl3), δ: 13.75 (Me), 14.3 (CO2CH2CH3), 61.2 (CO2CH2CH3), 71.1 (NC-CH), 93.8, (S-CH), 104.3, 105.2, 107.8, and 109.4 (4 aromatic carbons), 116.5 (CN), 117.9, 120.2, 120.8, 121.3, 122.3, 123.1, 126.2, 136.4, 147.1, and 148.4 (10 aromatic carbons), 169.8 (C=O).
Ethyl 2-(2-(1H-indol-3-yl)-2-methylbenzo[d]thiazol-3(2H)-yl)-2-cyanoacetate (6c)
Yellow powder, yield 90%, 0.34 g; m.p. 126–128 °C, IR (KBr) (vmax/cm−1): 1731. MS, m/z (%) = 362 (M-Me, 93), 347 (M-2Me, 91), 317 (M-OEt and Me, 85), 304 (M-CO2Et, 68), 261 (M-C8H6N, 59), 116 (C8H6N, 73). Anal. calcd for C21H19N3O2S (377.38): C, 66.84; H, 5.07; N, 11.13%, Found: C, 66.78; H, 5.01; N, 11.17%. 1H NMR (250.1 MHz, CDCl3), δ: 1.10 (3H, t, 3JHH = 7.3 Hz, CO2CH2CH3), 2.11 (3H, s, Me), 4.13 (2H, q, 3JHH = 7.3 Hz, CO2CH2CH3), 6.65–8.30 (10H, m, 9 ArH, 1 from NC-CH), 8.79 (1Hbr, s, HN).13C NMR (62.9 MHz, CDCl3), δ: 13.9 (CO2CH2CH3), 29.7 (Me), 61.6 (CO2CH2CH3), 70.8 (NC-CH), 97.4, (S-CMe), 106.3, 108.9, 110.5, and 112.4 (4 aromatic carbons), 118.2 (CN), 119.7, 120.4, 121.2, 122.7, 123.1, 123.9, 135.1, 136.5, 147.3, and 149.0 (10 aromatic carbons), 168.4 (C=O).
Ethyl 2-cyano-2-(2-methyl-2-(2-methyl-1H-indol-3-yl)benzo[d]thiazol-3(2H)-yl)acetate (6d)
Pale brown powder, yield 89%, 0.35 g; m.p. 119–121 °C, IR (KBr) (vmax/cm−1): 1730. MS, m/z (%) = 376 (M-Me, 92), 361 (M-2Me, 87), 346 (M-OEt, 70), 318 (M-CO2Et, 66), 261 (M-C9H8N, 76), 130 (C9H8N, 69). Anal. calcd for C22H21N3O2S (391.4): C, 67.51; H, 5.41; N, 10.73%, Found: C, 67.60; H, 5.30; N, 10.79%. 1H NMR (250.1 MHz, CDCl3), δ: 1.17 (3H, t, 3JHH = 7.4 Hz, CO2CH2CH3), 2.16 and 2.50 (6H, 2s, 2Me), 4.09 (2H, q, 3JHH = 7.4 Hz, CO2CH2CH3), 6.58–8.28 (9H, m, 8 ArH, 1 from NC-CH), 8.76 (1Hbr, s, HN). 13C NMR (62.9 MHz, CDCl3), δ: 13.9 (CO2CH2CH3), 14.4 (Me), 29.5 (Me), 62.2 (CO2CH2CH3), 68.8 (NC-CH), 98.6, (S-CMe), 105.6, 109.1, 110.4, and 111.8 (4 aromatic carbons), 117.3 (CN), 120.3, 120.9, 122.2, 122.6, 123.0, 124.1, 134.7, 136.9, 148.2, and 148.8 (10 aromatic carbons), 168.9 (C=O).
Ethyl 2-(2-(1H-indol-3-yl)-2,5-dimethylbenzo[d]thiazol-3(2H)-yl)-2-cyanoacetate (6e)
Brown powder, yield 90%, 0.35 g; m.p. 129–131 °C, IR (KBr) (vmax/cm−1): 1733. MS, m/z (%) = 376 (M-Me, 81), 361 (M-2Me, 77), 346 (M-OEt, 66), 318 (M-CO2Et, 57), 275 (M-C8H6N, 62), 116 (C8H6N, 51). Anal. calcd for C22H21N3O2S (391.4): C, 67.51; H, 5.41; N, 10.73%, Found: C, 67.64; H, 5.50; N, 10.70%. 1H NMR (250.1 MHz, CDCl3), δ: 1.21 (3H, t, 3JHH = 7.5 Hz, CO2CH2CH3), 1.80 and 2.30 (6H, 2s, 2Me), 4.20 (2H, q, 3JHH = 7.5 Hz, CO2CH2CH3), 6.64–8.20 (9H, m, 8 ArH and 1 from NC-CH), 8.73 (1Hbr, s, HN).13C NMR (62.9 MHz, CDCl3), δ: 13.7 (Me), 14.1 (CO2CH2CH3), 14.6 (Me), 60.7 (CO2CH2CH3), 67.5 (NC-CH), 97.2, (S-CMe), 103.7, 107.4, 109.9, and 112.3 (4 aromatic carbons), 117.7 (CN), 119.4, 120.8, 121.6, 122.7, 123.0, 125.2, 133.6, 138.3, 147.2, and 149.1 (10 aromatic carbons), 171.2 (C=O).
Ethyl 2-cyano-2-(2, 5-dimethyl-2-(2-methyl-1H-indol-3-yl)benzo[d]thiazol-3(2H)-yl)acetate (6f)
Brown powder, yield 88%, 0.36 g; m.p. 135–137 °C, IR (KBr) (vmax/cm−1): 1732. MS, m/z (%) = 390 (M-Me, 93), 375 (M-2Me, 85), 360 (M-OEt, 61), 345 (M-OEt and Me, 51), 332 (M-CO2Et, 53), 275 (M-C9H8N, 59). Anal. calcd for C23H23N3O2S (405.43): C, 68.14; H, 5.72; N, 10.36%, Found: C, 68.02; H, 5.80; N, 10.29%. 1H NMR (250.1 MHz, CDCl3), δ: 1.27 (3H, t, 3JHH = 7.1 Hz, CO2CH2CH3), 1.85, 2.27 and 2.50 (9H, 3s, 3Me), 4.23 (2H, q, 3JHH = 7.1 Hz, CO2CH2CH3), 6.54–8.25 (8H, m, 7 ArH and 1 from NC-CH), 8.77 (1Hbr, s, HN).13C NMR (62.9 MHz, CDCl3), δ: 13.7 (Me), 14.4 (CO2CH2CH3), 21.6 and 27.8 (2Me), 61.3 (CO2CH2CH3), 68.6 (NC-CH), 99.1, (S-CMe), 103.3, 106.9, 108.7, and 113.2 (4 aromatic carbons), 118.1 (CN), 118.9, 120.7, 122.3, 123.2, 123.8, 126.1, 133.5, 137.4, 148.3, and 149.4 (10 aromatic carbons), 170.5 (C=O).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received financial support from the Research Council of Chabahar Maritime University.

