Abstract
The complex (Ho(IA)3(H3O)(H2O)) n (0.5nHg2I6) (HIA = isonicotinic acid) has been synthesized via a hydrothermal reaction and has a one-dimensional (1D) chain-like structure. It has an emission band in the green region of 542 nm, which could originate from the 5S2 → 5I8 transition of the Ho3+ ion. The compound possesses Commission Internationale de I’Éclairage chromaticity coordinates of (0.2065, 0.4969) and has a wide optical band gap of 2.32 eV, as found by solid-state UV-Vis diffuse reflectance measurements.

A novel holmium-mercury complex was reported. It features a 1-D chain-like structure. The solid-state photoluminescence measurement reveals that it has a green emission peak which is originated from the 5S2 → 5I8 transition of the Ho3+ ion. The energy transfer mechanism is explained by using the energy level figure of Ho3+ ions and isonicotinic acid. It displays remarkable CIE chromaticity coordinates. It is a potential candidate for green light emitting materials. It possesses a wide semiconductor band gap of 2.32 eV.
Introduction
Due to their fascinating photoluminescence, magnetic, semiconductive and other properties, lanthanide compounds have received more and more interest in exploring the potential application of these compounds in the domain of light-emitting diodes (LEDs), luminescent probes, electrochemical displays, magnets and so on.1–3 To our knowledge, in comparison with the great number of explorations on the photoluminescence and magnetic properties of the lanthanide compounds, investigation on the semiconductor properties of these compounds is still underdeveloped and needs more study. 1
Mercury has attracted interest in its coordination geometries; photoelectric, semiconductive and photoluminescence behaviour; and so on.2,3 Recently, we have been exploring lanthanide–mercury compounds to gather new findings on their crystal structures, photoluminescence and semiconductive performance. In this work, we report the preparation, crystal structure, photoluminescence, energy transfer mechanism and semiconductor properties of a holmium–mercury complex (Ho(IA)3(H3O)(H2O))
n
(0.5nHg2I6) (HIA = isonicotinic acid) (
Experimental
Reagents were used as purchased for the synthesis of the title complex. Elemental microanalyses of carbon, hydrogen and nitrogen were carried out on an Elementar Vario EL elemental analyzer. Photoluminescence measurements were conducted on a F97XP photoluminescence spectrometer. Solid-state UV-Vis diffuse reflectance spectra were performed on a TU1901 UV-Vis spectrometer and infrared (IR) measurements on a Perkin Elmer Spectrum-One Fourier-transform infrared (FT-IR) spectrophotometer using KBr pellets.
Synthesis and characterization of 1
Ho(NO3)3·6H2O (1 mmol, 459 mg), HgI2 (1 mmol, 455 mg), isonicotinic acid (3 mmol, 369 mg) and distilled water (10 mL) were mixed into a 25 mL Teflon-lined stainless steel vessel. The vessel was heated to 473 K and kept at that temperature for 7 days. Once the vessel had slowly cooled down to room temperature, yellow crystals were obtained. The yield was 30% based on Ho(NO3)3·6H2O. Anal. calcd for C18H17HgHoI3N3O8: C, 18.81; H, 1.49; N, 3.66; found: C, 18.74; H, 1.45; N, 3.61%. IR peaks (cm−1): 3445(vs), 3081(w), 1600(m), 1547(m), 1495(w), 1385(s), 1271(w), 1236(s), 1196(vs), 1140(w), 1096(s), 1043(s), 907(m), 832(m), 767(m), 714(w), 662(s), 623(s), 570(w), 543(w), 482(w) and 443(w).
X-ray crystallographic study
A single crystal was adhered to the tip of a glass fibre and mounted on a SuperNova charge-coupled device (CCD) diffractometer with X-ray source being graphite monochromated Mo-Kα radiation. The crystal data set was obtained with an ω scan mode. CrystalClear software
4
was used for data reduction and the empirical absorption correction. The crystal structure was solved with direct methods and the final structure was refined on F2 with full-matrix least-squares by virtue of Siemens SHELXTLTM V5 crystallographic software.
5
All non-hydrogen atoms were found based on difference Fourier maps and refined anisotropically. Crystal data as well as the details of data collection and refinement are presented in Table 1; selected bond lengths and bond angles are shown in Table 2. Crystallographic data for the structural analysis have been deposited with the Cambridge Crystallographic Data Centre (CCDC No. 1872603). Copies of this information may be obtained free of charge from the Director, CCDC, 12 Union Road, Cambridge, CBZ 1EZ, UK (Fax: +44-1223-336033; email:
Crystal data and structural refinements.

