Abstract
A hydrothermal reaction results in the formation of a novel [Pr2(2,5-PA)2(2,5-HPA)2(H2O)4] n ·2nH2O complex (2,5-H2PA = 2,5-pyridinedicarboxylic acid). The complex is structurally characterized by single-crystal X-ray diffraction and crystallizes in the space group P21 of the monoclinic system with two formula units in one cell. This praseodymium complex is characterized by a two-dimensional layered structure. A solid-state photoluminescence experiment reveals that the praseodymium complex shows an emission in the red region. The complex has Commission Internationale de I’Éclairage chromaticity coordinates of 0.5495 and 0.4492. The photoluminescence emission bands could be assigned to the characteristic emission of the 4f electron intrashell transition of the 3P0 → 3H5, 1D2 → 3H4, 3P0 → 3H6, 3P0 → 3F2, and 3P1 → 3F3 of the Pr3+ ions. The energy transfer mechanism is explained by the energy level diagrams of the praseodymium ions and the 2,5-H2PA ligand. A solid-state diffuse reflectance measurement shows that the complex possesses a wide optical band gap of 3.48 eV.
A novel praseodymium complex is prepared and characterized. This praseodymium complex is characterized by a two-dimensional layered structure. It shows an emission in the red region, and it has Commission Internationale de I’Éclairage chromaticity coordinates of 0.5495 and 0.4492. The emission bands are resulted from the characteristic emission of the 3P0 → 3H5, 1D2 → 3H4, 3P0 → 3H6, 3P0 → 3F2, and 3P1 → 3F3 of the Pr3+ ions. It possesses a wide optical band gap of 3.48 eV.

Introduction
In recent years, lanthanide compounds have been a research hotspot in materials and chemical sciences.1–9 This is mainly because lanthanide compounds are widely used in sensors, luminous materials, magnetic materials, cell imaging, electroluminescent devices, luminous probes, catalysts, and many other fields.10–19 These broad applications have resulted from the versatile properties of lanthanide compounds. Among these properties, photoluminescence is particularly attractive. The photoluminescence of lanthanide compounds is mainly caused by lanthanide ions because lanthanide ions are rich in 4f electrons. If 4f electrons can be transferred effectively, lanthanide compounds usually emit ideal photoluminescence. However, due to the low absorption coefficient of lanthanide ions, 4f electrons cannot be transferred effectively. Therefore, to promote the transition of 4f electrons of lanthanide ions and improve their absorption coefficient, many kinds of organic molecules with conjugated structures have been used as coordination ligands. Aromatic carboxylic acids and heterocyclic derivatives are such organic compounds. It is considered that these organic compounds can absorb light, then transfer light energy to the lanthanide ions, and promote the transition of 4f electrons, that is, the so-called “antenna effect.”20,21 Compared with a large number of structures of photoluminescence properties, there are few studies on the properties of lanthanide semiconductors, which need to be further explored. 22
The 2,5-H2PA is an aromatic carboxylic acid and a heterocyclic derivative. It is a linear molecule with two carboxyl groups at both ends and a nitrogen atom on the heterocycle. Therefore, 2,5-H2PA has five coordination atoms, which enable it to bind to multiple metal ions to form different coordination configurations or extended structures. It is considered that lanthanide compounds containing 2,5-H2PA ligands may have interesting extended structures and new physicochemical behavior. Recently, the crystal engineering of lanthanide compounds with 2,5-H2PA ligand is studied in my laboratory. In this paper, the hydrothermal synthesis, crystal structure, photoluminescence, energy transfer mechanism, Commission Internationale de I’Éclairage (CIE), and solid UV-Vis diffusion reflectance of a new praseodymium compound [Pr2(2,5-PA)2(2,5-HPA)2(H2O)4]
n
·2nH2O (
Results and discussion
The molecular structure of

