Abstract
In this study, the MPW1PW91 method is applied to analyze the quantum theory of atoms in molecules, the electron localization function, and the localized-orbital locator in trans-(NHC)PtI2(para-NC5H4X) (X = H, F, COOH, CN, NO2, Me, OH, NH2) complexes. The substituent effect is assessed in the presence of electron-withdrawing groups and electron-donating groups and their influence on the Pt–C and Pt–N bonds of the molecules is analyzed using quantum theory of atoms in molecules, electron localization function, and localized-orbital locator methods. In addition, the eta index (η) is used to evaluate the Pt–C and Pt–N bonds in the studied complexes. The correlations between electron localization function, localized-orbital locator, and the η index values of Pt–C and Pt–N bonds with Hammett constants (σp) and dual parameters (σI and σR) are given.
Keywords

Introduction
Among inorganic drugs, the widely used cisplatin (cis-diaminedichloroplatinum (II), CDDP) is a highly successful agent for the treatment of cancer.1–4 This drug has been used in combination chemotherapy to treat ovarian, testicular, head, neck, and bladder cancer. Extensive experimental and computational investigations have led to the emergence of various platinum-based complexes having different levels of anticancer activity.5–9 These include the second-generation clinical anticancer agents, carboplatin and nedaplatin, the third-generation platinum complexes, oxaliplatin and lobaplatin, and other cisplatin/transplatin analogues such as fatty amine ligands, planar heterocycle ligands, iminoether ligands, and so on.10–13 Initial structure–activity connection research proposes that the leaving groups, commonly chloride and two amine ligands in the platinum complexes, must be in a cis-configuration and that the corresponding trans-compounds are inactive. However, various trans-platinum compounds have been established as potential drugs, numerous researchers have published on trans-Pt compounds with in vitro growth inhibitory and in vivo antitumor properties.12,13 More importantly, some of these complexes have been found to retain considerable efficacy against tumor cells resistant to cisplatin.14–18 For instance, the synthesis of trans-diiodo(pyridine)(1,3-dimethylimidazol-2-ylidene) platinum as a possible anticancer compound has been reported. 19 N-Heterocyclic carbene (NHC) platinum complexes have been introduced as novel platforms for providing new cytotoxic drugs of the cisplatin series. The syntheses of mixed NHC-amine Pt(II) complexes have been reported via a simplistic and modular two-step sequence leading to trans-configured square planar species. Spectroscopic methods and X-ray diffraction studies are used for the characterization of these complexes. In vitro experiments were employed to illustrate their effectiveness against both resistant (A2780/DDP, CH1/DDP, and SK-OV-3) and cisplatin-sensitive (CEM and H460) cell lines. We have previously reported theoretical studies of the substituent effect on the nuclear quadrupole resonance (NQR) and nuclear magnetic resonance (NMR) parameters in a platinum-based, anticancer, trans-(NHC)PtI2(para-NC5H4X) complexes. 20 Numerous theoretical researches on this subject have been conducted on evaluating the mechanism of action of cisplatin and its analogues.21–29
Computational investigations have been reported regarding substituent effects on the structures and properties of inorganic and organometallic complexes.20,30–38 In a further study, quantum chemical topology (QCT) descriptors as substitutes for appropriate Hammett constants have been reported. 39
In this article, we report quantum theory of atoms in molecules (QTAIM), electron localization function (ELF), and localized-orbital locator (LOL) investigations for the purpose of studying of the Pt–N and Pt–C bonds in trans-(NHC)PtI2(para-NC5H4X) complexes.
Computational methodology
All calculations were performed with the Gaussian 09 suite program 40 and employing the modified Perdew–Wang exchange and correlation (MPW1PW91) method. 41 The calculations of systems containing the main group elements (C, F, H, N, I, and O) were evaluated by the standard 6-311G(d, p) basis set.42–45 For Pt, the Def2-TZVPPD basis set 46 was used, and Pt is described by the effective core potential (ECP) of the Wadt and Hay pseudo-potential using the Def2-TZVPPD basis set. 47 Multiwfn 3.5 was used for topological analysis of the electron density, the ELF, and the LOL. 48
Results and discussion
QTAIM analysis
The QTAIM-based analysis of electron density is effective for the illustration of the chemical and physical properties of molecular systems.49–55 The topological properties of the electron density (ρ), the Laplacian of the electron density (∇2ρ), the potential electron energy density (V), the Lagrangian kinetic electron energy density (G), and the total electron energy density (H) at bond critical points (BCPs) of trans-(NHC)PtI2(para-NC5H4X) (X = H, F, COOH, CN, NO2, Me, OH, NH2) complexes (Figure 1) are calculated in this investigation.
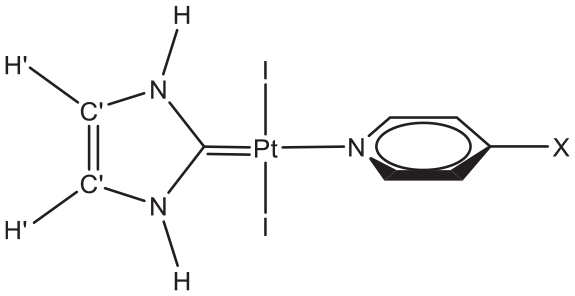
The structure of trans-(NHC)PtI2(para-NC5H4X) (X = H, F, COOH, CN, NO2, Me, OH, NH2) complexes.
Electron density
The electron density values at the BCPs of the Pt–C and Pt–N bonds in trans-(NHC)PtI2(para-NC5H4X) (X = H, F, COOH, CN, NO2, Me, OH, NH2) complexes are listed in Table 1. The QTAIM calculations indicate a higher density of electrons at the BCP of Pt–C compared to the Pt–N bond. This result is compatible with changes in the bond distances. Table 1 shows that the Pt–N bonds are longer than the Pt–C bonds in the studied complexes.
Hammett constant and the inductive (σI) resonance (σR) dual parameters of substitutions, Pt–C and Pt–N bond distances (r, in pm), electron density (ρ, in e.Å–3) and Laplacian of electron density (∇2ρ, in e.Å–5) values at the BCP of Pt–C and Pt–N bond in the trans-(NHC)PtI2(para-NC5H4X) complexes.

