All commercially available reagents were used as received unless otherwise stated. Reactions were monitored by thin layer chromatography (TLC) and visualized under UV light (254 nm). Melting points were determined using a Büchi B-540 capillary melting point apparatus. Nuclear magnetic resonance (NMR) spectra were recorded on Varian spectrometer (400 MHz) with CDCl3 as the solvent and tetramethylsilane (TMS) as an internal standard. High-resolution mass spectra (HRMS) were recorded on an electrospray ionization quadrupole time-of-flight (ESI-Q-TOF) System ESI spectrometer.
General procedure for the synthesis of 3
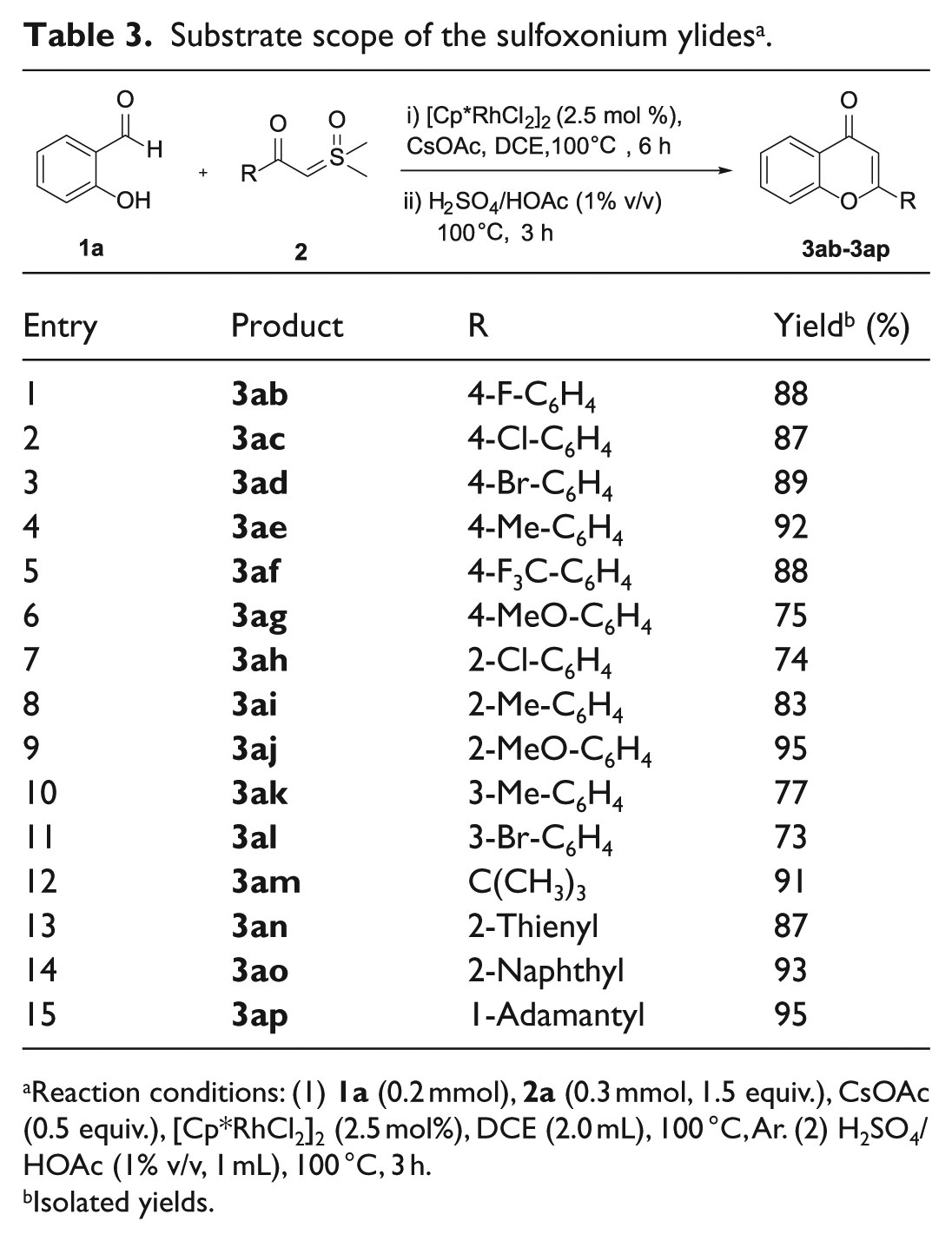
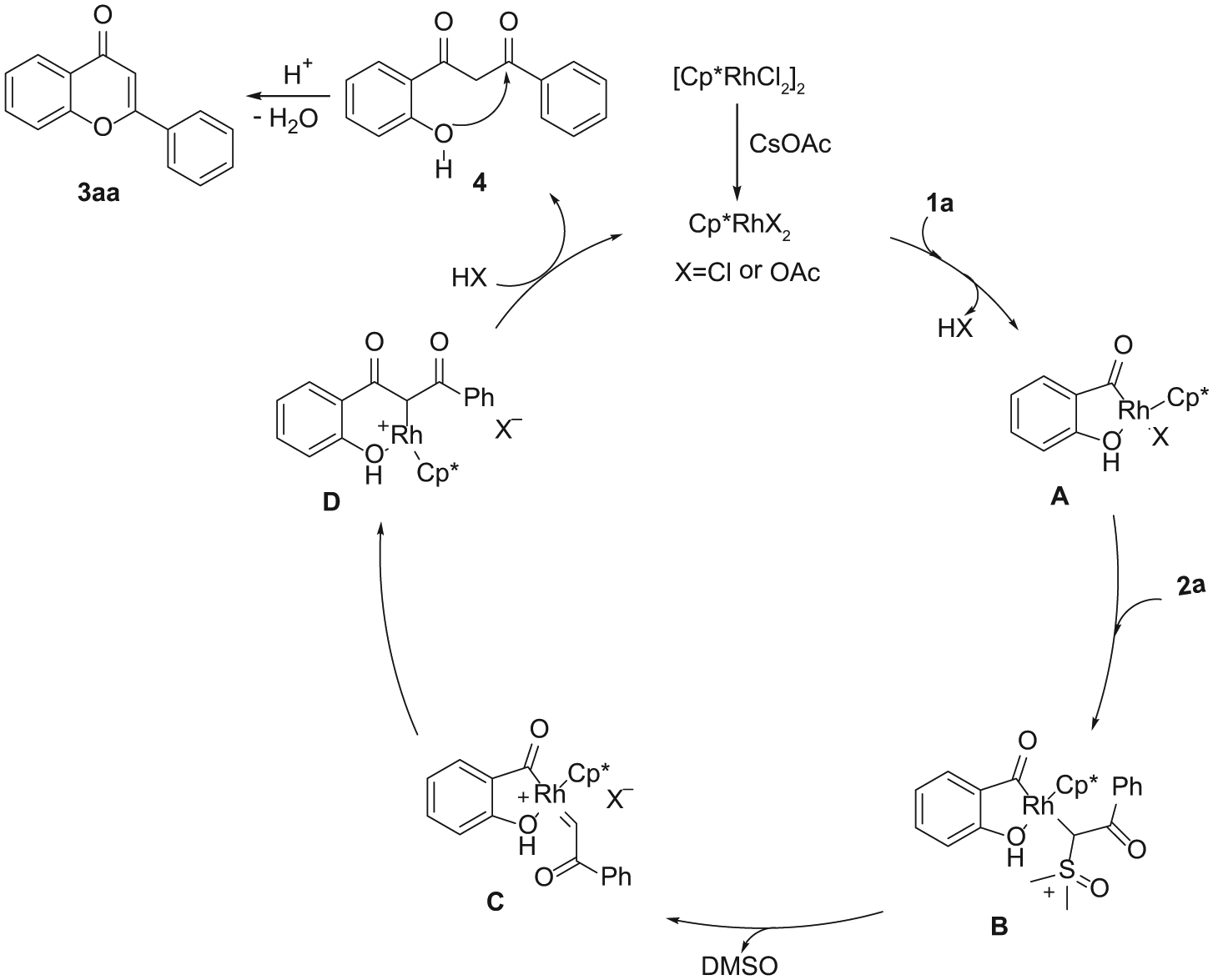
A sealed tube was charged with 1a (0.2 mmol, 1.0 equiv.), 2a (0.3 mmol, 1.5 equiv.), [Cp*RhCl2]2 (0.005 mmol, 2.5 mol%), and CsOAc (0.1 mmol, 0.5 equiv.) in DCE (2 mL). After the reaction mixture had been stirred at 100 °C under Ar for 6 h, 1 mL of H2SO4/HOAc (10 μL of H2SO4 dissolved in 