Abstract
The ground and lowest singlet excited state geometries of selected (para-C5H4X)Ir(PH3)3 iridabenzene complexes (para-substituent = NH2, OMe, Me, H, F, Cl, CCl3, CF3, NO2) are optimized using the MPW1PW91 procedure employing the LanL2DZ(Ir) and 6-311G(d, p) (C, H, N, O, P, F, Cl, P) basis sets. The excited state is generated using the time-dependent density function method. The effects of electron-donating groups and electron-withdrawing groups on the energy, atomization energy, rotational constants, and frontier orbital energies in the first singlet excited state (S1) of iridabenzene are investigated and compared to those of the ground state (S0). The Ir–C and Ir–P bonds in the studied molecules are analyzed by electron localization function and localized-orbital locator methods. The correlations between the Ir-C and Ir–P bond distances, electron localization function, and localized-orbital locator values Hammett constants (σp) and dual parameters (σI and σR) are given for the two studied states. The para-delocalization index is used for investigation of the aromaticity of the studied complexes.
Keywords

Introduction
Metallabenzenes are a type of metallacycle that consists of six-membered ring. Metallabenzenes are similar to benzene with one CH unit substituted by an isolobal transition-metal fragment.1–5 Over the years, the preparation and characterization of various metallabenzenes have been reported.6–13 The aromaticity of metallabenzenes compared to benzene and cyclic heteroaromatics has been investigated.14–18
As an example, replacing a CH of benzene with isolobal fragments of iridium leads to the synthesis of different iridabenzenes.19–22
Iridabenzene was the initial synthetic sample of a metallabenzene with a structure like that suggested by Thorn and Hoffmann. 23 Crystal structures have been described for high oxidation state iridabenzenes. 24 These 16-electron complexes have an open coordination site at iridium. In other studies, the nonlinear optical (NLO) properties of these complexes have been explored. 25 The polarity effect of the solvent on the structure and electronic properties of these complexes have been reported using computational methods. 26 Also, in the other theoretical studies, the structure, chemical reactivity, aromaticity, and 14N nuclear quadruple resonance (NQR) parameters of iridapyridine isomers have been investigated. 27
It is well known that electron-withdrawing groups (EWGs) and electron-releasing groups (ERGs) influence molecular behavior. In other words, the presence of different substituents affects the physical, electronic, and spectroscopic features of molecules. This is why many theoretical studies have been conducted to show how various substituents influence the properties of organometallic complexes.26,28–39 Moreover, it has been shown that quantitative substituent parameter scales can be used to explain how molecular properties are influenced by the polar or steric effects of the substituent. For example, the Hammett constant (σp) 40 explains the electronic effects of substituents on the equilibrium and rate constants of a reacting molecule. The substituent effects in the structure and the properties of the para-substituted iridabenzene in the ground state have been studied. 26
Investigation of variations in the structures and properties of molecules in the electronic excited states is possible by quantum chemical methods. The time-dependent density functional theory (TDDFT) approach 41 is one of the most commonly employed computational methods. Numerous computational studies on the excited states of molecules have been reported.42–54
The absence of any theoretical or experimental investigations on the first singlet excited states of iridabenzene complexes encouraged us to study substitution effects on the structural, electronic properties, and aromaticity of para-substituted iridabenzenes at the first singlet excited state. We compare the obtained results with those of the ground states of the complexes.
Results and discussion
Atomization energies
Figure 1 displays the structures of the studied para-substituted iridabenzene molecules in our investigations. The atomization energy is the energy difference between a molecule and its constituent ground state atoms. To compute the atomization energies of the investigated iridabenzene molecules in the ground state (S0) and first singlet excited state (S1), molecules are considered as closed shell singlet and the multiplicities assumed for H, C, N, O, F, Cl, and P atoms are doublet, triplet, quartet, triplet, doublet, doublet, and quartet, respectively. The calculated atomization energies of the investigated molecules are listed in Table 1. Larger atomization energy values (Eatom) are observed for the S0 state compared to the S1 state.

(a) Aromatic structure and (b) non-aromatic quinonoid structure of the (para-C5H4X)Ir(PH3)3 iridabenzene complexes, where X represents an acceptor. For donors, the zwitterionic form has the opposite charge separation.
Total energy (E, a.u), excitation energy with respect to the ground state (ΔE, eV), atomization (Eatom, kcal/mol), dipole moment (µ, Debye), and para-delocalization index (PDI) values in the ground state (S0) and first singlet excited state (S1) of (para-C5H4X)Ir(PH3)3 iridabenzene complexes.

