Abstract
Two novel RE–Hg (RE = rare earth) complexes [RE(IA)3(H3O)2]2n·2n(HgCl4)·n(HgCl5)·nH2O·3nH3O (RE = Y,
Two novel lanthanide compounds were synthesized and characterized. They are isostructures and feature a one-dimensional chain-like structure and three-dimensional supramolecular networks. They show photoluminescence emission bands in the blue or red region with remarkable CIE chromaticity coordinates of (0.1172, 0.182) and (0.623, 0.3765), respectively. They show wide optical band gaps of 3.29 eV and 2.89 eV.
Introduction
Rare earth materials have gained more and more attention in recent years because of their fascinating magnetic, semiconductor, and photoluminescence performance.1–9 Many researchers have devoted their efforts toward the exploration of the design, preparation, and characterization of novel rare earth materials. To date, scientists have accomplished a large number of studies on rare earth materials in order to reveal the practical applications of such materials in the field of light-emitting diodes (LEDs), magnets, electrochemical displays, luminescent probes, and so on.10–13 In comparison with the many studies on the magnetic and photoluminescence performance of the rare earth materials, reports on the semiconductor performance of the rare earth materials are still rare and needs to be explored further.
Zinc, cadmium, and mercury are group 12 (IIB) elements and they have also gained increasing interest for many decades due to the following reasons: their various coordination numbers, their photoluminescence and photoelectric performance, as well as the vital role played by zinc in the biosystem.14,15 Moreover, IIB elements are also an important component for semiconductor materials. To date, many semiconductor materials using IIB element have been prepared.16–19 We have focused on exploring photoluminescence and semiconductor materials for a long time. In recent years, our main aim was investigating RE–IIB (RE = rare earth) materials in order to obtain new insights into their structures, photoluminescence, and semiconductor performance. We report in the present work the syntheses, structures, photoluminescence, and semiconductor performance of two novel RE–Hg materials [RE(IA)3(H3O)2]2n·2n(HgCl4)·n(HgCl5)·nH2O·3nH3O (RE = Y,
Results and discussion
The title compounds

An ORTEP plot of

A one-dimensional (1D) chain of

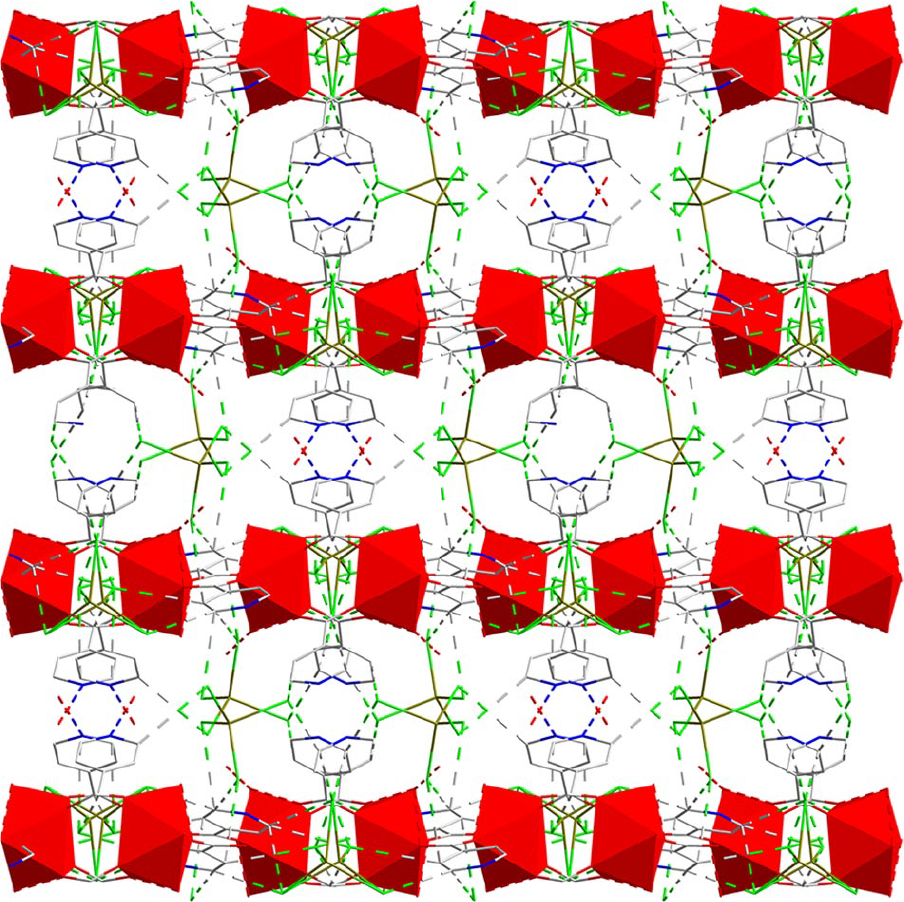
(a) A three-dimensional (3D) supramolecular network of
It is well known that rare earth materials usually exhibit photoluminescence performance, which endows these materials with important applications in the areas of LEDs, chemical sensors, electrochemical displays, and so on.26,27 To date, a large number of rare earth materials have been reported due to their fascinating photoluminescence performance.28–30 Furthermore, mercury materials and isonicotinic acid compounds can also display photoluminescence performance.31–33 Therefore, based on the above considerations, we deem that both lanthanide complexes could display interesting photoluminescence performance. The photoluminescence performance for both complexes in the present work was studied in the solid state at room temperature. The results of the photoluminescence measurements are given in Figures 4 and 5. For compound