0.99 mL of HOAc) was added at room temperature. After completion of the addition, the reaction mixture was then stirred at 100 °C for 3 h before being cooled to room temperature. The mixture was diluted with EtOAc (20 mL), washed with brine, and dried over anhydrous Na2SO4. After removal of the EtOAc, the residue was purified by chromatography on basic silica gel (PE/EtOAc = 8/1) to afford 3aa (40 mg, 89%) as a white solid.
2-Phenyl-4H-chromen-4-one (3aa): Product 3aa was obtained as a white solid (40 mg, 89%); m.p.: 96–98 °C. 1H NMR (400 MHz, CDCl3): δ = 8.24 (1H, d, J = 8.0 Hz, CH), 7.97–7.89 (2H, m, 2CH), 7.71 (1H, t, J = 7.8 Hz, CH), 7.61–7.49 (4H, m, 4CH), 7.43 (1H, t, J = 7.6 Hz, CH), 6.84 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.48 (Cq), 163.40 (Cq), 156.24 (Cq), 133.77 (CH), 131.75 (Cq), 131.59 (CH), 129.03 (CH), 126.28 (CH), 125.69 (CH), 125.22 (CH), 123.95 (Cq), 118.07 (CH), 107.58 (CH). HRMS (ESI): m/z calcd for C15H11O2 [M + H]+: 223.0754; found: 223.0743.