PDI: para-delocalization index.
Hammett substituent constants (σp) and dual parameters (σI and σR) for substituents.
Since in atomization processes, we break all bonds in the molecule and form separated atoms; it seems plausible that the change in electronic energy for atomization can be estimated as the sum of the energies associated with each bond in the molecule. Therefore, the atomization energy is equivalent to the total binding energy.
Energetic aspects
The absolute energy values of the investigated complexes in the S0 and S1 states are collected in Table 1. The excitation energies with respect to the ground states (ΔE) are calculated (Table 1). The Hammett substituent constants (σp) 55 and dual parameters (σI and σR) 55 are also listed in Table 1. The σR and σI values are the resonance and inductive constants of the substituents, respectively.
The plot of ΔE values versus the Hammett constants (σp) values is shown in Figure 2. Surprisingly, the ΔE values demonstrate a linear relation with respect to the σp values


Linear correlation between the ΔE = E(S1) − E(S0) values and Hammett constants (σp) in (para-C5H4X)Ir(PH3)3 iridabenzene complexes.
It is found that these values are lower in the presence of electron-donating groups (EDG) compared to EWG.
Rotational constants
The calculated rotational constants for the investigated iridabenzene molecules in the S0 and S1 states are shown in Table 2.
Rotational constants (GHz) in the ground state (S0) and first singlet excited state (S1) of (para-C5H4X)Ir(PH3)3 iridabenzene complexes.

These values show that rotational constants in the excited state are less than those of the corresponding ground state rotational constants. Since the moment of inertia (and consequently the rotational constant) of a molecule is directly related to the nuclear coordinate, 56 it is reasonable that the rotational constant changes in the S0 and S1 states.
By taking a closer look at the data in Table 2, it is revealed that the studied iridabenzenes are asymmetric top as expected (namely, A ≠ B ≠ C). With the electron number (Ne) of the molecule becoming larger, the rotational constants become smaller. It is interesting to note, however, that there are good linear correlations between the A values and number of electrons

Dipole moment
The dipole moment values of the studied para-substituted iridabenzene molecules in the S0 and S1 states are shown in Table 1. Larger dipole moment values are observed in the S1 state compared to the S0 state. Therefore, the electronic distributions of the first singlet excited state differ compared to the ground state because of the large charge transfer excitation. These electronic distributions are dependent on the substituent character.
However, the dipole moment values are larger in the presence of EWGs compared to EDGs in both the S0 and S1 states. It is interesting to note, however, that there are good linear correlations between the dipole moment values and the Hammett constants (Figure 3)


Linear correlation between the dipole moment values and Hammett constants in (para-C5H4X)Ir(PH3)3 iridabenzene complexes in the S0 and S1 states.
Bond distances
The C–C and Ir–C bond distances of the studied para-substituted iridabenzene molecules are listed in Table 3 for the S0 and S1 states.
Selected bond distances (in Å) in the ground state (S0) and first singlet excited state (S1) of (para-C5H4X)Ir(PH3)3 iridabenzene complexes.

These values show that the Ir–C2, C3–C4, and Ir–Pax bond distances are longer in the S1 state than in the S0 state. In contrast, the C2–C3 and Ir–Peq bond distances are shorter in S1 state than S0 state.
The study of changes in bond distances in S1 state compared to S0 state shows that the most changes occurs in Ir–Pax bond distance. These changes are larger in the presence of EWGs.
As shown, the Ir–Pax bond lengths are longer in the presence of EDGs than in the EWGs in the S0 state. In contrast, the Ir–Pax bond lengths are longer in the presence of EWGs compared with EDGs in the S1 state.
The Ir–C and C–C bond lengths in the ground state are compatible with the experimental data of similar compounds. 57 The shorter C2–C3 bond in the S1 state compared to that in the S0 state indicates a larger contribution of the zwitterionic resonance structure to the S1 state (Figure 1(b)).
The plots of the Ir–C and Ir–P bond distances versus Hammett constants give the following equations:
S 0 state

S 1 state

It can be found that there are better Hammett correlations for the Ir–Peq bonds compared to Ir–Pax bonds.
Next, we studied the multiple linear regression which considers the inductive (σI) and resonance (σR) dual parameters. The results of these correlations are as follows:
S 0 state

S 1 state

These equation show good correlations for r(Ir–P) bonds in the S0 and S1 states. In these equations, the major contribution of the σR parameter is demonstrated in the Ir–Peq bonds.
Frontier orbital energies and HOMO-LUMO gap
The computed frontier orbitals energies and highest occupied molecular orbital (HOMO)-lowest unoccupied molecular orbital (LUMO) gap energies of the studied para-substituted iridabenzene complexes in the S0 and S1 states are shown in Table 4.
Frontier orbital energies and HOMO-LUMO gap energies (eV) in the ground state (S0) and first singlet excited state (S1) of (para-C5H4X)Ir(PH3)3 iridabenzene complexes.

HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital.
These values show that the energy of stability of the frontier orbitals increases in the presence of EWGs in both the S0 and S1 states. In comparison, the stability of the frontier orbitals decreased in the presence of EDGs in both the S0 and S1 states.
Good linear correlations can be observed between the frontier orbital energy values and Hammett constants in both the S0 and S1 states:
S 0 state

S 1 state

However, there are larger HOMO-LUMO gap values in the presence of EDGs compared with EWGs in both the S0 and S1 states. This trend is due to an electron-withdrawing inductive effect which concentrates the electronic density over the substituted region of the complex. The HOMO-LUMO gap values are larger in the S0 state compared to the S1 state.
Electron localization function analysis
We analyzed the nature of the chemical bonding in the Ir–C and Ir–P bonds on the basis of the electron localization function (ELF) 58 distribution, which is indicative for concentrations of valence electron density in regions of chemical bonds.
A large ELF value corresponds to largely localized electrons which indicates that a covalent bond, a lone pair, or inner shells of the atom are involved. Table 5 reveals larger ELF values for the Ir–C bond compared to Ir–P bonds. There are larger ELF values for the Ir–C bond in the presence of EWGs. However, the ELF values of the Ir–C bond are larger in the S0 state than the S1 state. There are larger ELF values for the Ir–Pax bond compared to the Ir–Peq bond in the S0 state. The plots of ELF values of the Ir–C and Ir–P bonds versus the Hammett constants give the following equations:
ELF and LOL values of selected bonds in the ground state (S0) and first singlet excited state (S1) of (para-C5H4X)Ir(PH3)3 iridabenzene complexes.

ELF: electron localization function; LOL: localized-orbital locator.
S 0 state

S1 state
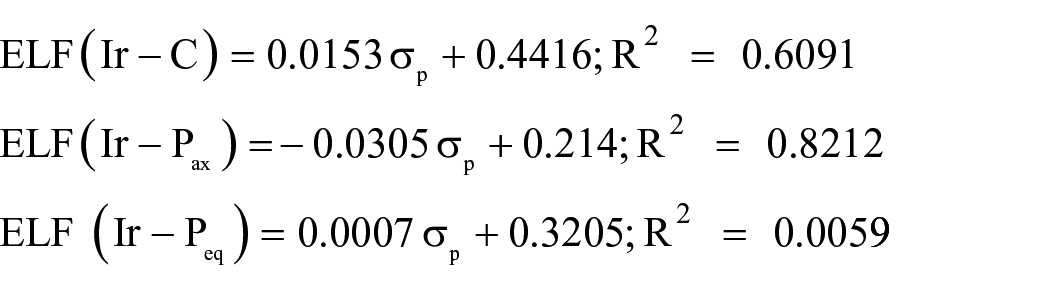
It can be seen that there are poor Hammett correlations for these equations.
We also studied the multiple linear regression which considers the inductive (σI) and resonance (σR) dual parameters. The results of these correlations are:
S0 state

S1 state

These equations show good correlations of ELF(Ir–C) for the S0 and S1 states and ELF(Ir–Pax) for the S0 state. In these equations, the major contribution of the σR parameter is demonstrated.
Figure 4 presents shaded surface maps with projections of the ELF in the (para-C5H5)Ir(PH3)3 iridabenzene complex for S0 and S1 states.

Shaded surface maps with projection of (a) the electron localization function (ELF) and (b) the localized-orbital locator (LOL) in the (para-C5H5)Ir(PH3)3 iridabenzene complex in the S0 and S1 states.
Localized-orbital locator analysis
A developed descriptor for electron localization is the localized-orbital locator (LOL).59,60 The LOL gives simple, recognizable patterns in classic chemical bonds and proves useful in interpreting the structures of unusual materials. LOL analysis focuses on the topological properties of kinetic energy density. 61
A large LOL value corresponds to largely localized electrons which indicates that a covalent bond, a lone pair, or inner shells of the atom are involved. Table 5 reveals larger ELF values for Ir–C bonds compared to Ir–P bonds. There are larger LOL values for Ir–C bonds in the presence of EWGs. However, the LOL values of Ir–C bonds are larger in the S0 state than in the S1 state. There are larger LOL values for the Ir–Pax bond compared to the Ir–Peq bond in the S0 state. In contrast, there are larger LOL values for the Ir–Peq bond compared to the Ir–Pax bond in the S1 state.
The plots of the LOL values of the Ir–C and Ir–P bonds versus Hammett constants give the following equations:
S0 state