BCP: bond critical points; NHC: N-heterocyclic carbene.
Laplacian of electron density
Table 1 indicates that the ∇2ρ values of the Pt–C and Pt–N bonds are positive at related BCPs, as was observed for interactions of closed shells.
Energy density
According to Table 2, the energy density (H) has negative values at the BCP for the Pt–C and Pt–N bonds. Hence, shared interactions exist at these BCPs. This is consistent with findings for the M–C bonds in the organometallic complexes 56 and transition-metal carbonyl clusters, 57 where a mixture of parameters of the closed-shell and shared interactions were shown by the metal–ligand bonding. The total energy density (H) is defined as

where, G and V are Lagrangian kinetic energy and potential energy density, respectively.
Total energy density (H, in a.u), Lagrangian kinetic energy (G, in a.u), potential energy density (V, in a.u) and η index values at the BCP of Pt–C and Pt–N bond in the trans-(NHC)PtI2(para-NC5H4X) complexes.

BCP: bond critical points; NHC: N-heterocyclic carbene.
Eta index
The eta index is defined as58,59

where λ1 and λ3 are the smallest and largest eigenvalues of the Hessian matrix of electron density, respectively. It was argued that the η < 1 and η > 1 values at BCP correspond to the closed-shell and covalent interactions, respectively. A stronger covalent character of the bond is conveyed with more positive η index values.
The η index values at the BCPs of the Pt–C and Pt–N bonds are listed in Table 2. These values show greater η index values for Pt–C bonds compared with Pt–N bonds in the studied complexes. Therefore, the Pt–C bond is more covalent compared to the Pt–N bond in these complexes. This result is compatible with variations in the corresponding bond distances. The dependencies of the X-character on the eta index values show that the eta index values of the Pt–C bonds are larger in the presence of electron-withdrawing groups (EWGs) compared to electron-donating groups (EDGs). On the contrary, the eta index values of the Pt–N bond are larger in the presence of EDGs compared to EWGs. Investigations of the dependency of the eta index values on the Hammett constant (σp) give the following equations

It can be observed that there is good linear correlation between η(Pt–C) and the Hammett constant. In contrast, a good linear correlation between η(Pt–N) and the Hammett constant was not observed.
Next, we studied the multiple linear regression which considers the inductive (σI) and resonance (σR) dual parameters. The results of these correlations are

These equations show good correlations for the eta indices of the Pt–C and Pt–N bonds. In these equations, a major contribution of the σR parameter is demonstrated in the Pt–C and Pt–N bonds.
ELF
The ELF values of the Pt–C and Pt–N bonds in the trans-(NHC)PtI2(para-NC5H4X) (X = H, F, COOH, CN, NO2, Me, OH, NH2) complexes are listed in Table 3. These values show greater ELF values for the Pt–C bonds compared to the Pt–N bonds in the studied complexes. Therefore, the Pt–C bond is more covalent in comparison to the Pt–N bond in these complexes. This result is compatible with variations in the corresponding bond distances. The dependencies of the X-character on the ELF values show that those of the Pt–C bond are larger in the presence of EWGs in compared with those with EDGs. On the contrary, the ELF values of the Pt–N bond are larger in the presence of EDGs compared to EWGs. Investigations of the dependency of ELF values on the Hammett constant (σp) show the following equations

ELF and LOL values of Pt–C and Pt–N bond in the trans-(NHC)PtI2(para-NC5H4X) complexes.