6-Fluoro-2-phenyl-4H-chromen-4-one (3ba): Product 3ba was obtained as a yellow solid (45 mg, 93%); m.p.: 105–107 °C. 1H NMR (400 MHz, CDCl3): δ = 7.96–7.83 (3H, m, 3CH), 7.63–7.50 (4H, m, 4CH), 7.43 (1H, t, J = 8.0 Hz, CH), 6.82 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 177.63 (Cq), 163.69 (Cq), 160.81 (Cq), 158.35 (Cq), 152.45 (CH), 131.65 (d, 2JC–F = 28.2 Hz, Cq), 129.09 (CH), 126.31 (CH), 125.16 (d, 3JC–F = 7.2 Hz, Cq), 121.91 (d, 2JC–F = 25.6 Hz, CH), 120.16 (d, 3JC–F = 8.0 Hz, CH), 110.64 (d, 2JC–F = 23.8 Hz, CH), 106.90 (CH). HRMS (ESI): m/z calcd for C15H9FNaO2 [M + Na]+: 263.0479; found: 263.0473.
6-Chloro-2-phenyl-4H-chromen-4-one (3ca): Product 3ca was obtained as a yellow solid (48 mg, 94%); m.p.: 178–180 °C. 1H NMR (400 MHz, CDCl3): δ = 8.19 (1H, d, J = 2.4 Hz, CH), 7.97–7.84 (2H, m, 2CH), 7.64 (1H, dd, J = 9.0, 2.4 Hz, CH), 7.58–7.50 (4H, m, 4CH), 6.83 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 177.19 (Cq), 163.65 (Cq), 154.53 (Cq), 133.93 (CH), 131.84 (Cq), 131.36 (CH), 131.17 (Cq), 129.09 (CH), 126.29 (CH), 125.15 (Cq), 124.91 (CH), 119.79 (CH), 107.46 (CH). HRMS (ESI): m/z calcd for C15H9ClNaO2 [M + Na]+: 279.0183; found: 279.0175.
6-Bromo-2-phenyl-4H-chromen-4-one (3da): Product 3da was obtained as a yellow solid (55 mg, 92%); m.p.: 192 –194 °C. 1H NMR (400 MHz, CDCl3): δ= 8.35 (1H, d, J = 2.4 Hz, CH), 7.91 (2H, dd, J = 8.0, 1.8 Hz, 2CH), 7.78 (1H, dd, J = 9.0, 2.6 Hz, CH), 7.58–7.50 (3H, m, 3CH), 7.47 (1H, d, J = 9.0 Hz, CH), 6.83 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ= 177.04 (Cq), 163.65 (Cq), 154.96 (Cq), 136.69 (CH), 131.85 (CH), 131.32 (Cq), 129.08 (CH), 128.33 (CH), 126.29 (CH), 125.27 (Cq), 120.01 (CH), 118.64 (Cq), 107.52 (CH). HRMS (ESI): m/z calcd for C15H9BrNaO2 [M + Na]+: 322.9678; found: 322.9686.
6-Methyl-2-phenyl-4H-chromen-4-one (3ea): Product 3ea was obtained as a yellow solid (38 mg, 81%); m.p.: 124–126 °C. 1H NMR (400 MHz, CDCl3): δ = 8.02 (1H, s, CH), 7.97–7.89 (2H, m, 2CH), 7.59–7.43 (5H, m, 5CH), 6.82 (1H, s, CH), 2.47 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.62 (Cq), 163.24 (Cq), 154.53 (Cq), 135.19 (CH), 134.98 (Cq), 131.88 (CH), 131.48 (Cq), 128.99 (CH), 126.25 (CH), 125.03 (CH), 123.62 (Cq), 117.82 (CH), 107.44 (CH), 20.94 (CH3). HRMS (ESI): m/z calcd for C16H12O2 [M + H]+: 237.0910; found: 237.0903.
6-Nitro-2-phenyl-4H-chromen-4-one (3fa): Product 3fa was obtained as a yellow solid (19 mg, 36%); m.p.: 195–197 °C. 1H NMR (400 MHz, CDCl3): δ = 9.11 (1H, d, J = 2.8 Hz, CH), 8.55 (1H, dd, J = 9.0, 2.8 Hz, CH), 7.94 (2H, dd, J = 8.0, 1.4 Hz, 2CH), 7.74 (1H, d, J = 9.0 Hz, CH), 7.67–7.51 (3H, m, 3CH), 6.90 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 176.64 (Cq), 164.10 (Cq), 159.03 (Cq), 144.80 (Cq), 132.35 (CH), 130.72 (Cq), 129.26 (CH), 128.11 (CH), 126.41 (CH), 124.05 (CH), 122.48 (Cq), 119.81 (CH), 107.83 (CH). HRMS (ESI): m/z calcd for C15H10NO4 [M + H]+: 268.0604; found: 268.0608.