A molecular structure of
In compound

A 1D chain runs along the c direction.
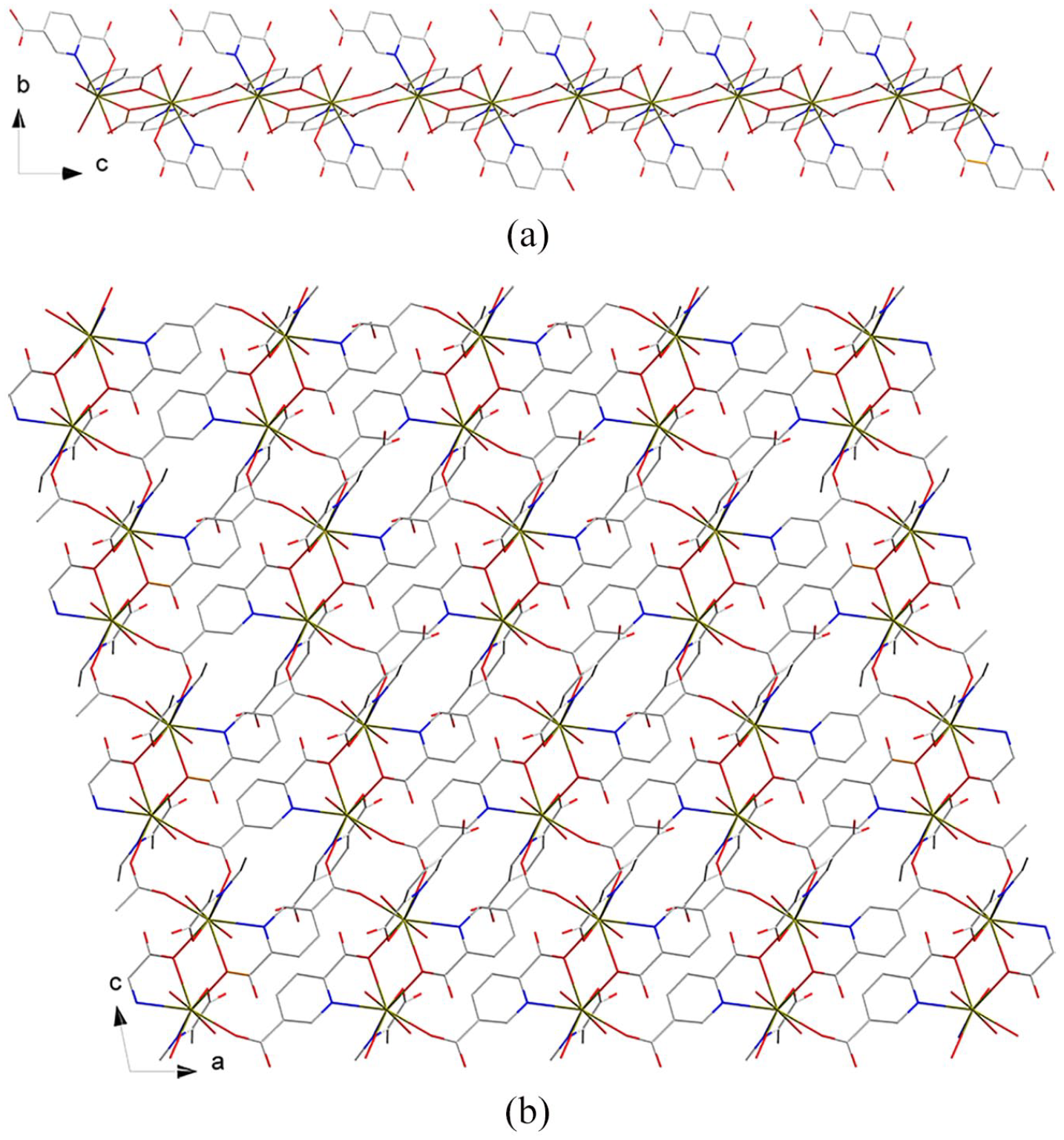
A 2D layer viewed from different directions: (a) Viewed from the a-axis and (b) from the b-axis.

