Abstract
Small azomethine molecules (

Introduction
The conjugation in organic molecules is the basis of many applications, including fluorescent sensors, organic lasers, fluorescent biological labels, organic light-emitting diodes (OLEDs), photovoltaic cells, organic field-effect transistors (OFET), and so on.1–4 The use of azomethine linkers in conjugated structures has allowed the assembly of highly useful compounds.5–9 The use of these compounds as photovoltaic materials has increased due to the fact that the electron transfer is good and the azomethine group provides the electron donor.10–12
Azomethines are also very suitable components of conjugated compounds for reasons such as their easy synthesis under mild reaction conditions. Compounds containing polyazomethines have also been studied as organic photovoltaic materials, and their power conversion capacities were found to be in the range of about 0.1%–0.3% in these studies. Besides, the results of recent studies have shown that the photovoltaic performance in non-polymer small molecules is able to compete with that of polymers. Moreover, small molecules have many advantages in photovoltaics because their molecular structures can be determined precisely and easily, and their solubility is high.13–16
Many azomethines have been studied for organic optoelectronic applications. Indeed, due to the ease of obtaining azomethines and their useful photovotaic performance, a great deal of work has been done on these compounds.17–21 Thus, two new azomethines (
Results and discussion
UV-Vis and fluorescence spectroscopic analysis
The UV-Vis and fluorescence spectra of

Experimental UV-Vis and fluorescence spectra of (a)
As can be seen from Figure 1(a),
The experimental and computed UV-Vis spectral parameters and the related electronic transitions of
The experimental and calculated UV-Vis parameters of

DFT: density functional theory;
The strong absorption wavelengths for
As can be seen from Figure 1(c), P3HT has two absorption maxima, one of which is about 275 nm and the other is about 450 nm. Its fluorescence spectrum spans between 500 and 800 nm and has a maximum about 580 nm.
When Figure 1(a) and (b) are compared, P3HT’s absorption and
Theoretical HOMO-LUMO calculation
The molecular structures, the HOMO energies, and the LUMO energies of

Optimized molecular structures of (a)
HOMO-LUMO orbitals are important tools to understand the chemical reactivity and kinetic stability of molecules. 24 From photovoltaic point of view, the HOMO-LUMO relationship between the donor and the acceptor is significant for an effective charge transfer as well. There should be an energy difference of at least 0.3 eV between the LUMOs of the acceptor and donor for efficient charge transfer. The frontier orbital and HOMO-LUMO energy gaps of the azomethines are presented in Figure 3.
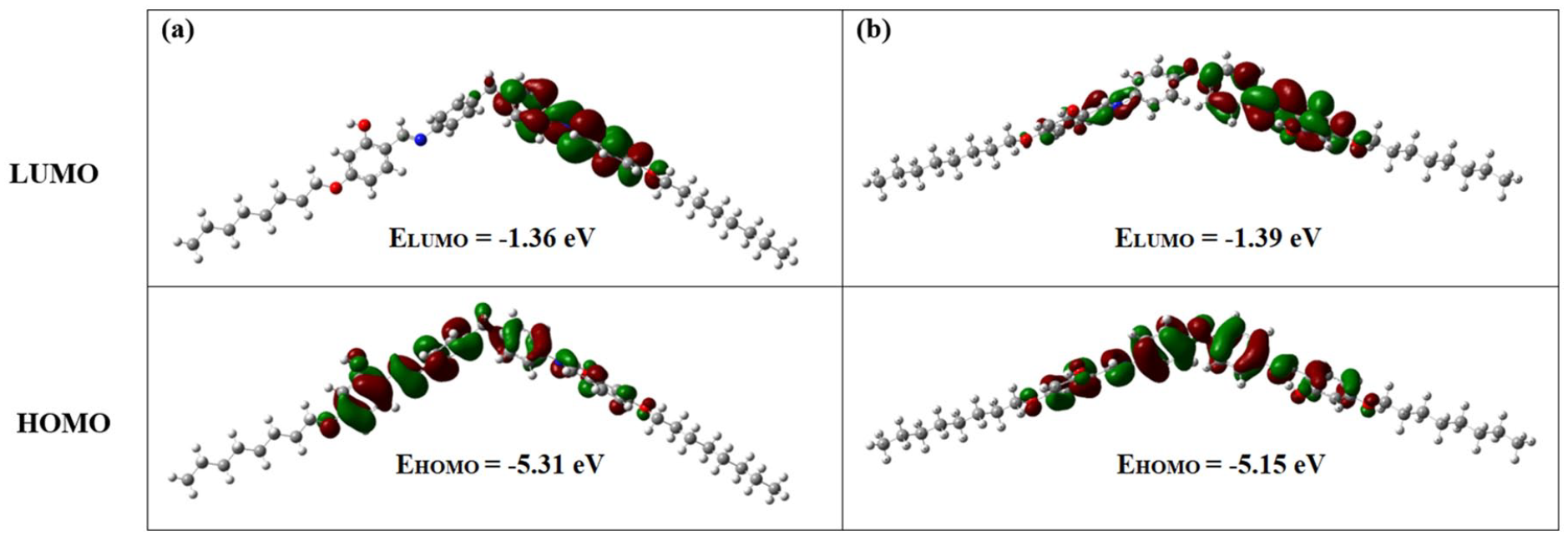
The plots of the frontier molecular orbitals and the HOMO-LUMO energy diagrams for (a)
According to these values, charge transfer interactions may take place in these molecules. Due to the existence of electron-withdrawing azomethine groups and the central linker atoms in the chemical structures of
MEP surface analysis
The chemical reactivity, electronegativity, and structure activity of compounds can be investigated using their MEP which is a method for mapping electrostatic potential on the iso-electron density surface.
29
The MEP surfaces were simulated at the B3LYP/6-31G(d) level for