The solid-state photoluminescence emission spectrum of
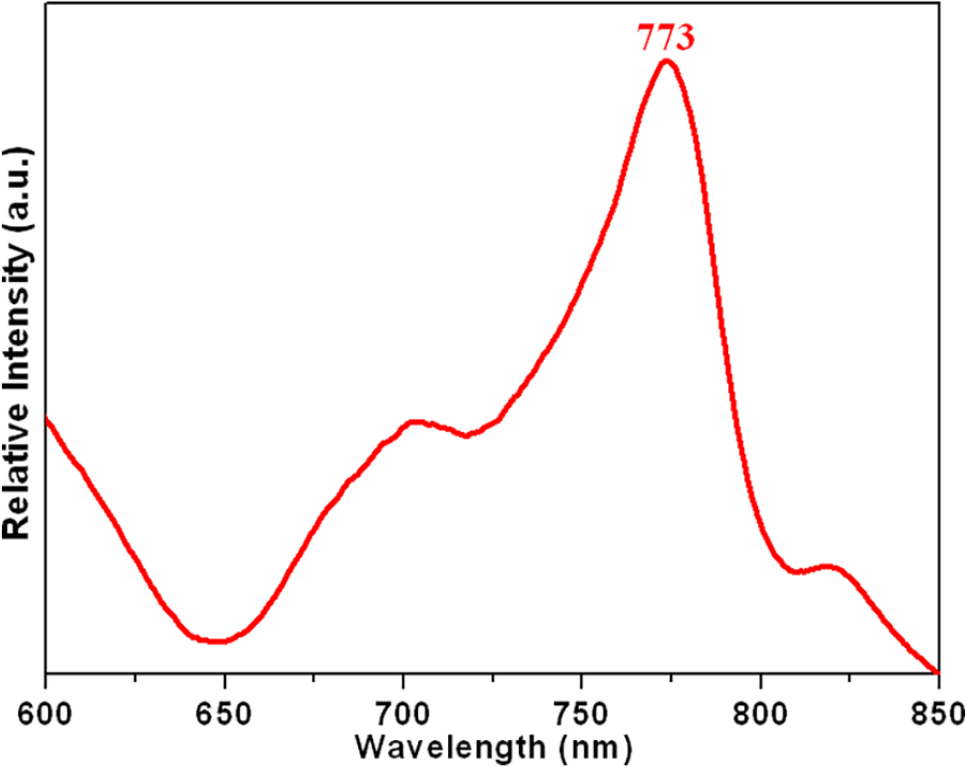
The solid-state photoluminescence emission spectrum of

The CIE chromaticity diagram and chromaticity coordinate of the photoluminescence emission spectrum of

The CIE chromaticity diagram and chromaticity coordinate of the photoluminescence emission spectrum of
The group 12 or IIB elements are zinc, cadmium, and mercury; they are well known in the field of semiconductor materials. Both compounds