A packing diagram of
To our knowledge, praseodymium compounds25,26 generally show photoluminescence emission. Based on this consideration and with efforts directed to discover the potential photoluminescent performance, solid-state photoluminescent measurements were conducted on complex

Solid-state photoluminescent emission spectra of

The CIE chromaticity diagram and chromaticity coordinates of the photoluminescence emission spectrum of
To reveal the energy transfer mechanism of the photoluminescence emission of the title compound, the phosphorescence emission spectrum of 2,5-H2PA at 77 K was measured and is given in Figure 7. The onset of the emission spectrum of 2,5-H2PA is around 525 nm, while the lowest triplet state energy of 2,5-H2PA is 19,048 cm−1. The energy difference between the lowest triplet state of 2,5-H2PA and the resonant energy level of Pr3+ ions (3P1, 21,100 cm−1) is −2052 cm−1, as presented in Figure 8. According to the intramolecular energy transfer theory proposed by Dexter 27 and Sato and Wada, 28 the intramolecular energy transfer efficiency mainly depends on two processes. One process is to transfer from the lowest triplet energy level of the ligand to the resonance energy level of the lanthanide ion through the resonance exchange interaction of Dexter. Another process is the reverse energy transfer from the lanthanide ion to the ligand by thermal inactivation. When the energy difference is too small, the reverse energy transfer process may be more likely to occur. These two energy transfer processes are obviously opposite, although they depend on the energy gap between the lowest triplet energy level of the ligand and the resonance energy level of the lanthanide ion. The intramolecular energy transfer theory points out that the assumed optimal energy gap should be about 2500–3500 cm−1. 29 A small or large energy gap may weaken the photoluminescence properties of the material.
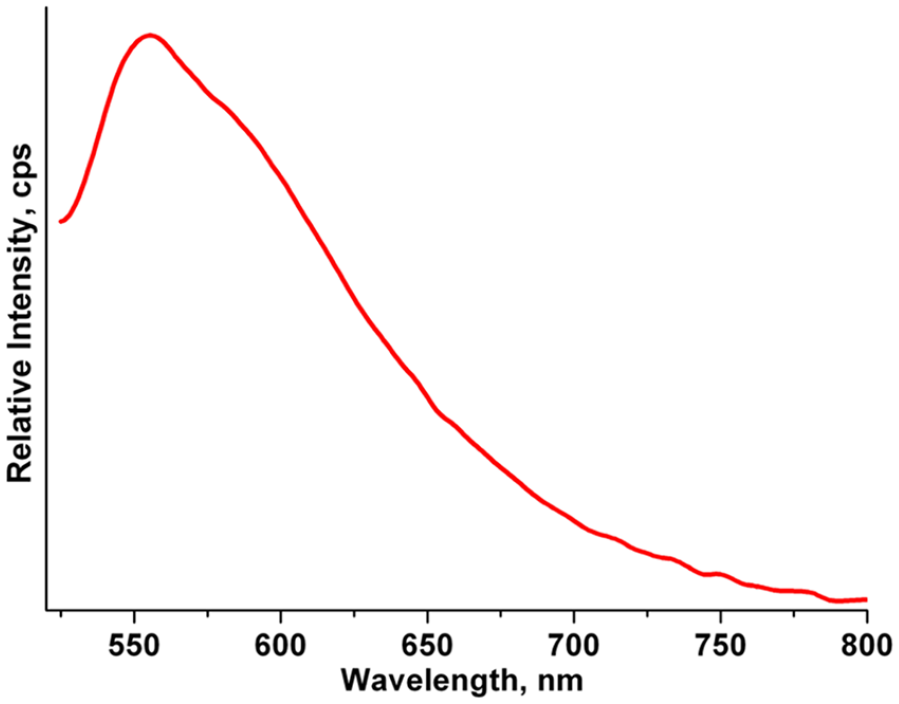
Phosphorescence spectrum of 2,5-H2PA carried out at 77 K with λex = 357 nm.

