Abstract
Two new phosphorescent iridium(III) complexes,
Keywords
In this paper, two new 5,7-difluoro-2-phenylbenzothiazole-based iridium(III) complexes have been synthesized and characterized. Their photophysical properties, theoretical calculations, and electrochemical behaviors have been investigated.

Introduction
Due to the favorable optoelectronic properties, cyclometalated iridium(III) complexes have been widely used to fabricate organic light-emitting diodes (OLEDs), especially white light OLEDs.1–4 In general, the realization of white emission requires a combination of either three primary colors (red, green, and blue) or two efficient complementary colors (yellow/orange and blue). During the past decades, red-, green-, and blue-light emitters for full-color display applications have been extensively studied.5–7 However, there is also a great demand for yellow/orange emitters in the two-color white OLEDs (WOLEDs).8–12 Therefore, novel yellow- or orange-emitting iridium complexes with high luminescent quantum yields and excellent device performance are still need to be developed.13–16
Since the first yellow-emitting (bt)2Ir(acac) complex (bt = 2-phenylbenzothiazole) was reported,
17
a number of bt derivatives with different substituents have been developed for OLED applications.16,18–21 In 2004, Laskar and Chen
18
modified the bt ligand by attaching –F and –CF3 groups on the 2-phenyl ring. The resulting complexes, (4-F-bt)2Ir(acac), (3-F-bt)2Ir(acac), and (4-CF3-bt)2Ir(acac), displayed yellow emission in the range of 544–567 nm. In 2011, Wang et al.
16
reported two yellow Ir(III) complexes, (CF3-bt)2Ir(acac) (λmax = 564 nm) and (F-bt)2-Ir(acac) (λmax = 554 nm), by adding –CF3 and –F groups to the benzothiazole ring in the bt ligand. Both complexes displayed moderate quantum efficiencies of 0.36 and 0.32 in CH2Cl2 solution, respectively.16,22 In 2018, we reported a series of yellow-emitting fluorinated bt-based iridium (III) complexes with tetraphenylimidodiphosphinate (tpip) as ancillary ligand.
23
Among them, the quantum yield of 5,7-positions fluorinated bt-based iridium (III) complex [(dfbt)2Ir(tpip)] is the highest one (48.4%). The device based on (dfbt)2Ir(tpip) displays the best device performance with a maximum current efficiency (
In order to further develop new dfbt-based iridium complexes, we tried to use different types of ancillary ligands, such as picolinate (pic) and acetylacetonate (acac). Hence, two corresponding iridium(III) complexes were synthesized (Scheme 1), that is,