6-Methoxy-2-phenyl-4H-chromen-4-one (3ga): Product 3ga was obtained as a yellow solid (47 mg, 93%); m.p.: 161–163 °C. 1H NMR (400 MHz, CDCl3): δ = 7.97–7.85 (2H, m, 2CH), 7.60 (1H, d, J = 3.0 Hz, CH), 7.55–7.48 (4H, m, 4CH), 7.30 (1H, dd, J = 9.0, 3.0 Hz, CH), 6.83 (1H, s, CH), 3.91 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.41 (Cq), 163.12 (Cq), 156.97 (Cq), 151.06 (Cq), 131.84 (Cq), 131.46 (CH), 128.99 (CH), 126.21 (CH), 124.64 (Cq), 123.80 (CH), 119.49 (CH), 106.86 (CH), 104.78 (CH), 55.92 (CH3). HRMS (ESI): m/z calcd for C16H13O3 [M + H]+: 253.0859; found: 253.0849.
7-Chloro-2-phenyl-4H-chromen-4-one (3ha): Product 3ha was obtained as a yellow solid (45 mg, 88%); m.p.: 159–161 °C. 1H NMR (400 MHz, CDCl3): δ = 8.17 (1H, d, J = 8.6 Hz, CH), 7.90 (2H, d, J = 6.6 Hz, 2CH), 7.61 (1H, s, CH), 7.54 (3H, d, J = 6.8 Hz, 3CH), 7.39 (1H, d, J = 8.2 Hz, CH), 6.82 (1H, s, CH).
13
C NMR (101 MHz, CDCl3): δ = 177.94 (Cq), 163.51 (Cq), 156.34 (Cq), 139.75 (Cq), 131.82 (Cq), 131.32 (CH), 129.09 (CH), 127.06 (CH), 126.26 (CH), 126.06 (CH), 122.83 (Cq), 118.17 (CH), 107.87 (CH). HRMS (ESI): m/z calcd for C15H9ClNaO2 [M + Na]+: 279.0183; found: 279.0174.
7-Methyl-2-phenyl-4H-chromen-4-one (3ia): Product 3ia was obtained as a yellow solid (38 mg, 81%); m.p.: 124–126 °C. 1H NMR (400 MHz, CDCl3): δ = 8.11 (1H, d, J = 8.0 Hz, CH), 7.96–7.87 (2H, m, 2CH), 7.52 (3H, d, J = 4.0 Hz, 3CH), 7.38 (1H, s, CH), 7.23 (1H, d, J = 8.0 Hz, CH), 6.80 (1H, s, CH), 2.51 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.42 (Cq), 163.10 (Cq), 156.37 (Cq), 145.10 (Cq), 131.87 (Cq), 131.46 (CH), 128.99 (CH), 126.70 (CH), 126.21 (CH), 125.41 (CH), 121.68 (CH), 117.83 (Cq), 107.50 (CH), 21.84 (CH3). HRMS (ESI): m/z calcd for C16H12NaO2 [M + Na]+: 259.0730; found: 259.0724.
8-Bromo-2-phenyl-4H-chromen-4-one (3ja): Product 3ja was obtained as a yellow solid (44 mg, 73%); m.p.: 174–176 °C. 1H NMR (400 MHz, CDCl3): δ = 8.18 (1H, d, J = 8.8 Hz, CH), 8.05–8.00 (2H, m, 2CH), 7.92 (1H, d, J = 8.4 Hz, CH), 7.60–7.51 (3H, m, 3CH), 7.30 (1H, t, J = 7.8 Hz, CH), 6.88 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 177.84 (Cq), 163.34 (Cq), 152.73 (Cq), 137.14 (CH), 131.95 (Cq), 131.20 (CH), 129.16 (CH), 126.46 (CH), 125.82 (CH), 125.32 (CH), 125.07 (Cq), 111.98 (Cq), 107.19 (CH). HRMS (ESI): m/z calcd for C15H9BrNaO2 [M + Na]+: 322.9678; found: 322.9669.