Solid-state diffuse reflectance spectra for
Conclusion
In summary, we have successfully synthesized two novel rare earth materials through hydrothermal reactions. The complexes are isostructures and are characteristic of a 1D chain-like structure as well as a 3D supramolecular network. Their solid-state photoluminescence emission bands are in the blue or red region. They have remarkable CIE chromaticity coordinates of (0.1172, 0.182) and (0.623, 0.3765), respectively. As a result, they are potential candidates for LEDs. They show wide optical band gaps of 3.29 eV and 2.89 eV, as revealed by the solid-state UV/Vis diffuse reflectance spectra.
Experimental
Reagents and chemicals were commercially purchased and directly used for the preparation of the title compounds. Elemental microanalyses of carbon, hydrogen, and nitrogen were carried out on an Elementar Vario EL elemental analyzer. The infrared spectra were measured on a Perkin-Elmer (PE) Spectrum-One Fourier-transform infrared (FTIR) spectrophotometer over the frequency range 4000~400 cm−1 by using the KBr pellet technique. Photoluminescence spectra were measured on an F97XP photoluminescent spectrometer. The solid-state UV/Vis diffuse reflectance spectra were measured on a TU1901 UV/Vis spectrometer with an integrating sphere.
Synthesis of compound 1
YCl3·6H2O (1 mmol, 304 mg), HgCl2 (3 mmol, 813 mg), isonicotinic acid (3 mmol, 369 mg) and distilled water (10 mL) were loaded into a 25 mL Teflon-lined stainless steel vessel. This vessel was heated to 433 K and kept at this temperature for 10 days. Once the vessel had slowly cooled to room temperature, yellow block-like crystals were collected. The yield was 33% based on YCl3·6H2O. C36H45Cl13Hg4N6O20Y2: calculated C, 18.61; H, 1.95; N, 3.62; found C, 18.68; H, 1.98; N, 3.66. Infrared (IR) (KBr) (cm−1): 3456 (vs), 3101 (w), 2923 (w), 1964 (w), 1608 (vs), 1551 (s), 1499 (w), 1420 (vs), 1236 (m), 1058 (m), 1006 (w), 870 (w), 775 (s), 692 (s), 561 (w) and 456 (m).
Synthesis of compound 2
LuCl3·6H2O (1 mmol, 389 mg), HgCl2 (3 mmol, 813 mg), isonicotinic acid (3 mmol, 369 mg) and distilled water (10 mL) were loaded into a 25 mL Teflon-lined stainless steel vessel. This vessel was heated to 433 K and kept at this temperature for 10 days. Once the vessel had slowly cooled to room temperature, colorless block-like crystals were collected. The yield was 40% based on LuCl3·6H2O. C36H45Cl13Hg4Lu2N6O20: calculated C, 17.33; H, 1.82; N, 3.37; found C, 17.37; H, 1.84; N, 3.41. IR (KBr) (cm−1): 3457 (vs), 3089 (w), 2894 (w), 1962 (w), 1620 (vs), 1578 (s), 1498 (w), 1420 (vs), 1232 (m), 1053 (m), 1002 (w), 850 (w), 766 (s), 683 (s), 556 (w) and 420 (m).
X-ray structure determination
Carefully selected single crystals were adhered on the tip of a glass fiber and mounted in a SuperNova CCD diffractometer with the X-ray source being graphite monochromated Mo-Kα radiation. The crystal data sets were measured using the ω scan mode. CrystalClear software was used for data reduction and empirical absorption correction. The crystal structures of both compounds were solved with direct methods and the final structures were refined on F
2
with full-matrix least-squares by employing Siemens SHELXTLTM V5 crystallographic software. All non-hydrogen atoms were found based on difference Fourier maps and refined anisotropically. Crystal data as well as the details of data collection and refinement are presented in Table 1; selected bond lengths and bond angles are listed in Table 2. Crystallographic data for the structural analysis have been deposited with the Cambridge Crystallographic Data Centre, CCDC No. 1858911 and 1858912 for
Crystal data and structure refinement details.

Selected bond lengths (Å) and bond angles (º).

Symmetry codes: #1 −x+3/2, −y+3/2, −z; #2 −x+1, y, −z−1/2; #3 −x+1, y, −z−3/2.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the Open Foundation of the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, CAS (20180008).