Synthetic routes of complexes
Results and discussion
The UV-Vis absorption and photoluminescence (PL) emission spectra of
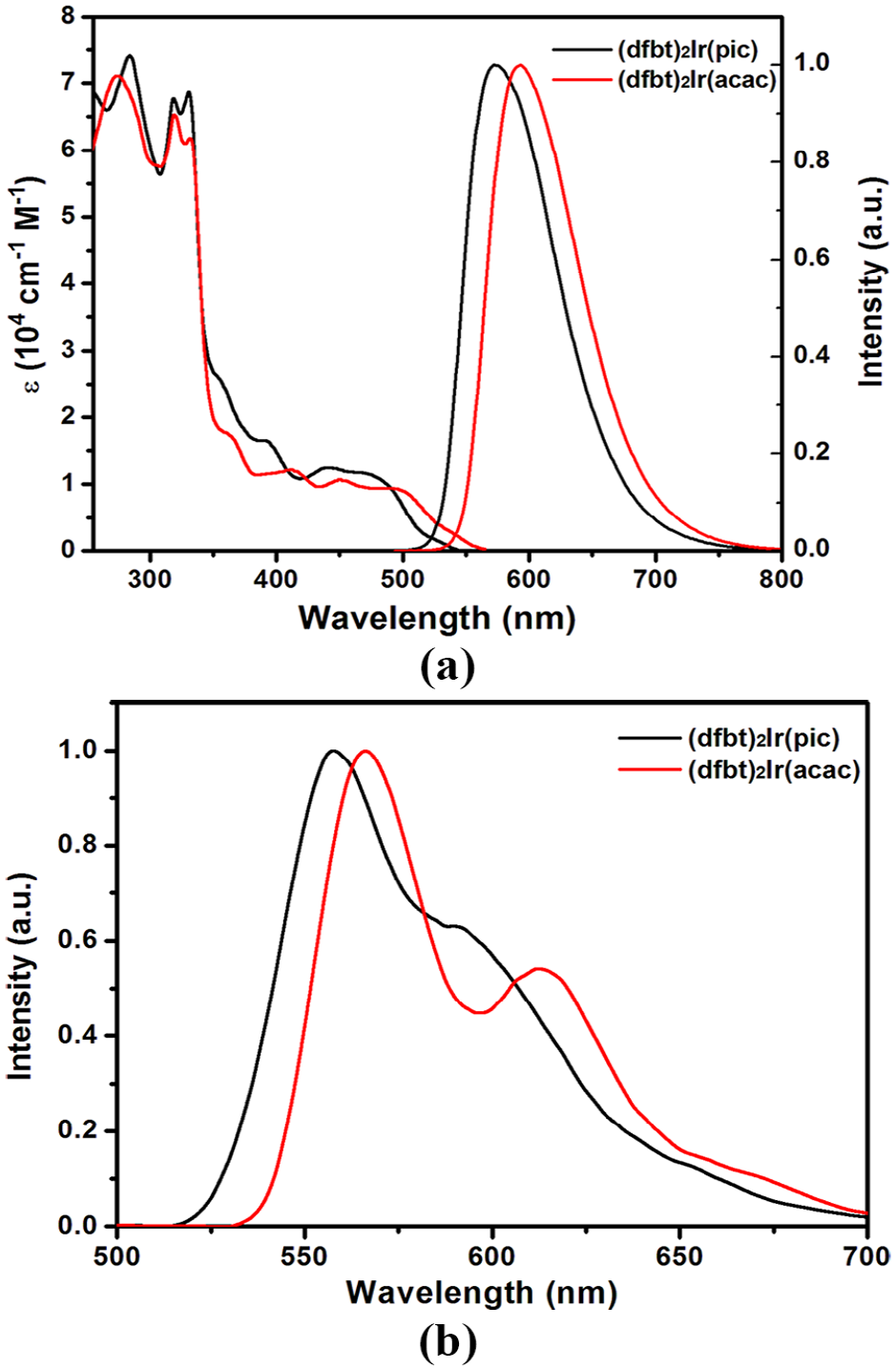
(a) UV-Vis absorption and PL emission spectra of
Photophysical data of complexes
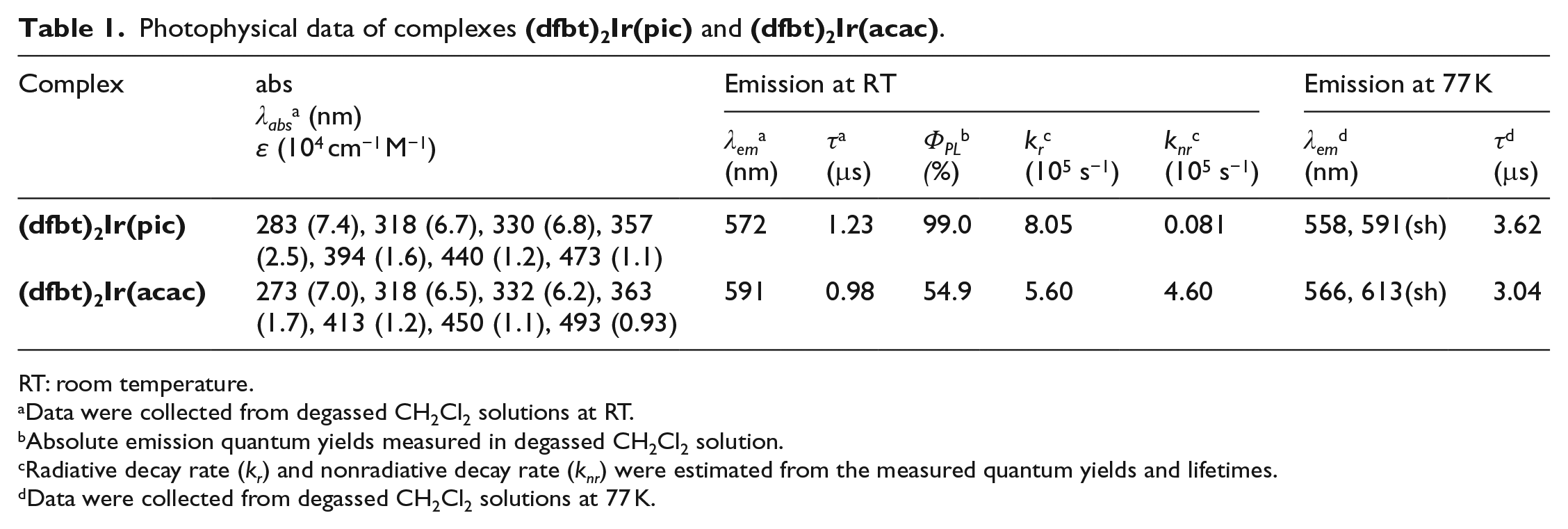
RT: room temperature.
Data were collected from degassed CH2Cl2 solutions at RT.
Absolute emission quantum yields measured in degassed CH2Cl2 solution.
Radiative decay rate (
Data were collected from degassed CH2Cl2 solutions at 77 K.
The emission spectra of the two Ir(III) complexes at RT (Figure 1(a)) show a broad band covering a range from 572 (full width at half maximum (FWHM) = 84 nm) to 591 nm (FWHM = 83 nm). Their emission colors are tuned from yellow [CIE (0.58, 0.42)] to orange [CIE (0.52, 0.48)] through the change of ancillary ligands. Moreover, their RT emission profiles are featureless, indicating that a 3MLCT excited-state emission is dominant. Upon cooling to 77 K, their low-temperature PL spectra in CH2Cl2 solution undergo a pronounced blue shift and display a well-resolved vibronic structure (Figure 1(b)), originating from the mixing between the 3MLCT and 3LC emission. These results suggest that the ligands should have significant contributions to the lowest lying excited stated. This speculation will be confirmed by DFT calculations below.
To investigate the effect of the ancillary ligand, the absolute photoluminescent quantum yields (
To deeply understand the photophysical properties of