8-Methyl-2-phenyl-4H-chromen-4-one (3ka): Product 3ka was obtained as a yellow solid (42 mg, 89%); m.p.: 164–166 °C. 1H NMR (400 MHz, CDCl3): δ = 8.07 (1H, d, J = 7.8 Hz, CH), 7.98–7.90 (2H, m, 2CH), 7.57–7.49 (4H, m, 4CH), 7.30 (1H, t, J = 7.6 Hz, CH), 6.84 (1H, s, CH), 2.61 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.85 (Cq), 162.86 (Cq), 154.66 (Cq), 134.69 (Cq), 132.01 (Cq), 131.53 (CH), 129.07 (CH), 127.49 (CH), 126.16 (CH), 124.73 (CH), 123.82 (CH), 123.28 (Cq), 107.29 (CH), 15.83 (CH3). HRMS (ESI): m/z calcd for C16H12NaO2 [M + Na]+: 259.0730; found: 259.0720.
8-Methoxy-2-phenyl-4H-chromen-4-one (3la): Product 3la was obtained as a yellow solid (46 mg, 93%); m.p.: 197–199 °C. 1H NMR (400 MHz, CDCl3): δ = 7.99–7.95 (2H, m, 2CH), 7.78 (1H, dd, J = 8.0, 1.0 Hz, CH), 7.54–7.50 (3H, m, 3CH), 7.33 (1H, t, J = 8.0 Hz, CH), 7.22–7.15 (1H, m, CH), 6.85 (1H, s, CH), 4.03 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.46 (Cq), 163.01 (Cq), 149.09 (Cq), 146.62 (Cq), 131.79 (Cq), 131.54 (CH), 129.00 (CH), 126.33 (CH), 124.91 (CH), 124.81 (Cq), 116.39 (CH), 114.40 (CH), 107.31 (CH), 56.35 (CH3). HRMS (ESI): m/z calcd for C16H12NaO3 [M + Na]+: 275.0679; found: 275.0672.
6-Phenyl-8H-[1,3]dioxolo[4,5-g]chromen-8-one (3ma): Product 3ma was obtained as a yellow solid (34 mg, 64%); m.p.: 207–209 °C. 1H NMR (400 MHz, CDCl3): δ 7.90–7.86 (2H, m, 2CH), 7.60–7.46 (4H, m, 4CH), 6.97 (1H, s, CH), 6.77 (1H, s, CH), 6.12 (2H, s, CH2). 13C NMR (101 MHz, CDCl3): δ = 177.41 (Cq), 162.70 (Cq), 153.49 (Cq), 152.76 (Cq), 146.18 (Cq), 131.71 (Cq), 131.34 (CH), 128.99 (CH), 126.04 (CH), 118.98 (Cq), 106.98 (CH), 102.42 (CH), 102.31 (CH2), 98.04 (CH). HRMS (ESI): m/z calcd for C16H11O4 [M + H]+: 267.0652; found: 267.0655.
2-(4-Fluorophenyl)-4H-chromen-4-one (3ab): Product 3ab was obtained as a yellow solid (42 mg, 88%); m.p.: 145–147 °C. 1H NMR (400 MHz, CDCl3): δ = 8.31–8.16 (1H, m, CH), 7.97–7.91 (2H, m, 2CH), 7.78–7.67 (1H, m, CH), 7.57 (1H, d, J = 8.4 Hz, CH), 7.43 (1H, t, J = 7.6 Hz, CH), 7.22 (2H, t, J = 8.6 Hz, 2CH), 6.78 (1H, s, CH).
13
C NMR (101 MHz, CDCl3): δ = 178.30 (Cq), 164.73 (d, 1JC–F = 253.2 Hz, Cq), 162.37 (Cq), 156.15 (Cq), 133.82 (CH), 128.48 (d, 3JC–F = 9.0 Hz, CH), 127.96 (d, 4JC–F = 3.2 Hz, Cq), 125.72 (CH), 125.30 (CH), 123.87 (Cq), 117.99 (CH), 116.28 (d, 2JC–F = 22.0 Hz, CH), 107.36 (CH). HRMS (ESI): m/z calcd for C15H10FO2 [M + H]+: 241.0659; found: 241.0662.
2-(4-Chlorophenyl)-4H-chromen-4-one (3ac): Product 3ac was obtained as a yellow solid (45 mg, 87%); m.p.: 185–187 °C. 1H NMR (400 MHz, CDCl3): δ = 8.23 (1H, dd, J = 8.0, 1.4 Hz, CH), 7.87 (2H, d, J = 8.6 Hz, 2CH), 7.75–7.67 (1H, m, CH), 7.56 (1H, d, J = 8.4 Hz, CH), 7.50 (2H, d, J = 8.6 Hz, 2CH), 7.43 (1H, t, J = 7.6 Hz, CH), 6.80 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.23 (Cq), 162.17 (Cq), 156.12 (Cq), 137.85 (Cq), 133.89 (CH), 130.20 (CH), 129.35 (CH), 127.51 (Cq), 125.71 (CH), 125.35 (CH), 123.87 (Cq), 118.01 (CH), 107.66 (CH). HRMS (ESI): m/z calcd for C15H10ClO2 [M + H]+: 257.0364; found: 257.0368.