The MEP surfaces of (a)
The negative electrostatic potential (nucleophilicity) of
Photovoltaic studies
Device fabrication
Initially, all indium tin oxide (ITO)-coated glass substrates were patterned by etching with a mixture of HCl:HNO3:H2O (4.6:0.4:5) for 40 min. Then, the glass substrates were cleaned in an ultrasonic bath with iso-propanol and acetone at each stage for 20 min, respectively. An aqueous solution of poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate; PEDOT:PSS, Clevios PH1000), to be used as the hole transport layer, was deposited on the ITO-coated glass substrates by spin coating at 1000 r min−1 for 45 s and annealed at 120 °C for 30 min under ambient conditions to remove any residual moisture.
Current density–voltage (J–V ) characterization
Figure 5 shows the current density–voltage (

Current density–voltage (
PV data of solar cells employing

PV data: photovoltaic data;
As shown in Figure 5, the devices employing
Figure 6 shows the current density–voltage (

Current density–voltage (
PV data of solar cells employing

FF: fill factor; PV data: photovoltaic data;
As can be seen from Figure 6, P3HT:PCBM-based devices in which
In literature,21,26–32 P3HT:PCBM solar cells exhibit PCEs of 3.5%. However, these efficiencies are achieved for the devices fabricated in a glovebox where the oxygen and humidity effects are omitted and also in which thermal annealing is not applied. 33 In this study, the devices have been fabricated in an ambient atmosphere. On the other hand, partial crystallization of P3HT upon postproduction annealing over 120 °C improves the device performance. Here, a postproduction treatment was not applied to either P3HT:PCBM or ternary blends.
Conclusion
Absorption–emission spectra, HOMO-LUMO energies, mo-lecular structures, and MEP surface properties of
Here, ternary blend systems were studied whose components’ absorption and fluorescence spectra are complementary which in turn may favor an energy transfer. Further investigation will be performed by changing the volume ratio of the blends, the choice of the solvent, and film thicknesses.
Experimental
Materials
2,4-Dihydroxybenzaldehyde,
Instruments and measurements
UV-Vis spectra were recorded using PerkinElmer Lambda 25 UV-Vis spectrometers in CHCI3. The fluorescence spectra were determined using a Jobin Yvon Horiba FluoroMax-P fluorescence spectrophotometer. The IR spectra were determined on a NICOLET iS10 Fourier-transform infrared spectroscopy (FTIR) spectrometer. 1H NMR and 13C NMR spectra were recorded on a Bruker Avance III 500 MHz spectrometer (in CDCl3). Mass spectra (liquid chromatography–mass spectrometry (LC-MS)) were obtained on an Agilent 1200 Infinity HPLC-Agilent 6460 Jet-Stream TripleQuad spectrometer. Melting points were determined with an Electrothermal 9100 Melting Point Apparatus.
The devices were performed on ITO-coated glass substrates with a sheet resistance of 12 Ω cm−2 from Kintec Company, Hong Kong. The ITO was patterned by etching of the glass sheet with the mixture of HCl:HNO3:H2O (4.6:0.4:5) for 30 min. The part of the substrate which forms the contact was covered with a scotch tape to prevent etching. The tape was removed after etching and the substrate was then cleaned by sonication with iso-propanol and acetone. The blends for the active layer with 1:2 wt ratio was prepared by 12.5 mg of P3HT, 6 mg of PCBM, and 5 mg of
Organic bulk heterojunction solar cells were prepared by spin coating the PEDOT:PSS layer on top of the ITO-coated glass surface. The PEDOT:PSS layers were tempered at 140 °C for 10 min. Then, active layers of P3HT:
The current–voltage (
Synthesis and characterization
General experimental procedure for small molecules
The azomethine compounds

Synthesis of new azomethine molecules.
2-Hydroxy-4-octyloxybenzaldehyde (1.0 mmol), which was synthesized by alkylation of 2,4-dihydroxybenzaldehyde with
The compounds were characterized by using UV-Vis, FTIR, 1H NMR, 13C NMR, and LC-MS data. All the spectroscopic data of the target compounds were fully in accordance with their assigned structures.
Footnotes
Acknowledgements
Thanks to Prof. Dr Serap Güneş and Dr Mehmet Kazici from Yıldız Technical University, Department of Physics, and Dr Halil Gokce from Giresun University for their support to photovoltaic characterization and theoretical calculation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
