Abstract
Cucurbit[8]uril (Q[8]) was used as the building block for the facile preparation of Q[8]-based supramolecular assemblies in aqueous HCl solutions (
Introduction
Cucurbit[n]urils (Q[n]s, n = 5–8, 10) are pumpkin-shaped macrocycles, each consisting of a rigid hydrophobic cavity and two open polar portals lined with carbonyl groups. Recently, our group described the facile preparation of Q[10]-based porous supramolecular assemblies by simply mixing two aqueous HCl solutions containing Q[10] and CdCl2, respectively. These Q[10]-based assemblies exhibited novel adsorption capacities for certain dyes, and the solid dye@Q[10] assembly displayed intense fluorescence emission. 1 Over a decade ago, Kim and co-workers described a simple Q[6]-based supramolecular assembly with honeycomb-like channels, permanent porosity, exceptional stability and special capacities for capturing acetylene molecules 2 and carbon dioxide. 3 We now know that the outer surface interaction of cucurbit[n]urils, Q[n]s, which is attributed to the positive electro-potential outer surface of Q[n]s, results in the formation of these novel Q[n]-based porous supramolecular assemblies. 4 A new field of Q[n]-based supramolecular chemistry in fields of relevance to porous functional materials and light emitters that can be used for detection of explosives, chemical agents, pesticides and other applications has been created. 5
It is well known that Q[n]s with an even number of units, such as Q[6], Q[8] and Q[10], are non-functionalized and barely dissolve in neutral water, whereas Q[n]s with odd numbers, such as Q[5] and Q[7], are water soluble. Compared with Q[n]s with odd numbers, Q[n]s with even numbers can more rapidly yield solid Q[n]-based supramolecular assemblies in the absence or presence of inorganic anions or polyaromatic compounds as structure-directing agents.6–8 For example, we found that Q[10] interacts with metal ions to form one-dimensional coordination polymers in aqueous HCl solutions, which are converted into metal-free Q[10]-based two-dimensional supramolecular assemblies through the outer surface interaction of Q[10]s in aqueous HNO3 solutions, making Q[10]-based systems suitable as metal-selective materials. 9 We also synthesized a series of Q[8]-based porous supramolecular assemblies under different conditions. Among these supramolecular assemblies, two compounds with similar structural features were prepared. One was generated from aqueous HCl solution in the presence of LnCl3 and CdCl2, which could form [CdCl4]2– anions in aqueous HCl solution (Figure S1a in the Supporting Information), 10 and the other was generated by crystallization of a ternary mixture of Q[8], CsCl and p-hydroxybenzoic acid in aqueous HCl solution (Figure S1b in the Supporting Information). 11
Ions such as depronated p-hydroxybenzoic acid and [CdCl4]2– are so-called structure-directing agents that promote the formation of Q[8]-based three-dimensional (3D) frameworks. However, our recent work revealed that a simple Q[8]-based supramolecular assembly could be obtained from aqueous HCl solutions without using any structure inducer, and the Q[8]-based assembly possesses similar structural features to the two aforementioned Q[8]-based 3D frameworks (Figure S1c in the Supporting Information). Further experiments revealed that all precipitates from HCl solutions, whether microcrystals or single crystals, shared the same structural features, suggesting that the formation of simple Q[8]-based 3D frameworks reflected the intrinsic attributes of the Q[8] molecule.
In the present work, the structural features of this simple Q[8]-based supramolecular assembly (
Experimental section
Materials
Q[8] was prepared in our laboratory according to a literature method.12,13 All other chemicals were of analytical grade, obtained from Aladdin and used as received without further purification.
Preparation of Q[8]-based supramolecular assembly A
Solid Q[8] was dissolved in aqueous HCl solutions (3–8 M). Colourless crystals of
Measurements
All 1H NMR spectra, including those for titration experiments, were recorded at 20 °C in D2O on a JEOL JNM-ECZ400s spectrometer.