2-(4-Bromophenyl)-4H-chromen-4-one (3ad): Product 3ad was obtained as a yellow solid (54 mg, 89%); m.p.: 176–178 °C. 1H NMR (400 MHz, CDCl3): δ = 8.29–8.18 (1H, m, CH), 7.80 (2H, d, J = 8.6 Hz, 2CH), 7.74–7.69 (1H, m, CH), 7.67 (2H, d, J = 8.6 Hz, 2CH), 7.57 (1H, d, J = 8.2 Hz, CH), 7.43 (1H, t, J = 7.6 Hz, CH), 6.81 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.28 (Cq), 162.25 (Cq), 156.13 (Cq), 133.91 (CH), 132.33 (CH), 130.67 (CH), 127.68 (Cq), 126.29 (Cq), 125.72 (CH), 125.37 (CH), 123.94 (Cq), 118.03 (CH), 107.70 (CH). HRMS (ESI): m/z calcd for C15H10BrO2 [M + H]+: 300.9859; found: 300.9867.
2-(p-Tolyl)-4H-chromen-4-one (3ae): Product 3ae was obtained as a yellow solid (43 mg, 92%); m.p.: 109–111 °C. 1H NMR (400 MHz, CDCl3): δ = 8.23 (1H, d, J = 8.0 Hz, CH), 7.82 (2H, d, J = 8.0 Hz, 2CH), 7.69 (1H, t, J = 7.8 Hz, CH), 7.56 (1H, d, J = 8.4 Hz, CH), 7.41 (1H, t, J = 7.6 Hz, CH), 7.32 (2H, d, J = 8.0 Hz, 2CH), 6.80 (1H, s, CH), 2.43 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.44 (Cq), 163.57 (Cq), 156.20 (Cq), 142.22 (Cq), 133.62 (CH), 129.73 (CH), 128.90 (CH), 126.19 (Cq), 125.63 (CH), 125.09 (CH), 123.94 (Cq), 118.02 (CH), 106.93 (CH), 21.52 (CH3). HRMS (ESI): m/z calcd for C16H13O2 [M + H]+: 237.0910; found: 237.0914.
2-[4-(Trifluoromethyl)phenyl]-4H-chromen-4-one (3af): Product 3af was obtained as a yellow solid (51 mg, 88%); m.p.: 139–141 °C. 1H NMR (400 MHz, CDCl3): δ = 8.24 (1H, d, J = 8.0 Hz, CH), 8.05 (2H, d, J = 8.2 Hz, 2CH), 7.80 (2H, d, J = 8.2 Hz, 2CH), 7.74 (1H, t, J = 7.8 Hz, CH), 7.60 (1H, d, J = 8.4 Hz, CH), 7.45 (1H, t, J = 7.6 Hz, CH), 6.88 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.17 (Cq), 161.58 (Cq), 156.17 (Cq), 135.16 (d, 4JC–F = 1.0 Hz, Cq), 134.10 (Cq), 133.12 (d, 2JC–F = 33.0 Hz, CH), 126.62 (CH), 126.03 (q, 3JC–F = 3.8 Hz, CH), 125.78 (CH), 125.54 (CH), 123.92 (Cq), 123.58 (d, 1JC–F = 272.6 Hz, Cq), 118.10 (CH), 108.73 (CH). HRMS (ESI): m/z calcd for C16H10F3O2 [M + H]+: 291.0627; found: 291.0632. The NMR data agree with those in a literature report.
44
2-(4-Methoxyphenyl)-4H-chromen-4-one (3ag): Product 3ag was obtained as a yellow solid (38 mg, 75%); m.p.: 151–153 °C. 1H NMR (400 MHz, CDCl3): δ = 8.23 (1H, d, J = 8.0 Hz, CH), 7.89 (2H, d, J = 8.8 Hz, 2CH), 7.69 (1H, t, J = 7.8 Hz, CH), 7.55 (1H, d, J = 8.4 Hz, CH), 7.41 (1H, t, J = 7.6 Hz, CH), 7.03 (2H, d, J = 8.8 Hz, 2CH), 6.76 (1H, s, CH), 3.89 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.34 (Cq), 163.40 (Cq), 162.38 (Cq), 156.15 (Cq), 133.54 (CH), 127.98 (CH), 125.63 (CH), 125.05 (CH), 123.98 (Cq), 123.88 (Cq), 117.93 (CH), 114.43 (CH), 106.14 (CH), 55.48 (CH3). HRMS (ESI): m/z calcd for C16H12NaO3 [M + Na]+: 275.0679; found: 275.0685.