Schematic and partial energy level diagrams of the main energy absorption transfer and phosphorescence processes in
Based on the above discussion, for compound
The solid-state UV-Vis diffuse reflection spectra of complex

Solid-state UV-Vis diffuse reflectance spectrum of
Conclusion
In summary, a novel praseodymium compound with a 2D layer has been synthesized and characterized by single-crystal X-ray diffraction. A solid-state photoluminescence experiment revealed that complex
Experimental
Elemental microanalyses of carbon, hydrogen, and nitrogen were carried out on an Elementar Vario EL elemental analyzer. The infrared spectrum was measured on a PE Spectrum-One Fourier-transform infrared (FTIR) spectrophotometer over the frequency range 4000–400 cm−1 by using the KBr pellet technique. A solid-state photoluminescence experiment was performed on an F97XP photoluminescence spectrometer. Solid-state UV-Vis measurements were conducted on a TU-1901 UV-Vis spectrometer. Powder X-ray diffraction (PXRD) patterns were carried out on a Bruker D8 Advance powder diffractometer using Cu-Kα (λ = 1.54056 Å) with a step size of 0.02°. The simulated powder pattern was calculated using single-crystal X-ray diffraction data and processed by the free Mercury v1.4 program provided by the Cambridge Crystallographic Data Centre, as shown in Figure 10.
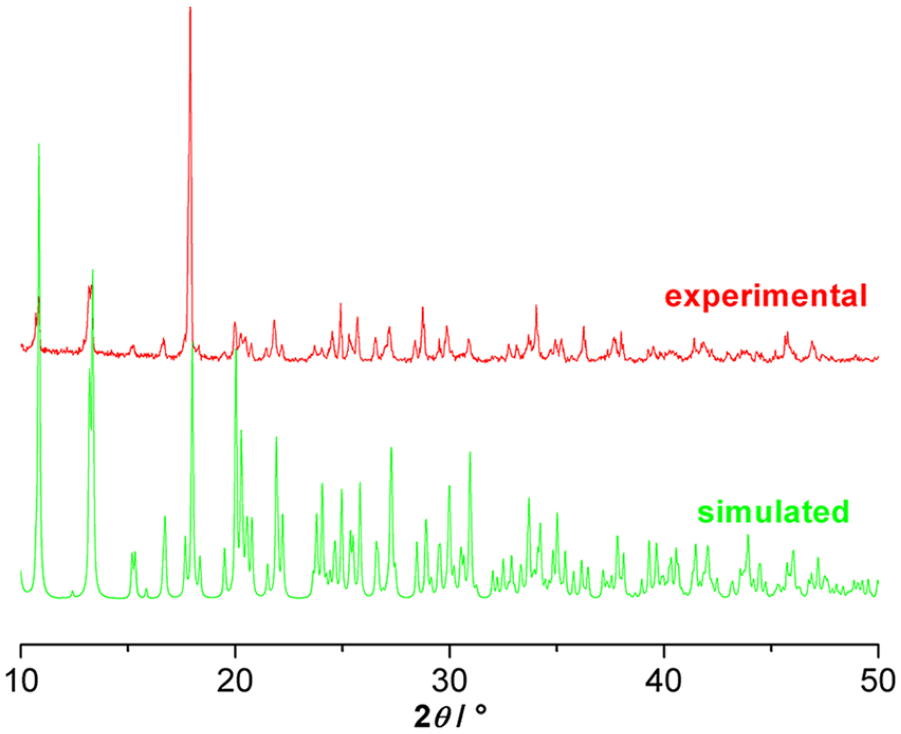
PXRD patterns for
Synthesis and characterization of [Pr2(2,5-PA)2(2,5-HPA)2(H2O)4]
n
·2nH2O (1 )
All reagents are analytical grade and commercially available and were used without further purification. Complex
X-ray crystallographic study
The X-ray diffraction dataset was measured on a SuperNova charge-coupled device (CCD) X-ray diffractometer by using a carefully selected single crystal of complex
Crystal data and structure refinement details.

Selected bond lengths (Å) and bond angles (°).

Symmetry transformations used to generate equivalent atoms: ax − 1, y, z − 1; bx + 1, y, z; cx − 1, y, z; dx + 1, y, z + 1.
Footnotes
Author’s note
This study is dedicated to Professor Jin-Shun Huang on the occasion of his 80th birthday.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Jiangxi Provincial Department of Education’s Item of Science and Technology (GJJ170637) and the Open Foundation (20180008) of the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, CAS.
