Abstract
Using a hydrothermal reaction, a novel
A novel

Introduction
It is well-known that most of lanthanide ions (except for La and Lu) are photoluminescent and, nowadays, lanthanide compounds with photoluminescence performance have received more and more attention.1–5 Up to date, a lot of researchers have devoted themselves into the design, preparation, and characterization of novel lanthanide compounds, for the sake of investigating the useful applications of the lanthanide compounds in the areas of electrochemical displays, light-emitting diodes, luminescent probes, and so on.6,7 In comparison with the large number of investigations on the photoluminescence performance of the lanthanide compounds, the exploration on the semiconductor performance of the lanthanide compounds is seldom yet and, therefore, it still needs to be investigated. 8
Mercury is an interesting element and it has also drawn lots of attention in recent years due to the following reasons: different coordination geometries of mercury ions, photoelectric performance, semiconductor behaviors, photoluminescence properties, and so on.9,10 These years we have kept focusing on the investigation of the compounds possessing photoluminescence and semiconductor properties, especially, the lanthanide mercury compounds, in order to obtain new findings on their crystal structures, photoluminescence, and semiconductor performance. In this work, we report the preparation, crystal structure, photoluminescence, energy transfer mechanism, and semiconductor property of a novel terbium–mercury complex {[Tb2(IA)6(H2O)4](Hg3Cl7)}
n
(nHgCl4)·nCl·4nH3O (
Results and discussion
The molecular structure of complex

Molecular structure of
There are two crystallographically independent Tb3+ ions, and both of them have an eight-coordination environment, coordinated by two oxygen atoms of two coordination water molecules and six oxygen atoms of six isonicotinic acid molecules to form a slightly contorted square anti-prismatic geometry. The bond lengths of Tb–Owater are in the range of 2.442(6)–2.515(7) Å, while those of Tb–Oisonicotinic acid are in the range of 2.287(7)–2.427(6) Å. The bond lengths of Tb–Owater are obviously longer than those of Tb–Oisonicotinic acid, indicating that terbium ions show weaker affinity to water molecules than to isonicotinic acid ligands. These Tb–O bond lengths are in the normal range and comparable with those found in the literature.17–20 The O–Tb–O bond angles locate in a very wide range of 69.0(2)°–145.6(2)°. The neighboring Tb3+ ions are interlinked by the isonicotinic acid molecules with the number of the isonicotinic acid molecules being of -2-4-2-4- to yield a one-dimensional (1D) infinite chain running along the b direction, as shown in Figure 2. The distances between the neighboring Tb3+ ions are 4.3638(1) and 5.1336(2) Å. The 1D infinite chains are interconnected by the Hg2 atom of the Hg3Cl7 moiety to yield a 2D layer extending along the ab plane, as presented in Figure 3. The electro-static interaction and van der Waals interaction among the {[Tb2(IA)6(H2O)4](Hg3Cl7)}
n
n– anionic 2D layers, isolated (HgCl4)2– anions, isolated Cl− anions, and lattice water molecules solidify the crystal packing structure of complex

The 1D chain running along the b axis. Pink: TbO8 polyhedra.

The 2D layer in

The packing diagram of
The photoluminescence spectrum for compound

The solid-state photoluminescence emission spectrum of

The CIE chromaticity diagram and chromaticity coordinate of the photoluminescence emission spectrum of
For the sake of unveiling the nature of the photoluminescence spectrum of compound

Phosphorescence spectrum of isonicotinic acid measured at 77 K.

A schematic and partial energy level diagram of the main energy absorption transfer and phosphorescence processes in compound
As mentioned previously, the energy gap between the isonicotinic acid’s lowest triplet state energy level and the Tb3+ ion’s resonant state energy level is 2541 cm−1 in the title compound. The 2541 cm−1 locates in the range of 3000 ± 500 cm−1. Therefore, this compound is expected to exhibit good photoluminescence performance. This conclusion is in good line with the photoluminescence emission curve of compound
Compound
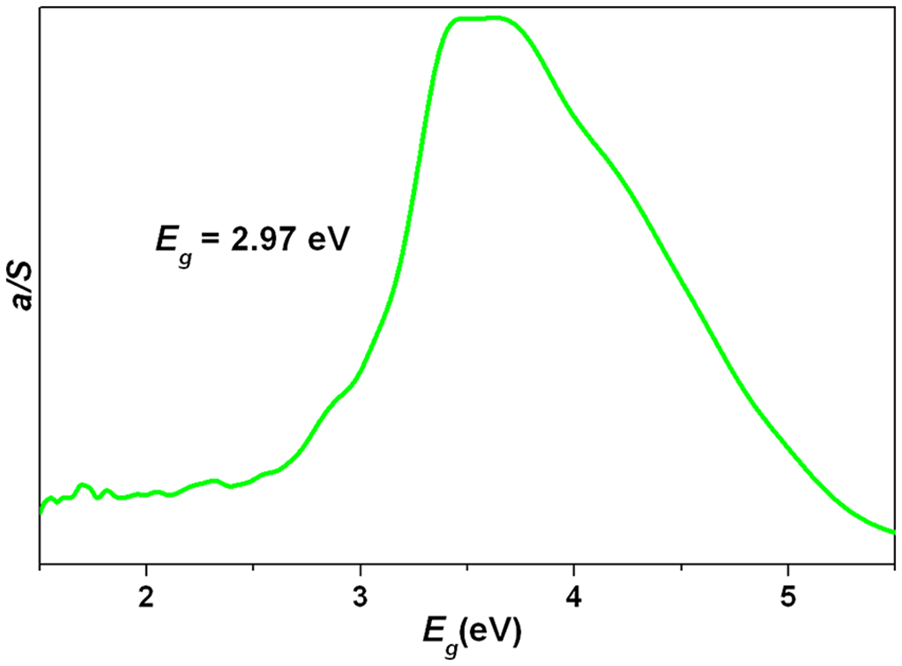
Solid-state diffuse reflectance spectrum of
The thermal stability measurement of compound