ELF: electron localization function; LOL: localized-orbital locator.
It can be observed that there is good linear correlation between the ELF(Pt–C) and Hammett constant. Conversely, the linear correlation between the ELF(Pt–N) and the Hammett constant is not good.
Next, we studied the multiple linear regression, which considers the inductive (σI) and resonance (σR) dual parameters. The results of these correlations are
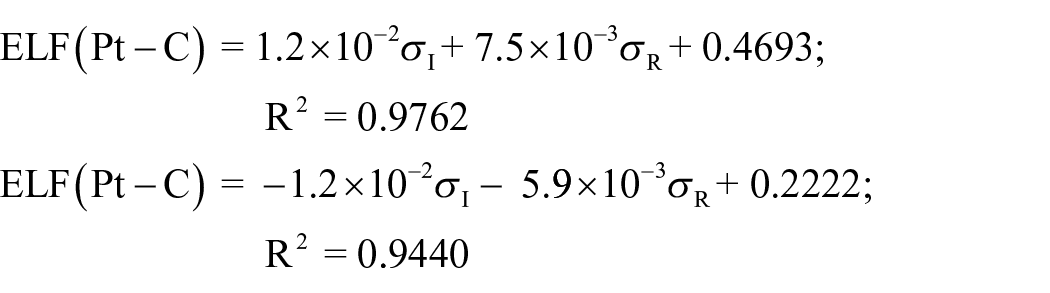
These equations show good correlations for the ELFs of the Pt–C and Pt–N bonds. In these equations, the major contribution of the σR parameter is demonstrated in the Pt–C and Pt–N bonds.
LOL
The LOL values of the Pt–C and Pt–N bonds in the trans-(NHC)PtI2(para-NC5H4X) (X = H, F, COOH, CN, NO2, Me, OH, NH2) complexes are listed in Table 3. These values show greater LOL values for the Pt–C bonds compared to the Pt–N bonds in the studied complexes. Therefore, the Pt–C bond is more covalent in character compared to the Pt–N bond in these complexes. This result is compatible with variations in the corresponding bond distances. The dependencies of the X-character on the LOL values show that the ELF values of the Pt–C bonds are larger in the presence of EWGs compared to EDGs. On the contrary, the LOL values of the Pt–N bonds are larger in the presence of EDGs compared to EWGs. An investigation of the dependency of the LOL values on the Hammett constant (σp) show the following equations

Hence, there is a good linear correlation between the LOL (Pt–C) values and the Hammett constant, while the opposite is the case for the correlation between the LOL(Pt–N) values and the Hammett constant.
As before, we have studied also the multiple linear regression, which considers the inductive (σI) and resonance (σR) dual parameters. The results of these correlations are

These equations show good correlations for the LOL values of the Pt–C and Pt–N bonds. In these equations, the major contribution of the σR parameter is demonstrated in the Pt–C and Pt–N bonds.
Conclusion
QTAIM investigations of several trans-(NHC)PtI2(para-NC5H4X) complexes have been shown larger ρBCP(Pt–C) values compared to the ρBCP(Pt–N), ρBCP(Pt–C) and ρBCP(Pt–N) values. These results were compatible with changes in the bond distances. The positive ∇2ρ(Pt–C) and ∇2ρ(Pt–N) values at the related BCPs reveal closed-shell interactions. Then, negative energy density values at the BCPs of the Pt–C and Pt–N bonds are compatible with shared interactions existing at these BCPs. The larger ELF, LOL, and η index values of the Pt–C bonds compared to the Pt–N bonds are compatible with the more covalent nature of the Pt–C bonds compared to the Pt–N bonds in these complexes. In addition, there were the good correlations between the LOL values of the Pt–C and Pt–N bonds and the σI and σR parameters, and the major contribution of the σR parameter was demonstrated for the Pt–C and Pt–N bonds.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