Selected bond lengths (Å) and bond angles (°).

Symmetry codes: #1 −x, −y + 1, −z + 3; #2 −x − 1, −y + 1, −z + 3.
Results and discussion
The crystal structure of complex

The crystal structure of

The 1D chain in

The packing diagram of
The photoluminescence spectrum of

The solid-state photoluminescence emission spectrum of
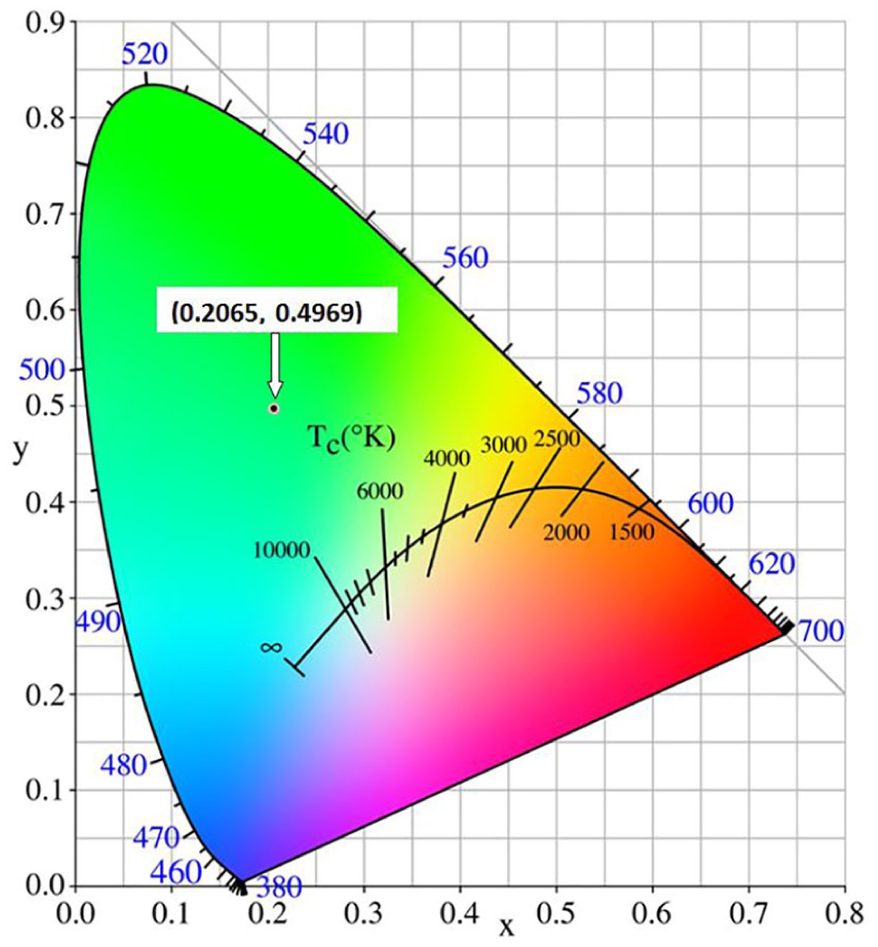
The CIE chromaticity diagram and chromaticity coordinate of the photoluminescence emission spectrum of
In order to reveal the nature of the photoluminescence emission spectrum of

Phosphorescence spectrum of isonicotinic acid measured at 77 K.

Schematic and partial energy-level diagram of the main energy absorption transfer and phosphorescence processes in
As discussed above, the energy gap between the isonicotinic acid’s lowest triplet energy level and the Ho3+ ion’s resonant energy level is 4741 cm−1 in compound
Some mercury-containing compounds are famous semiconductor materials, for instance, the MCT (HgxCd1-xTe) which is a very useful IR detector in the military area. Compound

Solid-state diffuse reflectance spectrum for
In summary, a novel holmium–mercury complex was obtained via a hydrothermal reaction and characterized by single-crystal X-ray diffraction. Compound
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: We are grateful for the financial support of the NSF of China (21361013, 31460488), NSF of Fujian (2018J01447), Jiangxi Provincial Department of Education’s Item of Science and Technology (GJJ170637) and the open foundation (20180008) of the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences.