2-(2-Chlorophenyl)-4H-chromen-4-one (3ah): Product 3ah was obtained as a yellow solid (38 mg, 74%); m.p.: 113–115 °C. 1H NMR (400 MHz, CDCl3): δ = 8.26 (1H, d, J = 7.8 Hz, CH), 7.71 (1H, t, J = 7.6 Hz, CH), 7.64 (1H, d, J = 7.0 Hz, CH), 7.53 (2H, t, J = 8.2 Hz, 2CH), 7.49–7.38 (3H, m, 3CH), 6.66 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.13 (Cq), 162.63 (Cq), 156.57 (Cq), 133.91 (CH), 132.91 (Cq), 131.90 (Cq), 131.76 (CH), 130.79 (CH), 130.63 (CH), 127.07 (CH), 125.74 (CH), 125.33 (CH), 123.83 (Cq), 118.18 (CH), 112.99 (CH). HRMS (ESI): m/z calcd for C15H9ClNaO2 [M + Na]+: 279.0183; found: 279.0184.
2-(o-Tolyl)-4H-chromen-4-one (3ai): Product 3ai was obtained as a yellow oil (39 mg, 83%). 1H NMR (400 MHz, CDCl3): δ = 8.26 (1H, d, J = 8.0 Hz, CH), 7.70 (1H, t, J = 7.8 Hz, CH), 7.51 (2H, dd, J = 13.8, 8.0 Hz, 2CH), 7.46–7.39 (2H, m, 2CH), 7.33 (2H, d, J = 7.2 Hz, 2CH), 6.50 (1H, s, CH), 2.49 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.29 (Cq), 166.08 (Cq), 156.42 (Cq), 136.75 (Cq), 133.74 (Cq), 132.56 (CH), 131.22 (CH), 130.68 (CH), 129.16 (CH), 126.17 (CH), 125.70 (CH), 125.20 (CH), 123.74 (Cq), 118.01 (CH), 111.90 (CH), 20.52 (CH3). HRMS (ESI): m/z calcd for C16H12NaO2 [M + Na]+: 259.0730; found: 259.0720.
2-(2-Methoxyphenyl)-4H-chromen-4-one (3aj): Product 3aj was obtained as a yellow solid (48 mg, 95%); m.p.: 101–102 °C. 1H NMR (400 MHz, CDCl3): δ = 8.23 (1H, dd, J = 8.0, 1.6 Hz, CH), 7.91 (1H, dd, J = 7.8, 1.6 Hz, CH), 7.70–7.65 (1H, m, CH), 7.53 (1H, d, J = 8.2 Hz, CH), 7.51–7.45 (1H, m, CH), 7.40 (1H, t, J = 7.6 Hz, CH), 7.15 (1H, s, CH), 7.11 (1H, t, J = 7.2 Hz, CH), 7.05 (1H, d, J = 8.4 Hz, CH), 3.94 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.89 (Cq), 160.84 (Cq), 157.96 (Cq), 156.46 (Cq), 133.50 (CH), 132.38 (CH), 129.25 (CH), 125.58 (CH), 124.87 (CH), 123.79 (Cq), 120.80 (Cq), 120.69 (CH), 118.00 (CH), 112.63 (CH), 111.72 (CH), 55.65 (CH3). HRMS (ESI): m/z calcd for C16H12NaO3 [M + Na]+: 275.0679; found: 275.0668.
2-(m-Tolyl)-4H-chromen-4-one (3ak): Product 3ak was obtained as a yellow solid (36 mg, 77%); m.p.: 108–110 °C. 1H NMR (400 MHz, CDCl3): δ = 8.23 (1H, d, J = 8.0 Hz, CH), 7.76–7.66 (3H, m, 3CH), 7.58 (1H, d, J = 8.4 Hz, CH), 7.45–7.38 (2H, m, 2CH), 7.35 (1H, d, J = 7.6 Hz, CH), 6.82 (1H, s, CH), 2.46 (3H, s, CH3). 13C NMR (101 MHz, CDCl3): δ = 178.50 (Cq), 163.60 (Cq), 156.25 (Cq), 138.81 (Cq), 133.69 (CH), 132.38 (CH), 131.70 (CH), 128.91 (Cq), 126.82 (CH), 125.66 (CH), 125.15 (CH), 124.00 (Cq), 123.48 (CH), 118.06 (CH), 107.54 (CH), 21.49 (CH3). HRMS (ESI): m/z calcd for C16H12NaO2 [M + Na]+: 259.0730; found: 259.0719.
2-(3-Bromophenyl)-4H-chromen-4-one (3al): Product 3al was obtained as a yellow solid (44 mg, 73%); m.p.: 90–92 °C.
1
H NMR (400 MHz, CDCl3): δ = 8.23 (1H, d, J = 7.0 Hz, CH), 8.08 (1H, s, CH), 7.83 (1H, d, J = 8.0 Hz, CH), 7.75–7.69 (1H, m, CH), 7.67 (1H, d, J = 8.0 Hz, CH), 7.59 (1H, d, J = 8.4 Hz, CH), 7.47–7.36 (2H, m, 2CH), 6.80 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.17 (Cq), 161.64 (Cq), 156.13 (Cq), 134.41 (CH), 133.98 (CH), 133.77 (CH), 130.53 (CH), 129.22 (Cq), 125.73 (CH), 125.42 (CH), 124.81 (Cq), 123.89 (Cq), 123.22 (CH), 118.08 (CH), 108.16 (CH). HRMS (ESI): m/z calcd for C15H10BrO2 [M + H]+: 300.9859; found: 300.9869.