Some frontier molecular orbitals of complexes
Contributions, oscillator strengths and transition characters of singlet and triplet transitions for complexes

HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital; MLCT: metal-to-ligand charge transfer; LC: ligand-centered.
Frontier orbital energy and electron density distribution for complexes

HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital.
The HOMOs are principally distributed on the iridium atom and phenyl moiety of cyclometalated ligands, whereas the LUMOs are primarily located on the whole cyclometalated ligands (Table 3). Therefore, the calculated lowest lying singlet and triplet transitions of
Through the DFT calculation, we found that the triplet energy of ancillary ligand (pic = 3.399 eV, acac = 3.548 eV) is higher than those of the cyclometalated ligand (dfbt = 2.758 eV) and the complexes ((dfbt)2Ir(pic) = 2.419 eV, (dfbt)2Ir(acac) = 2.387 eV). Thus, both complexes give emission from an excited state on the “dfbt2Ir” fragment, supporting the assignment of the lowest energy excited state. Meanwhile, we also calculated the triplet energies of bt ligand (2.731 eV) and the complexes ((bt)2Ir(pic) = 2.456 eV, (bt)2Ir(acac) = 2.425 eV). Likewise, the two complexes emit from states within the “C^N2Ir” fragment as discussed above.
The electrochemical properties of both Ir(III) complexes have been investigated by cyclic voltammetry (CV) using ferrocene as the internal standard (Fc+/Fc = +0.38 V vs SCE (saturated calomel electrode) in acetonitrile)
30
(Figure 3) and their data are listed in Table 4. For both the complexes, the first irreversible oxidation waves (0.79 V for

Cyclic voltammograms for complexes
Electrochemical and theoretical data of complexes

HOMO: HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital.
Redox measurements were carried out in CH3CN solution, and values are reported to Fc+/Fc.
Deduced from the equation
Obtained from theoretical calculations.
In conclusion, we report herein on the synthesis and photophysical properties of two new dfbt-based iridium(III) complexes,
Experimental
All reactions were performed under nitrogen atmosphere. All of the reagents were purchased from commercial sources and used without further purification, unless stated otherwise. 5,7-Difluoro-2-phenylbenzo[d]thiazole (dfbt) and the corresponding dichloro-bridged [Ir(dfbt)2Cl]2 were synthesized according to our previous literature. 23
1H NMR spectra were recorded on a Bruker AM 400 MHz instrument, and chemical shifts were reported in ppm relative to Me4Si as internal standard. MALDI-TOF-MS spectra were recorded on a Bruker Autoflex IITM TOF/TOF instrument. The elemental analyses were measured on a Vario EL Cube Analyzer system. UV-Vis spectra were recorded using a Hitachi U3900/3900H spectrophotometer. Fluorescence spectra were carried out on a Hitachi F-7000 spectrophotometer in degassed CH2Cl2 solutions at RT and 77 K. Luminescence lifetime curves were measured on a HORIBA FL-3 fluorescence spectrometer under 375 nm excitation of a Delta-Diode laser. The absolute PL quantum yields (
CV was performed on a CHI 1210B electrochemical workstation, with a glassy carbon electrode as the working electrode, a platinum wire as the counter electrode, an Ag/AgCl electrode as the reference electrode, and 0.1 M
The geometry of complexes
General procedure for the preparation of the Ir(III) complexes (dfbt)2Ir(pic) and (dfbt)2Ir(acac)
A mixture of the above dichloro-bridged dimer complex [Ir(dfbt)2Cl]2, the corresponding ancillary ligand (picolinic acid or pentane-2,4-dione, 2.5 equiv.), and Na2CO3 (5.0 equiv.) was dissolved in ethoxyethanol (6 mL) and was refluxed for 12 h under nitrogen. After cooled to RT, the mixture was diluted with water and extracted multiple times with DCM. The combined organic phases were washed with brine, dried over Na2SO4. After concentrated, the residue was purified by column chromatography with petroleum ether/DCM (5:1) eluent to afford pure Ir(III) complex.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Natural Science Foundation of Hainan Province (Nos. 219MS043 and 218QN236).