S1 state
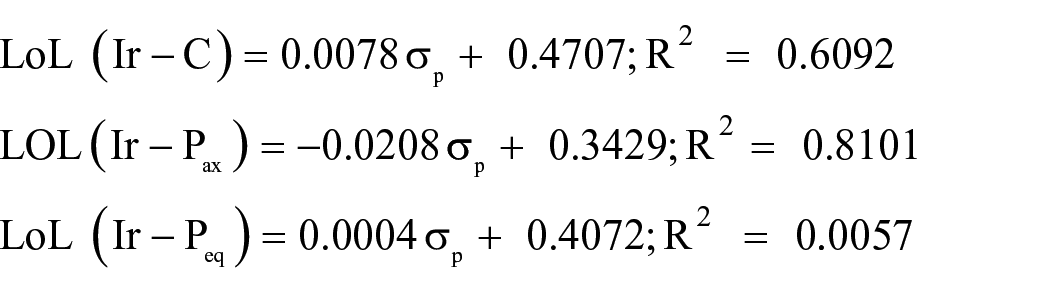
It is observed that there are poor Hammett correlations for these equations.
Studying the multiple linear regression, which considers the inductive (σI) and resonance (σR) dual parameters, gave the following correlations:
S0 state

S1 state

These equations show good correlations for LOL(Ir–C) in the S0 and S1 states and for LOL(Ir–Pax) in the S0 state. In these equations, the major contribution from the σR parameter is demonstrated.
Figure 4 reveals shaded surface maps with projection of the LOL in the (para-C5H5)Ir(PH3)3 iridabenzene complex for the S0 and S1 states.
Aromaticity
The para-delocalization index (PDI) is a quantity for measuring the aromaticity of six-membered rings.62,63 PDI is fundamentally the averaged para-delocalization index (para-DI) in six-membered rings. In the studied systems, this parameter is calculated as

The foundation of PDI was laid according to a study by Bader et al., who believed that the DI in benzene is greater for para-related than for meta-related carbon atoms. It is obvious that the PDI would mean a larger delocalization and stronger aromaticity. The main limitation of the definition of PDI is its applicability for studying the aromaticity of six-membered rings only; it was reported that the PDI is unsuitable for cases in which the ring plane has an out-of-plane distortion.
The PDI values of the studied molecules are listed in Table 1. These values show larger PDI values for EWGs compared to EDGs. This result is consistent with nucleus-independent chemical shift (NICS) values in previous studies on para-substituted iridabenzenes. 34
Conclusion
Computational investigations of the substitution effects on the structures and properties of para-substituted iridabenzenes in the ground state and first singlet excited state showed the following:
Atomization energy values (Eatom) for the S1 state were larger compared to the S0 state.
The energy difference values of the ground state and first singlet excited state were lower in the presence of EDGs compared to EWGs.
The rotational constants in the excited state were less than those of the corresponding ground state rotational constants.
The dipole moment values were larger in the presence of EWGs compared to EDGs in both the S0 and S1 states. There were good linear correlations between the dipole moment values and the Hammett constants.
The Ir–Pax bond lengths were longer in the presence of EDGs compared with EWGs in the S0 state. In contrast, the Ir–Pax bond lengths were longer in the presence of EWGs compared with EDGs in the S1 state.
The ELF and LOL values for the Ir–C bond are larger compared to the Ir–P bonds. There are larger ELF and LOL values for the Ir–C bond in the presence of EWGs. However, the ELF and LOL values of Ir–C bond are larger in the S0 state than the S1 state.
The larger PDI values for EWGs compared to EDGs indicate their enhanced aromatic properties in the presence of EWGs.
Computational methods
All calculations were carried out with the Gaussian (Version 09) software package 64 using the standard 6-311G (d, p) basis set computations for systems involving the main group elements.65,66 For Ir, the standard LANL2DZ basis set67–69 is applied and Ir is explained by the effective core potential (ECP) 70 with a double-ξ valance using the LANL2DZ basis set.
The ground state and first singlet state geometry optimizations were carried out by applying the parameter hybrid functional with adapted Perdew–Wang exchange and correlation (MPW1PW91). 71 According to the results of computations for transition-metal complexes, the MPW1PW91 functional outperformed B3LYP.72–75 A vibrational analysis at each stationary point verified its identity as an energy minimum. The first singlet excited states of the target compounds were determined by the TD-DFT method. 41
The Multiwfn 3.5 program was used for topological analysis of the ELF, the LOL, and for calculation of the PDI. 76
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