2-(tert-Butyl)-4H-chromen-4-one (3am): Product 3am was obtained as a yellow solid (37 mg, 91%); m.p.: 72–74 °C. 1H NMR (400 MHz, CDCl3): δ = 8.18 (1H, d, J = 8.0 Hz, CH), 7.66 (1H, t, J = 7.8 Hz, CH), 7.46 (1H, d, J = 8.4 Hz, CH), 7.38 (1H, t, J = 7.6 Hz, CH), 6.29 (1H, s, CH), 1.36 (9H, s, 3CH3). 13C NMR (101 MHz, CDCl3): δ = 178.94 (Cq), 176.04 (Cq), 156.44 (Cq), 133.43 (CH), 125.52 (CH), 124.78 (CH), 123.40 (Cq), 117.80 (CH), 106.65 (CH), 36.47 (Cq), 27.85 (CH3). HRMS (ESI): m/z calcd for C13H14NaO2 [M + Na]+: 225.0886; found: 225.0891.
2-(Thiophen-2-yl)-4H-chromen-4-one (3an): Product 3an was obtained as a yellow solid (40 mg, 87%); m.p.: 93–95 °C. 1H NMR (400 MHz, CDCl3): δ = 8.21 (1H, dd, J = 8.0, 1.6 Hz, CH), 7.73 (1H, d, J = 3.0 Hz, CH), 7.71–7.65 (1H, m, CH), 7.58 (1H, d, J = 5.0 Hz, CH), 7.53 (1H, d, J = 8.4 Hz, CH), 7.41 (1H, t, J = 7.6 Hz, CH), 7.21–7.17 (1H, m, CH), 6.71 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 177.89 (Cq), 158.99 (Cq), 155.87 (Cq), 135.11 (Cq), 133.71 (CH), 130.23 (CH), 128.46 (CH), 128.41 (CH), 125.64 (CH), 125.23 (CH), 123.95 (Cq), 117.90 (CH), 106.16 (CH). HRMS (ESI): m/z calcd for C13H9O2S [M + H]+: 229.0318; found: 229.0307.
2-(Naphthalen-2-yl)-4H-chromen-4-one (3ao): Product 3ao was obtained as a yellow solid (51 mg, 93%); m.p.: 157–159 °C. 1H NMR (400 MHz, CDCl3): δ = 8.47 (1H, s, CH), 8.29–8.22 (1H, m, CH), 8.01–7.86 (4H, m, 4CH), 7.75–7.69 (1H, m, CH), 7.63 (1H, d, J = 8.0 Hz, CH), 7.60–7.55 (2H, m, 2CH), 7.43 (1H, t, J = 7.2 Hz, CH), 6.96 (1H, s, CH). 13C NMR (101 MHz, CDCl3): δ = 178.41 (Cq), 163.27 (Cq), 156.29 (Cq), 134.61 (Cq), 133.77 (CH), 132.85 (Cq), 129.01 (CH), 128.90 (CH), 128.85 (CH), 127.98 (CH), 127.79 (CH), 127.04 (Cq), 126.88 (CH), 125.69 (CH), 125.21 (CH), 123.99 (CH), 122.47 (Cq), 118.08 (CH), 107.85 (CH). HRMS (ESI): m/z calcd for C19H13O2 [M + H]+: 273.091; found: 273.0922.
2-(Adamantan-1-yl)-4H-chromen-4-one (3ap): Product 3ap was obtained as a white solid (53 mg, 95%); m.p.: 100–102 °C. 1H NMR (400 MHz, CDCl3): δ = 8.18 (1H, dd, J = 8.0, 1.6 Hz, CH), 7.68–7.62 (1H, m, CH), 7.45 (1H, d, J = 8.2 Hz, CH), 7.40–7.33 (1H, m, CH), 6.20 (1H, s, CH), 2.13 (3H, s, 3CH), 2.00–1.96 (6H, m, 3CH2), 1.86–1.73 (6H, m, 3CH2). 13C NMR (101 MHz, CDCl3): δ = 179.03 (Cq), 175.77 (Cq), 156.45 (Cq), 133.34 (CH), 125.53 (CH), 124.70 (CH), 123.61 (Cq), 117.79 (CH), 106.59 (CH), 39.47 (CH), 38.17 (CH2), 36.41 (CH2), 27.94 (CH2). HRMS (ESI): m/z calcd for C19H21O2 [M + Na]+: 281.1536; found: 281.1529.