Single-crystal determination
A suitable single crystal (~0.2 × 0.2 × 0.1 mm3) was coated with paraffin oil and mounted on a Bruker SMART Apex II CCD diffractometer equipped with a graphite-monochromated Mo-Kα radiation source (λ = 0.71073 Å,
μ
= 0.828 mm−1) operating in the ω-scan mode and fitted with a nitrogen cold stream (–30 °C). Data were corrected for Lorentz and polarization effects using SAINT, and semi-empirical adsorption corrections based on equivalent reflections were also applied using SADABS. The structure was elucidated by direct methods and refined by the full-matrix least-squares method using F2 values with the SHELXS-97 and SHELXL-97 packages, respectively.14,15 All non-hydrogen atoms were refined anisotropically. Carbon-bound hydrogen atoms were introduced at calculated positions and were treated as riding atoms with an isotropic displacement parameter equal to 1.2 times that of the parent atom. Most of the water molecules in compound
Preparation of FG@A solid-state fluorescent materials
A 1.0 g sample of Q[8]-based
Adsorption and response studies of FG@A with VOC s
The required solid assembly (0.5–1.0 g) contained in a tared open glass vial was added to a sealed glass vessel, which was evacuated using a vacuum pump. Pumping was continued until the sample achieved a constant weight. A second open container containing few millilitre of the volatile liquid (methanol, ethanol, diethyl ether, acetonitrile, acetone, dichloromethane, trichloromethane, or tetrachloromethane) was then added and the vessel resealed. The weight change was measured, and the corresponding solid fluorescence spectra were determined at ~1 to 60 min intervals over several hours to obtain the vapour adsorption profiles.
Measurement of fluorescence spectra of solid FG@A assemblies
Fluorescence spectra of solid
Results and discussion
Description of the crystal structures of compound A