TG curve of
Conclusion
In summary, a novel terbium–mercury isonicotinic acid complex {[Tb2(IA)6(H2O)4](Hg3Cl7)} n (nHgCl4)·nCl·4nH3O was synthesized and characterized. It is characterized by a 2D layer-like structure. The solid-state photoluminescence measurement revealed that it shows four emission peaks at 489, 546, 587, and 622 nm, which could be assigned to the 5D4 → 7F6, 5D4 → 7F5, 5D4 → 7F4, and 5D4 → 7F3 transitions of the Tb3+ ions, respectively. The energy transfer mechanism was described by virtue of the energy level scheme of the Tb3+ ions and the isonicotinic acid ligand. This compound possesses remarkable CIE chromaticity coordinates of (0.3633, 0.5038) in the yellowish green region. So, it is a potential yellowish green photoluminescence material. It possesses a wide optical band gap of 2.97 eV, suggesting that it is probably a wide gap semiconductor compound.
Experimental
Materials and instrumentation
The chemicals and reagents are A.R. grade, purchased and directly used for the preparation of the title complex. IR was carried out with a KBr pellet on a Thermo Scientific Nicolet iS10 spectrometer. Solid-state photoluminescence measurement was measured on a F97XP photoluminescence spectrometer. Solid-state diffuse reflectance UV-Vis spectrum was conducted at room temperature on a computer-controlled TU1901 UV-Vis spectrometer equipped with an integrating sphere in the wavelength span of 190–1100 nm. BaSO4 powder was applied as a reference (100% reflectance), on which the finely ground powder sample was coated. TG measurement was carried out on a NETZSCH TG 209F3 analyzer in nitrogen atmosphere.
Synthesis of compound 1
TbCl3·6H2O (1 mmol, 374 mg), HgCl2 (3 mmol, 813 mg), isonicotinic acid (3 mmol, 369 mg), and 10 mL distilled water were mixed into a 25-mL Teflon-lined stainless steel vessel. The vessel was heated to 433 K and held there for 7 days. Once the vessel was slowly cooled down to room temperature, colorless crystals were obtained. The yield was 37% based on TbCl3·6H2O. IR peaks (cm−1): 3431(vs), 3068(w), 1594(vs), 1558(w), 1421(s), 1386(vs), 1248(w), 1058(w), 855(w), 759(m), 681(m), 547(w), and 413(w).
X-ray structure determination
A single crystal was adhered on the tip of a glass fiber and mounted to a SuperNova CCD diffractometer with X-ray source being graphite monochromated Mo–Kα radiation. The crystal dataset was obtained with ω scan mode. CrystalClear software
26
was used for data reduction and the empirical absorption correction. Crystal structure was solved with direct method and the final structure was refined on F2 with full-matrix least-squares by virtue of Siemens SHELXTLTM V5 crystallographic software.
27
All non-hydrogen atoms were found based on difference Fourier maps and refined anisotropically. Crystal data as well as the details of data collection and refinement are presented in Table 1, and selected bond lengths and bond angles are shown in Table 2. Crystallographic data for the structural analysis have been deposited with the Cambridge Crystallographic Data Centre, CCDC No. 1885107. Copies of this information may be obtained free of charge from the Director, CCDC, Cambridge, UK (email:
Crystallographic data and structural analysis.

Selected bond lengths (Å) and bond angles (°).

Symmetry codes: (1) –½ + x, 4 – y, z; (2) x, –1 + y, z; (3) x, 1 + y, z.
Supplemental Material
checkcif – Supplemental material for A novel terbium–mercury compound: Preparation, structure, and properties
Supplemental material, checkcif for A novel terbium–mercury compound: Preparation, structure, and properties by Wen-Tong Chen in Journal of Chemical Research
Supplemental Material
compound_1 – Supplemental material for A novel terbium–mercury compound: Preparation, structure, and properties
Supplemental material, compound_1 for A novel terbium–mercury compound: Preparation, structure, and properties by Wen-Tong Chen in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NSF of China (no. 21361013), Jiangxi Provincial Department of Education’s Item of Science and Technology (no. GJJ170637), and the open foundation of the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences (no. 20180008).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