Q[8]-based supramolecular assemblies can be formed easily from Q[8] molecules, or in the presence of inorganic anions or aromatic compounds through the outer surface interaction of cucurbit[n]urils under different conditions.10,11,16–19 In the present work, a simple Q[8]-based supramolecular assembly was generated from aqueous HCl solutions (

Crystal structures of
Powder X-ray diffraction analyses of
Adsorption properties of Q[8]-based porous supramolecular assemblies
Q[n]-based supramolecular assemblies present adsorbed various dyes and polyaromatic compounds to form solid fluorescent materials.1,18 In the present case, different channels and spaces were observed in the Q[8]-based porous supramolecular assembly (
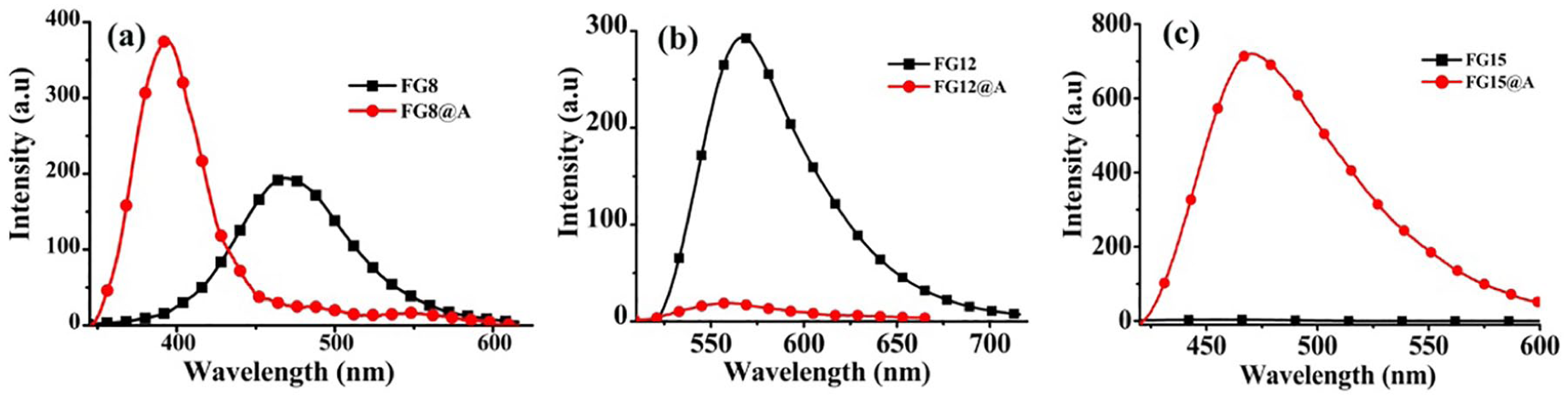
Comparison of the fluorescence spectra of (a)
Previous work showed that Q[n]-based supramolecular assemblies could adsorb various

Fluorescence spectra of (a)
We have noted that among these
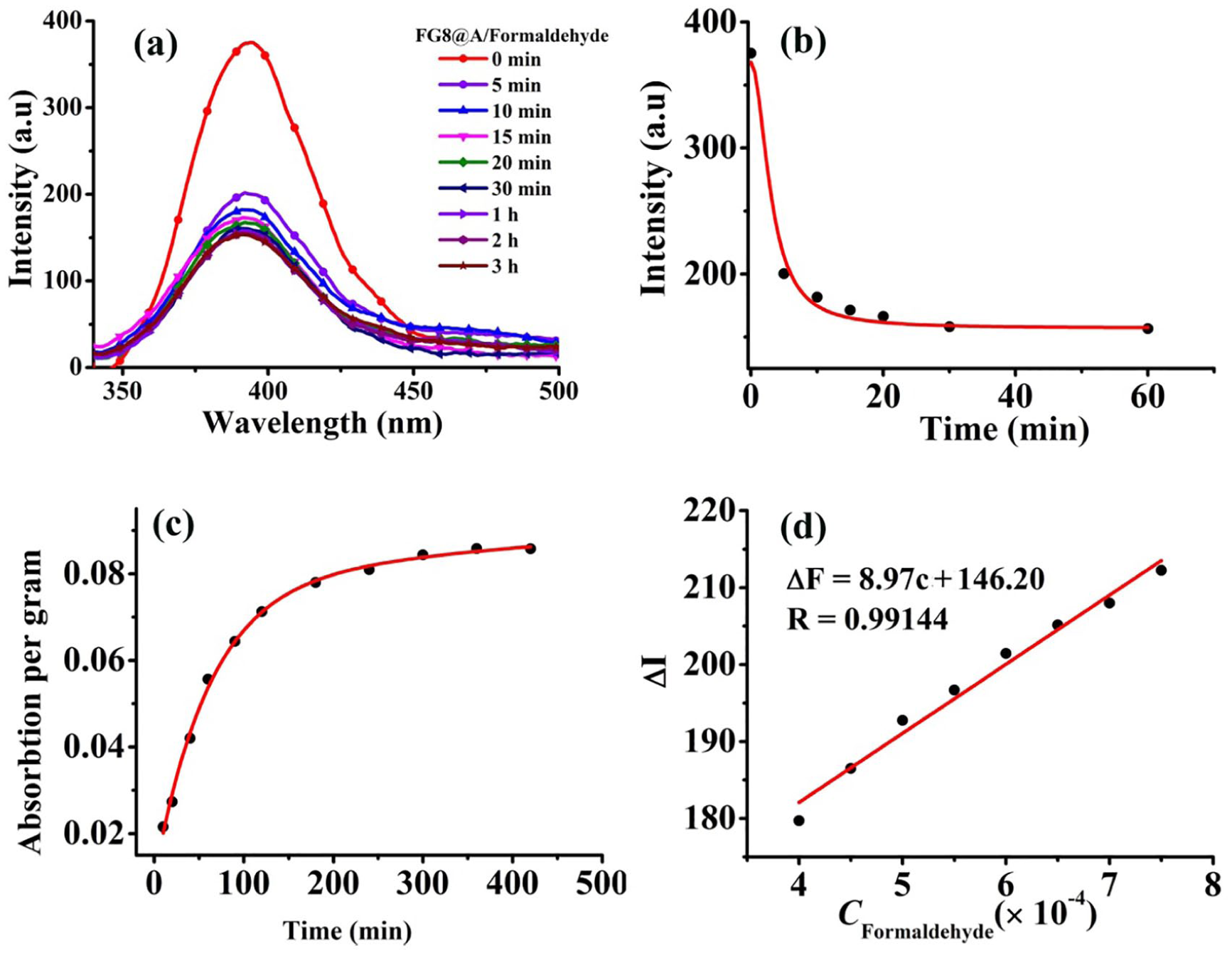
(a) Titration fluorescence spectra of the loading of
On the other hand, the titration fluorescent spectra of solid

(a) Titration fluorescence spectra of the loading of
In general, a change in the fluorescence emission of a
Out of curiosity, we investigated the adsorption of a neat
Conclusion
In summary, we selected Q[8] as a building block and prepared a Q[8]-based porous supramolecular assembly by simple crystallization in aqueous HCl solutions (
Supplemental Material
Supplementary_File-CHL-19-0072 – Supplemental material for Preparation and adsorption properties of a facile solid cucurbit[8]uril-based porous supramolecular assembly
Supplemental material, Supplementary_File-CHL-19-0072 for Preparation and adsorption properties of a facile solid cucurbit[8]uril-based porous supramolecular assembly by Chunchun Chen, Weiwei Ge, Kai Chen, Zhu Tao, Yunqian Zhang and Qian Jiang Zhu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant nos 21601090, 51663005 and 21761007) and the Natural Science Foundation of Jiangsu Province (grant no. BK20160943) and also supported by the Start-up Foundation of Nanjing University of Information Science & Technology (no. 2015r047).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
